
Rendall’s Serotine Bat
Pseudoromicia rendalli

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Pseudoromicia rendalli – (Thomas, 1889)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Pseudoromicia – rendalli
Common Names: Rendall’s Serotine, Rendall’s Pipistrelle, Rendall’s Pipistrelle Bat (English), Pipistrelle de Rendall, Serotine de Rendall (French)
Synonyms: Vesperugo (Vesperus) rendalli O. Thomas, 1889, Eptesicus phasma (G. M. Allen, 1911); Eptesicus faradjius (J. A. Allen in J. A. Allen, Lang & Chapin 1917); Eptesicus rendalli (Corbet & J. Edwards Hill 1980); Pipistrellus rendalli (Corbet & J. Edwards Hill 1991); P. r. phasma (Koopman 1994); P. r. rendalli (Koopman 1994); Neoromicia rendalli (Van Cakenberghe, Herrel & Aguirre 2002); Neoromicia rendalli phasma (Simmons 2005); Neoromicia rendalli rendalli (Simmons 2005); Vesperus hösemanni (Matschie in Turni & Kock 2008) (Mammal Diversity Database 2025).
Taxonomic Note:
Originally named in the genus Pipistrellus and later changed to Neoromicia (ACR 2024). Subsequent research placed it in a newly erected genus Pseudoromicia (Monadjem et. al. 2020b). Historically, two subspecies have reported: P. r. rendalli distributed within western Africa from the Gambia to Chad and P. r. phasma extending from the eastern Africa (Somalia and the Sudan) to southern Africa (Botswana, Mozambique, and likely including South Africa) (Simmons 2005). It is uncertain whether the South African population is contiguous with the nearest neighbouring population in Mozambique. The possibility that the South African and Mozambique population are distinct from those elsewhere in Africa should be investigated.
Red List Status: VU – Vulnerable D2(IUCN version 3.1)
Assessment Information
Assessors: Balona, J.1, Richardson, E.J. 2,3, & da Silva, J.M.4
Reviewers: Richards, L.R.5, Howard, A.6, Naidoo, T. 5 & Smith C. 7
Institutions: 1Gauteng and Northern Regions Bat Interest Group, 2Richardson & Peplow Environmental, 3Bat Interest Group of KwaZulu-Natal, 4South African National Biodiversity Institute, 5Durban Natural Science Museum, 6University of the Free State, 7Endangered Wildlife Trust
Previous Assessors and Reviewers: Monadjem, A., Taylor, P., Richards, L.R., White, W., Cohen, L., Jacobs, D., MacEwan, K., Schoeman, C. & Child, M.F.
Previous Contributors: Raimondo, D. & Sethusa, T.
Assessment Rationale
This is a widely distributed species outside of the assessment region, found in moist and dry woodland savannah and shrubland and commonly associated with swamps in degraded habitats and can thus occupy habitats that have been disturbed. In southern Africa, however, it occurs in only two known localities: Bonamanzi Game Reserve in northern KwaZulu-Natal Province, and in the southern Kruger National Park (Bats KZN data, J. Balona, unpubl. data, 2018, Rollier 2019), despite it being easily sampled with mist nets and multiple surveys having taken place in the region. However, it is possible that it may be present in the under surveyed iSimangaliso Wetland Park (in respect of bat surveys) and possibly found in southern coastal Mozambique where suitable habitat exists. This is an edge-of-range species that is common elsewhere in Africa, but the population from Mozambique and South Africa is isolated from the rest of the African range, this population could be taxonomically distinct. Additionally, as there are no recent records for the South African population, field surveys are required to determine its continued existence in the assessment region. Within the assessment region, the EOO is 62.75 km2. Given only two known locations within the assessment region with no further records in recent times, and the possibility that the southern African species is genetically isolated (given the disjunct distribution), we assess this species as Vulnerable D2.
Regional population effects: This species has been recorded from Palmiera in southern Mozambique and is suspected to occur more extensively there. However, it flies low to the ground and has low wing-loading so rescue effects are uncertain.
Red List Index
Red List Index: Uplisted
Reasons for change: The species, previously listed as Least Concern, is considered an edge-of-range taxon within the assessment region and is currently known from only two localities. No additional records have been documented in recent years. A precautionary yet liberal approach has been adopted in assigning its status as Vulnerable rather than Endangered or Critically Endangered, given that the species occurs exclusively within protected areas and no significant threats are anticipated that would lead to population decline.
Recommended citation: Balona J, Richardson EJ & da Silva JM. 2025. A conservation assessment of Pseudoromicia rendalli. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has been widely, but patchily, recorded over much of sub-Saharan Africa, ranging from Senegal in the west, through West and Central Africa to Somalia in the east, and as far south as South Africa. It occurs at a few widely separated localities in southern Africa, having been recorded from the southern Democratic Republic of the Congo (DRC), south-central Zambia, Chiromo in southern Malawi, the Okavango Delta in northern Botswana (Monadjem et al. 2020), Mana Pools National Park in Zimbabwe (Rautenbach & Fenton 1992), the Tete Province (Skinner & Chimimba 2005) and Palmiera in Mozambique (Monadjem et al. 2020). However, in the assessment region, the species is only recorded from Bonamanzi Game Reserve (Kearney & Taylor 1997), near Hluhluwe and iSimangaliso Wetland Park in KwaZulu-Natal (Figure 1), and in the southern Kruger National Park (J. Balona, unpubl. data, 2018; Rollier 2019). Surveys in suitable habitat across the range, including neighbouring protected areas, have revealed no further subpopulations.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): o m asl (Van Cakenberghe and Happold 2013; Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,641 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
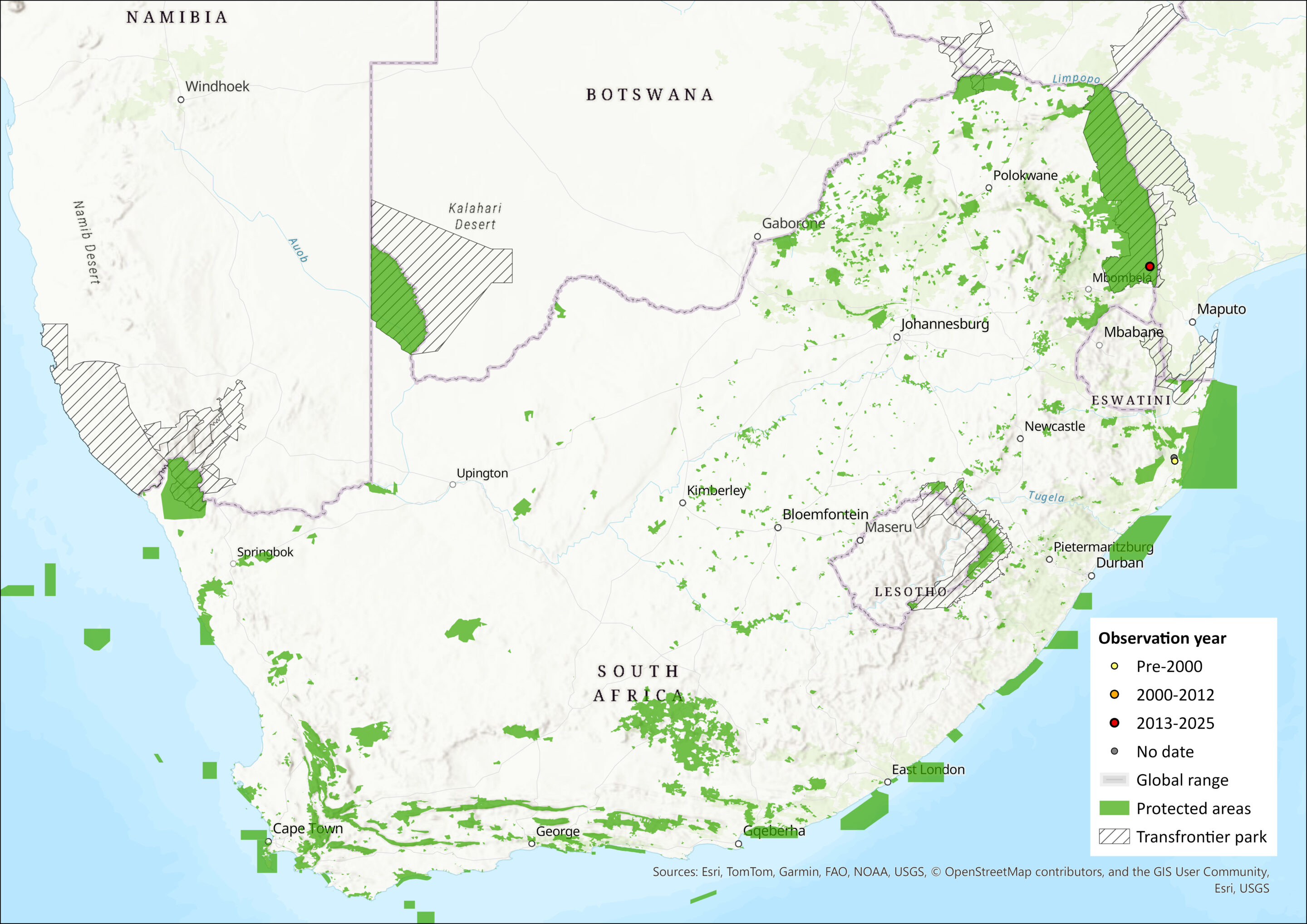 Figure 1. Distribution records for Rendall’s Serotine Bat (Pseudoromicia rendalli) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Rendall’s Serotine Bat (Pseudoromicia rendalli) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Cote d’Ivoire | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Republic of the Congo | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Sudan | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal study on the direct effects of climate change in this species has yet been conducted. However, due to the increasing temperatures across most of the species’ distribution (Mbokodo et al. 2020), the roosting preferences and foraging areas are predicted to shift. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
This species is always associated with water bodies, which will likely be threatened by increasingly common and severe droughts. As it occurs at the southernmost edge of its distribution in the assessment region, the South African locations might serve as refugia for the species.
Population information
In southern Africa, this species is very poorly represented in museums, with just 17 records examined in Monadjem et al. (2020). It appears to be a locally rare species, usually occurring as moderately small colonies of several individuals up to a few dozen animals (ACR 2024). While the species is widespread and common throughout the rest of the continent, it is only recorded from two localities in the assessment region.
Current population trend: Unknown
Continuing decline in mature individuals? Unknown
Number of subpopulations: One, possibly two.
Extreme fluctuations in the number of subpopulations: This cannot be determined without population genetic studies
Continuing decline in number of subpopulations: This cannot be determined due to the lack of available data on population size
All individuals in one subpopulation: This cannot be determined without population genetic studies
Number of mature individuals in largest subpopulation: Assumed to be ≤1000, given that the species is only know from two isolated localities within the assessment region.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: This cannot be determined due to the lack of available data on population size
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: This cannot be determined due to the lack of available data on population size
Probability of extinction in the wild within 100 years: This cannot be determined due to the lack of available data on population size
Population genetics
Given the distance between the two known localities, it is expected that two subpopulations exist within the assessment region. While the species is considered rare and expected to exist at fewer than 1,000 individuals within the assessment region, at least one subpopulation (KwaZulu-Natal) could be genetically connected to those in neighbouring countries (e.g. Mozambique), which could impact its effective population size. However, the subpopulation in Kruger appears to be isolated and likely occurs below the Ne 500 threshold.
Habitats and ecology
The species is easily discerned from other pipistrelle-like bats (with the exception of P. tenuipinnis), based on its translucent or white-coloured wing and tail membranes (Monadjem et al. 2020a). Pseudoromicia rendalli has been recorded from moist and dry woodland savannah, moist and dry tropical shrubland, and deforested areas of formerly tropical moist lowland forest. It is mostly associated with water bodies throughout its range (Skinner & Chimimba 2005; Van Cakenberghe and Happold 2013; Monadjem et al. 2020), and appears to be commensal with humans in some areas. In the assessment region, the species is recorded from the Lowveld Bioregion and Western Maputaland Bushveld. It appears to be solitary, although several individuals were observed feeding together over a pan at Bonamanzi (P. Taylor and T. Kearney unpubl. data). Nothing is known about its roosting habits in southern Africa, but elsewhere in its range it roosts singly or in small groups; roosting sites include tree holes, dense fronds of palm trees, thatched huts, brick walls and rafters (Allen et al. 1917: Rosevear 1965;). Similarly, there is no information about its diet in southern Africa. In Kenya, its diet comprised mainly Lepidoptera (Whitaker & Mumford 1978). It flies low, less than 2 m from the ground (Skinner & Chimimba 2005), and thus is easily netted.
Ecosystem and cultural services: None are currently reported; however, as an insectivorous species, it likely plays a role in regulating insect populations within its range.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Marginal | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Unknown | – |
| 14.6. Artificial/Terrestrial -> Artificial/Terrestrial – Subtropical/Tropical Heavily Degraded Former Forest | – | Marginal | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Sexes combined: mean forearm length = 3.56 ± 0.14 cm (Monadjem et al. 2020); Female mean forearm length = 3.27 cm (Van Cakenberghe and Happold 2013)
Size at Maturity (in cms): Male: Mean forearm length = 3.46 cm (Van Cakenberghe and Happold 2013)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Female maximum forearm length = 3.60 cm; Male maximum forearm length = 3.70 cm (Van Cakenberghe and Happold 2013)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Birthing appears to take place in the summer, with records of pregnant and lactating females documented in December from KwaZulu-Natal (Taylor 1998).
Average Annual Fecundity or Litter Size: 2 pups per litter (Van Cakenberghe and Happold 2013)
Natural Mortality: None recorded
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: None reported
Congregatory: No documented records of roosting or social behaviour within the assessment region (Monadjem et al. 2020, however, it is known to roost in groups of six individuals in Central Africa (Allen et al. 1917)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be utilised or traded in any form.
Local Livelihood: None reported
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
In some parts of its range, including the one of the localities in the assessment region, this species is possibly threatened by the conversion of its habitat to agricultural use. For example, clearing lala palms was listed as its major threat in Friedmann and Daly (2004). It is uncertain whether this is occurring within Bonamanzi Game Reserve. Additionally, climate change may represent an emerging threat as anecdotal evidence suggests that the small stream in Bonamanzi has not flowed more than intermittently since 2003 due to the extended Zululand drought (W. White unpubl. data).
Conservation
No specific conservation interventions can be recommended until more research is conducted to quantity habitat preferences, threats and identify further potential subpopulations. Although one of the records of this species is in a formal protected area (Kruger National Park), the only other known record in South Africa is outside the protected area iSimangaliso Wetland Park but inside Bonamanzi Game Reserve (private). Managers should determine current occupancy inside Bonamanzi and identify key roosting sites and implement measures to protect them.
Recommendations for land managers and practitioners:
- Establish a systematic monitoring programme.
- Protect key roosting sites on Bonamanzi Game Reserve.
Research priorities:
- Further research is needed into the distribution of this patchily recorded species (ACR 2024).
- Further phylogenetic and phylogeographic studies to determine the genetic relatedness of individuals within the assessment region to those elsewhere.
- Conduct systematic and targeted surveys in the northeastern portion of the assessment region to identify additional localities.
- Document reference echolocation calls to facilitate acoustic surveys aimed at identifying additional distribution records.
- Studies documenting roosting habits and subpopulation sizes and trends in southern Africa.
- Research quantifying habitat preferences and threats
Citizen Scientists:
- Upload sightings to iNaturalist and MammalMap.
- Deposit any deceased specimens with a biorepository.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Allen, J.A., Lang, H. and Chapin, J.P. 1917. The American Museum Congo expedition collection of bats. Order of the Trustees, American Museum of Natural History.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kearney, T. and Taylor, P.J. 1997. New distribution records of bats in KwaZulu-Natal. Durban Museum Novitates 22: 53–56.
Mammal Diversity Database. 2025. Pseudoromicia rendalli (O. Thomas, 1889) Rendall’s Serotine. https://mammaldiversity.org/taxon/1005774
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020a. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Demos, T., Dalton, D., Webala. P., Musila, S., Kerbis Peterhans, J. and Patterson, B. 2020b. A revision of Pipistrelle-like bats (Mammalia: Chiroptera: Vespertilionidae) in East Africa with the description of new genera and species, Zoological Journal of the Linnean Society.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Rautenbach, I.L. and Fenton, M.B. 1992. Bats from Mana Pools National Park in Zimbabwe and the first record of Eptesicus rendallii from the country. Zeitschrift für Säugetierkunde 57: 112–115.
Rollier. 2019. https://www.inaturalist.org/observations/130646186. Accessed 20 November 2025.
Rosevear, D.R. 1965. The Bats of West Africa. British Museum (Natural History), London, UK.
Skinner, J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P. 1998. The smaller mammals of KwaZulu-Natal. University of KwaZulu Natal Press, South Africa.
Whitaker, J.O. and Mumford, R.E. 1978. Foods and ectoparasites of bats from Kenya, East Africa. Journal of Mammalogy 59: 632–634.
Van Cakenberghe, V. and Happold, M. 2013. Pipstrellus rendalli Rendall’s Pipistrelle; pp645 – 647 in Happold M., Happold D.C.D. (eds) Mammals of Africa: Volume IV, Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
