Pygmy Rock Mouse

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Petromyscus collinus – (Thomas & Hinton, 1925)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Petromyscus – collinus
Common Names: Pygmy Rock Mouse, Minute Rock Mouse (English), Dwergklipmuis, Kleinklipmuis, Damaralandse Klipmuis (Afrikaans).
Synonyms: No Synonyms
Taxonomic Note: Three subspecies of Petromyscus collinus were originally listed by Meerster et al. (1986). However, more recently Monadjem et al. (2015) considered barbouri (previously P. c. barbouri) and shortridgei (previously P. c. shortridgei) distinct species. Although P. collinus occurs sympatrically with P. barbouri and P. monticularis in the assessment region, the Pygmy Rock Mouse can be distinguished by its longer, uniformly coloured tail, larger skull, and three pairs of nipples, as opposed to two in P. barbouri (Monadjem et al. 2015) Phylogenetic analyses are required to confirm the taxonomic status of these species.
Red List Status: LC – Least Concern
Assessment Information
Assessor: Russo, I.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions: 1Cardiff University,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors and Reviewers: Wilson, B., Bragg, C. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G. & Taylor, P.
Assessment Rationale
This species is listed as Least Concern because of its relatively wide distribution in habitat unlikely to be rapidly transformed, and because its population is not believed to be declining at present. There are no major land-use threats to this species within the assessment region, as it does not occur in the diamond mining areas. Climate change is, however, a major emerging threat to the species. The construction of renewable energy projects earmarked in some of its distributional range may represent another emerging threat. However, more research needs to be undertaken to explicitly define the impacts of this threat. This species is considered rare but could be locally common in some portions of its range, for example, large numbers have been collected on the Namibian Escarpment.
Regional population effects: Occasional dispersal of this species across the Orange River between South Africa and Namibia is likely, thus rescue effect is possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Russo I & da Silva JM. 2025. A conservation assessment of Petromyscus collinus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
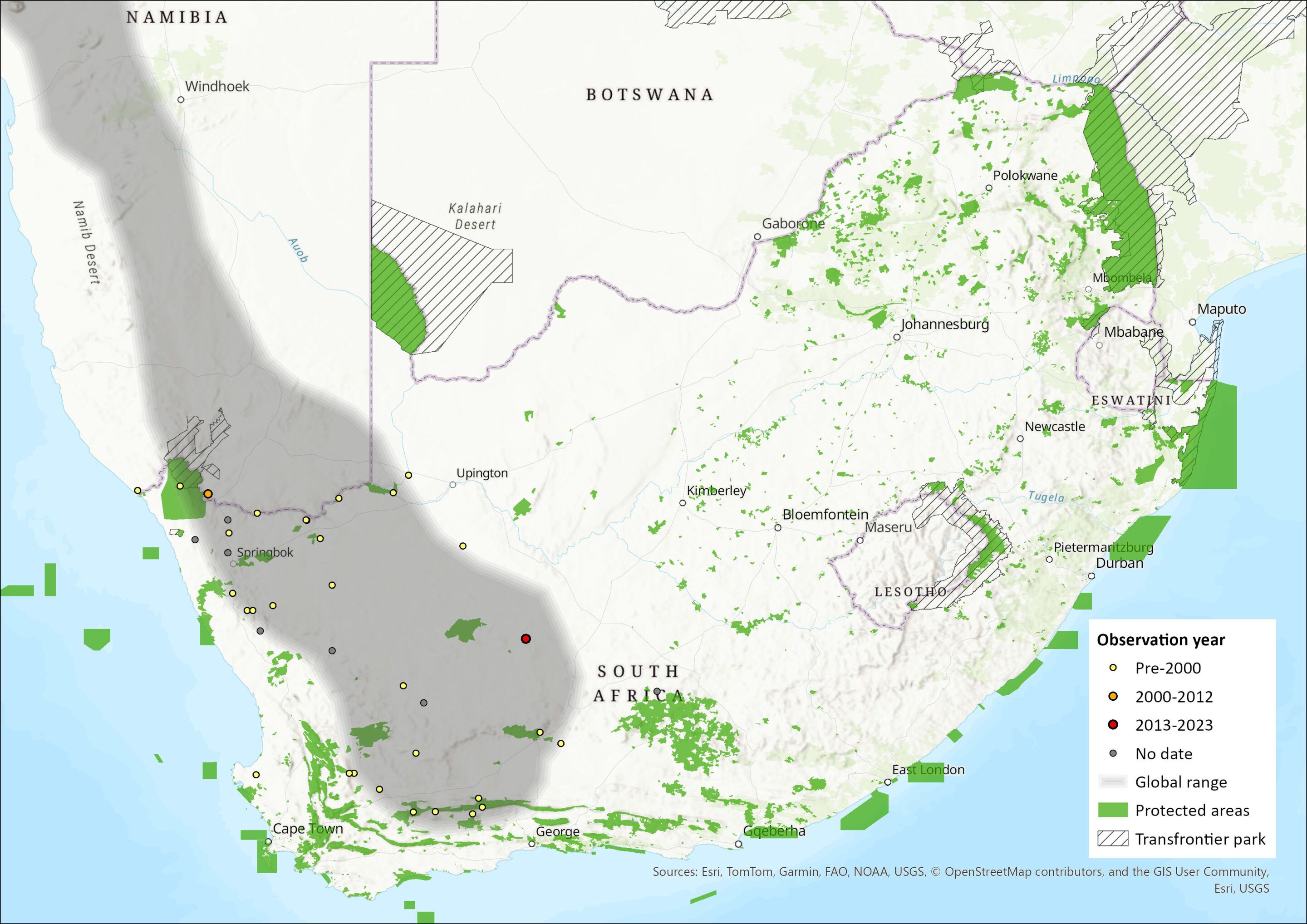
The Pygmy Rock Mouse has the most extensive range of all Petromyscus species and is distributed from South Africa’s Succulent- and Nama-Karoo biomes, northwards through western Namibia, including parts of the Namib Desert, and into the southwestern regions of Angola (Skinner & Chimimba 2005; Monadjem et al. 2015). Their altitudinal range limits are from 100 m to 2,000 m asl. Within the assessment region, they occur in rocky outcrops across the western part of the Northern and Western Cape Provinces. However, informal surveys have not recently recorded this species in rocky outcrops south of Springbok (C.S. Bragg unpubl. data) and so further vetting of museum records and field surveys should be undertaken to determine its current extent of occurrence.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 100
Elevation Upper Limit (in metres above sea level): 2,000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Pygmy Rock Mouse (Petromyscus collinus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
This species only occurs in the Western and Northern Cape provinces, South Africa. There is a possibility that it might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). This species uses overhangs and cracks in rocks for shelter. It also undergoes torpor to limit water loss during hot seasons. Rainfall is predicted to decrease in its range. These decreases will result in a more arid and drought-prone habitat which could affect food availability in the species distribution range.
Population Information
There are no population estimates available for the Pygmy Rock Mouse. It is generally considered rare but may be relatively abundant in certain locations in parts of its range (Coetzee 2013), such as in its preferred habitat of rocky outcrops. This species typically has a high survival rate, forming stable populations, and exhibiting population demographics based on a low reproductive potential (Withers et al. 1980).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: Yes. Naturally fragmented as it is restricted to isolated koppies and avoids the surrounding plains.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Given the limited information available on this species distribution (limited recent surveys) and the lack of genetic information, it is not possible to note whether this species exists as a single metapopulation throughout its range or whether distinct genetic subpopulations exist. However, given that its relatively wide distribution and the likelihood that its habitat has not been greatly or rapidly transformed, with no population declines perceived, it is fair to suggest that none of the species potential populations have gone extinct. Based on this, we can infer that all populations are maintained. Accordingly, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBFs) complementary genetic indicator – the proportion of populations maintained within a species – can be quantified and receive a value of 1.0 (all populations maintained).
Given the lack of population abundance and density estimates, it would not be possible to infer the effective population size (Ne). Consequently, the GBF’s headline indicator – proportion of populations with an effective population size (Ne) greater than 500 – cannot be quantified.
It is highly recommended that a fine scale population genetic study be undertaken to help clarify these uncertainties.
Habitats and ecology
The Pygmy Rock Mouse occurs within rocky outcrops (koppies) in arid and semi-arid shrubland (Monadjem et al. 2015). It prefers vegetated areas or overhangs and cracks in the rocks, which provide shelter (Withers 1979) to avoid predation (Brown et al. 1998). Breytenbach (1982) documented its preference for succulent Portulacaria afra shrubland in the southern portion of its range (Swartberg, Western Cape Province). The average body mass of Petromyscus collinus is 19.88 ± 1.04 g (n = 8) (Bragg 2000).
During the summers in the Namib Desert, this well-adapted, nocturnal species will undergo torpor, lowering its body temperature to limit water loss (Withers et al. 1980) and conserve energy (Bragg 2000). Although individuals can enter torpor in winter-acclimated laboratory conditions, this proved energetically risky for them, and they had difficulty arousing. It is likely that the species depends on natural ambient temperature cycles to aid its arousal in the field and may use torpor more frequently in the summer rather than winter as a means of energy-conserving adaptations to the spatially and temporally unpredictable environment of deserts. From laboratory records, it is presumed that in the wild this species is predominantly granivorous (Skinner & Chimimba 2005). Withers (1979) noted that it was an opportunistic feeder, incorporating insects (which have c. 70% water content) and plant material into its diet. Brown et al. (1998) recorded Pygmy Rock Mouse feeding on seeds deposited in the faecal pellets of hyraxes (that feed extensively away from the koppies), and insects or arthropods, which themselves were feeding on the faecal pellets. In the Namib Desert, Pygmy Rock Mice breed seasonally, and only in the presence of fog, producing one litter each summer, usually 2–4 young with a high survival rate (Withers 1983; Bragg 2000).
Ecosystem and cultural services: This species probably contributes towards seed dispersal in the semi-arid Succulent Karoo Biome of South Africa and, similar to other nocturnal rodents, may form a valuable prey species for nocturnal aerial predators, as well as several relatively sedentary reptile and mammal species (Coetzee 2013). Since they seem to utilise hyrax faeces as a food source, an obligate relationship might exist between the two species. They may also control some associated insect populations.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
6. Rocky areas (eg. inland cliffs, mountain peaks) |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species does not appear to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been identified for this species. However, in association with global climate change, the projected vegetation shift in the semi-arid Succulent Karoo may represent a future threat to this species (Rutherford et al. 1999). Increased temperatures, in conjunction with an enhanced frequency and duration of drought, are predicted to have negative effects on resource availability for small herbivorous mammals in the arid regions of southern Africa (Hoffman et al. 2009). The conservative metabolic rates and summer torpor shown by P. collinus suggest tolerance to limited, periodic supplies of food such as that experienced in the summer rainfall season of southern African arid regions. During summer, resources are unpredictable but might be of sufficient quantity and quality for the mouse to have adequate energy reserves to enter torpor. Bragg (2000) showed that it was extremely risky for P. collinus to enter torpor. Thus, any prolonged droughts and concomitant shortages of food resources during summer, such as climate change-exacerbated droughts, are likely to have severe energetic consequences on this species.
The rapidly expanding construction of major solar farms in the Northern Cape may signify an emerging threat to this species as its range partially overlaps areas in the west of South Africa earmarked for projects (van der Westhuizen 2013). However, it is unlikely that these solar farms will impact the species’ habitat as their habitat is mostly limited to rocky outcrops which is not suitable for the construction of solar farms, but the presence of these developments on the flats and plains might cause disruption in dispersal.
Overgrazing, which could be exacerbated by climate change, impacts on natural grazing capacity of the vegetation, which could lead to habitat loss for the species and in addition, increase predation risk as the vegetation cover decreases. It was observed that the species is not as aggressive as its habitat counterpart, Micaelamys namaquensis, and resource constraints, such as that caused by overgrazing or climate change-exacerbated drought periods, might lead to increased interspecific competition.
Conservation
No specific conservation initiatives have been identified for the Pygmy Rock Mouse. The range of this species extends into several protected areas, including Anysberg Nature Reserve, the Groot Swartberg Nature Reserve, Richtersveld National Park, Augrabies Falls National Park, and the Karoo National Park. However, field surveys are needed to confirm its presence in these protected areas. This species is likely to benefit from continued research into the potential threat of enhanced aridity associated with climate change, and how this may affect food resources for this species, as well as potential habitat loss in areas earmarked for alternative energy developments.
Recommendations for land managers and practitioners:
- Systematic surveys needed to gather information on population size and trends.
Research priorities:
- Population size, distribution and trend estimates.
- Population genetic study be undertaken.
- Analysis of potential impacts of alternative energy projects falling within the distributional area.
- Analysis of potential impacts of increased resource constraints and increased temperature extremes on energetics of the species under climate change scenarios.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNauralistt and MammalMAP), especially outside protected areas. However, due to their morphological similarities, misidentification of this species with other Petromyscus species is common. See taxonomic note for distinguishing characteristics.
- Encourage the inclusion of mitigation measures specific to the species in the Environmental Management Plans for new renewable energy developments overlapping with this species’ distribution.
Bibliography
Bragg CJ. 2000. Thermoregulation and torpor in the pygmy rock mouse, Petromyscus collinus – energy-conservative traits in a desert subject to the unpredictability of El Nino Southern Oscillations. Honours Thesis. University of KwaZulu-Natal.
Breytenbach, G.J. 1982. Small mammal responses to environmental gradients in the Groot Swartberg of the Southern Cape. University of Pretoria.
Brown JS, Kotler BP, Knight MH. 1998. Patch use in the pygmy rock mouse (Petromyscus collinus). Mammalia 62: 108–112.
Coetzee CG. 2013. Petromyscus collinus Pygmy Rock Mouse. In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits., pp. 205–207. Bloomsbury Publishing, London, UK.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J. and Midgley, G.F., 2024. Projections of future climate change in Southern Africa and the potential for regional tipping points. In Sustainability of Southern African Ecosystems under Global Change: Science for Management and Policy Interventions (pp. 169-190). Cham: Springer International Publishing.
Hoffman M.T., Carrick P.J., Gillson L, West A.G. 2009. Drought, climate change and vegetation response in the succulent karoo, South Africa. South African Journal of Science 105: 54–60.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Rutherford MC, Powrie LW, Schulze RE. 1999. Climate change in conservation areas of South Africa and its potential impact on floristic composition: a first assessment. Diversity and Distributions 5: 253–262.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Van der Westhuizen C. 2013. Determination of Development Potential: Department of Environmental Affairs National Wind and Solar PV Strategic Environmental Assessments – To facilitate the efficient and effective rollout of wind and solar PV energy in South Africa. Centre for Scientific and Industrial Research, Pretoria.
Withers PC, Louw GN, Henschel J. 1980. Energetics and water relations of Namib desert rodents. South African Journal of Zoology 15: 131–137.
Withers PC. 1979. Ecology of a small mammal community on a rocky outcrop in the Namib Desert. Madoqua 11: 229–246.
Withers PC. 1983. Seasonal reproduction by small mammals of the Namib Desert. Mammalia 47: 195–204.