
Pygmy Right Whale
Caperea marginata

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Caperea marginata – (Gray, 1846)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – NEOBALAENIDAE – Caperea – marginata
Common Names: Pygmy Right Whale (English), Dwergnoordkaper (Afrikaans), Baleine Pygmée (French), Ballena Franca Pigmea (Spanish; Castilian)
Synonyms: No Synonyms
Taxonomic Note:
Despite its common name, the Pygmy Right Whale (Caperea marginata) is not closely related to Right Whales (Eubalaena spp.). Currently assigned to its own family, the Neobalaenidae, it may be the last surviving member of the formerly diverse Cetotheriidae (Marx and Fordyce 2016). The family Neobalaenidae is an enigmatic taxon consisting of a single genus and species, the Pygmy Right Whale (C. marginata) (Fitzgerald 2012), which was initially described from three baleen plates from western Australia (Skinner & Chimimba 2005). Historically, there was a lack of described fossil records for this species, and it was previously included within the family Balaenidae. However, a range of distinct morphological characteristics that differ from other balaenids, and genetic analyses by Árnason and Best (1991), suggest that C. marginata is, in fact, more closely related to Balaenopteridae or the family Eschrichtiidae. Even more recently, Fordyce and Marx (2012) proposed that the Pygmy Right Whale may be the last surviving species of the allegedly extinct family Cetotheriidae, but further analyses are necessary before this can be confirmed. Currently, no subspecies have been described.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Elwen, S.1 & da Silva, J.2
Reviewer: Smith, C.3
Institutions: 1TBC, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors: Leeney, R., Findlay, K., Elwen, S., Meyer, M., Oosthuizen, H. & Plön, S.
Previous Reviewer: Best, P.B.
Previous Contributors: Relton, C. & Child, M.F.
Assessment Rationale
This species is widely distributed throughout the southern hemisphere, with records from Australasia, South Africa, Namibia and in the open ocean of the South Atlantic, very few records are known from South America. There are no known direct threats to this species, and it was not exploited commercially during the whaling era. Furthermore, its main habitats are outside of most fisheries’ operational boundaries. Thus, this species remains Least Concern within the assessment region. Similarly, at the global scale, while there is a lack of current data on population size and distribution, it is unlikely that the species qualifies for a threatened category. Therefore, this species should be reassessed once current data on population size and trend are available.
Regional population effects: Although it is unknown whether this species exhibits migratory behaviour, the Pygmy Right Whale has a circumpolar distribution and there are no known barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Elwen S & da Silva JM. 2025. A conservation assessment of Caperea marginata. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Pygmy Right Whales are restricted to the southern hemisphere, where they have a circumpolar distribution, predominantly within temperate regions, between ~30°S and ~50°S in sea temperatures between 5°C and 20°C. However, within the Benguela system, the range of this species has been documented extending to ~23°S (Leeney et al. 2013) to Walvis Bay, Namibia, where juvenile C. marginata strand annually during the austral summer and early autumn (November to March; Leeney et al. 2013). Additional strandings have been documented in South Africa, along the western, southern and eastern coasts of Australia, both the North and South Islands of New Zealand, Tasmania. Only three confirmed strandings occur in South America (Chile, Argentina and the Falkland Islands) and the species is likely vagrant there (Frainer and Elwen 2024). A single stranding in the northern Hemisphere in the Gambia (Tsai & Mead, 2018), is considered a vagrant (Elwen and Frainer 2024). Live sightings of the species are highly limited with only 10s of sightings globally (Frainer and Elwen 2024). Most sightings of the species at sea are in shallow coastal waters in the same area where strandings have occurred. Around Africa, only 13 live sightings have been recorded, all in very coastal waters (Frainer and Elwen 2024). contrast, many of the sightings from the open ocean have been of groups or aggregations of whales, sometimes quite large, with groups of 5, 7, 8 (Mikhalev & Budylenko, 2012), 14 (Matsuoka et al. 2005), 80 (Matsuoka et al. 1996) and at least a 100 (Gill et al. 2008) individuals reported. Most of these sightings were associated with either upwelling areas or the subtropical convergence, where high densities of copepods or euphausiid prey are known to aggregate, and feeding behaviour during these sightings was often observed or assumed. There is no direct evidence of migrations, but stranding and sightings records around southern Africa are biased towards summer, with 56 of 72 record occurring between November and March (Frainer and Elwen 2024), although records from Australia suggest year-round residency (Kemper 2018) with stable isotope analysis suggesting year-round foraging in coastal waters (Dedden et al. 2023). Additionally, scarring patterns from cookie-cutter Sharks (Isistius spp.) suggests possible movement between the Antarctic and warmer waters. Coastal upwelling and productivity increase during climatic phenomena such as El Niño and are likely to be quickly beneficial to plankton-feeding whales such as C. marginata (Kemper et al. 2013) and thus influence its distribution.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
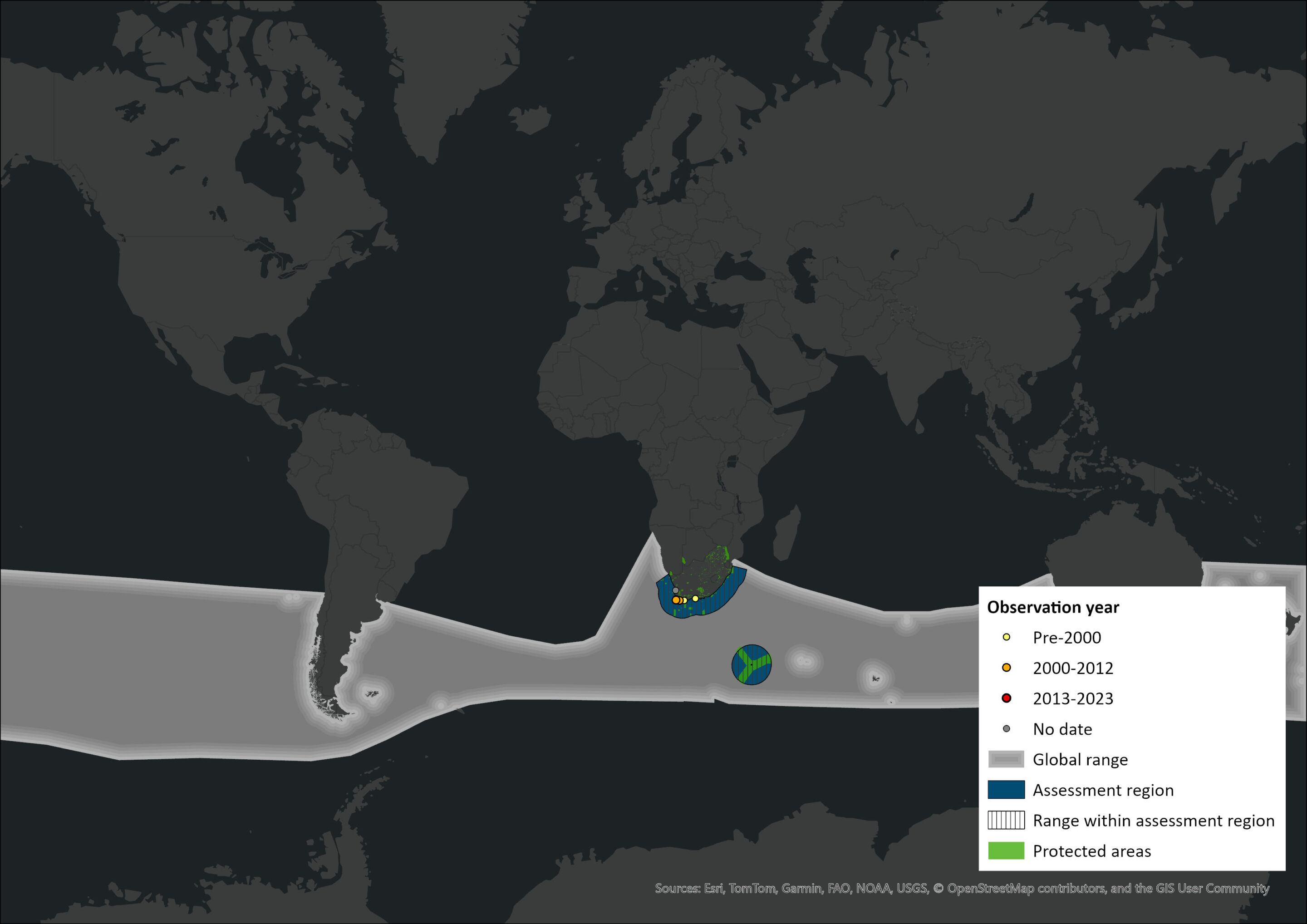 Figure 1. Distribution records for Pygmy Right Whale (Caperea marginata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Pygmy Right Whale (Caperea marginata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Antarctic, Australasian, Neotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Argentina | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Chile | Extant | Native | – | – |
| Falkland Islands (Malvinas) | Extant | Native | – | – |
| French Southern Territories | Presence Uncertain | Native | – | – |
| Namibia | Extant | Native | – | – |
| New Zealand | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 48. Atlantic – Antarctic | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 58. Indian Ocean – Antarctic | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
The main feeding habitat of Pygmy Right Whales in the sub Antarctic convergence, where they are known to prey primarily on krill. These subantarctic areas are especially vulnerable to climate change and significant warming has already taken place (Cai et al. 2023, Kubiszewski et al. 2024). These changes and others (krill overfishing) are already impacting the prey base for baleen whales, with changes in the population growth rate and feeding grounds of southern right whales (Eubalaena australis), linked to reduced prey availability southern of Southern Africa (Van den Berg et al. 2021).
Population Information
Of all baleen whales, the Pygmy Right Whale is the most unfamiliar, and the substantial lack of data prevents an accurate estimate of global or regional population size. Offshore observations of large presumed to be feeding aggregations of 80-100 individuals (Matsuoka et al. 1996, Gill et al. 2008) suggest that the species may be relatively abundant in some areas. However, this species is usually sighted in pairs or as single individuals and is occasionally associated with other cetacean species. Pygmy Right Whales are expected to spend long periods underwater (Davies and Guiler 1957), are difficult to identify by nonexperts, and have fairly inconspicuous, rapid blows, thus making them challenging to observe at sea (Reilly et al. 2008).
Within the assessment region, there is no evidence of population structure, and no reliable trend data, however no major threats have been identified for this species. Although Soviet whalers harpooned two individuals in the southern Atlantic in 1970, it was never commercially exploited. Thus, we suspect that the population is stable but have no evidence in support of this. Similarly, at a global scale, although listed as Data Deficient given the paucity of live records, given the lack of identified threats and presumed wide distribution, it is unlikely that the species is affected by any direct threats (Reilly et al. 2008, Frainer and Elwen 2024). However, as surface feeding skim feeders, pygmy right whales are particularly vulnerable to ingestion of floating plastic (e.g., Kahane- Rapport et al. 2022; Zantis et al. 2022), and the northern part of the species range, especially the areas north of 40S are predicted to be areas with a high density of floating plastics and of high concern for seabirds (Clark et al. 2023).
| Continuing decline in mature individuals? | Qualifier | Justification |
| Unknown | – | – |
Current population trend: Suspected to be stable.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
To date, no population genetic study has been carried out on this species; however, other molecular work has. Much of this work has centred around the evolutionary origins of the species, given that it is the sole representative of one of the four extant mysticete (sub)families and it is currently recognized as the only extant species of the family Neobalaenidae (sensu Gray, 1846) by the Society for Marine Mammalogy Taxonomy Committee, the International Whaling Commission, and the IUCN (Cooke, 2018). As their common name suggests, pygmy right whales were historically grouped with the right whales (balaenids) based largely on similar morphological features, such as a strongly arched and narrow upper jaw (rostrum) and narrow brain case. However, more recent molecular and morphological work has provided strong evidence that the genus Caperea is the last living taxon of the highly diverse family Cetotheriidae (Fordyce & Marx, 2013) which is more closely related to the Balaenopteroidea (including Balaenopteridae and Eschrichtidae) than to the Balaenidae (Fordyce & Marx, 2013; Park et al. 2017). The morphological similarities with right whales are now thought to have arisen as a result of convergent evolution for skim feeding (Dutoit et al. 2023).
The genome of C. marginata was recent sequenced (Wolf et al. 2023). The level of genome-wide heterozygosity was comparable to that of the blue and North Atlantic right whales (Wolf et al. 2022), but higher compared to other mysticetes (Árnason et al. 2018; Wolf et al. 2022) , Moreover, through simulations, Wolf et al (2023) showed that the population of C. marginata over time followed a similar trajectory to other baleen whales, starting from a high abundance around the Late-Pleistocene Transition (2.6 Mya) and slowly declining in effective population size (Ne) over time until reaching a lower, but stable population size after the Mid-Pleistocene Transition (1.6 Mya–700 kya). After this point in time, the trajectory showed no sign of influence from major climatic oscillations that would have consequently affected marine circulation and productivity. Despite these demographic models providing an indication of historical Ne (~3,000-4,000 individuals about 100,000 years ago), contemporary Ne has not been quantified.
As noted in the Population section, no structure is believed to exist within the assessment region; hence the single population is considered to be maintained (PM Indicator: 1/1=1.0); however, due to the lack of data on population size Ne cannot be estimated.
A population genetic study utilising samples from strandings might be useful in obtaining more concrete data on genetic diversity and structure for this species.
Habitats and ecology
Although very little is known about the habitat use of the Pygmy Right Whale, they have been documented utilising both oceanic and neritic habitats (Kemper 2002, 2009, Frainer and Elwen 2024). When sighted along coastal regions, this species is commonly located within shallow, sheltered bays. In a recent global review of the species (Frainer and Elwen, 2024), only 13 cases of live sightings could be confirmed around coastal southern Africa with an additional 5 in deeper waters (Mikhaley & Budylenko 2012) combined with 78 strandings, 2 bycaught animals and 4 taken during whaling (Frainer and Elwen, 2024) The availability of food during spring and summer may be a driver for seasonal movements inshore (Ross et al. 1975, Sekiguchi et al. 1992b). In Namibia several individuals have been observed in the coastal waters of Walvis Bay by dolphin-watching tour operators (RH Leeney pers. obs.) and the regular occurrence of live strandings, particularly of juveniles, in Walvis Bay suggests that this region may be an important habitat for young Pygmy Right Whales, and may possibly extend into the offshore environment towards the Walvis Ridge (Leeney et al. 2013).
The stomach contents of a juvenile female discovered in False Bay, South Africa comprised of a range of copepods (such as Calanoides, Carinatus, Centropages brachiatus, Paracartia africana, Oithonia spp. and Oncaea spp.) and the ampipod Themisto gaudichaudi (Sekiguchi et al. 1992a). Similarly, the stomachs of two individuals caught by Soviet whalers contained mostly calnoid copepods (Ivashin et al. 1972). Best (2007) documents that Pygmy Whales also feed on euphausiids.
As the smallest of all baleen whales, reaching a total length of just over 6 m, the term “Pygmy” was aptly attached to this species. Although very little information is available for the reproductive biology of this species, Ross et al. (1975) used information from other baleen whales to approximate the calf length at birth as 1.6–2.2 m, and at weaning as 3.2–3.8 m. Additionally, their gestation and lactation periods are estimated to last 10 and 5 months, respectively (Pavey 1992).
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales, and therefore they are prime sentinels of marine ecosystem change (Moore 2008).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
There is no contemporary trade or use of this species in South Africa.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
This species has very rarely been exploited (Clapham et al.1999) and has never been a target species by the commercial whaling industry. This species is likely to be naturally rare within its range, or may be difficult to recognise, due to its inconspicuous nature. Alternatively, the core regions of Pygmy Right Whale concentration may not yet have been identified. However, there is currently no evidence of any direct anthropogenic threats (Reilly et al. 2008), thus there is no reason to suppose that the conservation status of this species is threatened.
Although its principal habitat (40–50°S) is remote from most fishing activity, within the Benguela system, juvenile Pygmy Right Whales are known to utilise some of the bays in the region including Walvis Bay, False Bay and potentially others. These areas contain relatively high levels of commercial and recreational boat traffic and marine tourism. Since this species may also utilise regions offshore of this area, it probably also overlaps with hake trawl fisheries, which operate off the Namibian and South African coasts. Thus, similar to other baleen whales, this species may be somewhat vulnerable to entanglement in fishing gear, or collision and disturbance by boats. As surface-feeding skim feeders, pygmy right whales are particularly vulnerable to ingestion of small particles of floating plastic (e.g., Kahane- Rapport et al. 2022; Zantis et al. 2022), and the northern part of the species range, especially the areas north of 40S are predicted to be areas with a high density of floating plastics and of high concern for seabirds (Clark et al. 2023). The impacts of a changed Sub Antarctic environment which has suffered from the massive impacts of whaling and, more recently, fishing effort and climate change are unknown.
Current habitat trend: Unknown, but suspected to be stable.
Conservation
No specific conservation initiatives have been identified for this rare and enigmatic species; however, they are recorded on Appendix I and Appendix II of CITES (Convention on International Trade in Endangered Species of Wild Fauna and Flora) and CMS (Convention on the Conservation of Migratory Species of Wild Animals), respectively. Additionally, this species is likely to be somewhat covered by generic whale protection interventions both regionally and internationally. Similarly, this species is fully protected in South African waters through national legislation (Friedmann and Daly 2004).
The current lack of ecological data for this cryptic species, and its associated distribution and population trends within the assessment region, currently prevents the implementation of species-specific mitigation actions. However, this species is likely to benefit from the collection of sightings records during ship-based surveys directed at other marine species. Furthermore, data collected from stranded individuals may be a valuable means of assessing Pygmy Right Whale distributional and reproductive seasonality, diet and additional potential threats to this species.
Recommendations for managers and practitioners:
- Although species-specific monitoring is deemed unnecessary for this species in the assessment region, sightings data should be recorded during systematic monitoring of other cetacean species.
- Enforce regulations associated with artisanal fisheries, especially regarding the collection of bycatch data.
Research priorities:
- Population size and trend estimates.
- Distributional limits, and the identification of core concentration areas, including seasonality and diving behaviour.
- Diet, reproduction and general biology.
- Identification of potential threats.
Encouraged citizen actions:
- Report strandings and sightings in all range states to relevant local authorities. This is in place in Namibia through the NSN and the network of marine tour operators in Walvis Bay.
- Whale watching operators could contribute to photo ID catalogues and behavioural observations.
Bibliography
Árnason U, Best PB. 1991. Phylogenetic relationships within the Mysticeti (whalebone whales) based upon studies of highly repetitive DNA in all extant species. Hereditas 114: 263-269.
Árnason, Ú., Lammers, F., Kumar, V., Nilsson, M.A. and Janke, A., 2018. Whole-genome sequencing of the blue whale and other rorquals finds signatures for introgressive gene flow. Science advances, 4(4), p.eaap9873.
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Cabrera, E., Carlson, C. A., Galletti, V. M., Cardenas, J. C. and Brownell Jr., R. L. 2005. A pygmy right whale (Caperea marginata) from Chiloe Island, Chile. Scientific Committee of the International Whaling Commission, Ulsan, Korea.
Wenju Cai, Libao Gao, Yiyong Luo, Xichen Li, Xiaotong Zheng, Xuebin Zhang, Xuhua Cheng, Fan Jia, Ariaan Purich, Agus Santoso, Yan Du, David M. Holland, Jia-Rui Shi, Baoqiang Xiang, Shang-Ping Xie, Southern Ocean warming and its climatic impacts, 2023 Science Bulletin, Volume 68, Issue 9,
pp 946-960
Clapham, P.J., Young, S.B. and Brownell Jr., R.L. 1999. Baleen whales: conservation issues and the status of the most endangered populations. Mammal Review 29: 35–60.
Clark, B.L., Carneiro, A.P., Pearmain, E.J., Rouyer, M.M., Clay, T.A., Cowger, W., Phillips, R.A., Manica, A., Hazin, C., Eriksen, M. and González-Solís, J., 2023. Global assessment of marine plastic exposure risk for oceanic birds. Nature communications, 14(1), p.3665.
Cooke, J. G. (2018), Caperea marginata. The IUCN red list of threatened species 2018 e.T3778A50351626.
Davies JG, Guiler ER. 1957. A note on the pygmy right whale, Caperea marginata, Gray. Proceedings of the Royal Society of London 129: 579-589.
Dedden, A.V., Kemper, C.M., Truong, G., McCurry, M.R., van Ruth, P.D. and Rogers, T.L., 2023. Stable isotopes infer the diet and habitat of the enigmatic pygmy right whale (Caperea marginata) off southern Australia. Frontiers in Marine Science, 10, p.1190623.
Dutoit, L., Mitchell, K. J., Dussex, N., Kemper, C. M., Larsson, P., Dalén, L., Rawlence, N. J., & Marx, F. G. (2023). Convergent evolution of skim feeding in baleen whales. Marine Mammal Science, 39, 1337.
Fitzgerald EM. 2012. Possible neobalaenid from the Miocene of Australia implies a long evolutionary history for the pygmy right whale Caperea marginata (Cetacea, Mysticeti). Journal of Vertebrate Paleontology 32: 976-980.
Fordyce, R. E., and Marx, F. G. 2012. The pygmy right whale Caperea marginata: the last of the cetotheres. Proceedings of the Royal Society B 280: 20122645.
Frainer, G. and Elwen, S., 2024. Pygmy Right Whale Caperea.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
IUCN (International Union for Conservation of Nature). 2008. Caperea marginata. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org.. Downloaded on 21 February 2016.
Gill, P.C., Kemper, C.M., Talbot, M. and Lyons, S.A., 2008. Large group of pygmy right whales seen in a shelf upwelling region off Victoria, Australia. Marine mammal science, 24(4).
Ivashin, M. V., Shevchenko, V. I. and Yuchov, V. L. 1972. The pygmy right whale Caperea marginata (Cetacea). Zoological Journal 51(11): 1715-1723.
Kahane-Rapport, S.R., Czapanskiy, M.F., Fahlbusch, J.A., Friedlaender, A.S., Calambokidis, J., Hazen, E.L., Goldbogen, J.A. and Savoca, M.S., 2022. Field measurements reveal exposure risk to microplastic ingestion by filter-feeding megafauna. Nature communications, 13(1), p.6327.
Kemper, C. M. 2009. Pygmy Right Whale Caperea marginata. In: W.F Perrin, B. Würsig and J.G.M. Thewissen (eds), Encyclopedia of marine mammals, pp. 939-941. Academic Press, Amsterdam.
Kemper, C.M., Middleton, J.F., and van Ruth, P.D. 2013. Association between pygmy right whales (Caperea marginata) and areas of high marine productivity off Australia and New Zealand. New Zealand Journal of Zoology 40(2): 102-128. doi: 10.1080/03014223.2012.707662.
Kubiszewski, I., Adams, V.M., Baird, R. et al. 2024. Cascading tipping points of Antarctica and the Southern Ocean. Ambio .
Leeney RH, Post K, Best PB, Hazevoet CJ, Elwen SH. 2013. Pygmy right whale Caperea marginata records from Namibia. African Journal of Marine Science 35: 133-139.
Matsuoka, K., Fujise, Y. and Pastene, L.A. 1996. A sighting of a large school of the Pygmy Right Whale, Caperea marginata, in the southeast Indian Ocean. Marine Mammal Science 12: 594–597.
Matsuoka, K., Pitman, R. L. and Marquez, F. F. C. 2005. A note on a pygmy right whale (Caperea marginata) sighting in the southwestern Pacific Ocean. Journal of Cetacean Research and Management 7(1): 71-74.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Mikhalev, Y., & Budylenko, G. (2012). Карликовый гладкий кит Caperea marginata Gray, 1846, Южной Атлантики (in Russian) [Pygmy right whale Caperea marginata Gray, 1846, South Atlantic]. Український антарктичний журнал (in Ukrainian). [Ukrainian Antarctic Magazine], 10–11, 144–160.
Park, T., Marx, F. G., Fitzgerald, E. M. G., & Evans, A. R. (2017). The cochlea of the enigmatic pygmy right whale Caperea marginata informs mysticete phylogeny. Journal of Morphology, 278(6), 801–809.
Pavey CR. 1992. The occurrence of the pygmy right whale, Caperea marginata (Cetacea: Neobalaenidae), along the Australian coast. Australian Mammalogy 15: 1-6.
Reilly, S. B., Bannister, J. L., Best, P. B., Brown, M., Brownell Jr., R. L., Butterworth, D. S., Clapham, P. J., Cooke, J., Donovan, G. P., Urbán, J., and Zerbini, A. N. 2008. Caperea marginata. In ‘The IUCN Red List of Threatened Species’. Version 2008. Available at: www.iucnredlist.org. (Accessed: 1 February 2009).
Rice, D.W. 1998. Marine mammals of the world: systematics and distribution. Allen Press, Lawrence, Kansas, USA.
Ross, G. J. B., Best, P. B. and Donnelly, B. G. 1975. New records of the pygmy right whale (Caperea marginata) from South Africa, with comments on distribution, migration, appearance, and behavior. Journal of the Fisheries Research Board of Canada 32: 1005-1017.
Sekiguchi, K., Best, P. B. and Kaczmariuk, B. Z. 1992. New information on the feeding habits and baleen morphology of the pygmy right whale Caperea marginata. Marine Mammal Science 8: 288-293.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Tsai, C.H. and Mead, J.G., 2018. Crossing the equator: a northern occurrence of the pygmy right whale. Zoological Letters, 4(1), p.30.
van Den Berg, G.L., Vermeulen, E., Valenzuela, L.O., Bérubé, M., Ganswindt, A., Gröcke, D.R., Hall, G., Hulva, P., Neveceralova, P., Palsbøll, P.J. and Carroll, E.L., 2021. Decadal shift in foraging strategy of a migratory Southern Ocean predator. Global Change Biology, 27(5), pp.1052-1067.
Wolf, M., De Jong, M., Halldórsson, S.D., Árnason, Ú. and Janke, A., 2022. Genomic impact of whaling in North Atlantic fin whales. Molecular biology and evolution, 39(5), p.msac094.
Wolf, M., Zapf, K., Gupta, D. K., Hiller, M., Árnason, Ú., & Janke, A. (2023). The genome of the pygmy right whale illuminates the evolution of rorquals. BMC Biology, 21(1), 79.
Zantis, L. J., Bosker, T., Lawler, F., Nelms, S. E., O’Rorke, R., Constantine, R., Sewell, M., & Carroll, E. L. (2022). Assessing microplastic exposure of large marine filter-feeders. Science of the Total Environment, 818, 151815.
