Riverine Rabbit
Bunolagus monticularis

2025 Red list status
Endangered
Regional Population Trend
Unknown
Change compared
to 2016
Downlisted
Overview
Bunolagus monticularis (Thomas, 1903)
ANIMALIA – CHORDATA – MAMMALIA – LAGOMORPHA – LEPORIDAE – Bunolagus – monticularis
Common Names: Riverine Rabbit, Bushman Hare, Bushman Rabbit (English), Boshaas, Oewerkonyn, Pondhaas, Vleihaas, Doekvoet (Afrikaans), Buschmannhase (German)
Synonyms: Lepus monticularis Thomas, 1903
Taxonomic Note: Generic synonym Lepus.
| Red List Status |
|
EN – Endangered, B2ab(iii) (IUCN version 3.1) |
Assessment Information
Assessors: Bragg, C.1, Matthew, E.E.2,3, Mynhardt, S.2, Woodgate, Z.A.4, Brassine, A.5, da Silva, J.6, Collins, K.7 & Roxburgh, L.2
Reviewers: Schumann, B,2 & de Villiers, M.8
Contributors: Raimondo, D6. & Patel, T.2
Institutions: 1Private, Bohemian Scientist, 2Endangered Wildlife Trust, 3IUCN Lagomorph Specialist Group, 4Institute of Communities and Wildlife in Africa, 5Independent Professional Natural Scientist, 6South African National Biodiversity Institute, 7Mammal Research Institute, University of Pretoria, 8CapeNature
Previous Assessors & Reviewers: Collins, K., Bragg, C., Birss, C. & Child, M.F.
Previous Contributors: Matthee, C.M., Nel, V., Hoffmann, M., Smith, A.T. & Roxburgh, L.
Assessment Rationale
Riverine Rabbits are endemic to the semi-arid Karoo regions of South Africa. They are confirmed to be distributed within three smaller sub-populations across the Nama Karoo, Succulent Karoo and Fynbos Biomes of the Northern and Western Cape Provinces. Extent of occurrence (EOO) is estimated at 99,960 km², [up from 54,227 km² for the 2016 assessment], and area of occupancy (AOO) is estimated between 500 and 3,050 km². The AOO was estimated as 2,943 km² in the previous assessment. Previous population estimates of 157-207 mature individuals indicated an alarmingly small species population size, with no subpopulation having > 50 mature individuals. A recent population genetic study based on mitochondrial DNA (mtDNA) estimated the effective population size (Ne) at approximately 5000 mature individuals, and although this suggests a larger population size, mtDNA is a reflection of past evolutionary events, rather than current nucleic diversity or population and thus is likely an overestimate of population size. Although new knowledge has led to larger EOO and AOO’s, there remains a lack of information on inter-population connectivity, with no known locality supporting more than 50 mature individuals, or a connected habitat size (estimated at 1000 ha) that could sustain a viable population. Since the discovery of a previously undetected population in 2018, there are believed to be 3 distinct subpopulations (one in the northern range, one in the south, and the new subpopulation east of the historically known southern range)- substantiated by mtDNA analysis. Numerous surveys throughout the species’ distribution have failed to detect individuals in some areas of its historic range (as indicated by museum records), which indicates a decrease in the distribution range (specifically for the northern subpopulation). Subsequently, only current subpopulations are used to estimate population size. Due the cryptic nature of the species, and often low densities observed at specific localities, monitoring work should be continued to improve the accuracy of subpopulation estimates and trends. This species should therefore be reassessed once substantially more data has been generated by relevant research. Subpopulations are presumably further fragmented and isolated by anthropogenic barriers to dispersal, such as impoundments in river channels, fencing, farming activities, and infrastructure developments. Furthermore, existing subpopulations are threatened by illegal hunting, road related mortality and predation by domestic animals. Climate change and other high-impact developments, such as fracking, are emerging as potential threats: an initial assessment of climate change indicates that a net reduction of 89% in habitat is likely for the northern subpopulation. Thus, even though the recent mtDNA analysis indicated a significantly higher population size than previously believed, it is important to evaluate the impact of threats (specifically habitat loss and fragmentation), population trends, habitat utilisation and other factors such as the impact of road collisions and energy infrastructure, for each subpopulation, to establish the appropriate assessment status for the species. The current assessment suggests that the Riverine Rabbit should be downlisted to Endangered based on criterion B2 due to its area of occupancy and inferred continuing decline from habitat loss, degradation and climate change.
Red List Index
Red List Index: Downlisted
Recommended citation: Bragg C, Matthew EE, Mynhardt S, Woodgate ZA, Brassine A, da Silva JM, Collins K & Roxburgh L. 2025. A conservation assessment of Bunolagus monticularis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
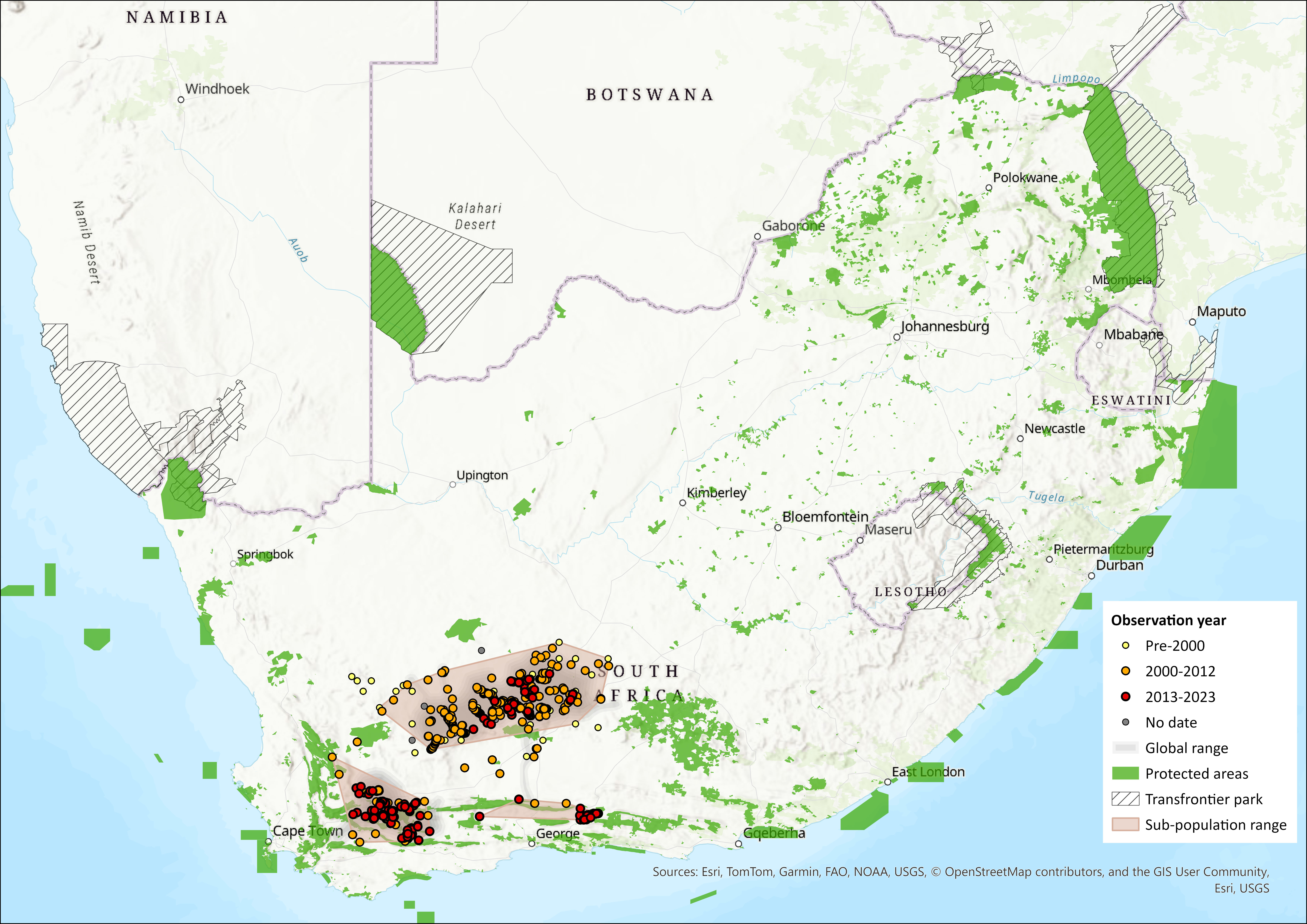
This species is distributed across the Nama Karoo, Succulent Karoo and Fynbos Biomes of the Northern and Western Cape Provinces of South Africa. It is known from three subpopulations: one in the Nama Karoo (northern subpopulation), where it was first discovered near Richmond in 1902, one in the Succulent Karoo, in the vicinity of Touwsriver, where the second population (southern subpopulation) was discovered in 2003, and a third population (eastern subpopulation), discovered in 2018, just west of the Baviaanskloof, approximately 250 kilometres east of the southern subpopulation. These subpopulations have been delineated in the past by their habitat parameters, because of the seemingly tight habitat specificity of the first subpopulation discovered (the Nama Karoo sub-population, where the species is found only in alluvial floodplain vegetation). However, genetic factors show some discrepancy in these subpopulation delineations, as one genetic cluster groups the northern and southern subpopulations.
The northern subpopulation is associated with the dense, discontinuous vegetation adjacent to seasonal rivers. The Riverine Rabbit is the only indigenous burrowing rabbit in Africa and is thought to be dependent on soft and deep alluvial soils along the river courses for constructing stable breeding stops. In addition, in Nama Karoo, they inhabit dense riparian growth along the seasonal rivers. However, in the Little Karoo, they are not restricted to alluvial floodplains and can also occur in old lands not associated with riverine vegetation. Historically, this species was known to occur in five localities towards the northwestern portion of its range, along the Vis and Renoster Rivers, as well as their tributaries near Calvinia (Duthie 1989). However, their continues presence in historical areas within the northern subpopulation, such as localities near Richmond and Calvinia, has not recently been confirmed, the lack of sightings data during the last 30 years, suggests that Bunolagus monticularis is now locally extinct in these regions (Collins and Toit 2016), which indicates a likely reduction in distribution within this subpopulation. This is likely to be a direct consequence of the extensive agricultural expansion along riverine floodplains (Duthie et al. 1989).
Numerous surveys from 1999-2024 throughout the distribution range have improved estimates of location and sizes of the various subpopulations and facilitated the discovery of and confirmation of the southern- (2003) and eastern subpopulations (2018). Surveys with the Endangered Wildlife Trust’s scent detection dog (Matthew, 2021) and camera traps have confirmed the presences of the species at three historical sites that were predicted to have the species by Duthie et al. (1989) within the northern subpopulation, and new records that extend the previous range estimates.
Extent of occurrence was calculated, using a minimum convex polygon around all occurrence points from 2000 onwards. This value is 99,960 km2, up from 54,227 km2 in the previous assessment, due to the new occurrence points found for the eastern subpopulation. AOO was calculated using the IUCN’s 2×2 km grid method using all occurrence points from 2000 onwards, which came to 1600 km2, and a more conservative estimate using just records from 2013 to 2023 yielded an AOO of 500 km2 but is probably an underestimate as fewer new sites were sampled in this period. Of the 1600 km2, the northern subpopulation had an AOO of 1068 km2, the southern subpopulation had an AOO of 440 km2 and the eastern of 92 km2.
An analysis of AOO similar to the previous assessment was also done using rivers (NBA 2018) that were buffered by a 1 km radius that intersected with riverine rabbit occurrence records from 2000 onwards (also buffered by 1 km). AOO was calculated as 3,050 km². This is slightly higher than previous estimate of 2,943 km², as it includes the newly discovered eastern subpopulation. This estimate of AOO comes with a caveat that rabbits in the southern subpopulation are not restricted to riverine habitat, and thus the AOO would be underestimated for this subpopulation using this methodology. A second more conservative calculation using just occurrence records collected between 2013 and 2023 yields an AOO of 875 km2.
In summary, our calculations suggest that the AOO lies between 500 and 3,050 km2. This is too large for the species to qualify as Critically Endangered based on criterion B2 but would allow the species to qualify for Endangered (using the precautionary principle), especially considering that continuing declines are predicted by climate change models (see climate change section). Given that we have little evidence that the rabbit subpopulations are contiguous, and that there are different habitat requirements across the northern and southern subpopulations, it is considered a precautionary measure to consider the lower AOO estimates as being more representative of the status quo.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Riverine Rabbit (Bunolagus monticularis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change is starting to impact our environment, our species and human communities. In South Africa, the arid drylands are already seeing glimpses of what the future holds. Traditional livelihoods have been changing in the Karoo for the past few decades and climate change will exacerbate these changes, such as increasing the need for hunting for bushmeat and expanding the pressures of farming practices on natural resources. Karoo vegetation is likely to undergo shifts with changing climate. For example, there is evidence that grass cover has been replacing shrubland across the transition between the Nama-Karoo and Grassland Biomes over the last few decades (Arena et al. 2023). In the northern subpopulation, this has implications for the specific shrub-dominated habitat requirement for rabbits. Karoo ecosystems will be placed under pressure and species may be at risk of extinction. Water resources are already stressed and climate change will increase this stress through increasing evapotranspiration, more variable rainfall and increasing crop demand. From 2014 to 2019, the Karoo experienced a long drought. For Riverine Rabbits it will likely put even more pressure on the species that is already restricted by habitat availability and activity patterns. An initial assessment of climate change indicates that a net reduction of 89% in habitat is likely for the northern subpopulation (Hughes et al. 2008). Results from more refined models of the entire population completed for this assessment using 3 different climate prediction models under scenarios SSP126 and SSP 585 for 2011 to 2040, show that there will be a 7 to 56% or a 12 to 60% decline in range respectively (Cowan 2025). As the species occupies an unevenly distribution within its range, the relationship between projected habitat loss and range change may not be linear. The rabbit may be considered particularly vulnerable to climate change due to its rarity, inferred lack of dispersal between subpopulations and within the northern subpopulation, sensitivity to land use changes, slow reproductive capacity and habitat specialisation (particularly within the northern subpopulation) (Foden & Young 2016).
Population information
Riverine Rabbits were previously estimated as having a critically low population size; however, concerted monitoring efforts have uncovered an additional subpopulation (see Geographical Distribution) and overall sightings suggest that past estimates were likely underestimates. Several methods have been applied for estimating the population size of this species, with varying outcomes.
Using the estimated EOO and AOO values, we calculated population size estimates for Riverine Rabbits based on the density estimates of Duthie et al. (1989), which were for rabbits in the northern subpopulation and Woodgate et al. (2024), which were for rabbits in the southern subpopulation. These density estimates were 1) 6-17 rabbits per km2, and 2) 0.11-0.12 rabbits per km2 respectively. The Woodgate et al. (2024) density estimate was based on the entire sample area and not just on those areas that were suitable for rabbits, and we consider this a more appropriate density estimate to use with the EOO. In contrast, the Duthie estimate was based on areas of suitable habitat and is thus more appropriate to use with the AOO.
Our population estimates based on Duthie et al. (1989) are 3000-8500 (northern: 768-2176, southern: 1848-5236 and eastern: 384-1088) individuals for records from 2013-2024. Assuming 70% adults, this would be 2100-5950. The Woodgate value for the entire EOO is 10,995 to 11,995, or 7.697 to 8,397 mature individuals. However, the EOO does include vast tracts of habitat not considered suitable for Riverine Rabbits. We therefore calculated EOOs for each of the subpopulations, with resulting areas of 37,784, 11,813 and 3,614 km2 for the northern, southern and eastern populations respectively. The associated population estimates for each of these, based on the Woodgate estimate, was 2,909-3,174, 909-992 and 278-304 mature individuals respectively, with a total estimated mature population of 4,097-4,470. However, the density estimate was for the southern subpopulation and likely overestimates densities in the northern subpopulation. Thus, the Woodgate estimates for the southern and eastern subpopulations, and the Duthie estimates for the northern populations would be our recommended best estimates. Combining these estimates gives a minimum mature population size of 1,955 and a maximum of 3,472. Thus, based on all these estimates, it seems likely that the mature population is below 5,000 individuals, and possibly below 2,500.
Population Information
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown, no numbers for any subpopulation
QUANTITATIVE ANALYSIS
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A recent population genetic study based on mitochondrial DNA (mtDNA) identified two predominant genetic groups of the Riverine Rabbit: a northern cluster comprised of individuals from the Nama and Succulent Karoo biomes, and a southern cluster of individuals from the Fynbos (Renosterveld) biome (Matthee et al. 2022). While these groups are not reciprocally monophyletic, the two groups are highly supported by various statistical metrics (AMOVA, pairwise ΦST comparisons, genetic sequence divergence greater between the two populations than the diversity within). This study included limited samples from the newly discovered southeastern range of the species (five from Uniondale). It is noteworthy that of all 12 localities included in this study, only Uniondale showed significant genetic differentiation despite its low sample size, and was also found to be monophyletic, indicating that this sample supports the recognition of a third, unique genetic cluster/subpopulation.
The same study examined the effective population size (Ne) for the species as a whole (based on mtDNA), which was estimated at 4,986 (1,300 – 21,400) individuals contributing to the gene pool, greatly exceeding previous numbers (Duthrie et al. 1989; Collins and Toit 2016); however, more closely reflecting current estimates using various methods (see Population Section). With that said it is important to note that Ne based on mtDNA data and for the species as a whole (rather than separate subpopulations) may overinflate results. Moreover, the results likely reflect past evolutionary events and historical population structure and sizes. Recent genetic diversity would be better identified using markers such as microsatellites or SNPs. It is therefore possible that additional genetic structure and diversity exists (or conversely may be lacking) within this species. Until more fine scale studies are conducted, efforts should be made to conserve the known diversity within the species (i. e., each subpopulation). Importantly, locations showing additional unique structure and diversity and/or high genetic diversity should be managed and conserved appropriately (e.g., Uniondale (Western Cape), Fraserburg (Northern Cape)).
Based on the population estimates extrapolated above, and applying Ne/Nc conversion ratios between 0.1-0.3, a current effective population size can be estimated. With 768-2176 estimated for the northern subpopulation, Ne for this subpopulation may be between 76 – 652. For the southern subpopulation, we estimated a population size between 909-992 mature individuals, resulting in Ne between 90 – 297. And the eastern subpopulation, with 278-304 mature individuals, is estimated at having an Ne between 27 – 91. Apart from the northern subpopulation, which shows Ne estimates on either side of the Ne 500 threshold, above which populations are thought to be genetically healthy and stable, all other subpopulations show indications of being well below this threshold. It is recommended that more accurate measures be acquired through finescale molecular analyses to verify these estimates.
Based on the available information, both of the population genetic indicators adopted into the Kunming-Montreal Biodiversity Framework, can be quantified. Three subpopulations exist and no additional ones are known to have gone extinct; therefore, the proportion of populations maintained within the species receives a score of 1.0 (3/3 population are maintained). The headline indicator, the proportion of populations with an effective population size (Ne) greater than 500, would receive a score between 0.0 and 0.17, to accommodate the uncertainty related to the northern subpopulation and whether it is above or below the Ne 500 threshold (i.e., averaging of 0 [0/3 subpopulations > Ne500] and 0.33 (1/3 subpopulations > Ne 500]).
Habitats and ecology
The Riverine Rabbits within the northern subpopulation occurring in the Central Karoo (Nama-Karoo shrubland) inhabits dense riparian growth along the seasonal rivers. Specifically, it occurs in riverine vegetation on alluvial soils adjacent to seasonal rivers (flood plains). This habitat is highly fragmented and transformed, with (Duthie 1989) in the Central Karoo. Observations from the southern subpopulation demonstrates that most localities are managed as game reserves/ private nature reserves, compared to livestock farms in the northern subpopulation. With the discovery of the eastern subpopulation, For the southern subpopulation found within the Little Karoo, the presence occurs within the vegetation variants, as described by Vlok and Schutte-Vlok (2010), of the Transitional Shrublands Vegetation Type, Arid Renosterveld Habitat Type, Succulent Karoo Vegetation Type, Apronveld Habitat Type and the Randteveld Habitat Type. However, they are not restricted to the alluvial floodplains in the southern Cape (C. Bragg pers. obs. 2014, Eichenberger pers obs, Woodgate et al. 2021) and can also occur in old lands not associated with riverine vegetation. Further habitat studies are required.
It should be noted that these are whereas subpopulations in the northern part of the distribution are always associated with alluvial floodplains and narrow belts of riverine vegetation adjacent to seasonal rivers on a scale that is unlikely to fit within these broader habitat types. They are thus highly reliant on the critical resource areas of Karoo riparian ecosystems. These descriptions do, however, give an indication of the general vegetation structure and composition within various parts of its range. whereas subpopulations in the northern part of the distribution are always associated with alluvial floodplains and narrow belts of riverine vegetation adjacent to seasonal rivers on a scale that is unlikely to fit within these broader habitat types. They are thus highly reliant on the critical resource areas of Karoo riparian ecosystems. These descriptions do, however, give an indication of the general vegetation structure and composition within various parts of its range.
Home range has been estimated as 12 ha (Duthie 1989) in a study restricted to one location in the Central Karoo and based on a sample size of 8 individuals. This species is elusive and crepuscular, spending daylight hours in a scrape beneath riparian vegetation. They are solitary and will only be found in breeding pairs for short periods, or in female-juvenile pairs for rearing purposes (Duthie 1989).
This species is predominantly a browser but is known to occasionally feed on grasses during the early wet rainy season when short, green grasses become available (Duthie 1989). When browsing, they have been found to show a particular selection for Pteronia erythrochaetha, Kochia pubescens, Salsola glabrescens and species of Aizoacae. They are unable to survive on heavily overgrazed or agriculturally transformed habitats but have been found feeding on lucerne fields at night.
Generation length for this species is two years (Collins et al. 2004). This species has a single litter per year with 1-2 young per litter in a fur- and grass-lined subterranean chamber excavated in stable soils (Duthie 1989). Reproductive periodicity occurs from August through May (Duthie and Robinson 1990). Gestation time is 35-36 days (Duthie 1989). Longevity in captivity is five years (K. Collins unpubl. data).
Ecosystem and cultural services: It is both a flagship species for the Karoo, as well as an indicator species of riparian habitat fringing the rivers of the Nama (Upper and Central Karoo) where its presence is associated with ecosystem integrity (healthy ecosystem services, such as water infiltration, vegetation cover, and soil health).
IUCN Habitats Classification Scheme
Life History
Generation Length: (Not specified)
Age at maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 5 Years
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 33.7-47.
Size at Birth (in cms): (Not specified)
Gestation Time: 35-36 Days
Reproductive Periodicity: August through May
Average Annual Fecundity or Litter Size: LS=1-2, 1 litter/yr.
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs?: No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding?: No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: Solitary
Systems
System: Terrestrial
General Use and Trade Information
The Riverine Rabbit is suspected to be opportunistically hunted for sport and for bushmeat by farm workers (Duthie and Robinson 1990, Coetzee 1994).
National Commercial Value: No
International Commercial Value: No
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: (Not specified)
Threats
The Riverine Rabbit faces significant threats from ongoing habitat degradation and fragmentation due to detrimental land-use practices and habitat-transforming land uses, such as climate change and energy development (Ahlmann et al. 2000). Over the last century, ca within the distribution of the northern subpopulation has been lost as a result of cultivation (for example, winter wheat production, mostly in the past) and livestock farming (ongoing) (Duthie 1989; Duthie et al. 1989, Duthie and Robinson 1990, Coetzee 1994, Ahlmann et al. 2000). Other threats to the species include hunted for sport and for bushmeat by farm workers), and accidental mortality in traps set for ‘pest’ animals on farmlands. However, some traps may actually target rabbits that are considered pests by some farmers (A. Brassine, pers. obs.). There is also the potential that the dominance of livestock farming and emerging wildlife ranching for economically valuable species may increase the frequency of both overgrazing (and thus reduction in vital vegetation cover) and predation rates (for example, by higher Black-backed Jackal Lupulella mesomelas densities). Similarly, habitat degradation through fuel-wood collecting and overgrazing may have led to an increase in predation (Ahlmann et al. 2000). Its unique habitat is of economic importance to landowners in terms of cultivation and small-stock grazing. Threats to the river ecosystems include overgrazing and anthropogenic land and river transformation, which leads to the degradation and fragmentation of Riverine Rabbit habitat.
There are also several emerging threats that could hamper the survival of this species. These include climate changes (see climate change section of the assessment), wind farms, fracking and rabbit haemorrhagic disease virus (RHDV2). It is believed that RHDV2 originated in China in 1984 and has since spread to many countries. This virus is highly contagious but only affects lagomorphs. It spreads very easily, besides direct contact between lagomorphs, probably by flies and scavengers feeding on carcasses, on clothes, shoes and car tyres. It is suspected that the disease reached South Africa in early 2022. It is reported that RHDV2 disease caused widespread mortalities of wild hares, and domestic rabbits across South Africa, including within the Northern and Western Cape, between October 2022 and to date, the EWT has not received any reported Riverine Rabbit mortalities, even with reports in close proximity to known Riverine Rabbit locations. However, there has been significant number of reports of dead wild hares and domestic rabbits, and only a few reports of Red Rock Rabbits. Even though no Riverine Rabbit deaths have been confirmed to be linked to RHDV2, this could likely be attributed to the fact that the species is rarely seen due to their elusive behaviour in general, and as such no carcasses have been discovered yet. It is possible that a reduced impact could be attributed to the fact that Riverine Rabbits fall into its own genes (Champman & Flux, 2008), or that the species was previously exposure to a similar virus. Needless to say, RHDV2 poses a potential significant threat to the survival of the species and should be closely monitored, as the possibly impact is still unknown, and it can easily be spread by humans and other wildlife.
The rapid and widespread expansion of renewable energy is likely to have a significant impact on the species and monitoring programs are urgently required to better understand these impacts. The identified potential risks of adverse impacts of wind energy facilities to Riverine Rabbits and their habitat include direct mortality from road collisions, noise impact, visual effect and possible complex and unpredictable cascading impact on the ecosystem and biodiversity as well as population-level impacts from the cumulative impacts of other wind energy facilities in the region (A Brassine, unpubl.). This threat is exacerbated by the lack of a proactive strategy to implement research-based and stakeholder-approved guidelines for mitigation, non-scientific and ad hoc pre- and post-monitoring techniques as well as lack of coordination around the cumulative impacts. Fracking of the Karoo region is also a major emerging threat to Riverine Rabbit habitat due to ancillary activities associated with fracking, which could lead to increased roadkill mortalities, habitat fragmentation and altered hydrology of Karoo river systems, which in turn impacts on the species’ habitat.
Habitat quantity and quality is declining through overgrazing by livestock, resulting in reduced cover from predators and lack of sufficient forage. Reductions in streamflow owing to the construction of dams upstream has presumably also reduced habitat quality (Ahlmann et al. 2000). High livestock, and in some cases, wildlife, stocking rates have resulted in large areas of the Karoo having reduced vegetation cover and becoming dominated by unpalatable species, which have further reduced vegetation biodiversity and stocking potential for livestock and game. Additionally, continued rural settlement expansion on the Northern Cape Province, estimated to be 9% from 2000 to 2013 (GeoTerraImage 2015), will presumably increase rates of poaching and predation by dogs. However, stewardship schemes for this species in the Karoo have also been initiated and cover 350,000 ha in the Greater Karoo. Anecdotal evidence suggests improved biodiversity and Riverine Rabbit populations on these farms, as well as greater landowner awareness of the requirements to protect Riverine Rabbits (C. Bragg unpubl. data). The EWT- DCP teams’ continued biodiversity stewardship expansion will help to mitigate habitat loss and deterioration.
Habitat quality might also be characterised by the presence of lagomorph competitors. Woodgate et al. (2021) found that, contrary to prior studies, Riverine Rabbits were not restricted to riparian habitat in Sanbona Wildlife Reserve, and that their occurrence was conditional on Scrub Hare (Lepus saxatilis) and Cape Hare (Lepus capensis) absence and was negatively affected by terrain ruggedness – a result substantiated by Duthie et al. (1985)’s original findings of less rabbits at sites where hares were present. However, the EWT camera trap surveys found that spatial overlap with hare species was spatially variable.
Loss of habitat due to infestation of invasive Prosopis trees and thickets in riparian habitats is also a cause of concern. Prosopis spp., commonly known as Mesquite, have become serious invasive species in many arid and semi-arid areas of South Africa. Prosopis invasions causes a change in habitat, reducing plant species richness, density and diversity, and outcompeting indigenous plants. This is particularly concerning for the Riverine Rabbit that has specific riparian habitat requirements which would be altered and lost because of Prosopis infestations.
A more underrated threat to the survival of the species is road collisions. Even though roadkill is very rare within the Northern subpopulation distribution (likely due to habitat restrictions), roadkills have been frequently observed in both the Southern- and Eastern subpopulations (EWT-DCP records). From our current knowledge, the highest impact has been seen in the Sanbona Wildlife Reserve. Roadkills have also been recorded within the newly discovered eastern subpopulation. As such, road signs have been erected at specific sites by the reserve and EWT (respectively) to try to mitigate the problem. The successfulness of the signboards has yet to be determined.
Conservation
Apart from the Anysberg Nature Reserve (CapeNature unpubl. data), this species is only found outside state owned conservation areas; consequently, the future conservation of the species is reliant on efforts of private landowners (see Collins et al. 2019). There is a healthy subpopulation present in the private Sanbona Wildlife Reserve, which is in the process of achieving formal protected status through the CapeNature biodiversity stewardship programme. They are also recorded on Witteberg Contract Nature Reserve, Lettaskraal and other properties around Anysberg. substantial proportion of the species’ habitat from the northern subpopulation is informally protected within Riverine Rabbit conservancies.
Although captive breeding was recommended by Collins et al. (2004), captive breeding trials in the 1990s were largely unsuccessful (Dippenaar and Ferguson 1994, Ahlmann et al. 2000). Thus, the current conservation strategy for the species does not include captive breeding as an option due to the difficulty of breeding this sensitive species in captivity and the higher chance of improving populations in the wild through concerted conservation interventions such as stewardship and managing and restoration of existing habitat.
The Drylands Conservation Programme of the Endangered Wildlife Trust (EWT-DCP) was established in August 2003 (originally known as the Riverine Rabbit Working group), and coordinates all conservation efforts on the Riverine Rabbit and its habitat, across all three subpopulations. The programme aims to conserve the biodiversity of the Karoo region, to encourage private landowners to participate in conservation stewardship and to promote integrated land management practices that can sustain the Riverine Rabbit, its habitat and many other species while providing employment for communities and facilitating ecosystem restoration on landowners’ farms. The current projects are:
- Expanding Riverine Rabbit habitat conservation through biodiversity stewardship: little of the Riverine Rabbit’s remaining habitat is protected within nature reserve or national parks; the survival of the Riverine Rabbit is in the hands of private landowners, consisting mostly of farmers. Therefore, the EWT‐DCP works in partnership with provincial conservation departments to involve private landowners in biodiversity conservation through the process of ‘biodiversity stewardship’. Stewardship refers to the wise use, management and protection of that which has been entrusted to the landowner. Within the context of conservation, stewardship means protecting important ecosystems by effectively managing aspects such as invasive alien species, erosion, fires, and grazing and cultivation practices. A critical component of the work is to promote and encourage sustainable land-use practices amongst landowners and land managers in the Karoo through developing partnerships with the relevant stakeholders. Thus, the EWT-DCP strives to mainstream sustainable land management principles into agricultural practices in the Karoo and into decisions made by policymakers.
- Environmental Education and Awareness: The EWT-DCP creates awareness about Riverine Rabbit conservation efforts with farmers, farm workers and their families, school learners, teachers, and the general public. This is typically achieved through presentations, school events, posters and social media outputs. It aims to establish custodianship by cultivating a sense of ownership for the Riverine Rabbit, its habitat and the river ecosystems of the Karoo.
- The EWT-DCP continues to conduct surveys, using camera traps, detection dogs, eDNA and human observations, to refine the distribution of the species, monitor trends and gather other information to implement better conservation interventions.
- The team also encourages citizen science reports (from the public), with photos for verification, to incorporate into distribution and predictive modelling mapping. This is done in parallel with recording accurate(confirmed/verified) iNaturalist observations.
- The EWT-DCP in collaboration with Stellenbosch University is also on the forefront of the Riverine Rabbits genetic research, using a variety of samples and techniques. The results enable a better understanding of the species and population dynamics.
- The EWT team also works closely with other stakeholders and partners to share knowledge and experience to facilitate a better understanding of the species and combine conservation efforts for a greater impact.
Recommendations for conservation practitioners:
- Development of a legal tool to address Riverine Rabbit conservation: a Biodiversity Management Plan for the species in terms of the National Environmental Management: Biodiversity Act (No. 10 of 2004).
- Conduct surveys in previously known subpopulation to identify any changes in status.
- are needed to identify Riverine Rabbit habitat and occupancy. Hughes et al. (2008) developed a habitat model that showed potential suitable habitat east of Victoria West that might contain isolated subpopulations or offer re-introduction opportunities. Further, Cowan (2025) identified up to a 60% decline in range due to climate change. The results of such surveys should feed into provincial and/or national spatial conservation planning. For example, the work of the Drylands Conservation Programme has succeeded in mitigating impacts of high-risk wind farms through dedicated interventions, particularly in regard to stopping development within suitable habitat. These data would also be used to develop a broad habitat map of the entire distribution range.
- Develop public recognition schemes to encourage private landowners to protect the Riverine Rabbit. Presence on private land should lead to the exclusion of over-utilisation of natural resources in key habitat areas. This should include the use of the EWT riparian grazing guidelines and best practice for riparian area to inform sustainable management of riparian areas, and a user-friendly manual of cost-effective sustainable land management technologies to encourage farmers to adopt restoration and sustainable farming practices. Finally, incentives should be identified to encourage landowners to adopt such practices that benefit the species (sensu Pasquini et al. 2010).
- Develop a strategic action plan and guidelines to mitigate the cumulative impacts of expanding anthropogenic developments on the species.
Encouraged citizen actions:
- Landowners can join biodiversity stewardship initiative or other OECMs to protect this species and its habitat.
- Landowners can lower stocking levels to improve riparian ecosystem grazing systems; reduce the number of dogs on farms; and restore Riverine Rabbit habitats through Working for Water and/or the EWT-DCP.
- Landowners and the public can report sightings of individuals on virtual museum platforms (for example, iNaturalist and MammalMAP) or provincial conservation authorities to enhance the distribution map. For example, new sightings recorded by the public and landowners are recorded in the CapeNature database.
Recommendation for future research:
- Long-term monitoring of population trends is required for this species. There has been no monitoring of the species over time (repeat surveys) at designated sites. Long-term data is required to establish trends in the populations. We recommend samples sites within each subpopulation
- Further ongoing surveys are required to investigate presence of the species in new locations, that may shed light on the possible connectivity between sites and would serve to inform protect area expansion/ landscape protection strategies.
- Genomic studies need to be done to more robustly determine recent population genetic structure and trends.
- Research to investigate the genetic and ecological differences between the southern and northern subpopulations is required to inform the development of separate conservation strategies for all subpopulations.
- Research is needed to understand the impact of the Rabbit Haemorrhagic Disease Virus (RHDV2) on the species
- Pre-, during and post-development monitoring Riverine Rabbit habitat and populations, to understand energy infrastructure development impacts on species.
- Behavioural (ecology) study to look at movement and activity, focusing on habitat utilisation, social interaction and home ranges within each subpopulation (prioritising eastern – and southern subpopulations).
Bibliography
Ahlmann, V., Collins, K. and Seal, U.S. 2000. Riverine Rabbit (Bunolagus monticularis): A population and habitat viability assessment workshop. Conservation breeding specialist group (SSC/IUCN), Apple Valley, MN.
Arena, G., Hoffman, M., van der Merwe, H., & O’Connor, T. (2023). Expansion of the Grassland Biome in the eastern Karoo corresponds with changes in rainfall and livestock numbers. African Journal of Range & Forage Science, 40(1), 1–19. https://doi.org/10.2989/10220119.2023.2175035
Berndt, J. 2015. Martial Eagles and the national power grid in South Africa: the implications of pylonnesting for conservation management. MSc thesis, University of Cape Town, South Africa
Boshoff, A., Palmer, G., & Avery, G. 1990. Variation in the diet of Martial Eagles in the Cape Province, South Africa, South African Journal of Wildlife Research, 20: 57-68.
Boshoff, A., & Palmer. G., (1980). Macro-analysis of prey remains from Martial Eagle nests in the Cape Province. Ostrich, 51: 7–13.
Chapman, J.A. and Flux, J.E., 2008. Introduction to the Lagomorpha. In Lagomorph biology: evolution, ecology, and conservation (pp. 1-9). Berlin, Heidelberg: Springer Berlin Heidelberg.
Coetzee, K. 1994. The Riverine Rabbit (Bunolagus monticularis) and its habitat: Conservation implications of an unnaturally fragmented distribution. M Tech. Diploma. Port Elizabeth Technicon, South Africa.
Collins, K. and Du Toit, J.T., 2016. Population status and distribution modelling of the critically endangered riverine rabbit (Bunolagus monticularis). African Journal of Ecology, 54(2), pp.195-206.
Collins, K., Ahlman, V., Matthee, C., Taylor, P., Keith, M., Palmer, G. and van Jaarsveld, A. 2004. Bunolagus monticularis. In: Y. Friedman and B. Daly (eds), Red Data Book of the Mammals of South Africa: A Conservation Assessment, pp. 412-413. CBSG Southern Africa, Conservation Breeding Specialist Group (SSC/IUCN), Endangered Wildlife Trust.
Collins, K., Ahlmann, V., Matthee, C., Taylor, P.J., Keith, M. and van Jaarsveld, A. 2003. Bunolagus monticularis. Available at: www.redlist.org.
Cowan, O.S. 2025. Modelling the change in potential species distribution of Riverine Rabbit (Bunolagus monticularis) under predicted future climate change scenarios. Technical Report, Endangered Wildlife Trust.
Department Agriculture, Land Reform and Rural Development. 2024. Rabbit haemorrhagic disease outbreak update report. Directorate Animal Health, 27 February 2024.
Dippenaar, S.M. and Ferguson, J.W.H. 1994. Towards a captive-breeding programme for the riverine rabbit Bunolagus monticularis. South African Journal of Science 90: 381–385.
Duthie, A.G. 1989. The ecology of the riverine rabbit (Bunolagus monticularis). Thesis, University of Pretoria.
Duthie, A.G. and Robinson, T. J. 1990. The African rabbits. In: J.A. Chapman and J.E.C. Flux (eds), Rabbits, Hares and Pikas: Status Survey and Conservation Action Plan, pp. 121-127. IUCN, Gland, Switzerland.
Duthie, A.G., Skinner, J.D. and Robinson, T.J. 1989. The distribution and status of the Riverine Rabbit, Bunolagus monticularis, South Africa. Biological Conservation 47: 195-202.
Foden, W.B. and Young, B.E. (eds.) (2016). IUCN SSC Guidelines for assessing species’ vulnerability to climate change. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Hatfield, R. 2018. Diet and space use of the martial eagle (Polemaetus bellicosus) in the Maasai Mara region of Kenya. Theses and Dissertations — Forestry and Natural Resources.
Hughes, G.O., Thuiller, W., Midgley, G.F. and Collins, K. 2008. Environmental change hastens the demise of the critically endangered riverine rabbit (Bunolagus monticularis). Biological Conservation 141: 23–34.
IUCN. 2019. The IUCN Red List of Threatened Species. Version 2019-1. Available at: www.iucnredlist.org. (Accessed: 21 March 2019).
Machange, R. W., Jenkins, A. R. and Navarro, R. A. 2005. Eagles as indicators of ecosystem health: Is the distribution of Martial Eagle nests in the Karoo, South Africa, influenced by variations in land-use and rangeland quality? Journal of Arid Environments. 63(1):223–43
Matthee, C.A., de Wet, N. and Robinson, T.J., 2022. Conservation genetics of the critically endangered riverine rabbit, bunolagus monticularis: structured populations and high mtdna genetic diversity. Journal of Mammalian Evolution, 29(1), pp.137-147.
Matthew, E.E. and Relton, C.E., 2021. Training methodology for canine scent detection of a critically endangered lagomorph: a conservation case study. Journal of Vertebrate Biology, 69(3), pp.20092-1.Mucina, L. and Rutherford, M.C. 2006. The vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institue, Pretoria.
Pasquini, L., Cowling, R.M., Twyman, C. and Wainwright, J. 2010. Devising appropriate policies and instruments in support of private conservation areas: lessons learned from the Klein Karoo, South Africa. Conservation Biology 24: 470–478.
Robinson, T.J. 1981. Systematics of the South African Leporidae. University of Pretoria.
Robinson, T.J. 1981. The bushman hare. Proceedings of the Wild Lagomorph Conference, University of Guelph, Ontario, Canada: 934–938.
Robinson, T.J. and Dippenaar, N.J. 1987. Morphometrics of the South African Leporidae II: Lepus Linnaeus, 1758, and Bunolagus Thomas, 1929. Annals of the Transvaal Museum 34: 379–404.
Robinson, T.J. and Skinner, J.D. 1983. Karyology of the riverine rabbit, Bunolagus monticularis, and its taxonomic implications. Journal of Mammalogy 64: 678–681.
South African National Biodiversity Institute (2006-2018). The Vegetation Map of South Africa, Lesotho and Swaziland, Mucina, L., Rutherford, M.C. and Powrie, L.W. (Editors), Online, http://bgis.sanbi.org/Projects/Detail/186, Version 2018.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Vlok, J. and Schutte-Vlok, A.-L. 2010. Plants of the Klein Karoo. Umdaus Press, Hatfield.
Woodgate, Z., Distiller, G., O’Riain, M.J., 2021. Woodgate, Z., Distiller, G. and Riain, M.J.O., 2021. Hare today, gone tomorrow: the role of interspecific competition in shaping riverine rabbit occurrence. Endangered Species Research 44:351–361
Woodgate, Z., Distiller, G.B., Bragg, C. and Justin O’Riain, M., 2024. Using camera-trapping to estimate riverine rabbit, Bunolagus monticularis, population density. African Journal of Wildlife Research, 54: 94–100.