Red Bush Squirrel
Paraxerus palliatus

2025 Red list status
Paraxerus palliatus
Near threatened

2025 Red list status
P. p. ornatus (Ngoye Forest subpopulation)
Vulnerable
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Paraxerus palliatus – (Peters, 1852)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – SCIURIDAE – Paraxerus – palliatus
Common Names: Red Bush Squirrel, Red Squirrel, Red-bellied Coast Squirrel (English), Rooi Eekhoring (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note: This species is in urgent need of revision as there are large amounts of variation in size and pelage between isolated populations, and at least seven subspecies are currently recognised, some of which may represent valid species (Monadjem et al. 2015). While Meester et al. (1986) listed six subspecies from sub-Saharan Africa, including P. p. ornatus (Gray 1864) from Ngoye Forest and P. p. tongensis (Roberts, 1931) from Manguzi Forest in KwaZulu-Natal, it is unclear whether these taxa have distinct phenotypes or genotypes (Mugo et al. 1995). Similarly, Thorington Jr. et al. (2012) listed seven subspecies of P. palliatus but only listed P. p. ornatus as occurring in South Africa (Ngoye Forest, Eshowe District, Zululand). While Viljoen (Viljoen 1980, 1989) suggested a rise to species status for both taxa, which was taken up by Friedmann and Daly (2004), this has not been corroborated by molecular or morphometric research. Thus, given the confusion around the taxonomy due to the lack of genetic evidence and the species’ propensity for varied morphology, we assess at the species level but recognise that P. p. ornatus is a distinct form that distinguishes it from the rest of its African range.
Red List Status:
Paraxerus palliatus
NT, Near Threatened
P. p. ornatus (Ngoye Forest subpopulation)
VU, Vulnerable
Assessment Information
Assessors: Patel, T.,1 Ross, N.2 & da Silva, J.3
Reviewer: Gazzard, A.4
Institutions: 1Endangered Wildlife Trust,2Stellenbosch University,3South African National Biodiversity Institute,4IUCN SSC Small Mammal Specialist Group
Previous Assessors: Louw, S. & Coverdale, B.
Previous Reviewer: Child, M.F.
Previous Contributors: Roxburgh, L., Mondajem, A., Taylor, P., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G. & Wilson, B.
Assessment Rationale
The Red Squirrel has a restricted and fragmented range within the assessment region. It exists at the edge of its range, where it is common in suitable habitats and occurs in well-protected forest reserves, such as iSimangaliso Wetland Park, Ongoye Forest Reserve, Mhkuze Game Reserve and Ndumo Game Reserve. The northern KwaZulu-Natal forests are largely secure, however, there is a continuing decline in natural habitat. Although habitat loss and degradation are unlikely to be affecting the core protected population, there is still an inferred absolute ongoing decline in suitable habitat. Overall, the estimated extent of occurrence (EOO) is 12,239 km2 (or 6,676 km2 if we exclude the Ngoye Forest subpopulation) and the area of occupancy (AOO) (based on remaining forest patches) is 568-720 km2. Although the species qualifies for Vulnerable B1,2ab(ii,iii), we downlist it to Near Threatened B1,2ab(ii,iii) based on the rescue effect (see below).
Although molecular research is needed to resolve the taxonomy of this potential species complex, the Ngoye Forest subpopulation (P. P. ornatus) is suspected to be a source of genetic diversity and warrants special attention. The Ngoye Forest subpopulation is restricted to one location with an AOO of 33 km2 and an estimated total number of mature individuals of 14,190. Although there has been no net decline in the area under formal protection, excessive fires, leading to increased patchiness of the forest, specifically on the forest periphery and extending outwards of the formally protected area, have occurred within recent years. This indiscriminate burning is resulting in increased alien plant infestation and habitat transformation, which may reduce the available habitat for the Ngoye Forest subpopulation. Ngoye Red Squirrels are rare or absent in such areas and further research is needed to determine whether such degradation is causing subpopulation decline, the results of which will require reassessment as the subpopulation may qualify for Critically Endangered B1,2ab(iii). Recent research suggests that current levels of fuelwood harvesting do not affect canopy structure, but increased harvesting levels will threaten the forest structure. Thus, we list as Vulnerable D2, with the provison that wood harvesting and fire levels should be closely monitored and managed. As such, this subpopulation remains conservation dependent.
Interventions for this species include protected area expansion to connect forest fragments and enforcement on regulation to prevent disturbances and degradation inside existing forests. If P. p. tongensis is proved to be restricted to Maputaland then it will need reassessment. We recommend molecular research to resolve the complicated taxonomy of this potential species complex within the assessment region.
Regional population effects: Dispersal is possible through the northern border of KwaZulu-Natal into Mozambique, particularly through the Lubombo Transfrontier Conservation and Resource Area. Habitat is largely connected in these areas, especially as the species is an adequate disperser and can use secondary habitats. The species is also very common throughout the rest of its range. For example, it is abundant in Maputo Elephant Reserve and throughout the iSimangaliso Wetland Park. However, the Ngoye subpopulation may be isolated from the rest of the species, and not enough is known about its dispersal ability, thus its status is not altered in this regional-level assessment.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: (Not specified)
Recommended Citation: Patel T, Ross N & da Silva JM. 2025. A conservation assessment of Paraxerus palliatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
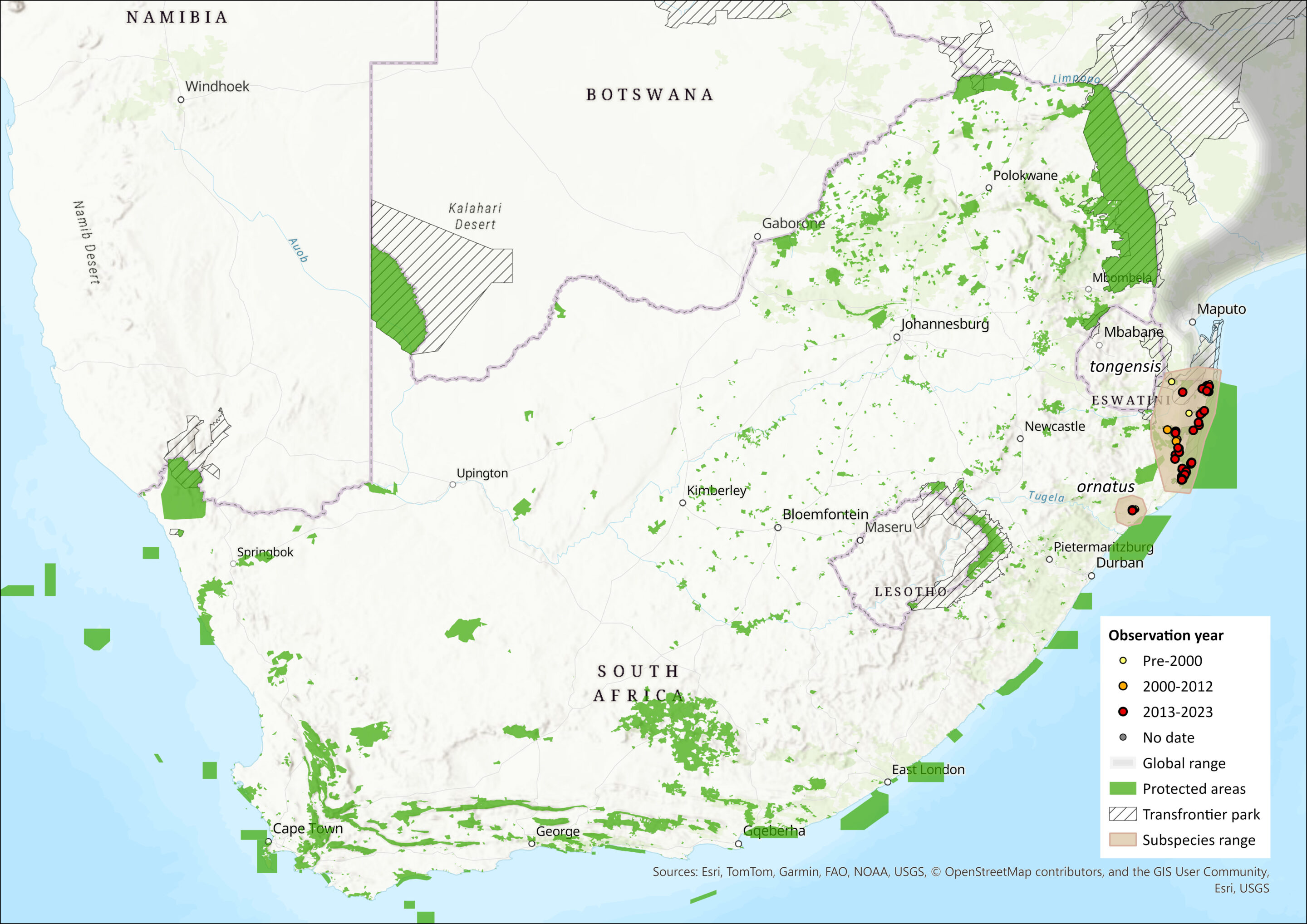
This species is widely distributed along the coastal forests of southern and eastern Africa (Monadjem et al. 2015), ranging from southern Somalia in the north, southwards to KwaZulu-Natal in South Africa, being found as far south along the coast as Lake Saint Lucia, with an isolated subpopulation in the Ngoye Forest (Viljoen 1989), which marks the southern limit. Within this range, their occurrence is patchy and discontinuous due to the fragmented nature of remaining forest where deforestation has previously occurred (Lawes et al. 2004; Skinner & Chimimba 2005). Within the assessment region, it occurs in evergreen coastal forests (Mugo et al. 1995; Skinner & Chimimba 2005), such as sand and scarp forests, exclusively in KwaZulu-Natal, and is common and widespread in the secure habitats of the northeastern regions and Maputaland. It does not occur in Eswatini (Monadjem 1998), despite potentially similar and connected habitat being found in the Lubombo region.
The potential subspecies are relict populations from habitats that were once connected to larger tracts of forests that continued northwards into Mozambique (Mugo et al. 1995). The Ngoye Red Squirrel, P. p. ornatus, is an Afromontane relic, isolated from other subpopulations for at least 6,000 years and is distinct morphologically. It may be a separate species, but molecular work is needed to confirm this. However, Ongoye Forest Reserve is certainly a key protected area for this species as a reservoir of genetic diversity. Although the subspecies P. p. tongensis has been recognised (Meester et al. 1986), with the type specimen coming from Manguzi Forest, and assessed separately before (Friedmann & Daly 2004), there is no evidence for its taxonomic uniqueness and it is not considered to be a Maputaland endemic in this assessment, until further taxonomic research has occurred.
The extent of the occurrence (EOO) for the species is 6,676–12,239 km2 while the combined area of occupancy (AOO), based on remaining natural forest habitat (2018 Vegetation map of South Africa), is between 568–720 km2 (range is calculated from the area of all natural forest patches with occurrence records since 2000 versus all forest patches intersecting the EOO). It does not include nearby forests outside of the EOO, because, despite the adequate dispersal abilities of the species (Viljoen 1986), rates of colonisation of forest fragments within KwaZulu-Natal are low (Lawes et al. 2000a), which means that patches of forest for which there are no records for the species are probably unoccupied in reality (true negatives). Similarly, as P. p. ornatus is restricted to Ngoye Forest (which includes Ongoye, Dengweni, Ezigwayini and Impeleshu Nature Reserves and the respective extensions falling within the Mzimela Traditional Council area), its AOO is calculated as 33 km2. The AOO for forest patches with confirmed presence of P. palliatus (since the year 2000) (excluding Ongoye Nature Reserve) is 568 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Red Bush Squirrel (Paraxerus palliatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
In South Africa, P. palliatus is largely confined to forested areas of Northern Kwa-Zulu Natal. The main threats to these areas are ongoing habitat loss due to logging and risk of forest fires. Climate change is predicted to increase the intensity of droughts and change rainfall patterns (Lawal et al. 2019). This could increase the fire risk to these forested areas and limit the water resources available to the species, leading to increased population fluctuations and potentially threatening the overall population. However, as this species has a viable population and no known population decline, severe population fluctuations and climate change does not pose an immediate threat to the species.
Population information
Overall, the species is common and abundant throughout northeastern KwaZulu-Natal, such as in Ndumo Game Reserve and iSimangiliso Wetland Park, and throughout the rest of its range. As forest habitat is currently stable, we infer that the population trend is stable. Viljoen (1986) calculated a density of 2.2 mature individuals / ha (or 449 g / ha), which equates to a population size of 124,960–158,400 based on a range of AOO estimates for the species (568–720 km2). Similarly, the density of P. p. ornatus was calculated as 4.3 individuals / ha (again excluding juveniles), which yields a mature population size of 14,190 for Ngoye Forest (33km2). Thus, the population size for this species is estimated to be large, although Viljoen (1986) cautions that juvenile recruitment is low. This direct extrapolation may, however, overestimate the current size for this subpopulation as a result of varying habitat assemblages, created through anthropogenic influence (fire, grazing, wood extraction), within the forest complex and limited number of reported sightings by persons working in the area. Although the core population is suspected to have remained stable since at least the 1990s, due to strong forest protected areas in the region, ongoing habitat loss is inferred to be causing a net decline in the population.
Population Information
Current population trend: Declining, inferred from ongoing habitat loss in the region although there are secure forest habitats in northeastern KwaZulu-Natal.
Continuing decline in mature individuals? No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
There are several subpopulations of P. palliatus within South Africa, all of them occurring within Northern KwaZulu-Natal. P.p. ornatus is only known to have one subpopulation estimated to be between 13,000 to 15,000 mature individuals solely within the Ngoye Forest area. There are several subpopulations of P.p. tongensis in the forests and thickets between St. Lucia and Mozambique. These could exist as a single metapopulation, but this will need to be confirmed using molecular techniques. There is no census data for individual populations, but it is estimated that there are between 97,000 and 155,000 total mature individuals. There are also no known extinct populations of this species, and no known phylogenetic studies have been carried out. However, P.p tongensis is morphologically and very likely genetically different from P.p ornatus with no known gene flow occurring between the subpopulations.
Based on the available information, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (2/2 subpopulations remaining). The GBF’s headline indicator – proportion of species with an effective population size (Ne) greater than 500 – can be estimated using a conversion ratio of Net/Nc between 0.1-0.3. P.p. ornatus is therefore estimated between 1 300 and 4 500; while P.p. tongensis is estimated between 9 700 and 46 500, far exceeding the threshold of 500. Consequently, the Ne 500 indicator would receive a value of 1.0 (2/2 populations with Ne > 500).
Habitats and ecology
This species has been recorded from a variety of habitat types including dry or moist evergreen forests, woodlands, riverine forest and thickets with an understorey of leafy vegetation (Skinner & Chimimba 2005). They do not occur in savannah woodlands unlike Tree Squirrel (Paraxerus cepapi). Logging followed by coppice regeneration provides suitable habitat (Skinner & Chimimba 2005), although there is no formal evidence that they utilise secondary habitats within the assessment region. They obtain their moisture from the fleshy fruits they eat or from dripping water on leaves and branches or water trapped in holes on trees (Skinner & Chimimba 2005). They are arboreal and diurnal. Generally solitary, but members of a nesting group will be within vocal contact distance. The female is the focal point of the group, whose males may change from time to time (Skinner & Chimimba 2005). The species has one or two litters annually of one or two young.
Fruit trees are focal points of activity as their diet is comprised primarily of wild fruits, nuts, and berries, with small amounts of roots, leaves, flower buds, bark, lichen and insects. A study from Ongoye Forest Reserve and Mkwankwa forests revealed male home ranges to be 3.2–4.2 ha and females to be 1.2–2.2 ha, with maximum dispersal distance of 1,065–1,142 m and 685–699 m for males and females, respectively (Viljoen 1980). It is a locally abundant and conspicuous species, due largely to its habit of tail-flicking and staccato calling when perturbed (Skinner & Chimimba 2005).
Ecosystem and cultural services: It is possible that it provides seed dispersal services, much like Samango Monkeys (Cercopithecus albogularis) (Linden et al. 2015), although no formal studies have evaluated this. It has the potential to be a flagship species of KwaZulu-Natal forests and biodiversity stewardship schemes.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.5. Forest -> Forest – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations |
– |
Unknown |
– |
Life History
Generation Length: 3.8 years (Pacifici et al. 2013)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not traded or utilised. There is no evidence for its use as bushmeat.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The major threats to this species are habitat loss and degradation, although the extent to which these affect the species currently needs research. Although much of its coastal forest habitat has been deforested or degraded historically, it currently occurs in well-protected habitat and, since the 1990s, there has been no real evidence for population decline (Mugo et al. 1995). Although the northern KwaZulu-Natal forests are largely secure, there is a continuing decline in natural habitat (19.7% loss of natural habitat in KwaZulu-Natal from 1994 to 2011, with an average loss of 1.2% per annum, slowing slightly to an overall loss of 4.5% between 2014 and 2020, with an annual rate of 0.57%), with the losses mainly occurring in small (< 0.5 ha) forest patches. Additionally, habitat degradation from human encroachment such as fuelwood extraction, may indicate a continuing decline in habitat quality along protected area edges if large canopy gaps are produced.
This species is probably not as threatened as ground-dwelling small mammals, such as shrews, because overgrazing and small-scale fuelwood collection will not alter habitat structure as drastically. For example, in a study assessing the impact of human utilisation of woodlands and sand forests in Maputaland, while utilisation decreased averaged stem diameter, the height structure of the forest remained largely unchanged (Gaugris & Van Rooyen 2010). Similarly, in Ongoye Forest Reserve specifically, no instance of canopy tree logging has been recorded (understorey species harvested exclusively at a harvesting pressure of 11.6% of available pole-size trees) and most species harvested for building materials and fuelwood regenerate over small spatial scales, suggesting that the level of harvesting is sustainable (Boudreau et al. 2005), yet this is not the case in adjacent areas within the Ngoye forest complex. A further study concluded that such understorey harvesting at low levels of harvesting (11.6%) does not affect the regeneration of canopy species and will not detrimentally affect the overall species composition of scarp forest (Boudreau & Lawes 2005). Similarly, Louw (2010) found that, while creating small gaps from harvesting pole-sized trees will not affect forest dynamics and species composition, harvesting in excess of eight adjacent trees, and so creating larger gaps, will potentially lead to successional shifts and alternate states in the ecosystem, potentially negatively affecting habitat suitability for the Ngoye Red Squirrel. Thus, harvest management should be regulated accordingly. Other factors that could cause habitat degradation are wildfires and alien plant infestations. For example, over recent years, forest patches have been lost in Ngoye Forest from excessive fires and the Ngoye Red Squirrel has neither been seen in such degraded patches nor the outer-lying eastern forest patches (S. Louw pers. obs. 2016). Furthermore, the protected areas and forest patches extending into the Mzimela Traditional Council area have minimal control, face the same challenges and run the risk of agricultural and residential developments together with their associated infrastructure, and are thus impacted to a greater extent.
Overall, although current rates of habitat loss and degradation appear not to be negatively affecting the population, we take a precautionary purview as an absolute net decline in habitat in the region and increased levels of harvesting pressure could rapidly affect the species in future.
Conservation
This species occurs in well protected coastal forest reserves, such as, iSimangaliso Wetland Park, Ongoye Forest Reserve, Mhkuze Game Reserve and Ndumo Game Reserve. Further field studies should document in which unprotected forest patches the species occurs, so as to assess the threat of losing small patches (Lawes et al. 2004), and to inform protected area expansion strategies. For example, a transfrontier conservation area in Maputaland that connects with the existing Lubombo Transfrontier Conservation Area was proposed and would add 480 km2 of linking corridors between forest habitats and potentially generate US$18.8 million from game ranching (Smith et al. 2008). Such a protected area network would undoubtedly benefit this species. Biodiversity stewardship schemes that protect remaining patches of forest would also help to create corridors.
Conservationists should also work with local communities to regulate the harvesting of pole-sized trees in forests. For example, recent research suggests that small-scale harvesting of understorey trees (around 11% of available stems) should be sustainable and not alter forest structure if fewer than eight trees are harvested adjacently in any one area (Boudreau et al. 2005; Boudreau & Lawes 2005; Louw 2010).
Recommendations for land managers and practitioners:
- Conservation authorities should develop harvesting strategy guidelines for understorey trees and engage with local communities.
- Enforce regulations on developments that potentially impact on the habitat integrity of forests.
- Conservation authorities should control and manage livestock grazing within forest protected areas.
- Conservation authorities and local Fire Protection Associations should monitor the burning regimes adjacent to indigenous forest patches and provide necessary support to burn appropriate fire breaks around such areas.
Research priorities:
- Further field studies are needed to determine the distribution of this species in forest patches within the matrix between protected areas.
- More research and systematic reviews are needed to determine the net effects of forest land cover change on this species.
- Molecular research is necessary to resolve the taxonomy of this potential species complex.
Encouraged citizen actions:
- Report sightings and photograph this species, especially outside protected areas, on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Boudreau S, Lawes MJ. 2005. Small understorey gaps created by subsistence harvesters do not adversely affect the maintenance of tree diversity in a sub-tropical forest. Biological Conservation 126: 279-286.
Boudreau, S., Lawes, M.J., Piper, S.E. and Phadima, L.J. 2005. Subsistence harvesting of pole-size understorey species from Ongoye Forest Reserve, South Africa: Species preference, harvest intensity, and social correlates. Forest Ecology and Management 21: 149-165.
Corrigan BM, Kneen M, Geldenhuys CJ, van Wyk BE. 2010. Spatial changes in forest cover on the KwaNibela Peninsula, St Lucia, South Africa, during the period 1937 to 2008. Southern Forests: a Journal of Forest Science 72: 47–55.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Gaugris JY, Van Rooyen MW. 2010. Woody vegetation structure in conserved versus communal land in a biodiversity hotspot: a case study in Maputaland, South Africa. South African Journal of Botany 76: 289-298.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Lawal, S., Lennard, C., Hewitson, B. 2019 Response of southern African vegetation to climate change at 1.5 and 2.0° global warming above the pre-industrial level. Journal of Climate Services 16: 1-16
Lawes MJ, Eeley HAC, Piper SE. 2000. The relationship between local and regional diversity of indigenous forest fauna in KwaZulu-Natal Province, South Africa. Biodiversity & Conservation 9: 683–705.
Lawes MJ, Macfarlane DM, Eeley HA. 2004. Forest landscape pattern in the KwaZulu-Natal midlands, South Africa: 50 years of change or stasis? Austral Ecology 29: 613-623.
Lawes, M.J., Meakin, P.E. and Piper, S.E. 2000. Patch occupancy and potential metapopulation dynamics of three forest mammals in fragmented Afromontane forest in South Africa. Conservation Biology 14: 1088-1098.
Linden B., Linden J., Fischer F., Linsenmair K.E. 2015. Seed dispersal by South Africa’s only forest-dwelling guenon, the samango monkey (Cercopithecus mitis). African Journal of Wildlife Research 45: 88–99.
Louw SL. 2010. The effect of the spatial scale of tree harvesting on woody seedling establishment and tree dynamic at Ongoye Forest Reserve. M.Sc. Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Mugo, D.N., Lombard, A.T., Bronner, G.N. and Gelderblom, C.M. 1995. Distribution and protection of endemic or threatened rodents, lagomorphs and macrosceledids in South Africa. Journal of Zoology 30: 115-126.
Pacifici M, Santini L, Di Marco M, Baisero D, Francucci L, Grottolo Marasini G, Visconti P, Rondinini C (2013) Database on generation length of mammals. 5427 data records. Online at http://doi.org/10.5061/dryad.gd0m3, version 1.0 (last updated on 2013-08-27), Resource ID: 10.5061/dryad.gd0m3, Data Paper ID: doi: 10.3897/natureconservation.5.5734
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smith RJ et al. 2008. Designing a transfrontier conservation landscape for the Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
Thorington Jr., R.W, Koprowski, J.L., Steele, M.A. and Whatton, J.F. 2012. Squirrels of the World. The John Hopkins University Press, Baltimore.
Viljoen S. 1980. A comparative study on the biology of two subspecies of tree squirrels, Paraxerus palliatus tongensis (Roberts. 1931) and Paraxerus palliatus ornatus (Gray. 1864) in Zululand. Ph.D. Thesis. University of Pretoria.
Viljoen S. 1986. Use of space in southern African tree squirrels. Mammalia 50: 293–310.
Viljoen S. 1989. Taxonomy and Historical Zoogeography of the Red Squirrel Paraxerus palliatus (Peters, 1852) in the Southern African Subregion (Rodentia: Sciuridae). Annals of the Transvaal Museum 35: 49–60.