Rock Hyrax
Procavia capensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Procavia capensis – (Pallas, 1766)
ANIMALIA – CHORDATA – MAMMALIA – HYRACOIDEA – PROCAVIIDAE – Procavia – capensis
Common Names: Rock Hyrax, Rock Dassie (English), Klipdas, Klipdassie (Afrikaans), Imbila (Ndebele, Swati, Xhosa, Zulu), Pela, Thobela, Thewbela (Sepedi), Pela (Sesotho, Setswana), Mbila (Xitsonga, Tshivenda)
Synonyms: Cavia capensis Pallas, 1766; Heterohyrax antineae Heim de Balsac & Bégouen, 1932
Taxonomic Note:
While as many as 17 subspecies have been described across its range by Hoeck and Bloomer (2013), only one (P. c. capensis), is recognised from the assessment region (Meester et al. 1986). However, Prinsloo and Robinson (1992) discovered a genetic break between the north-western and south-central populations in South Africa, indicating a possible species complex. Similarly, Visser (2013) found a genetic break across the Knersvlakte region; however, this was only evident in the mitochondrial DNA (mtDNA, for females). Thus, separation across this barrier has not been long enough to warrant a taxonomic revision of the two lineages flanking it. We treat Rock Hyrax from South Africa as monotypic.
UAE Taxonomic Note
The subspecies present is assumed to be either P. c. syriaca from the north of the region or P. c. jayakari from the south (Aspinall et al. 2005).
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Smith, C.1, & da Silva, J.2
Reviewer: Patel, T.1
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute
Previous Assessors: Visser, J. & Wimberger, K.
Previous Reviewers: Child, M.F.
Previous Contributors: Relton, C. & Bloomer, P.
Assessment Rationale
Listed as Least Concern in view of its wide distribution within the assessment region, the wide range of habitats it occurs in, its occurrence in many protected areas and generally high abundance. Additionally, the rocky habitats in which it occurs are unlikely to be transformed, it adapts readily to urban areas and there are no threats that could cause widespread population decline. Thus, no direct conservation interventions are necessary. However, local declines are possible due to bushmeat consumption. This is an important forage species for a number of predators and subpopulations should be sustained for ecosystem functioning. Indiscriminate reintroductions should be discouraged pending the outcome of more detailed phylogeographic research.
Regional population effects: Continuous distribution with the rest of the African range so rescue effects are possible. Molecular research within the assessment region suggests secondary contact and gene flow between the north-eastern and south-central parts of South Africa (Maswanganye et al. 2017).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Procavia capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa
Regional Distribution and occurrence
Geographic Range
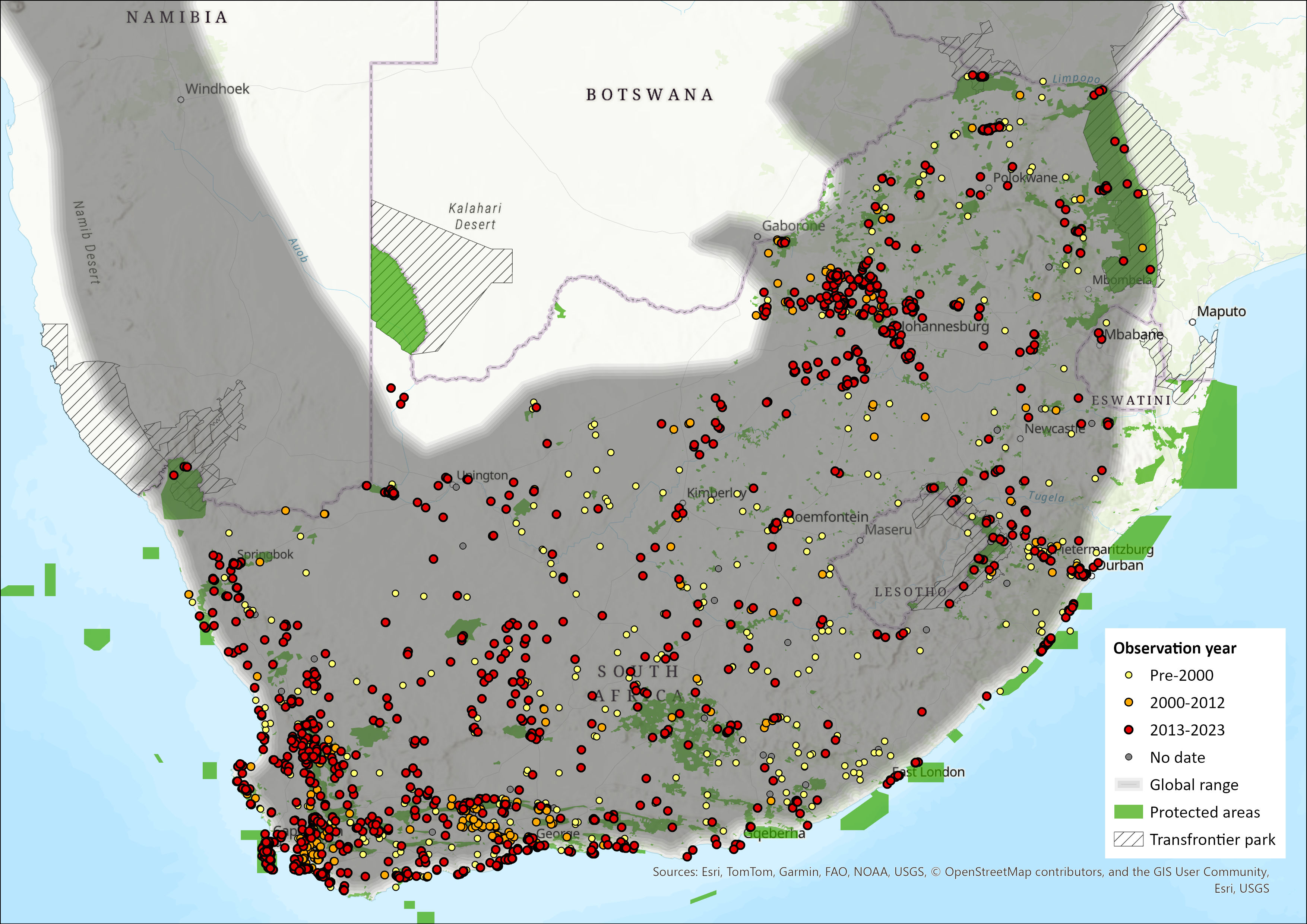
Rock Hyraxes are widely distributed across the African continent (throughout sub-Saharan Africa and North East Africa) and parts of the Middle East, excluding the Congo Basin forests and Madagascar (Olds & Shoshani 1982; Hoeck & Bloomer 2013; Lake 2016). Within the assessment region, they occur across the inland escarpment and adjacent rocky areas wherever there is suitable habitat (Skinner & Chimimba 2005). They occur in all provinces, as well as Eswatini (Skinner & Chimimba 2005), and have been sighted across Lesotho (Lynch 1994). In the North West Province, they are most abundant in the Norite Koppies and Pilanesberg Mountain Bushveld types and appear to have expanded their range, as they have been recorded at Wolwespruit Nature Reserve for the first time in 2013 (Power 2014) but were previously absent from this region (Newbery 1995). They have been observed on isolated koppies along the Molopo and in the calcrete hills of the Kalahari (P. Bloomer pers. obs.).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4300
Depth Lower Limit (in metres below sea level): (Not specified) NA
Depth Upper Limit (in metres below sea level): (Not specified) NA
Depth Zone: (Not specified) NA
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Map
Figure 1. Distribution records for Rock Hyrax (Procavia capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | Resident |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | Resident |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | Resident |
| Djibouti | Extant | Native | – | Resident |
| Egypt | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | Resident |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lebanon | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Malawi | Extant | Native | – | Resident |
| Mali | Presence Uncertain | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Rwanda | Extant | Native | – | Resident |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | Resident |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Sudan | Extant | Native | – | Resident |
| Sudan | Extant | Native | – | – |
| Syrian Arab Republic | Presence Uncertain | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change may become an increasing threat, especially in the western areas of the assessment region as drought becomes more frequent (for example, Erasmus et al. 2002). There are extreme local population fluctuations in some areas (Hoeck 1989; Barry & Mundy 1998; Hoeck & Bloomer 2013; Barry et al. 2015), which may follow rainfall patterns (drought).
Population information
They are widespread and common. As mentioned above, in some areas, they are characterised by extreme local population fluctuations (Hoeck 1989; Barry & Mundy 1998; Hoeck & Bloomer 2013; Barry et al. 2015), which may follow rainfall patterns (drought) and possibly disease outbreaks. Across the Namaqualand and Western Fynbos regions populations are stable, with the Namaqualand region having previously showed a demographic population expansion (Visser 2013). In the Free State, the population numbers of rock hyrax in residential areas doubled compared to wild populations. This was due to abundant food and a lack of predators. However, certain dog breeds had some degree of influence over population numbers, resulting in some declines (Wiid & Butler 2014).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: No. Although in some areas utilised for bushmeat.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Seven clusters were retrieved in the microsatellite data and two clusters in the mtDNA data across the Namaqualand/western Fynbos regions (Visser 2013). The number of subpopulations would therefore be at least seven across this region. However, the rest of the assessment region requires investigation.
Severely fragmented: No. When overpopulated or during food shortages, they are capable of traversing great distances between suitable rocky habitats, at least 20 km away (Skinner & Chimimba 2005).
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Genetic structure within P. capensis has been detected using a variety of molecular techniques. Molecular studies investigating the phylogeographic structure within P. capensis detected clear mitochondrial differentiation (no shared haplotypes) within the eastern parts South Africa, with two distinct groups identified: a subpopulation along the southern and eastern sections of the Great Escarpment and another across several mountain ranges in the northern parts of South Africa extending to the Matopo Hills of Zimbabwe (Prinsloo & Robinson 1992; Maswanganye et al. 2017). This structure was not detected within the nuclear DNA, likely associated with periods of secondary contact at different periods of time when conditions were more favourable/suitable (Maswanganye et al. 2017). Due to a limited microsatellite dataset and insufficient sampling within each mountain range, it was not possible to ascertain whether more recent population genetic structure exists within the two broader subpopulations, but it is possible that it does given their presence on disjunct mountain ranges (e.g., north: Magaliesberg, Waterberg and Soutpansberg mountains; south: Cape Fold, Great Escarpment and Karoo mountains along with other isolated mountains and rock outcrops). Moreover, substructure was identified across the western seaboard of South Africa (within Namaqualand and western Fynbos areas), supporting this notion (Visser et al. 2020). In particular, five geographically distinct genetic clusters were detected associated with Namaqualand, Olifants River, West Coast, Cape Peninsula and Overberg. All of these clusters/subpopulations are separated by one or more physical barriers to dispersal.
Patterns of gene-flow are influenced by the polygynous social system of this species in addition to the landscape connectivity (amount of intervening suitable habitat) between isolates in a specific region (Visser 2013). As such, male-biased dispersal and female philopatry characterises population genetic structure with areas devoid of suitable rocky habitat acting to limit gene-flow between isolated populations.
Based on the available information, there is a minimum of seven genetic distinct subpopulations within the assessment region (with high probability of over 11), which should be investigated further to ensure all available genetic diversity is accounted for and maintained. Assuming there has been no loss of subpopulations, the proportion of populations maintained (PM) indicator is estimated at 1.0 (regardless of the actual number of subpopulations confirmed). While general statements indicate that the species is abundant, the effective population sizes (Ne) for the known populations have not been quantified, and no population size data exists for any subpopulation. Moreover, the species is known to undergo extreme population fluctuations, making any assumptions difficult, Consequently, the Ne 500 indicator cannot be quantified at this time.
Habitats and ecology
This species occupies a wide range of habitats, but is typically associated with rocky outcrops, cliffs, or piles of boulders with bushes (Skinner & Chimimba 2005; Hoeck & Bloomer 2013), also occurring in fynbos and karroid habitats. Granite formations with boulders and dolomite intrusions in the Karoo are especially favoured (Skinner & Chimimba 2005). This species occurs in modified or disturbed habitats, such as erosion gullies in the Karoo, culverts under roads and holes in stone walls (Olds & Shoshani 1982; Rübsamen et al. 1982; Skinner & Chimimba 2005). Some land uses may actually create habitat for hyrax, which also adapt well to human areas, leading to overpopulation and often becoming pests (Wimberger et al. 2009). This could be from easily accessible food resources in urban gardens and increased habitat availability in the form of houses or garden rockeries (especially in Pietermaritzburg and Ladysmith in the KwaZulu-Natal Province). It appears to be more adaptable than the Yellow-spotted Rock Hyrax (Heterohyrax brucei), with which it is sympatric in the northern areas of the assessment region (Skinner & Chimimba 2005).
They are predominantly diurnal and gregarious, living in colonies that vary in size according to food availability, and consisting of a dominant male with a harem of 3–17 females (Fourie & Perrin 1987). Males are forced to disperse when mature whereas females disperse voluntarily. Males, especially juveniles, are therefore highly vulnerable to predation when dispersing (Hoeck 1982). They are generalist herbivores, and their diet comprises a variety of grasses, forbs and shrubs, with a predilection for new shoots, buds, fruits and berries (Hoeck & Bloomer 2013). They consume a wide variety of plant species (Fourie & Perrin 1989).
Shelters are used to hide from predators but also play an important role in the regulation of hyrax body temperature (Brown & Downs 2006). Hyrax use the shelters in summer to lower their body temperature relative to the high ambient temperatures as well as to prevent excessive water loss. However, body temperatures were found to be lower in winter than in summer, with lower ambient temperatures, such that they spend more of their time outside their shelter to increase their body temperatures by sunbathing (Brown & Downs 2006). Body temperatures during winter nights were low but constant, presumably by individuals huddling within the shelter (Brown & Downs 2006), but there was no evidence to support this theory (Downs et al. 2013).
This species, although known previously, was first described in 1766 by Pallas, who first saw one in a tavern in Cape Town where it was kept as a pet (Skinner & Chimimba 2005).
Ecosystem and cultural services: The Rock Hyrax may be seen as a keystone species as they are a source of food for many of the larger predators such as the Verreaux’s (Black) Eagle (Aquila verreauxii) (for example, constituting 74% of its diet in the Karoo, Davies 1989), Martial Eagle (Polemaetus bellicosus), Crowned Eagle (Stephanoaetus coronatus) (Boshoff et al. 1994), Leopard (Panthera pardus), Caracal (Caracal caracal) (for example, comprising 22–55% of its diet, Grobler 1981; Palmer & Fairall 1988), jackals (Canis spp.), African Wild Cat (Felis silvestris) and various snakes (Olds & Shoshani 1982; Davies 1989, 1994, 1999; Barry & Barry 1996; Kotler et al. 1999; Druce et al. 2006; Chiweshe 2007; Kruger 2010). The importance of Rock Hyrax as a forage species has been demonstrated by Fourie (1983) who estimated that 11% and 4% of the mature population (N = 24,553 individuals) in an area were eaten by Caracal and Verreaux’s Eagle respectively in one year (see also Davies 1989).
Rock Hyraxes may also cause ecosystem damage, especially if predators have been removed and colonies are overpopulated: Heavy grazing around colonies can cause a preponderance of unpalatable plants (such as Hermannia burkei), can compete with livestock exceeding the recommended stocking rate by a factor of 15 (Skinner & Chimimba 2005), and may also reduce the regeneration of trees. Furthermore, their latrines can cause a health risk when they occupy spaces below houses (for example, in Pietermaritzburg) and in roofs of hospitals (for example, Ladysmith, KwaZulu-Natal Province). Thus, sustaining predators in landscapes is a sound management tool, as well as preventing access to buildings.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | No |
| 2.2. Savanna -> Savanna – Moist | – | Marginal | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | No |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | No |
| 8.1. Desert -> Desert – Hot | – | Marginal | – |
| 8.2. Desert -> Desert – Temperate | – | Suitable | No |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: 16 months
Age at Maturity: Male: 16 months
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: 45-60 cm
Longevity: 9-10 years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 6-8 months
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
The Rock Hyrax is snared for skins and meat. This is not expected to impact on the population overall. However, local declines or extinction may occur. For example, in Lesotho they are hunted with dogs (Lynch 1994), and in Pietermaritzburg with small snares (K. Wimberger pers. obs. 2007).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat use. | Yes | Local trade in skins. |
National Commercial Value: No
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: No ranched or domesticated populations.
Threats
There are no major threats to this species. However, it is hunted locally for bushmeat which may lead to local subpopulation declines. Power (2014) notes that concerns over the species have been raised in the Magaliesberg by landowners who suspect that numbers have declined recently. However, this may be conflated with natural population fluctuation due to drought, disease and predator eradication. For example, entire subpopulations have become locally extinct due to drought (Barry & Mundy 1998) and sarcoptic mange may have caused local extinctions in the KwaZulu-Natal Province in the late 1990s (Wimberger et al. 2009).
Current habitat trend: Stable. They occupy rocky outcrops that are largely inaccessible and not under threat of extensive transformation. They also occur in modified habitats.
Conservation
The species occurs in many large, well-protected areas across much of its range within the assessment region. No specific conservation interventions are necessary at present. Visser (2013) found low genetic diversity in two conservation areas in the Western Cape (Table Mountain National Park and Boulders Penguin Colony), likely due to the poor habitat connectivity of this region in addition to an anthropogenically influenced landscape. Thus, protected area expansion and possibly reintroduction/translocation (see below) will benefit this species, especially biodiversity stewardship programmes that connect suitable rocky outcrops.
Reintroduction as a management tool has had mixed success: Since 2004, this species has been bought from local conservation authorities (for example, Ezemvelo-KZN Wildlife) by private landowners and reintroduced into various areas. Similarly in Gauteng, Rock Hyraxes have been removed from overpopulated urban nature reserves and reintroduced into areas where they are thought to have declined, sometimes with the dual benefit of ensuring the survival of threatened species such as the Verreaux’s Eagle subpopulation at Walter Sisulu National Botanical Gardens (“Hyrax Operation Project”). However, post-release observations suggest that only three of six reintroductions remain self-sustaining today, providing the eagles with much needed dassie sustenance (B. van der Lecq pers. comm. 2012; Wimberger et al. 2009). Indeed, reintroduction attempts have been met with only limited success (Crawford 1984). Wimberger et al. (2009) described an unsuccessful reintroduction attempt where captive individuals (N = 16) and wild individuals (N = 9) were released into Umgeni Valley Nature Reserve, KwaZulu-Natal Province. After three months captive individuals showed no site fidelity and could not be found while wild individuals were dead within 18 days, mostly due to predation (Wimberger et al. 2009). This failure is attributed mainly to predation and group disintegration. Hyraxes are vulnerable to predation when foraging away from cover (Druce et al. 2006) which is similarly true of the post-release period. Indeed, 78% of the released individuals were probably eaten by Caracal. Small group sizes thus hinders vigilance (Hoeck 1975) and a lack of social cohesion (poor group composition) may also contribute to failure.
To improve the success of future reintroduction attempts, Wimberger et al. (2009) recommend an estimation of predator density in the release site and active predator deterrent (if predator density is high) for a period after release, or the consideration of another release site. Also, the use of soft releases and post release monitoring with radio telemetry are recommended.
Recommendations for land managers and practitioners:
- No special land management is necessary to sustain populations, given that they inhabit rocky, untransformed habitats and can utilise human structures. The eradication of predators may however cause higher population densities in affected areas (Fairall & Hanekom 1987). In such areas (or areas of naturally high population density), the reintroduction of predators is recommended as a holistic management strategy (and even necessary to maintain the natural vegetation; see Fairall & Hanekom 1987).
- Monitoring of the genetic diversity of populations in conservation areas of poor habitat connectivity across South Africa may inform appropriate conservation interventions. Given the low genetic diversity in two conservation areas of the Western Cape (Visser 2013), animal numbers/genetic diversity may have to be augmented via translocation of genetically closely related animals from surrounding (wild or protected) areas to curb any future loss of genetic diversity through population fluctuations. Conservation planning should also redress the lower landscape connectivity of certain areas under formal protection (for example, in the Western Cape: Tankwa Karoo National Park, West Coast National Park, Table Mountain National Park, Silvermine National Park, Cape Peninsula National Park, Boulders Penguin Colony, Betties Bay penguin colony and the Kogelberg Nature Reserve) and consider strategies that would establish corridors.
- Finally, post-release monitoring of reintroduction attempts is necessary to build an evidence base on reintroduction techniques (Wimberger et al. 2009).
Research priorities:
- A phylogenetic study is necessary for the genus across its African distribution to identify lineages with separate evolutionary histories. This may also inform a taxonomic revision of the genus Procavia.
- Studies of diseases between populations close to human settlements versus populations further away may be valuable. Additionally, the consequences of disease on population cycles could be investigated.
- The extent of its use as bushmeat should be examined.
- Finally, an examination into the use of translocation as a reintroduction tool may be required as it has proven to be ineffective in the past (Wimberger et al. 2009).
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Avoid feeding or keeping as pets.
Bibliography
Barry RE, Barry LM. 1996. Species composition and age structure of remains of hyraxes (Hyracoidea: Procaviidae) at nests of black eagles. Journal of Mammalogy 77: 702-707.
Barry RE, Chiweshe N, Mundy PJ. 2015. Fluctuations in bush and rock hyrax (Hyracoidea: Procaviidae) abundances over a 13-year period in the Matopos, Zimbabwe. South African Journal of Wildlife Research 45: 17-27.
Barry, R.E. and Mundy, P.J. 1998. Population dynamics of two species of hyraxes in the Matobo National Park, Zimbabwe. African Journal of Ecology 36: 221-233.
Boshoff AF, Palmer NG, Vernon CJ, Avery G. 1994. Comparison of the diet of crowned eagles in the savanna and forest biomes of south-eastern South Africa. South African Journal of Wildlife Research 24: 26-31.
Brown KJ, Downs CT. 2006. Seasonal patterns in body temperature of free-living rock hyrax (Procavia capensis). Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 143: 42-49.
Chiweshe N. 2007. Black Eagles and hyraxes – the two flagship species in the conservation of wildlife in the Matobo Hills, Zimbabwe. Ostrich 78: 381-386.
Crawford RJM. 1984. Male rock Hyraxes Procavia capensis return to former home ranges after translocation. Koedoe 27: 151-153.
Davies RAG. 1989. Where dassies dare. Custos 17: 53-56.
Davies RAG. 1994. Black eagle (Aquila verreauxii) predation on rock hyrax (Procavia capensis) and other prey in the Karoo. Ph.D Thesis. University of Pretoria, Pretoria, South Africa.
Davies RAG. 1999. The extent, cost and control of livestock predation by eagles with a case study on black eagles (Aquila verreauxii) in the Karoo. Journal of Raptor Research 33: 67-72.
Downs CT, Wimberger K, Wilson A-L. 2013. No effects of huddling on core body temperature in rock hyrax, Procavia capensis. African Zoology 48: 173-176.
Druce DJ, Brown JS, Castley JG, Kerley GIH, Kotler BP, Slotow R, Knight MH. 2006. Scale-dependent foraging costs: habitat use by rock hyraxes (Procavia capensis) determined using giving-up densities. Oikos 115: 513-525.
Erasmus, B.F.N., van Jaarsveld, A.S., Chown, S.L., Kshatriya, M. and Wessels, K.J. 2002. Vulnerability of South African animal taxa to climate change. Global Change Biology 8: 679-693.
Fairall N, Hanekom N. 1987. Population dynamics and possible management options for the rock dassie Procavia capensis population in the Tsitsikamma Coastal National Park. Koedoe 30: 139-148.
Fourie LJ, Perrin MR. 1987. Social behaviour and spatial relationships of the rock hyrax. South African Journal of Wildlife Research 17: 91-98.
Fourie LJ, Perrin MR. 1989. Quantitative and qualitative aspects of the diet of the rock hyrax (Procavia capensis Pallas, 1766) in the Mountain Zebra National Park. Journal of African Zoology 103: 361-370.
Fourie LJ. 1983. The population dynamics of the rock hyrax Procavia capensis (Pallas, 1766) in the Mountain Zebra National Park. Ph.D. Thesis. Rhodes University, Grahamstown, South Africa.
Grobler JH. 1981. Feeding behaviour of the caracal Felis caracal Schreber 1776 in the Mountain Zebra National Park. South African Journal of Zoology 16: 259-262.
Hoeck HN, Bloomer P. 2013. Procavia capensis Rock Hyrax. In: Kingdon J, Happold D, Hoffmann M, Butynski, Happold M, Kalina J (ed.), Mammals of Africa Volume 1, pp. 166-171. Bloomsbury Publishing, London, UK.
Hoeck HN. 1975. Differential feeding behaviour of the sympatric hyrax Procavia johnstoni and Heterohyrax brucei. Oecologia 22: 15-47.
Hoeck HN. 1982. Population dynamics, dispersal and genetic isolation in two species of hyrax (Heterohyrax brucei and Procavia johnstoni) on habitat islands in the Serengeti). Zeitschrift für Tierpsychologie 59: 177-210.
Hoeck HN. 1989. Demography and competition in hyrax. Oecologia 79: 353-360.
Kotler BP, Brown JS, Knight MH. 1999. Habitat and patch use by hyraxes: there’s no place like home? Ecology Letters 2: 82-88.
Kruger TL. 2010. Long term prospects for the persistence of breeding Verreaux’s Eagles (Aquila verreauxii) at the Walter Sisulu National Botanical Garden, Johannesburg. M.Sc. Thesis. University of Witwatersrand, Johannesburg, South Africa.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Maswanganye, A., Cunningham, M., & Bennett, N., Chimimba, C. & Bloomer, P. 2017. Life on the rocks: Multilocus phylogeography of rock hyrax (Procavia capensis) from southern Africa. Molecular Phylogenetics and Evolution. 114. 10.1016/j.ympev.2017.04.006.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Newbery CH. 1995. Mammal Checklist of the Provincial Nature Reserves. North West Parks Board, Mafikeng, South Africa.
Olds N, Shoshani J. 1982. Procavia capensis. Mammalian Species 171: 1-7.
Palmer R, Fairall N. 1988. Caracal and African wild cat diet in the Karoo National Park and the implications thereof for hyrax. South African Journal of Wildlife Research 18: 30-34.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rübsamen K, Hume ID, Engelhardt WV. 1982. Physiology of the rock hyrax. Comparative Biochemistry and Physiology Part A: Physiology 72: 271-277.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Visser JH. 2013. Gene-flow in the rock hyrax (Procavia capensis) at different spatial scales. M.Sc. Thesis. University of Stellenbosch, Stellenbosch, South Africa.
Wiid, R.E. and Butler, H.J., 2015. Population management of rock hyraxes (Procavia capensis) in residential areas. Pest management science, 71(2), pp.180-188.
Wimberger K, Downs CT, Perrin MR. 2009. Two unsuccessful reintroduction attempts of rock hyraxes (Procavia capensis) into a reserve in the KwaZulu-Natal Province, South Africa. South African Journal of Wildlife Research 39: 192-201.