
Rufous Hairy Bat
Myotis bocagii

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Myotis bocagii – (Peters, 1870)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Myotis – bocagii
Common Names: Rufous Hairy Bat, Bocage’s Mouse-eared Bat, Bocage’s Hairy Bat, Rufous Mouse-eared Bat, Rufous myotis (English), Rooi langhaarvlermuis (Afrikaans), Liputiputi/Ndemiya (Malawian)
Synonyms: Vespertiliio bocagii (W. C. H. Peters, 1870), dogalensis (Monticelli, 1887), bocagei (du Bocage, 1889) [incorrect subsequent spelling}, cupreolus O. Thomas, 1904, hildegardeae O. Thomas, 1904
Taxonomic Note:
Originally Vespertilio bocagii. Listed as bocagei by Koopman (1993), but this spelling is incorrect. Two subspecies are recognised in Africa; where the nominate subspecies occurs in southern Africa, while M. b. cupreolus Thomas 1904 occurs in West Africa (Monadjem et al. 2020).
Red List Status: LC – Least Concern
Assessment Information
Assessors: Howard, A.1, Richards, L.R.2 & da Silva, J.M.3
Reviewer: Smith, C.4
Institutions: 1University of the Free State, 2Durban Natural Science Museum, 3South African National Biodiversity Institute, 4Endangered Wildlife Trust
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., Cohen, L., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Raimondo, D. & Child, M.F.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (estimated extent of occurrence within the assessment region is 288,645 km2), its occurrence in multiple protected areas (including Great Limpopo Transfrontier Park), and because there are no major identified threats that could be causing widespread decline. It has been shown to occur more extensively in and beyond the assessment region (recorded from KwaZulu-Natal and the Eastern Cape provinces of South Africa) and can tolerate transformed and human disturbed habitats, including cultivated plantations (L. Richards, pers obs.). Further field surveys and research are necessary to delimit distribution, population size, reproductive ecology and habitat selection more accurately.
Regional population effects: The range of this species is continuous across the borders of the assessment region into Zimbabwe through its occurrence in Great Limpopo Transfrontier Park and Greater Mapungubwe Transfrontier Conservation Area. However, it has low wing loading, so rescue effects are uncertain (Schoeman & Jacobs 2008).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Howard A, Richards LR & da Silva JM. 2025. A conservation assessment of Myotis bocagii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widespread but patchily distributed throughout much of sub-Saharan Africa. It ranges from Sierra Leone and Senegal in West Africa, eastwards through Cameroon and Central Africa, to Ethiopia and East Africa, being recorded as far south as northeastern South Africa (ACR 2024). It occurs in Malawi, Mozambique, Zambia, the Democratic Republic of the Congo and Angola (Monadjem et al. 2020). It is probably more widespread in Mozambique and eastern Zambia than is currently documented (Monadjem et al. 2020). Outside of Africa, it has been recorded from southern Yemen (ACR 2024). Riparian fringes along the Limpopo and Zambezi rivers explains outlying records of this species in semi-arid savannahs of southern and northern Zimbabwe (Monadjem et al. 2010). Within the assessment region, it is restricted to the eastern lowveld regions, having been recorded from Limpopo, Mpumalanga, KwaZulu-Natal and Eastern Cape provinces of South Africa. It has been confirmed to occur in Eswatini where two individuals were sampled at localities 75 km apart at Mlawula weir in Mlawula Nature Reserve in 2007 and at the Ngonini Citrus Estate in 2010 (Shapiro & Monadjem 2016). Its current estimated extent of occurrence within the assessment region is 288,645 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 7 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,497 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
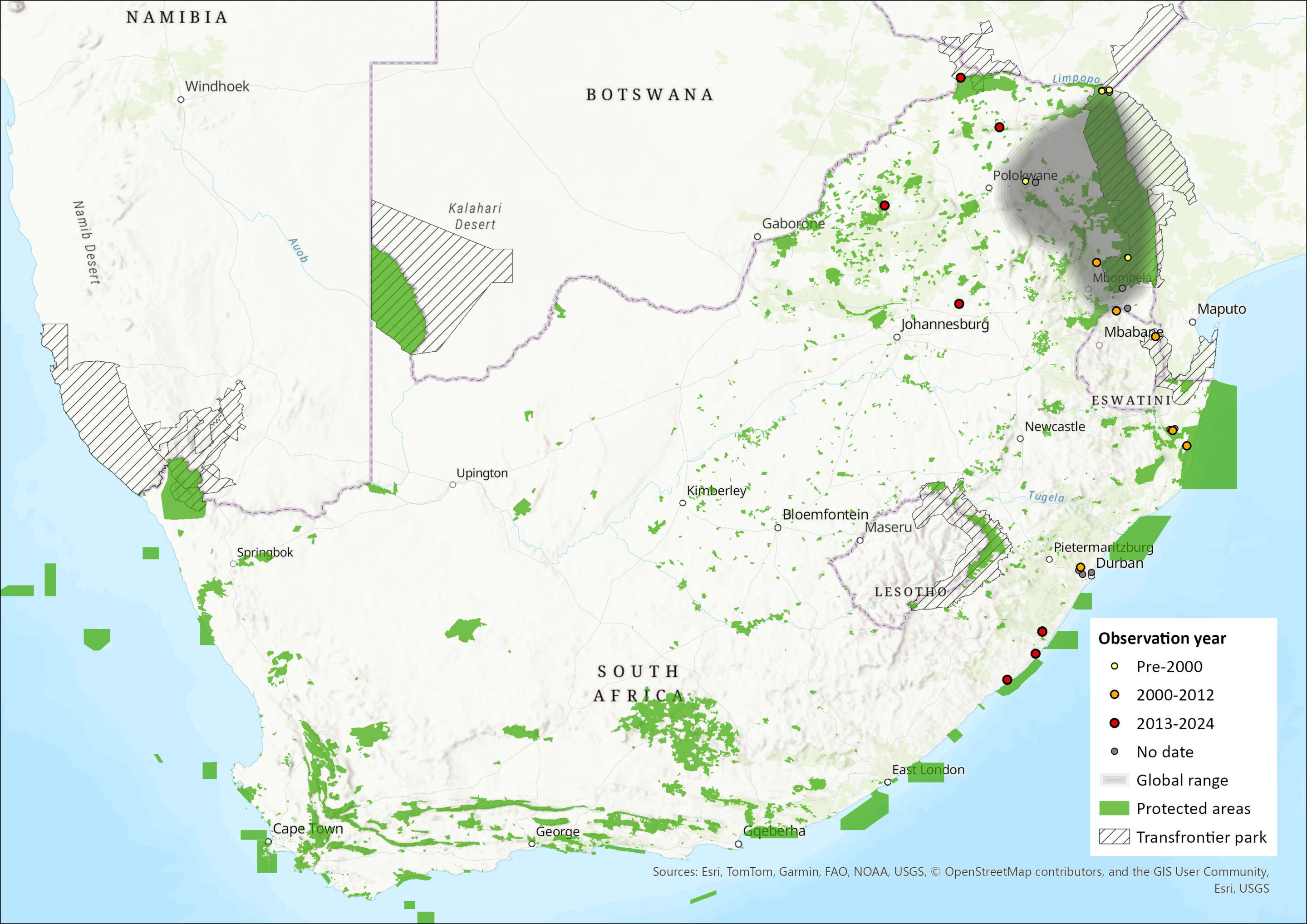 Figure 1. Distribution records for Rufous Hairy Bat (Myotis bocagii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Rufous Hairy Bat (Myotis bocagii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No formal study on the direct effects of climate change in this species has been conducted. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as maximum temperatures are expected to increase over much of southern Africa for this water-dependent species (Adams & Hayes 2021; Mbokodo et al. 2020; Monadjem et al. 2020).
Population Information
This species forages low over open water and has a close association to wetlands and riverine habitats (Monadjem et al. 2020), meaning that it may be more common that current records suggest. It is not well represented in museums, with only 60 records examined in Monadjem et al. (2020). It is often mentioned in southern African studies that this species occurs singly or in pairs (Happold 1987; Skinner & Chimimba 2005). However, in Central and West Africa, it lives in harem groups (Brosset 1976; Monadjem & Fahr 2007) and this may be the case in southern Africa (Monadjem et al. 2020).
Current population trend: Stable
Continuing decline in mature individuals? Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Suspected
Number of mature individuals in largest subpopulation: Cannot be determined
Number of Subpopulations: Suspected to be one
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Not likely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years:
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic studies have been conducted on this species, and considering M. bocagii is somewhat difficult to survey owing to its propensity for flying close to the surface of waterways, it is possible its distribution is more continuous, connecting eastern and northern parts of South Africa than currently depicted based on records. As the population size is unknown; the effective population size cannot be estimated. See Patterson et al. (2019) for the phylogeographic relationships of Afrotropical Myotis species.
Habitats and ecology
In southern Africa, it appears to be associated with low-lying riverine habitats or wetlands within a savannah or woodland vegetation matrix (Monadjem et al. 2020). This species has also been recorded from tropical moist forest (Rosevear 1965). Populations are also often found close to rivers and streams bordered by forest (Happold et al. 1987). Allen (1917) noted that the species tended to avoid human settlements. However, it appears to tolerate disturbed habitats to a degree. For example, an individual from Ngonini Citrus Estate in Eswatini was sampled along a small perennial stream with disturbed riparian forest (Shapiro & Monadjem 2016). Similarly, six individuals were sampled along the polluted Umbilo River in the Durban region in 2008 (Naidoo et al. 2011). However, this may have been influenced by the presence of nearby Paradise Valley Nature Reserve, which possibly provided roosting sites such as tree cavities and wild banana (Strelitzia nicolai) (Naidoo et al. 2011). A grounded pup was located in a complex alongside the Umgeni River, in the greater Durban North region (L. Richards, pers. obs.). Adult bats were noted flying above the grounded pup.
Its roosting habits are not known in southern Africa. However, in West Africa it has been captured singly or in groups of up to eight in furled banana leaves (Monadjem & Fahr 2007), or other plants with broad leaves and hollow trees (Rosevear 1965; Happold et al. 1987). It has been netted in mixed banana and macadamia plantations in KwaZulu-Natal and is likely able to utilise the furled banana leaves as roosting sites (L. Richards pers. obs.; Durban Natural Science Museum records) In northern Mozambique, it has been netted in a banana plantation (A. Monadjem unpubl. data). Very little is known regarding the reproductive behaviour of the species within southern Africa (Monadjem et al. 2020). It is a clutter-edge and clutter forager; feeding mainly on Trichoptera, Lepidoptera, Coleoptera, Hemiptera, and Diptera (Monadjem et al. 2020; Naidoo et al. 2011). Along the Umbilo River, this species fed opportunistically on Diptera during winter (Naidoo et al. 2011). Morales et al. (2024) assigned M. bocagii to the “trawler” ecomorph group as a water-surface forager.
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations that damage crops (Boyles et al. 2011; Kunz et al. 2011).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: specified) Mean Forearm = 4.04 cm
Size at Maturity (in cms): Male: Mean Forearm = 3.91 cm
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms):
Size at Birth (in cms):
Gestation Time: Unknown
Reproductive Periodicity: Unknown
Average Annual Fecundity or Litter Size: Assumed one
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
A recent study by Tarango et al. (2025) listed this species as imported into the U.S.A so online e-commerce platforms should be monitored for illegal trade in bat taxidermy and specimens from the assessment region
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species (ACR 2024). It appears to be able to utilise semi-disturbed vegetation or landscapes for roosting and foraging. However, there is ongoing habitat loss from agricultural transformation, especially in KwaZulu-Natal (Jewitt et al. 2015). Selective logging of trees for fuelwood and charcoal production may also cause local declines. Pollution of slow-flowing rivers, decline of aquatic invertebrate prey and altered precipitation patterns for this water-associated species are some potential future threats to be investigated.
Conservation
There are no direct conservation measures currently needed for this species (ACR 2024). In the assessment region, the species is recorded from several protected areas including the Great Limpopo Transfrontier Park, Greater Mapungubwe Transfrontier Conservation Area, Tzaneen Dam and iSimangaliso Wetland Park. It is also known from the Oribi Gorge and Umtamvuna Nature Reserves in KwaZulu-Natal. No direct interventions can be put in place until more data on subpopulation size and trends, as well as local threat severity, is produced. This species would benefit from holistic land management that reduces pesticide use, river pollution and conserves buffer strips of natural vegetation to sustain insect biomass. Identification and protection of key roost sites is also necessary.
Recommendations for land managers and practitioners:
- Reduce pesticide use and pollution in agricultural landscapes and maintain buffer strips of natural vegetation.
- Ensure slow-flowing rivers, riparian habitats and wetlands are protected adequately
Research priorities:
- More research is needed on the distribution and population sizes of the species. Monitoring of known subpopulations should be performed to establish population size and trend.
- Studies into the reproductive behaviour and general ecology of the species are also needed.
- Quantification of severity of local threats.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report 2015. Pretoria, South Africa.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Allen, J.A., Lang, H. and Chapin J. 1917. The American Museum Congo Expedition collection of bats. Bulletin of the American Museum of Natural History 37: 405-478.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Brosset, A. 1976. Social organization in the African bat, Myotis bocagei. Z. Tierpsychol. 42: 50–56.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Happold, D.C.D., Happold, M. and Hill, J.E. 1987. The bats of Malawi. Mammalia 51: 337–414.
Happold, D.C.D., Happold, M. and Hill, J.E. 1987. The bats of Malawi. Mammalia 51: 337–414.
Happold, D.C.D. 1987. The Mammals of Nigeria. Oxford University Press, London, UK.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Koopman, K.F. 1993. Order Chiroptera. In: D.E. Wilson and D.M. Reeder (eds), Mammal species of the world: a taxonomic and geographic reference, pp. 137–241. Smithsonian Institution Press, Washington, D. C., USA.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem, A. and J. Fahr. 2007. Rapid survey of bats of North Lorma, Gola and Grebo National Forests, with notes on shrews and rodents.” In A Rapid Biological Assessment of North Lorma, Gola and Grebo National Forests, Liberia. In: Hoke, P., R. Demey and A. Peal (eds), RAP Bulletin of Biological Assessment 44:47-58 and 101-106. Conservation International, Arlington, Virginia.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Morales, A.E., Burbrink, F.T., Segall, M., Meza, M., Munegowda, C., Webala, P.W., Patterson, B.D., Thong, V.D., Ruedi, M., Hiller, M. and Simmons, N.B. 2024. Distinct Genes with Similar Functions Underlie Convergent Evolution in Myotis Bat Ecomorphs. Molecular Biology and Evolution, 41(9), p.msae165.
Naidoo, S., Mackey, R.L. and Schoeman, C.M. 2011. Foraging ecology of insectivorous bats (Chiroptera) at a polluted and an unpolluted river in an urban landscape. Durban Museum Novitates 34: 21–28.
Patterson, B.D., Webala, P.W., Kerbis Peterhans, J.C., Goodman, S.M., Bartonjo, M. and Demos, T.C. 2019. Genetic variation and relationships among Afrotropical species of Myotis (Chiroptera: Vespertilionidae). Journal of Mammalogy, 100(4), pp.1130-1143.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Rosevear, D.R. 1965. The Bats of West Africa. British Museum, London, UK.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defences on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS one 3(11): e3715.
Shapiro, J.T. and Monadjem, A. 2016. Two new bat species for Swaziland and a revised list for the country. Mammalia 80: 353–357.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith, R.J. et al. 2008. Designing a transfrontier conservation landscape for the Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
Tarango, M., Kolby, J.E., Goodman, O.L., Anderson, C.J., Tinsman, J., Kirkey, J., Liew, A., Jones, M., Firth, C. and Reaser, J.K. 2025. Going batty: US bat imports raise concerns for species conservation and human health. One Health, 20, p.100999.
