Rusty-spotted Genet
Genetta maculata

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Genetta maculata – (Gray, 1830)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – VIVERRIDAE – Genetta – maculata
Common Names: Large-spotted Genet, Rusty-spotted Genet, Blotched Genet, Central African Large-spotted Genet (English), Grootkolmuskeljaatkat (Afrikaans), Insimba (Ndebele, Swati, Zulu), Tshipa-thoko (Sepedi), Tshipa, T’sipa, Tsipa e Matheba a Maholo (Sesotho), Thokolo (Tswana), Msimba-mangovo, Nsimba (Tsonga), Tshipathokolo, Tsimba (Venda), Inyhwagi (Xhosa), Gemeine Grossfleckgenette Rostfleckgenette (German) Genetta maculata , Genetta panterina (Italian), Genette commune à grandes taches, Genette à taches rousses (French)
Synonyms: aequatorialis Heuglin 1866; albiventris Roberts 1932; deorum Funaioli and Simonetta 1960; erlangeri Matschie 1902; fieldiana Du Chaillu 1860; gleimi Matschie 1902; insularis Cabrera 1921; matschiei Neumann 1902; pumila Hollister 1916; schraderi Matschie 1902; soror Schwarz 1929; stuhlmanni Matschie 1902; zambesiana Matschie 1902
Taxonomic status: Species complex (but probably only one species in the assessment region).
Taxonomic Note:
Previously considered part of Genetta tigrina. The species epithet “maculata” is no longer valid according to the International Commission on Zoological Nomenclature, and thus should only be used as a provisional naming (ICZN 2007). Genetta maculata should be recognised as an invalid synonym for Genetta pardina (Grubb 2004). The correct name for the Rusty-spotted genet, which has been called Genetta rubiginosa, has yet to be determined (Grubb 2004). Genetta “maculata” is part of the large-spotted genet complex, also including G. pardina (western Africa), G. tigrina (South Africa) and other forest forms with uncertain taxonomic status (Gaubert 2003), and it is suggested to Designate it as a neotype (Gaubert et al. 2003). For discussion on the complex taxonomic history of this taxon see Crawford-Cabral and Fernandes (2001), Gaubert (2003), Gaubert et al. (2005a, b), Wozencraft (2005), Angelici and Gaubert (2013) and Matoso Silva et al. (2016). In Southern Africa there are three species of Genetta that can be considered under the colloquial name Rusty-spotted genets: G. letabae, G.zambesiana and G.mossambica.The first two are parapatric and most likely constitute a superspecies (Crawford-Cabral and Fernandes 2001). Genetta tigrina has an isolated distribution in the Western Cape and Eastern Cape and a plausible contact with G. letabae in Kwazulu-Natal and Free State (Crawford-Cabral and Fernandes 2001). A specimen of the panther genet Genetta maculata, the most common and widespread taxon of the large-spotted genet complex, was sampled in Gauteng Province, and it could be assigned to the subspecies G. m. letabae on geographic grounds (Matoso Silva et al. 2016). Based on museum specimens, cryptic hybridisation has been detected in hybrids with Genetta tigrina from Kwazulu-Natal, though it is rare apparently (Gaubert et al. 2005b).
Through the naked eye, Rusty-spotted Genet differs from Cape Genet by the presence of rusty spots and the absence of “black socks” on both the front and hind legs. In addition, it does not possess a mid-dorsal crest (i.e. longer hairs along the spine), and it has shorter tail hairs (2–3 cm vs 4–4.5 cm) than the Cape Genet, but these differences cannot always be recognised during fleeting encounters in the field or even on pictures. Although both species present additional morphological and genetic differences, further molecular studies are required to solve the taxonomic status of G. maculata relative to G. tigrina; and to establish how many species are in fact present in the G. maculata complex.
Interestingly, melanism in Rusty-spotted genets has been recorded (Camacho et al. 2023), specifically some records from Mpumalanga (Camacho et al. 2023; Forssman and Attwood 2023).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1, da Silva, J.M.2 & Patel, T.3
Reviewer: Do Linh San, E.4
Institutions: 1North West Province’s Directorate of Biodiversity Management; 2South African National Biodiversity Institute,3Endangered Wildlife Trust;4Sol Plaatje University
Previous Assessor & Reviewers: Roux, R., Zemouche, J., Blomsterberg, S., Strauss, M., Madikiza, Z., Somers, M.J., Gaubert, P. & Do Linh San, E.
Previous Contributors: Angelici, F.M., Monadjem, A., Collins, R. & Roxburgh, L.
Assessment Rationale
The Rusty-spotted Genet is listed as Least Concern. It is possible that this species may be undergoing some localised declines in a few areas due to road collisions, direct or accidental persecution by farmers, hunting for skins, meat and trophies, and predation by feral/domestic cats and dogs, it has a wide distribution range, occurring in a variety of habitats, and it is present in many protected areas within the assessment region.
Regional population effects: This species’ range within the assessment region is continuous with the rest of its African range, and we suspect that there is dispersal across regional boundaries.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Power RJ, da Silva JM & Patel T. 2025. A conservation assessment of Genetta maculata. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and Occurrence
Geographic Range
This species is widely distributed in sub-Saharan Africa, ranging from east of the Volta River (Ghana) to east of Eritrea and Somalia (its presence in Djibouti is uncertain) and southwards to central Namibia and KwaZulu-Natal Province in South Africa (Skinner and Chimimba 2005; Angelici & Gaubert 2013). It occurs to high elevations, up to 3,400 m in the Simien Mountains of Ethiopia (Yalden et al. 1996). As recently argued by Hoffmann et al. (2015), it is unlikely that this species is present on Bioko Island, Equatorial Guinea.
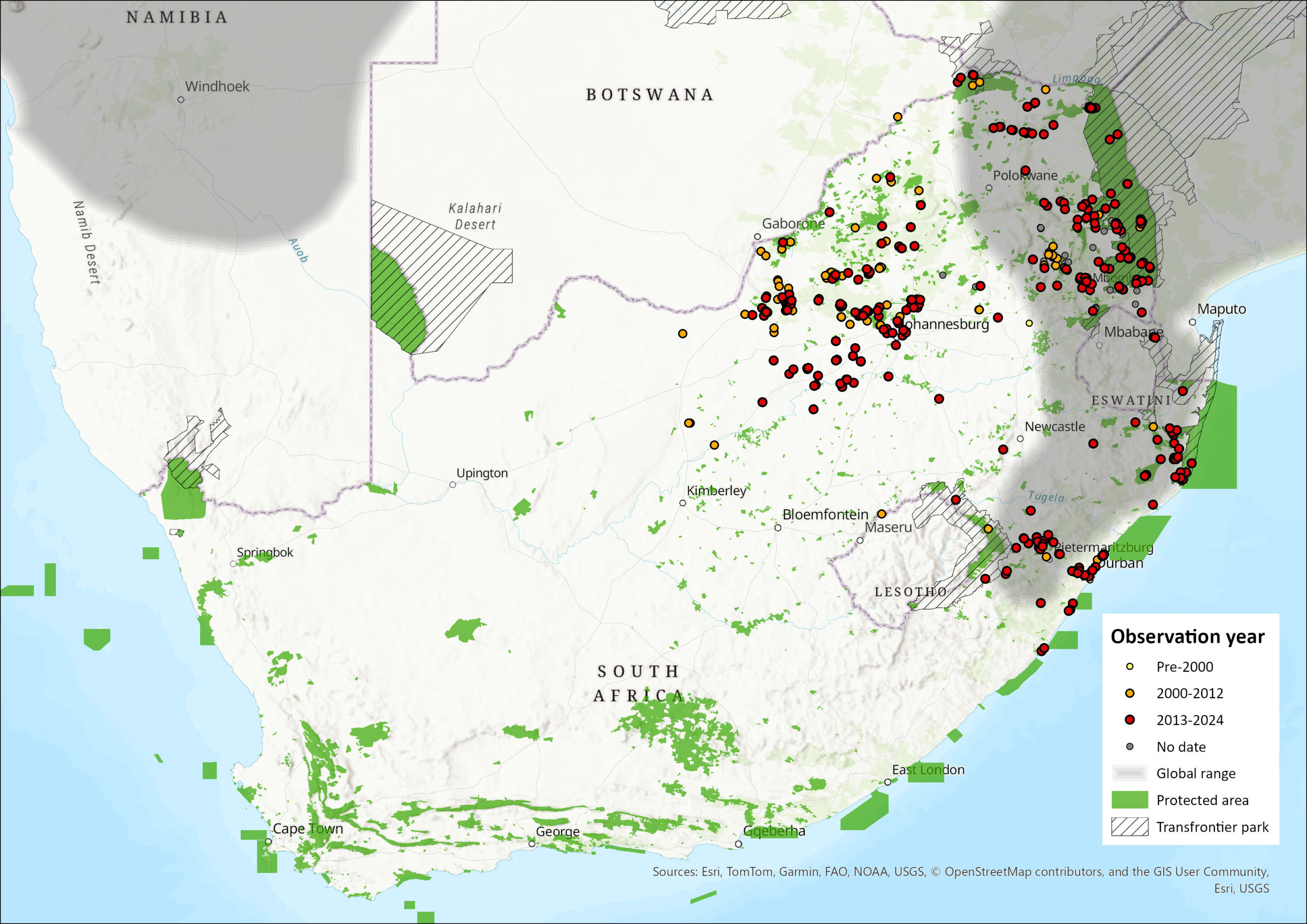
Within the assessment region, this species occurs across Limpopo, eastern Mpumalanga, Eswatini (Mondadjem 1998), KwaZulu-Natal, as well as Gauteng (Roux et al. 2016), and North West provinces (mostly in the east, Power et al. 2019), and it may even marginally enter the Northern Cape (Wilson, B. pers. Comm.). The species seems to be largely absent from the Free State Province (Figure 1). In Mpumalanga, the species occurs well within the Highveld (Loock and Swanepoel 2019), and along the Highveld edge (Roux 2017; Webster et al. 2021), while it has expanded its range onto the Highveld of the North West, as well as marginally into the Kalahari even (Power et al. 2019).
They are typically detected in riparian associations, and the species’ ecological requirements are facilitated by the nation’s cadastral system where farms are allotted into portions. The underlying mechanism for support for this hypothesis is that every portion of farm would invariably have a farmstead with associated woodlots, providing birds and small mammal prey as well as cover. The same principle probably holds true for woodland birds that advance into otherwise inhospitable dry or open ecosystems (Power 2014).
The species has not been recorded in Lesotho, but it might be present (Figure 1). In summary, the species occurs from eastern North West, through Gauteng, Mpumalanga, Eswatini, and KwaZulu-Natal.
Elevation / Depth / Depth Zones
- Elevation Lower Limit (m a.s.l.): (Not specified)
- Elevation Upper Limit (m a.s.l.): (Not specified)
- Depth Lower Limit (m b.s.l.): (Not specified)
- Depth Upper Limit (m b.s.l.): (Not specified)
- Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Rusty-spotted Genet (Genetta maculata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
|---|---|---|---|---|
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, DRC | Extant | Native | – | – |
| Djibouti | Presence Uncertain | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Biogeographic Realms
-
Biogeographic Realm: Afrotropical
FAO Area Occurrence
-
FAO Marine Areas: (Not specified)
Climate change
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify these using expert knowledge (Foden & Young 2016). An increase in shrub cover as predicted by many climate change models (Foden and Young 2016), may enhance the species prevalence, and some models have supported range expansion hypotheses for this species as a consequence of climate change for the species (Power 2014), and mammals in general (Power et al. 2019).
Population
This species is relatively common, though its nocturnal and elusive habits mean that it may be infrequently recorded. For example, in southeastern Nigeria, sightings are rare, but Rusty-spotted Genets are commonly recorded in bushmeat markets and frequently trapped by local hunters (Angelici et al. 1999a,b).
Within the assessment region, we suspect that the species is widespread and fairly common and is often recorded as being commensal with humans and adapting to human-modified landscapes (see Williams et al. 2018). We suspect that there are > 10,000 mature individuals. It is widespread and common in Eswatini (Monadjem 1998), both inside and outside of protected areas (Roux et al. 2016).
Population Information
Current population trend: Unknown but probably stable based on wide habitat tolerance and lack of significant threats.
Continuing decline in mature individuals: Unknown but probably not.
Number of mature individuals in population: Unknown, but probably > 10,000.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: No. Rusty-spotted Genets have a broad habitat tolerance and can exist in agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
To date, no comprehensive population genetic study has been done on this species. Consequently, the genetic structure is being inferred based on other available information. Given the known distribution of the species in the assessment region and its dispersal capabilities, it is likely that the species exists as a single genetic population (metapopulation), linked with neighbouring countries. There have been no reported extinctions of populations within the region; hence, as per the Convention on Biological Diversity’s population genetic indicators, this species has all of its populations maintained (1 continuous metapopulation; PM indicator = 1/1 = 1.0).
Based on the assumption that greater than 10 000 individuals remain and applying a census size to effective population size (Ne) conversion ratio ranging between 0.1-0.3, the Ne for this metapopulation is estimated above 1000-3000 individuals – above the Ne 500 threshold. Given that this species is not known to have experienced any sever population bottlenecks this estimate is likely to be adequate; however, given that the ‘census’ size was a broad estimate, it is highly recommended this be validated using population genetic metrics, if at all possible.
Such a population genetic study would also shed more light on hybrid zones where multiple genet species co-occur (Gaubert et al. 2005b), and the impact or proliferation of these hybrids into the greater genet population.
Habitats and ecology
The Rusty-spotted Genet is present in a variety of habitats, including rainforest, swampy areas, riverine vegetation, open and closed woodlands, moist forests, savannah–forest mosaics, thickets and even grassy savannah, but avoids extremely dry savannah and truly arid regions (Smithers 1971; Skinner and Chimimba 2005; Angelici & Gaubert 2013). It also occurs in cultivated areas (plantations), farmlands and suburban areas (Roux et al. 2016). An ecological study found that the presence of this species in Nigeria is positively correlated with “derived savannah”, “oil palm plantations” and other altered habitats, but negatively correlated with various types of forests (Angelici & Luiselli 2005), suggesting that the species adapts well to human–modified habitats (Roux et al. 2016). The species is known to utilise dense vegetation (Ramesh et al. 2017; Roux 2017; Webster et al. 2021). In Maputaland, KwaZulu-Natal, this genet species was more likely detected in closed habitats (Ramesh et al. 2017). In the Telperion Nature Reserve, on the edge of the Highveld in Mpumalanga, the Rusty-spotted Genet selected bushveld vegetation over and above grassland (Roux 2017), in particular riparian areas (Webster et al. 2021).
Resting sites are often located in trees, but also in dense shrubs, disused burrows of other animals such as Aardvark (Orycteropus afer) or Springhare (Pedetes spp.), rock crevices and overhangs, and even inhabited or abandoned man-made structures (Angelici & Roux 2017). Rusty-spotted Genets are both terrestrial and arboreal and can sometimes be seen taking shelter in trees or other elevated areas (Angelici & Gaubert 2013).
This species is mainly carnivorous (Smithers 1971; Skinner and Chimimba 2005), but will also eat vegetable matter such as fruits, seeds and berries (Angelici & Gaubert 2013). Mainly mammals and insects are caught, but centipedes and millipedes, spiders, scorpions, amphibians and reptiles (including lizards and snakes) as well as small birds and eggs are also hunted (Smithers 1971; Angelici & Luiselli 2005; Martinoli et al. 2006; Angelici & Gaubert 2013; Zemouche 2018). Across their range murids, and insects are common in their diet (Smithers 1971; Skinner and Chimimba 2005; Zemouche 2018), At Telperion Nature Reserve, small mammals and arthropods were found to be the dominant prey of the species, with birds, reptiles, seeds/fruit pulp, plant material and amphibians serving as supplementary food sources (Zemouche 2018). These genets will catch mammals up to the size of a hare and even aquatic animals such as gastropods and fish (Angelici & Gaubert 2013). They will also scavenge on human leftovers (Roux et al. 2016). The Rusty-spotted Genet is an opportunistic feeder and will eat whatever is mostly available in its area during the season (Roux et al. 2016; Zemouche 2018). In Tanzania, fruits were an important food source (Martinoli et al. 2006), probably due to their availability being higher than in South Africa. The percentage occurrence of a range of food items in the stomachs of 136 Rusty-spotted Genets in Zimbabwe (Smithers & Wilson 1979) and 30 in Botswana (Smithers 1971) was assessed. Results showed that, in Zimbabwe, 68% of the stomach contents consisted of murids, followed by 40% insects and 15% birds (Smithers & Wilson 1979). In Botswana, insects were the main food source comprising 90% of the diet followed by 47% murids, 27% arachnids and 17% fruits (Smithers 1971). There only 7% birds occurred (Smithers 1971). Insects that are eaten are usually Coleoptera, Orthoptera and Isoptera (Angelici & Gaubert 2013).
It is a generalist opportunist feeder which supplements it diet when preferred food items become less abundant and varying its diet according to season in response to seasonal changes in prey availability (Zemouche 2018). Interestingly, G. maculata overlaps in its diet with the Slender Mongoose (Herpestes sanguineus) (Zemouche 2018).
The Rusty-spotted Genet is mostly nocturnal (Smithers 1971; Skinner and Chimimba 2005; Ramesh et al. 2017; Roux 2017’), and part crepuscular (Webster et al. 2021) and spends the day in their resting sites (Angelici & Gaubert 2013; Roux 2017).
At the Telperion Nature Reserve, Rusty-spotted Genets are more active during the first half of the night and often have a short resting bout halfway through their active period. Activity usually starts with sunset and ends before sunrise. Males are more active than females, possibly due to higher metabolic requirements and/or different reproductive strategies. During winter nights there, they terminated activity earlier than in summer due to the cold temperatures. Predation risk may play an important role in the spatial ecology, as they select areas with enough vegetation cover and often in proximity of safe refuges while they are out hunting (Roux 2017). Genet activity there, was affected by cold temperature (Roux 2017; Webster et al. 2021), and they were active only above 5˚C (Webster et al. 2021).
The Rusty-spotted Genet is solitary and territorial although home ranges can sometimes overlap (Carpenter 1970). In Kenya, the home ranges of five radio-tracked genets varied between 0.1 and 1.0 km2 (Angelici & Gaubert 2013). In Telperion Nature Reserve, the home ranges of 15 individuals were found to be on average 3.3 km2 (range 2.1–7.0 km2) in size (Roux 2017). Male genet home-ranges there, averaged 2.9 ± 2.3 km2 (n= 6), while females were on average 3.3 ± 2 km2 (n=6) (Roux 2017), and thus there was minimal inter-sexual difference. Carpenter (1970) reported that male home ranges are usually larger than that of females, but no difference or even the opposite tendency was observed in Telperion (see Roux 2017). At Telperion, the daily movement distance for male rusty-spotted genets was on average 4.9 km, while females it was 4.3 km (Roux 2017). It is not clear to what extent these genets defend their territories, but they do mark them with secretions from the perineal glands, urine and faeces (Angelici & Gaubert 2013). Rusty-spotted Genets share latrine sites with other individuals (Blomsterberg 2016) and possibly also with other species such as African Civets (Civettictis civetta) and several mongoose species (Engel 2000). They could use these latrine sites for olfactory communication although specific scent-marking behaviour was not observed by Blomsterberg (2016). The breeding peak is from October to December, but a second peak can occur between March and May (Angelici & Gaubert 2013). Two to five kittens are generally born (Skinner & Chimimba 2005). Males may produce grumbling and coughing calls when courting a female and meowing has been recorded during mating (Dücker 1965). Soft growls and hissing were observed when wild Rusty-spotted Genets were captured in Telperion Nature Reserve (Roux 2017).
Rusty-spotted Genet crosses a canal, unusually so during the day in the Marico valley of the North West (John Power).
Ecosystem and cultural services: None have been described specifically. However, it is likely that, together with other small carnivores, this species plays a role in controlling rodent and arthropod populations, notably in agricultural areas (Roux et al. 2016).
The Rusty-spotted Genet features rodents in its diet, so it may be a useful agroecosystem service (Williams et al. 2018). In north-eastern Limpopo, in communal lands, the species has been frequently detected in croplands, grazing rangelands and in the vicinity of some settlements (Williams et al. 2018).
Rusty-spotted Genets, together with the two other genet species occurring in the assessment region, have the potential to become a symbol/indicator of urban wildlife and integration of development with natural landscapes (Roux et al. 2016).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Use as nutrition
There are a few reports mentioning that this species can be utilised as bushmeat, especially in western Africa (Angelici et al. 1999b). In Eswatini, genets are killed for food (Monadjem 1998).
Medicinal uses trade
The species is traded in Gauteng (Whiting et al. 2013), KwaZulu-Natal (Cunningham & Zondi 1991), and Eswatini (Monadjem 1998). In southern Africa, body parts may also be used for medicinal purposes, with pieces of genet skin used as stick-fight charms and parts of the body are used to treat eye ailments (Cunningham & Zondi 1991).
Body parts for decorative purposes
Genet hides and tails are sometimes used in Zulu culture as traditional adornments (Roux et al. 2016). In Eswatini, genets are killed for their skins which are used as clothing attire, especially for males, i.e. ‘emajobo’ (Monadjem 1998). Such practices, however, are localised and limited, and thus should not have a negative impact on the population (Roux et al. 2016).
Trophy hunting
There has been a steady increasing trend in use of the species by hunters in the last decade and a half, and stabilisation since the last assessment period (prior to 2016). However, there has been a decrease noted in 2019.
In the North West Province, hunting of the species is less prominent than that of G. genetta. Since 2020, there have been 5 hunting applications for the species, and 4 of these have been approved, owing inter alia to the revised quota policy for these species (North West Directorate of Biodiversity Management Records, 2020-2023). The species can only be hunted between April and September, and only one animal per property per year would be allowed, and distributional occurrence is furthermore taken into account. The species is provincially protected in most provinces, thus it requires a permit to utilise it.
Captive keeping
It is becoming increasingly popular to keep several genet species as pets, specifically in the USA. In southern Africa, it is not common, however, and since it is expected that such animals originate from captive bred populations and not from the wild, it should not influence wild population numbers excessively. The number and proportion of Rusty-spotted Genets kept as pets both globally and in the assessment region is unknown (Roux et al. 2016). However, as an example in the North West Province, as of the end of 2023, there were 40 Rusty-spotted Genets kept in captivity across 10 permitted facilities (North West Biodiversity Management Directorate records, 2022-2023). In the same province, there has only been one case, at least since 2020, where ‘genets’ have been kept illegally, though it may have been easy to identify the correct species, this was unfortunately not done (North West Biodiversity Management Directorate records, 2020-2023). Although they are endearing, the trade in keeping the species is probably not widespread.
Ecotourism
The species is very endearing to see and can be seen on guided night drives, and also in rest camps, where they may be semi-commensal with tourists there.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used as bushmeat, medicine or for their skins. | – | Selling of individuals as pets (or breeding stock). Local commercial use in traditional medicine trade and trophy hunting. |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 3. Medicine – human & veterinary | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Traditional medicine probably stable; trophy hunting predicted to increase.
Threats
There are no major threats to the species (Roux et al. 2016).
Road kills
Rusty-spotted Genets have been recorded in the Endangered Wildlife Trust’s road collision database (Roux et al. 2016), but the extent of road mortality on this species within the assessment region is unknown. There appear to be hotspots where this species frequently falls victim to road traffic (A. Halijian pers. comm. 2015–2016, In: Roux et al. 2016), and this might be related to both habitat features and higher local abundance.
Collinson et al. (2015b) drove a total of 14 400 km over 120 days spanning 10 months across 2011 and 2012, on a 120 km circuit around the Venetia Limpopo Nature Reserve, and a stretch that abutted the Mapungubwe National Park. On this circuit, only one genet was reported to have been killed on roads (Collinson et al. 2015). Given the 10-month study period, this would amount to 0.38 Genets killed by roads per annum, which may not be that severe. In the North West Province and more recently, from 2020 to 2023, six Rusty-Spotted Genet road kills have been recorded across the whole province. This is while driving non-systematic routes of 85,078 km, with no clear trend across the time period, though ˜30% of driving was done in the Bushveld ecoregion (NWPG 2024, and earlier annual reports). where the species is most found. Across the species there may be local variation in the incidence of road-related mortalities.
Body parts trade
Rusty-spotted Genets have been recorded in bushmeat markets; are locally used for traditional medicine and cultural purposes (Roux et al. 2016), and since they are traded for such use (Whiting et al. 2013), they may be locally threatened.
Trophy hunting
The species is locally hunted for their trophy, notably in Limpopo, but the numbers may be small in comparison with those killed on roads, and from other means.
Persecution owing to predation
Since they do have a reputation as poultry thieves, farmers sometimes poison or trap them (Smithers 1971; Skinner and Chimimba 2005; Roux et al. 2016). In the North West Province, from 2020 to 2023, there have been 22 cases where ‘genets’ have been responded to, and of these, three were said to be of the specific species. Two of the cases involved poultry predation, and one non-conflict related issue. In the cases of poultry, the perpetrating genets were relocated nearby (North West Biodiversity Management Directorate records, 2020-2023).
Habitat degradation
In some rural community areas, Rusty-spotted Genet appear to be impacted by anthropogenic threats, as there are fewer in such landscapes (se Da Rosa 2019), while they seem less impacted in similar areas of north-eastern Limpopo (Williams et al. 2018). In human-modified landscapes of KwaZulu-Natal the species is often collected and taken to rehabilitation centres (Wimberger & Downs 2010), which points to how urbanisation may impact upon them, despite them being relatively adapted to this environment
Domestic dogs/cats
Finally, they are occasionally killed by domestic cats and dogs in both peri-urban and rural areas (Angelici & Gaubert 2013; Roux et al. 2016).
Conservation
This species is present in a large number of protected areas, where the Kruger National Park is probably the most important one. Within the assessment region, no major and urgent conservation interventions are necessary. However, education should be used to raise the profile of this species and encourage farmers to live with rather than against genets. Marketing and awareness campaigns can also be used to position the presence of this species as a point of pride for urban and rural landowners, and conservationists should encourage better land management to facilitate genet conservation.
Compared with mammals worldwide, the overall conservation status of small carnivores in Africa appears relatively favourable (Do Linh San et al. 2013). It has been impressive that some reserves have risen to the challenge and contributed the site for research on the species, where Telperion Nature Reserve has seen furthering of knowledge of the species’ diet (Zemouche 2018), novel work on spatial ecology (Roux 2017), and their activity patterns (Roux 2017; Webster et al. 2021).
In the North West, several orphans have been sent to rehabilitation centres (North West Directorate of Biodiversity Management records, 2020-2023), and the intention is for them to be returned to their origins, but this is not always followed up on. In KwaZulu-Natal, of carnivores admitted to rehabilitation centres, most were orphans, and genet were the second most commonly admitted of all carnivores (Wimberger and Downs 2010). Though the actual genet species was not confirmed, it can be suspected that this species was likely admitted there too.
Recommendations for land managers and practitioners:
- To reduce collisions with vehicles, mitigation measures such as road fencing and improvement of habitat near road crossing structures (for example, underpasses) should be implemented whenever possible (Collinson et al. 2015a).
- Due to the reported predation or killing of Rusty-spotted Genets by feral and domestic dogs and cats (Angelici & Gaubert 2013; Roux et al 2016), it might be necessary to control the number of feral dogs and cats in both urban and rural areas, and encourage dog and cat owners to put collar-mounted bells, sonic bleepers or “pounce protectors” on their pets (Nelson et al. 2005; Calver et al. 2007).
- There needs to be an improved culture of correctly identifying the different genet species.
Research priorities:
- Taxonomic status of G. maculata versus G. tigrina.
- Competition and hybridisation patterns with sympatric species of genets (G. genetta and G. tigrina).
- Population estimates, demographic parameters and possible barriers to dispersal across the species’ distribution range.
- Home range and habitat use of Rusty-spotted Genets in a variety of landscapes (protected areas, agricultural areas and urban landscapes).
- Determine genetic health and diversity of both rural and urban populations.
- Testing of rabies in Rusty-spotted Genets and their potential role as vectors of the disease.
- Effect – if any – of commercial hunting on local populations.
- Encourage population density estimates of them, given their likely unique spot patterns, i.e. spatially explicit capture recapture, or otherwise use by-catch data from studies aimed at more high-profile species, even if relative abundance is all that can be garnered.
- Study the urban population of the species in Johannesburg, in the context of urban wildlife ecology.
Encouraged citizen actions:
- Report sightings of any genet species on virtual museum/social platforms (for example, iNaturalist and MammalMAP), especially outside protected areas, as well as to Emmanuel Do Linh San (emmanuel.dolinhsan@gmail.com). GPS locations and photographs would be of great assistance.
Bibliography
Angelici, F. M., Luiselli, L. and Politano, E. 1999. Distribution and habitat of selected carnivores (Herpestidae, Mustelidae, Viverridae) in the rainforests of southeastern Nigeria. Zeitschrift für Säugetierkunde 64: 116-120.
Angelici, F.M. and Gaubert, P. 2013. Genetta maculata Large-spotted Genet (Blotched Genet). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 232-236. Bloomsbury, London, Uk.
Angelici, F.M. and Luiselli, L. 2005. Habitat associations and dietary relationships between two genets, Genetta maculata and Genetta cristata. Revue d’Écologie (La Terre et la Vie) 60: 341-354.
Angelici, F.M., Luiselli, L., Politano, E. and Akani, G.C. 1999. Bushmen and mammal fauna: A survey of the mammals traded in bush-meat markets of local people in the rainforests of southeastern Nigeria. Anthropozoologica 30: 51-58.
Blomsterberg SE. 2016. The temporal use of latrines by rusty-spotted genet (Genetta maculata Gray 1830) in Telperion Nature Reserve. B.Sc. Honours Thesis. University of Pretoria.
Calver M, Thomas S, Bradley S, McCutcheon H. 2007. Reducing the rate of predation on wildlife by pet cats: the efficacy and practicability of collar-mounted pounce protectors. Biological Conservation 137: 341–348.
Camacho G, Attwood S, Tensen L. 2023. Melanistic Large-Spotted Genets (Genetta maculata) in South Africa. African Journal of Wildlife Research 53(1).
Carpenter GP. 1970. Some observations on the rusty spotted genet (Genetta rubiginosa zuluensis). Lammergeyer 11: 60–63.
Collinson W, Parker D, Patterson-Abrolat C, Alexander G, Davies-Mostert H. 2015a. Setjhaba SA, South Afrika: a South African perspective on an emerging transport infrastructure. In: van der Ree R, Smith DJ, Grilo C. (ed.), Handbook of Road Ecology, pp. 439–447. Wiley, Oxford, UK.
Collinson WJ, Reilly BK, Parker DM, Bernard RT, Davies-Mostert HT. 2015b. An inventory of vertebrate roadkill in the greater Mapungubwe Transfrontier conservation area, South Africa. African Journal of Wildlife Research 45(3): 301-311.
Crawford-Cabral J, Fernandes CA. 2001. The Rusty-spotted genets as a group with three species in Southern Africa (Carnivora: Viverridae). African small mammals 2001:65-80.
Cunningham, A.B. and Zondi, A.S. 1991. Use of animal parts for the commercial trade in traditional medicines. Institute of Natural Resources, University of Natal, Pietermaritzburg, South Africa.
Da Rosa BRP. 2019. Optimizing Small Mammal Relative Abundance Measures Using Non-Invasive Sampling and Assessment of its Contribution to Occupancy Modelling of Small Carnivores in Dry Woodland Savannah of South Africa. Master’s thesis, University of Lisbon, Lisbon.
Do Linh San E, Ferguson AW, Belant JL, Schipper J, Hoffmann M, Gaubert P, Angelici FM, Somers MJ. 2013. Conservation status, distribution and species richness of small carnivores in Africa. Small Carnivore Conservation 48:4-18.
Dücker G. 1965. Das Verhalten der schleichkatzen (Viverridae). Handbuch der Zoologie, Berlin 8: 1–48.
Engel TR. 2000. Seed Dispersal and Forest Regeneration in a Tropical Lowland Biocoenosis (Shimba Hills, Kenya). Logos Verlag, Berlin, Germany.
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Forssman K, Attwood S. 2023. A record of a melanistic rusty-spotted genet (Genetta maculata) from the White River area, Mpumalanga Province, South Africa. African Journal of Wildlife Research 53(1).
Gaubert, P. 2003. Description of a new species of genet (Carnivora; Viverridae; genus Genetta) and taxonomic revision of forest forms related to the Large-spotted Genet complex. Mammalia 67: 85-108.
Gaubert, P., Taylor, P.J. and Veron, G. 2005a. Integrative taxonomy and phylogenetic systematics of the genets (Carnivora, Viverridae, genus Genetta): a new classification of the most speciose carnivoran genus in Africa. In: B.A. Huber, B.J. Sinclair and K.-H. Lampe (eds), African Biodiversity: Molecules, Organisms, Ecosystems, pp. 371-383. Springer, New York, USA.
Gaubert, P., Taylor, P.J., Fernandes, C.A., Bruford, M.W. & Veron, G. 2005b. Patterns of cryptic hybridization revealed using an integrative approach: a case study on genets (Carnivora, Viverridae, Genetta spp.) from the southern African subregion. Journal of the Linnean Society. 86(1):11-33.
Gaubert P, Trainier M, Veron G, Kock D, Dunham AE, Taylor PJ, Stuart C, Stuart T, Wozencraft CW. 2003. Nomenclatural comments on the Rusty-spotted Genet (Carnivora, Viverridae) and designation of a neotype. Zootaxa 160(1):1-4.
Grubb P. 2004. Controversial scientific names of African mammals. African Zoology 39(1):91-109.
Hoffmann, M., Cronin, D.T., Hearn, G., Butynski, T.M. and Do Linh San, E. 2015. A review of evidence for the presence of Two-spotted Palm Civet Nandinia binotata and four other small carnivores on Bioko, Equatorial Guinea. Small Carnivore Conservation 52&53: 13-23.
International Commission of Zoological Nomenclature. 2007. OPINION 2183 (Case 3204). Viverra maculata Gray, 1830 (currently Genetta maculata; Mammalia, Carnivora): specific name not conserved. Bulletin of Zoological Nomenclature 64: 205–206.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Lynch CD. 1984. The large-spotted genet. Culna 27:16-17.
Martinoli A, Preatoni D, Galanti V, Codipietro P, Kilewo M, Fernandes CAR, Wauters LA, Tosi G. 2006. Species richness and habitat use of small carnivores in Arusha National Park (Tanzania). Biodiversity and Conservation 15: 1729–1744.
Matoso Silva R, Adega F, Kjöllerström HJ, Labuschagne K, Kotze A, Fernandes C, Chaves R, do Mar Oom M. 2016. Classical and molecular cytogenetics of the panther genet Genetta maculata (Mammalia, Carnivora, Viverridae). Cytogenetic and Genome Research 149(4):274-281.
Monadjem A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41(2): 45-59.
Nelson SH, Evans AD, Bradbury RB. 2005. The efficacy of collar-mounted devices in reducing the rate of predation of wildlife by domestic cats. Applied Animal Behaviour Science 94: 273–285.
NWPG (North West Provincial Government) 2024 The impact of roadkill on mammal populations in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Pringle JA. 1977. The distribution of mammals in Natal. Part 2. Carnivora. Annals of the Natal Museum 23: 93-115.
Ramesh T, Kalle R, Downs CT. 2017. Staying safe from top predators: patterns of co-occurrence and inter-predator interactions. Behavioral Ecology and Sociobiology. 71:1-4.
Rautenbach IL. 1978. The mammals of the Transvaal. Phd thesis, University of Natal, Pietermaritzburg.
Roux R. 2017. Spatio‐Temporal Ecology of the Rusty‐Spotted Genet, Genetta maculata, in Telperion Nature Reserve (Mpumalanga, South Africa). Msc thesis, UNISA, Johannesburg.
Roux R, Zemouche J, Blomsterberg SE, Strauss WM, Madikiza ZJK, Somers MJ, Gaubert P, Do Linh San E. 2016. A conservation assessment of Genetta maculata. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Webster AB, Pretorius ME, Somers MJ. 2021. The determinants of mesocarnivore activity patterns in highveld grassland and riparian habitats. African Journal of Wildlife Research. 51(1):178-92.
Whiting MJ, Williams VL, Hibbitts TJ. 2013. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. In:Alves RRN, Rosa IL, editors. Animals in traditional folk medicine: implications for conservation. Springer-Verlag Berlin Heidelberg, Pages 421-473.
Widdows CD. 2015. The ecology of large-spotted genets within an urban landscape. Doctoral dissertation, University of Kwazulu-Natal, Pietermaritzburg.
Williams ST, Maree N, Taylor P, Belmain SR, Keith M, Swanepoel LH. 2018. Predation by small mammalian carnivores in rural agro-ecosystems: an undervalued ecosystem service?. Ecosystem Services 30: 362-371.
Wimberger K, Downs CT. 2010. Annual intake trends of a large urban animal rehabilitation centre in South Africa: a case study. Animal Welfare. (4):501-513
Wozencraft, W.C. 2005. Order Carnivora. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World: A Taxonomic and Geographic Reference. Third Edition, pp. 532-628. Johns Hopkins University Press, Baltimore.
Yalden, D.W., Largen, M.J., Kock, D. and Hillman, J.C. 1996. Catalogue of the mammals of Ethiopia and Eritrea. 7. Revised checklist, zoogeography and conservation. Tropical Zoology 9: 73-164.
Zemouche J. 2018. Trophic ecology of rusty-spotted genet Genetta maculata and slender mongoose Herpestes sanguineus in Telperion Nature Reserve, with a focus on dietary segregation as a possible mechanism of coexistence. Msc thesis, University of the Witwatersrand, Johannesburg.