Red Hartebeest
Alcelaphus b. caama

2025 Red list status
Alcelaphus b. caama
Least Concern

2025 Red list status
Alcelaphus b. lichtensteinii
Not Applicable
Regional Population Trend
Unknown
Change compared to 2016
No Change
Overview
Alcelaphus buselaphus caama – (Geoffroy Saint-Hilaire, 1803)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Alcelaphus – buselaphus – caama
Common Names: Red Hartebeest (English), Rooihartbees (Afrikaans), Ihlezu, Indluzele, Iqhama (Ndebele), Thetele (Sepedi), Kgama, Khama (Setswana), Kgama, Khama Lethodile, Tlohela (Sesotho), Umzansi (Swati), Nondo, Nondzo (Tsonga), Thendele (Venda), Ixhama (Xhosa), Indluzela, Inkolongwane (Zulu)
Taxonomic status: Subspecies
Synonyms: No Synonyms
Taxonomic Note:
Following Gosling and Capellini (2013), and in contrast to Grubb (2005), this species is considered to include both Red Hartebeest A. caama and Lichtenstein’s Hartebeest A. lichtensteinii. A total of eight subspecies are recognised, of which Red Hartebeest occurs in the assessment region and possibly (see discussion below) Lichtenstein’s Hartebeest. The eighth and nominate subspecies, the Bubal Hartebeest A. b. buselaphus, from North Africa is now Extinct (IUCN SSC Antelope Specialist Group 2016).
Red List Status: Alcelaphus b. caama – LC – Least Concern
Red List Status: Alcelaphus b. lichtensteinii – NA – Not Applicable
Assessment Information
Assessors: Venter, J.A. 1 & da Silva, J.2
Reviewer: Shrader, AM.3
Contributor: Patel, T.4
Institutions: 1Nelson Mandela University, 2South African National Biodiversity Institute, 3University of Pretoria, 4Endangered Wildlife Trust
Previous Assessor: Venter, J.A
Previous Reviewer: Child, M.F.
Previous Contributors: Gaylard, A. & Eksteen, J.
Assessment Rationale
Globally, Red Hartebeest is the most numerous subspecies (c. 130,000 animals) and is increasing. Although historically reduced from overhunting, it is now common within the assessment region, having been reintroduced into a number of formal and private protected areas across its range. The wildlife ranching industry also harbors a large number of animals on privately owned game farms and reserves. The population is widespread on formally protected areas and private land. Previously, the population increased in formally protected areas over three generations (1992–2015). Within the assessment region, there were at least 14,849 mature animals (assuming a 70% mature population structure) on formally protected areas, which increased to 38,511 mature animals by including private lands (2013/14 counts). However, based on recent data, many protected areas are showing a decline in numbers since 2016. As data were not robust enough to infer total estimates across the assessment region, the Least Concern listing remains. While there are no major threats, local threats, such as poaching, may cause declines outside protected areas. However, the effects of the wildlife industry on this species should be monitored, and hybridisation between Red Hartebeest, other hartebeest subspecies and other antelope species should be carefully regulated (for example, there should be no movement from the private sector into formally protected areas without genetic testing). This species is a key species for sustainable, wildlife-based rural economies and incentives should be put in place to conserve this species as wild and free-roaming herds on private land.
Lichtenstein’s Hartebeest is Not Applicable within the assessment region, as there is much uncertainty over whether the subspecies was ever resident in both Kruger National Park (KNP) and northern KwaZulu-Natal Province (KZN) (for example, Pongola Nature Reserve). While they have been reintroduced into both KNP and private conservancies in the Lowveld, their numbers are currently very low within the assessment region (although their numbers were estimated to be 82,000 globally in 2008), probably comprising fewer than 50 individuals. Once further evidence has been produced to confirm or reject its historical residency in the assessment region, this subspecies should be reassessed.
Regional population effects: Most of the population is fragmented through fencing. However, there is some local dispersal in the Kgalagadi Transfrontier Park between South Africa and Botswana depending on rainfall and local climatic variability.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Venter JA & da Silva JM. 2025. A conservation assessment of Alcelaphus buselaphus caama. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
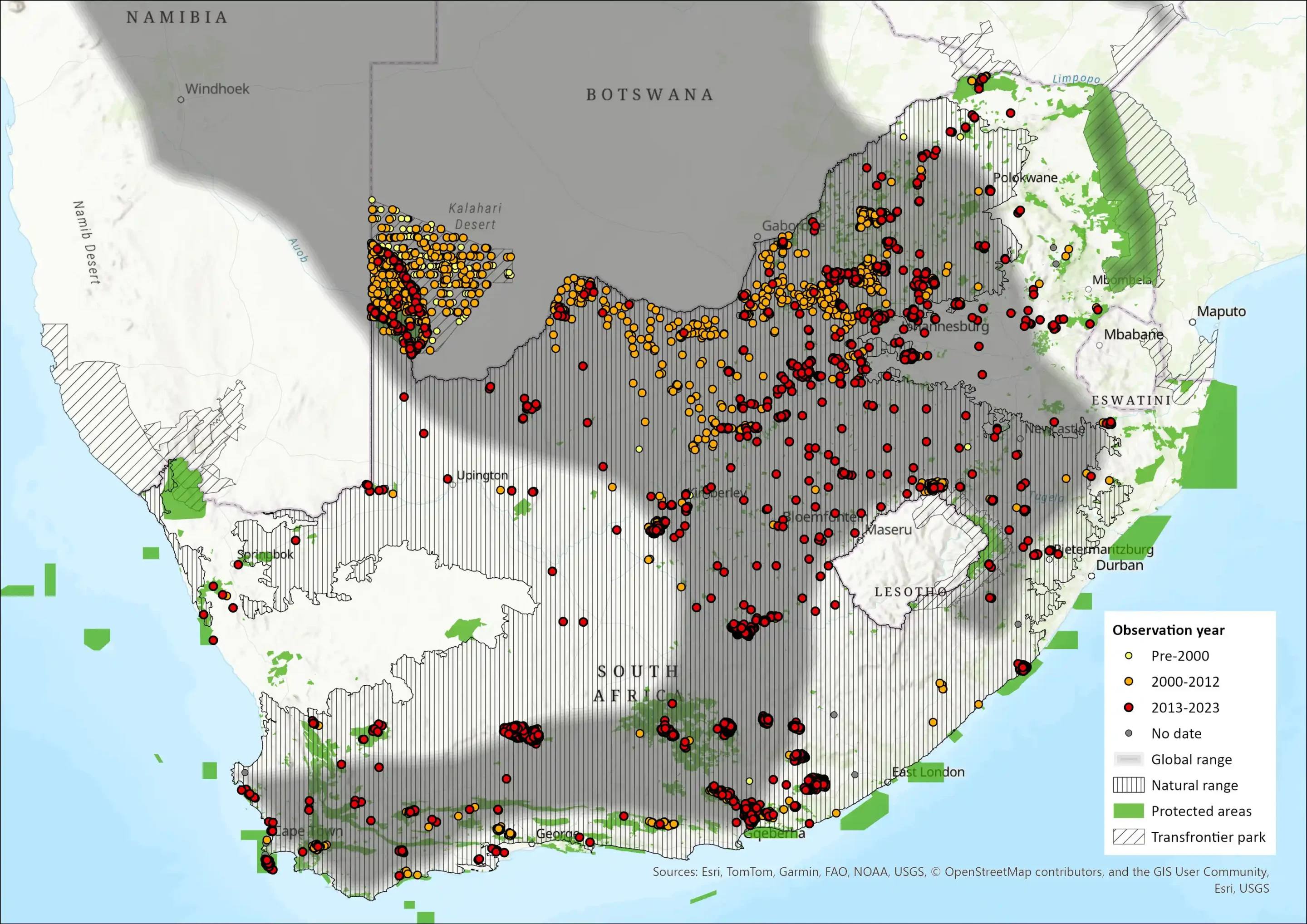
Red Hartebeest occur throughout much of southern Africa (and marginally into Angola near the Namibian border) and, although much reduced by European colonists, their range is expanding again as they have been reintroduced into many protected areas and private game farms (and widely introduced outside their former range) (IUCN SSC Antelope Specialist Group 2016); for example, in Namibia (Skinner & Chimimba 2005). The bulk of the population in Botswana is in the southwest of the country in the Kalahari but they occur widely south of the Okavango (Skinner & Chimimba 2005). While Red Hartebeest historically occurred in the western lowlands of Lesotho, intense hunting and depredation have eradicated them from this area (Lynch 1994). They are widespread throughout South Africa extending eastwards from the Western Cape coastal areas along the coast and into the hinterland into the semiarid and savannah regions as well as Highveld grasslands, but the natural range does not extend into the Lowveld of Mpumalanga and Limpopo and northern KZN. It is widespread in protected areas throughout its range, and there is an increasing tendency for this species to be introduced onto private conservation areas. Currently, the subspecies now occurs in all provinces, which is echoed historically as numerous writers documented their occurrence throughout the Northern, Western and Eastern Cape provinces and in parts of the Free State, North West, Gauteng, KZN and Limpopo provinces (Handley 1961; Skinner & Chimimba 2005). Extra-limital introductions have, however, occurred in parts of the eastern Limpopo as well as Mpumalanga (where they are currently present in three provincial protected areas; J. Eksteen pers. comm. 2015) using animals from Namibia (Skinner & Chimimba 2005). They have also been introduced into Eswatini (IUCN SSC Antelope Specialist Group 2016). The Red Hartebeest as a subspecies (A. b. caama) naturally occurs in South Africa, Namibia, Botswana and Zimbabwe.
The only part of their former range from which they were not entirely eliminated was the Northern Cape and presently herds move across the Botswana border onto farms in the province (Skinner & Chimimba 2005), which is corroborated by reports from the North West Province where the Moshita region harbours a naturally occurring, free-roaming subpopulation that probably originated from Botswana (Buijs 2010; Power 2014).
Lichtenstein’s Hartebeest formerly occurred widely in the miombo woodlands of south-central Africa, but now occur mainly in wildlife areas in Tanzania, Mozambique and Zambia; they are extinct in Burundi (IUCN SSC Antelope Specialist Group 2016). Within the assessment region, they were probably present in low numbers in the Lowveld and northern KNP and KZN (du Plessis 1969; Milstein 1989; Skinner & Chimimba 2005), but were perhaps mistaken for Tsessebe (Damaliscus lunatus lunatus) in southern KNP (Penzhorn 1985). In 1985, 18 hartebeest were reintroduced from Malawi to KNP, a further 91 captive-bred individuals were released into northern KNP during 1990–1994, and 31 to the southern regions in 1994, but there are no further planned translocations into KNP (S. Ferreira pers. comm. 2014). In KZN, there were reports of hartebeest in Pongola as early as 1895 (Skinner & Chimimba 2005). Being ecologically unsuitable for Red Hartebeest, it is reasonable to assume the subspecies was Lichtenstein’s. They have also been reintroduced to some private reserves in the Lowveld. However, there is debate around whether this subspecies ever truly occurred in the assessment region or whether they were occasional visitors from their core range. For example, it was excluded from the previous assessment (Friedmann & Daly 2004). Supporting the exclusion, several older texts do not mention the subspecies as occurring in South Africa (Roberts 1951; Rautenbach 1982; Meester et al. 1986). More research is necessary to determine whether this subspecies was, or should be, native to the assessment region.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map:
Figure 1: Distribution records for Red Hartebeest (Alcelaphus b. caama) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extinct Post-1500 | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Egypt | Extinct Post-1500 | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Introduced | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extinct Post-1500 | Native | – | – |
| Libya | Extinct Post-1500 | Native | – | – |
| Mali | Extant | Native | – | – |
| Morocco | Extinct Post-1500 | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Somalia | Extinct Post-1500 | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tunisia | Extinct Post-1500 | Native | – | – |
| Uganda | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change may make the western parts of South Africa drier in the coming years, and Red Hartebeest are particularly susceptible to drought. For example, many carcasses were found on Molopo Nature Reserve in North West Province following drought conditions (Nel 2015). Further research needs to be conducted on the possible effect of climate change on the vulnerability of this species.
Population information
Globally, East (1999) estimated the total population of all hartebeest at about 362,000 animals (including Lichtenstein’s), the majority of which are Red Hartebeest, estimated to number about 130,000 animals on both private and formally protected land. The population is widespread and thriving within the assessment region. On protected areas alone, there were a minimum observed number of 21,213 animals in 70 reserves (2013/14 counts), with the largest subpopulations occurring in the South African side of Kgalagadi Transfrontier Park (1,925 animals in the wet season of 2012; Ellis & Herbst 2013), Golden Gate Highlands National Park (1,646 animals in 2016; Bissett et al. 2016b), and Karoo National Park (1,650 animals in 2015; Gaylard et al. 2016). Interestingly, all of these protected areas have shown a decline in numbers since 2016 (SANParks data).
Some large private wildlife reserves (such as Tswalu Kalahari Reserve and Khamab Kalahari Reserve) also contained subpopulations in excess of 1,000 animals. Including animals on private lands increased the population estimate to a minimum number of 55,016 animals (2013/14 counts) on 726 properties. This yields a total estimated mature population size (assuming 70% mature population structure) of 14,849–38,511 animals in 2013/14. However, the most recent count for Khamab Kalahari Reserve showed 485 individuals, indicating a decline in numbers in both protected and private reserves.
Generation length has been calculated as 7.8 years (Pacifici et al. 2013), which yields a three-generation window of 23.5 years (1992–2015). During the 2016 assessment, trends on protected areas indicated a positive growth rate over three generations (Peinke & Gibisela 2014; for example, Nel 2015; Bissett et al. 2016a, 2016b; Ferreira et al. 2016; Gaylard et al. 2016). This trend has changed, however, and several state protected areas are showing significant declines since 2016. In North-West Province for example, the total population in provincial protected areas has declined by 7% with at least two protected areas losing more than 80% of their Red Hartebeest populations (NorthWest Parks and Tourism Board, 2023). This subspecies generally thrives in protected areas. Following the incorporation of QwaQwa National Park into Golden Gate Highlands National Park in 2008 (increasing the size of the protected area to 327 km2), Red Hartebeest increased from 346 in 2002 to 1,646 in 2016 (Bissett et al. 2016b). However, the population dropped to 762 in 2018, and further declined to 574 in 2020 (SANParks counts).
Population Information
Current population trend: Previously increasing, currently unknown – many protected and private areas are showing a decline in numbers since 2016.
Continuing decline in mature individuals? Unknown
Severely fragmented: Yes. Most subpopulations exist within fenced areas, relying on translocation for gene flow. However, some farms are fenced with cattle fencing which may allow herds to move between farms.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of subpopulations: 70
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
The eight recognised subspecies of A. buselaphus were described based on variation in horn shape and size, coat colour and body size (Sidney, 1965); however, these designations are not validated using molecular evidence. Instead, the eight subspecies have been shown to cluster within two evolutionarily significant units (ESUs) – northern and southern (Arctander et al. 1999; Flagstad et al. 2001; Ouma et al. 2011; Russo, unpublished data). The composition of each ESU is as follows: Northern – A. b. buselaphus, A. b. major, A. b. tora, A. b. lelwel, A. b. swaynei, A. b. cokii – and Southern – A. b. caama; A. b. lichtensteinii. Within the assessment region, a single ESU (Southern) is present.
Within this southern ESU, two Management Units (MUs) have been identified. The South-Western MU comprises A. b. lichtensteinii individuals and is believed to span South Africa (Kruger National Park only), Zimbabwe, Mozambique, Zambia, southern Democratic Republic of the Congo, Malawi, southern Tanzania.
Habitats and ecology
Red Hartebeest prefer open habitat and mainly occur in grasslands of various types (Skinner & Chimimba 2005). More tolerant of woodland areas and high grass than other alcelaphines, Hartebeest prefer the edge to the middle of open plains (Gosling & Capellini 2013). They thus appear to be an edge or ecotone species (Booth 1985), generally avoiding more closed woodland, and sometimes they occupy high-lying areas that are avoided by most other larger grazers (J. Eksteen unpubl. data). They occur on floodplain grassland, vleis, semi-desert savannah and open woodland (Skinner & Chimimba 2005).
Red Hartebeest are considered to be predominantly selective grazers that will make use of browse under limited resource conditions (Murray & Brown 1993). They feed selectively in medium-height grassland; they are less water-dependent than other alcelaphines, but nonetheless dependent on the availability of surface drinking water (IUCN SSC Antelope Specialist Group 2016). In areas with much moribund vegetation, the Red Hartebeest faces particular constraints because nearly all vegetation biomass is of low quality, which reduces food intake rates (Drescher et al. 2006a, 2006b; van Langevelde et al. 2008). Under these conditions grassland fire plays an important role in providing suitable grazing conditions for this species (Venter et al. 2014). Additionally, the hartebeest skull morphology is specially adapted to be very selective at times when good forage is scarce (Schuette et al. 1998). They are gregarious, occurring in herds of up to 20 but can occur in much larger herds (Skinner & Chimimba 2005). Among the various hartebeest subspecies, a positive correlation between mean body size and rainfall suggests that habitat productivity may drive morphological evolution between ecotypes (Capellini & Gosling 2007).
Ecosystem and cultural services: In Bushman folklore the hartebeest and the Eland (Taurotragus oryx) have magical power. A woman who has a young child does not eat hartebeest meat. The head and hide of a Hartebeest were sometimes worn over men’s shoulders when hunting large animals like African Elephants (Loxodonta Africana) or Eland. Whilst advancing towards their quarry through the grass, they would carefully mimic the actions of the hartebeest.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Marginal | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: 7.8 years (Pacifici et al. 2013)
Age at maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: 8 months
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: One calf
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is used in live animal trading at game auctions, and has a subsistence value as bushmeat or for recreational biltong hunters as they have high-quality meat (Gosling & Capellini 2013). The hartebeest also has national and international value as a species suitable for trophy hunting. This appears to have had no negative effect on the population as its value as a trophy animal ensures an increase in numbers due to reintroductions to game farms. Hunting quotas also benefit communal areas in some regions (Buijs 2010). There is also some captive breeding of hartebeest (IUCN SSC Antelope Specialist Group 2016), but this is limited.
Wildlife ranching and the private sector have generally had a positive effect on this species as it has been widely reintroduced onto private properties within its natural distribution range. Most populations on wildlife ranches are free-roaming. Ranches are generally a few hundred hectares in the central and northern parts of the country to several thousand hectares in the more arid areas of the Northern Cape. Some of the very large privately owned reserves in this area have populations in excess of 1,000 individuals and therefore contribute significantly to the regional population. Due to its value (medium priced) and popularity as a trophy hunting animal, it is a popular species to have amongst game farm owners. Captive breeding of this species is not common and currently hybrids or colour variants do not feature in the live sale market.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat and biltong hunting. | Yes | Trophy hunting and live sales. |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence |
| 1. Food – human | true |
| 15. Sport hunting/specimen collecting | true |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Most commercial use occurs in wildlife ranches and game farms. Limited captive breeding.
Threats
Globally, as the bushmeat trade escalates out of control (Lindsey et al. 2013), many hartebeest populations are being hunted to extinction (IUCN SSC Antelope Specialist Group 2016). The Red Hartebeest population within the assessment region occur in a number of protected and private reserves, however, many are experiencing declines. Poaching is a localised threat; for example, on Borakalalo National Park, North West Province (Nel 2015).
Ongoing threats to this subspecies are habitat loss and habitat degradation. Ongoing habitat conversion from agriculture, livestock farming and commercial development make habitat less available or less suitable for future reintroductions into such areas. The distributions of most hartebeest subspecies are likely to become increasingly fragmented until they are confined to those areas where there is effective control of poaching and encroachment by livestock and settlement (IUCN SSC Antelope Specialist Group 2016). However, the Songimvelo subpopulation showed an increasing trend despite an increase in competition from livestock; in the same period that the Sable (Hippotragus niger niger) subpopulation declined (J. Eksteen unpubl. data).
This subspecies is suspected to be hybridised with other hartebeest subspecies and with Blesbok (Damaliscus pygargus phillipsi), Bontebok (D. p. pygargus) and Tsessebe (Damaliscus lunatus lunatus) on private lands. This may make certain subpopulations ineligible for Red List assessment and may threaten the genetic integrity of the subspecies overall. This practice, while currently not common, should be disincentivised.
Conservation
Red Hartebeest are well protected across their range within the assessment region. No direct interventions are currently necessary for this subspecies. However, continued protected area expansion to connect fragmented subpopulations and restrictions on the introduction of extra-limital subspecies/species, especially on private lands, will increase the long-term resilience of the population.
Recommendations for land managers and practitioners
Drop internal fences to form conservancies so as to encourage greater movement within the landscape and thus less habitat degradation at the local scale.
Research priorities
Research is currently being conducted by Eastern Cape Parks and Tourism Agency on movement and feeding ecology of the subpopulation in Mkambati Nature Reserve. UNISA’s College of Agriculture and Environmental Sciences is conducting some research on the physiology of the subspecies in the Northern Cape.
Research priorities include:
- Determine the current estimate of Red Hartebeest within the assessment region.
- Quantifying the severity of bushmeat poaching.
- Quantifying vulnerability to climate change.
- Genetic work on wild subpopulations (Kalahari, Namibia and other regions) to establish if any genetically distinct subpopulations exist.
Encouraged citizen actions:
- Landowners should create conservancies for this species and engage local stakeholders to create sustainable, wildlife-based rural economies.
- Report sightings of free-roaming herds outside private lands or protected areas on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Bissett C, Ferreira S, Bezuidenhout H, Smit I, Daemane E, Mokoena V, Sikhosana T. 2016. Golden Gate Highlands National Park herbivore off-take recommendations 2016: An integrated approach combining local knowledge with data derived from animal census, herbivore models, vegetation field monitoring and satellite imagery. Scientific Services, South African National Parks.
Bissett, C., Ferreira, S., Bezuidenhout, H., Daemane, E., Smit, I., van Rooyen, F., du Plessis, N. and Moolman, L. 2016. Augrabies Falls National Park herbivore off-take recommendations 2016: An integrated approach combining local knowledge with data derived from animal census, herbivore models, vegetation field monitoring and satellite imagery. Scientific Services, South African National Parks.
Booth, VR. 1985. Some notes on Lichtenstein’s hartebeest, Alcelaphus lichtensteini (Peters). South African Journal of Zoology 20: 57-60.
Buijs, D. 2010. Aerial survey of wildlife and domestic stock on tribal land in North West Province 2010. DACERD, Mmabatho.
Capellini I, Gosling LM. 2007. Habitat primary production and the evolution of body size within the hartebeest clade. Biological Journal of the Linnean Society 92: 431-440.
Drescher M, Heitkoenig I, van den Brink PJ, Prins HH. 2006a. Effects of sward structure on herbivore foraging behaviour in a South African savanna: an investigation of the forage maturation hypothesis. Austral Ecology 31: 76-87.
Drescher M, Heitkönig IM, Raats JG, Prins HH. 2006b. The role of grass stems as structural foraging deterrents and their effects on the foraging behaviour of cattle. Applied Animal Behaviour Science 101: 10-26.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Ellis G, Herbst M. 2013. Kalahari Gemsbok National Park Aerial Census Report. Internal Field Report/2013. Scientific Services. South African National Parks.
Ferreira S, Gaylard A, Greaver C, Hayes J, Cowell C, Bissett C. 2016. Large vertebrate abundances in Parks: 2015/2016. Internal Report 14/2016, Scientific Services, SANParks, Skukuza, South Africa.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gaylard A, Ferreira S, Bezuidenhout H, Smit I. 2016. Karoo National Park aerial census report 2015. SANParks Internal Report 05 / 2016, Scientific Services
Gosling, L.M. and Capellini, I. 2013. Alcelaphus buselaphus Hartebeest. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, pp. 511-526. Bloomsbury Publishing, London, UK.
Grubb, P. 2005. Artiodactyla. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World. A Taxonomic and Geographic Reference (3rd ed), pp. 637–722. Johns Hopkins University Press, Baltimore, USA.
Handley GL. 1961. Extinction of the Red Haartebeest in Natal. Natal Wildlife 2: 6-8.
IUCN SSC Antelope Specialist Group. 2016. Alcelaphus buselaphus. The IUCN Red List of Threatened Species 2016: e.T811A50181009.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsing van die Nasionale Museum Bloemfontein 10: 177-241.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Milstein PLS. 1989. Historical occurrence of Lichtenstein’s hartebeest Alcelaphus lichtensteini in the Transvaal and Natal. Aepyceros 2: 1-141.
Murray MG, Brown D. 1993. Niche separation of grazing ungulates in the Serengeti: an experimental test. Journal of Animal Ecology 62: 380-389.
Nel, P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Peinke DM, Gibisela Z. 2014. Game management recommendations for 2014. Eastern Cape Parks and Tourism Agency, East London.
Penzhorn BL. 1985. An old reference to ʻHartebeestʼ in the Transvaal Lowveld. Koedoe 28: 69-71.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach IL. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Roberts, A. 1951. The Mammals of South Africa. Central New Agency, Johannesburg, South Africa.
Schuette JR, Leslie DM, Lochmiller RL, Jenks JA. 1998. Diets of hartebeest and roan antelope in Burkina Faso: support of the long-faced hypothesis. Journal of Mammalogy 79: 426-436.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Venter JA, Nabe-Nielsen J, Prins HH, Slotow R. 2014. Forage patch use by grazing herbivores in a South African grazing ecosystem. Acta Theriologica 59: 457-466.
du Plessis, SF. 1969. The past and present distribution of the Perissodactyla and Artiodactyla in southern Africa. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa
van Langevelde F, Drescher M, Heitkönig IM, Prins HH. 2008. Instantaneous intake rate of herbivores as function of forage quality and mass: Effects on facilitative and competitive interactions. Ecological Modelling 213: 273-284.