
Pantropical Spotted Dolphin
Stenella attenuata

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Stenella attenuata – (Gray, 1846)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Stenella – attenuata
Common Names: Pantropical Spotted Dolphin, Bridled Dolphin, Narrow-snouted Dolphin, Spotted Dolphin, Spotted Porpoise, Spotter, Sharpbeaked Dolphin, Slender-beaked Dolphin, Slender Dolphin, Cape Dolphin, White-spotted Dolphin (English), Gevlekte Dolfyn (Afrikaans), Dauphin tacheté Pantropical (French), Delfín Manchado, Delfín Pintado (Spanish; Castilian)
Synonyms: Steno attenuatus Gray, 1846; Stenella graffmani Lönnberg, 1934; Stenella capensis Gray 1865. The subspecies for this species have never been assessed; they are attached here to ensure the full cetacean taxonomy is accounted for in SIS, and to make these available to the Cetacean SG if they decide to assess these taxa in future.
Taxonomic Note:
The genus Stenella is paraphyletic, and it is likely that the Delphininae will be restructured in the coming years (LeDuc et al.1999, Perrin et al. 2013). Two subspecies are recognised: S. attenuata attenuata in oceanic tropical waters worldwide, and S. a. graffmani in the coastal waters of the eastern tropical Pacific (ETP) (Jefferson et al. 2015, Perrin 2018).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Plön, S.,1 & da Silva, J2
Reviewer: Purdon, J.3,4
Institutions: 1TBC, 2South African National Biodiversity Institute, 3TUT Nature Conservation, 4Whale Unit, University of Pretoria
Previous Assessors: Plön, S., Relton, C. & Cockcroft, V.
Previous Reviewers: Atkins, S. & Child, M.F.
Previous Contributors: Elwen, S., Findlay, K. & Meÿer, M.
Assessment Rationale
Stenella species have not been well studied in the assessment region and population size and trend estimates are unavailable. However, periodic, but regular strandings suggest that there is no major population decline of these species. Additionally, Pantropical Spotted and Spinner Dolphins are abundant in the Indian Ocean. Although general pelagic threats may apply and annual takes of Stenella species occur in regions outside of the assessment region, no major threats were identified for these species; thus, they are not considered conservation priorities. However, the potential emerging threat of a drift-net fishery requires monitoring. The listing as Least Concern is retained for S. attenuata, S. coeruleoalba and S. longirostris.
Regional population effects: No barriers to dispersal of these species have been identified, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Plön S & da Silva JM. 2025. A conservation assessment of Stenella attenuata. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Generally, Stenella species occur in tropical and subtropical waters, but the extent of their range is poorly known in many regions (Moreno et al. 2005). The Pantropical Spotted Dolphin ranges extensively across all oceans from approximately 40°N to 40°S, although it is considerably more plentiful within the lower-latitude regions of its distribution. It occurs in tropical and warm temperate waters. In the assessment region, strandings have been recorded along the KwaZulu-Natal coast in the region of St Lucia from 28°24’S to 29°50’S. Strandings have also occurred sporadically in the Western Cape, including as far up the west coast as St. Helena Bay, most likely being animals from the Indian Ocean population (Best 2007). Additionally, sightings from the Durban whaling ground have been documented in waters more than 200 m and up to 3,000 m or more in depth and as far south as 32–33°S (Findlay et al. 1992; Best 2007). The species has also been documented in the southwest Indian Ocean (20°43’S–35°30’S) in summer (Gambell et al. 1975) and sighting records exist from Mozambique, Tanzania, Seychelles, Comoro Islands, Madagascar, Mauritius and Réunion (Best 2007). Although, the species appears to be primarily restricted to South Africa’s east coast, exceptions have been documented as strandings near Gansbaai (34°40’S; 19°30’E), Yzerfontein (33°15’S; 18°07’E) and Varkvlei (32°46.5’S; 18°06’E) in the Western Cape (Findlay 1989). These exceptions may indicate that this species occurs offshore of South Africa’s west coast or may represent individual strays from the east coast population, as supported by the absence of additional sightings records from the west coast.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
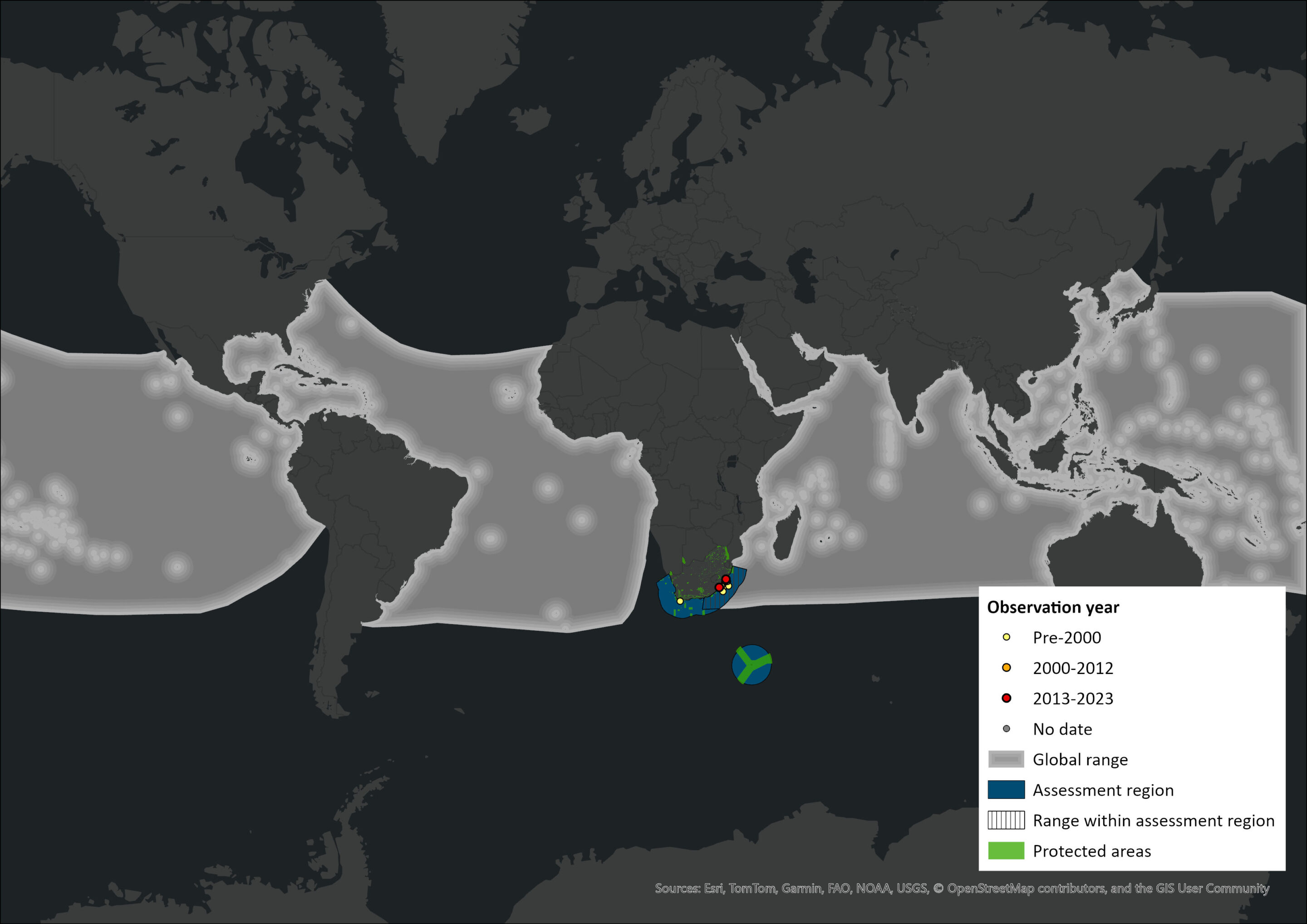 Figure 1. Distribution records for Pantropical Spotted Dolphin (Stenella attenuata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Pantropical Spotted Dolphin (Stenella attenuata) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| American Samoa | Extant | Native | – | Resident |
| Angola | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Anguilla | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Antigua and Barbuda | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Argentina | Extant | Native | – | Resident |
| Aruba | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Australia | Extant | Native | – | Resident |
| Bahamas | Extant | Native | – | Resident |
| Bahrain | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bangladesh | Extant | Native | – | Resident |
| Barbados | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Belize | Extant | Native | – | Resident |
| Benin | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bermuda | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | Resident |
| Brazil | Extant | Native | – | Resident |
| Brunei Darussalam | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cabo Verde | Extant | Native | – | Resident |
| Cambodia | Extant | Native | – | Resident |
| Cameroon | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cayman Islands | Extant | Native | – | Resident |
| China | Extant | Native | – | Resident |
| Cocos (Keeling) Islands | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Colombia | Extant | Native | – | Resident |
| Comoros | Extant | Native | – | Resident |
| Congo | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cook Islands | Extant | Native | – | Resident |
| Costa Rica | Extant | Native | – | Resident |
| Cuba | Extant | Native | – | Resident |
| Curaçao | Extant | Native | – | Resident |
| Côte d’Ivoire | Extant | Native | – | Resident |
| Djibouti | Extant | Native | – | Resident |
| Dominica | Extant | Native | – | Resident |
| Dominican Republic | Extant | Native | – | Resident |
| Ecuador | Extant | Native | – | Resident |
| Egypt | Extant | Native | – | Resident |
| El Salvador | Extant | Native | – | Resident |
| Equatorial Guinea | Extant | Native | – | Resident |
| Eritrea | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Fiji | Extant | Native | – | Resident |
| French Guiana | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| French Polynesia | Extant | Native | – | Resident |
| Gabon | Extant | Native | – | Resident |
| Gambia | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Ghana | Extant | Native | – | Resident |
| Grenada | Extant | Native | – | Resident |
| Guadeloupe | Extant | Native | – | Resident |
| Guam | Extant | Native | – | Resident |
| Guatemala | Extant | Native | – | Resident |
| Guinea | Extant | Native | – | Resident |
| Guinea-Bissau | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Guyana | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Haiti | Extant | Native | – | Resident |
| Honduras | Extant | Native | – | Resident |
| Hong Kong | Extant | Native | – | Resident |
| India | Extant | Native | – | Resident |
| Indonesia | Extant | Native | – | Resident |
| Iran, Islamic Republic of | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Iraq | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Israel | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Jamaica | Extant | Native | – | Resident |
| Japan | Extant | Native | – | Resident |
| Jordan | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Kenya | Extant | Native | – | Resident |
| Kiribati | Extant | Native | – | Resident |
| Korea, Democratic People’s Republic of | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Korea, Republic of | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Kuwait | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Liberia | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Madagascar | Extant | Native | – | Resident |
| Malaysia | Extant | Native | – | Resident |
| Maldives | Extant | Native | – | Resident |
| Marshall Islands | Extant | Native | – | Resident |
| Martinique | Extant | Native | – | Resident |
| Mauritania | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Mauritius | Extant | Native | – | Resident |
| Mexico | Extant | Native | – | Resident |
| Micronesia, Federated States of | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Montserrat | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Mozambique | Extant | Native | – | Resident |
| Myanmar | Extant | Native | – | Resident |
| Nauru | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| New Caledonia | Extant | Native | – | Resident |
| New Zealand | Extant | Native | – | Resident |
| Nicaragua | Extant | Native | – | Resident |
| Nigeria | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Niue | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Northern Mariana Islands | Extant | Native | – | Resident |
| Oman | Extant | Native | – | Resident |
| Pakistan | Extant | Native | – | Resident |
| Palau | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Panama | Extant | Native | – | Resident |
| Papua New Guinea | Extant | Native | – | Resident |
| Peru | Extant | Native | – | Resident |
| Philippines | Extant | Native | – | Resident |
| Pitcairn | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Puerto Rico | Extant | Native | – | Resident |
| Réunion | Extant | Native | – | Resident |
| Saint Barthélemy | Extant | Native | – | Resident |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | Resident |
| Saint Kitts and Nevis | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Saint Lucia | Extant | Native | – | Resident |
| Saint Martin (French part) | Extant | Native | – | Resident |
| Saint Vincent and the Grenadines | Extant | Native | – | Resident |
| Samoa | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Sao Tome and Principe | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Saudi Arabia | Extant | Native | – | Resident |
| Senegal | Extant | Native | – | Resident |
| Seychelles | Extant | Native | – | Resident |
| Sierra Leone | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Singapore | Extant | Native | – | Resident |
| Sint Maarten (Dutch part) | Extant | Native | – | Resident |
| Solomon Islands | Extant | Native | – | Resident |
| Somalia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Sri Lanka | Extant | Native | – | Resident |
| Sudan | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Suriname | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Taiwan, Province of China | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Thailand | Extant | Native | – | Resident |
| Timor-Leste | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Togo | Extant | Native | – | Resident |
| Tokelau | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Tonga | Extant | Native | – | Resident |
| Trinidad and Tobago | Extant | Native | – | Resident |
| Turks and Caicos Islands | Extant | Native | – | Resident |
| Tuvalu | Extant | Native | – | Resident |
| United Arab Emirates | Extant | Native | – | Resident |
| United States of America | Extant | Native | – | Resident |
| Uruguay | Extant | Native | – | Resident |
| Vanuatu | Extant | Native | – | Resident |
| Venezuela, Bolivarian Republic of | Extant | Native | – | Resident |
| Viet Nam | Extant | Native | – | Resident |
| Virgin Islands, British | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Virgin Islands, U.S. | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Wallis and Futuna | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Western Sahara | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Yemen | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 21. Atlantic – northwest | Extant | Native | – | – |
| 31. Atlantic – western central | Extant | Native | – | – |
| 34. Atlantic – eastern central | Extant | Native | – | – |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 51. Indian Ocean – western | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 61. Pacific – northwest | Extant | Native | – | – |
| 71. Pacific – western central | Extant | Native | – | – |
| 77. Pacific – eastern central | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
Climate change
Climate change affects cetaceans through a reduction in prey availability and a shift in the distribution of prey species (Kebke et al. 2022). This in turn results in a change in distribution range of cetaceans. In addition to affecting distribution range and abundance, climate change can also impact migration timing and behaviour (Kebke et al. 2022). With the reduction in food availability comes a change in body condition, leading to a decline in reproductive success. However, these impacts are all species-specific. Specific research needs to be carried on the impact of climate change on Spotted Dolphins.
Population
No estimates of abundance are available for Stenella species within the assessment region. Regular stranding data of Pantropical Spotted Dolphins suggest that there is no population decline: overall there have been 35 records of strandings or bycatych in KwaZulu-Natal since the 1980s. This is unlikely to be an isolated population as the species travels large distances and may interact with groups from Mozambique and the Mozambique Channel, where several thousands have been sighted. Model based estimates of generation time for the Pantropical Spotted Dolphin has been recorded at 23.1 years (Taylor et al. 2007).
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No study has looked into the genetic structuring of this species within and beyond the assessment region, however, because it can travel large distances, it is assumed to exist as a single population within the region, with its range likely extending into and beyond Mozambique waters.
Based on these assumptions, one of two genetic diversity indicators can be quantified. The one population exists and hence the proportion of populations maintained (PM) indicator is 1.0. The proportion of populations with an effective population size greater than 500 cannot be quantified at this time.
Habitats and ecology
Stenella species are relatively small dolphins, often inhabiting coastal and offshore tropical and subtropical waters, with distributions that overlap in many portions of their range (Moreno et al. 2005).
Pantropical Spotted Dolphins inhabit tropical, equatorial and occasionally warm temperate regions, and although much is known about this species in the Pacific and Atlantic Oceans, there is a paucity of information from the Indian Ocean. Off the KwaZulu-Natal coastline, group size ranged from 20 to ~ 300 individuals, with an average of about 100 (Ross 1984), while schools sighted in the subregion ranged in size from 4-1000 (n=39), but most (87.2%) contained less than 150 animals (Best 2007). In the southwest Indian Ocean, group size ranged from 25 to 200 animals (Gambell et al. 1975). Sightings of individuals caught in purse-seine nets suggest that schools are subdivided into smaller groups of female-calf pairs, mature males, or juveniles (Skinner & Chimimba 2005). In the eastern Pacific, S. attenuata have been recorded in association with S. longirostris (Hohn & Scott 1983). In the western Atlantic, S. attenuata occur in waters beyond the continental shelf in depths of more than 850 m (Moreno et al. 2005). The stomach contents from six South African S. attenuata specimens contained 68.6% fish and 31.4% cephalopods (Best 2007). Lantern fish (Symbolophorus 25.1%, Myctophum 16.5%, Ceratoscopelus 6.9% and Diaphus sp. 6.1%) were numerically the most abundant prey items, followed by the oceanic squids Chiroteuthis (12.5%) and Stenoteuthis (6.6%) (Best 2007; Ross 1984).
Similarly, Sekiguchi et al. (1992) identified ten species of cephalopods (primarily Sthenoteuthis spp., Chiroteuthis spp. and Todaropsis eblanae) and six species of fish from three Spotted Dolphin stomachs along South Africa’s coast. In the eastern Pacific Spotted Dolphins consumed both epipelagic species, such as flying fish (Oxyporhampus spp.), and mesopelagic species, for example Benthosema spp. (Fitch & Brownell 1968).
The Pantropical Spotted Dolphin reaches lengths of about 2 m, and adults weigh approximately 114 kg (Reference?). In the subregion, the largest individuals measured were a 2.25m male and a 2.15m female (n=14) (Best 2007). The length: weight relationship for 11 animals from the subregion would suggest that a 2.2m animal would weigh 93kg (Best 2007). Pantropical Spotted Dolphins are born without spots, acquiring them as they grow, until they are entirely covered with overlapping markings. Although, there is some evidence of seasonality in mating and calving in some regions (for example, off Japan: Kasuya et al. 1974, and in the eastern Pacific: Barlow 1984), this is not the case for all other regions. They experience gestation and lactation periods of 11–12 months (Kasuya et al. 1974; Perrin et al. 1976) and 20–24 months (Kasuya 1985; Myrick et al. 1986), respectively, and sexual maturity is reached at body lengths of approximately 1.82 m in females and 1.94 m in males (Skinner & Chimimba 2005).
Ecosystem and cultural services: Bycatch mitigation measures to reduce Spotted Dolphin entanglement in fishing gear in the Pacific led to the phrase “Dolphin friendly tuna”.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
There is no trade of these species within South Africa, although in certain regions Stenella spp. are hunted for food and as bait for fisheries.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: Yes
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The offshore distribution of Stenella species within the region suggests that industrial activity is not a major threat, additionally MacLeod (2009) predicted that due to the tropical distribution of S. attenuata, it is unlikely that this species would be negatively influenced by the effects of climate change. However, although it is largely undocumented, due to their diet and distribution, localised threats to these species include accidental bycatch and competition for prey resources associated with pelagic fisheries. Although within the assessment region there appears to be no major overlap between this species and major fishing operations, the Pantropical Spotted Dolphin in the eastern Pacific is severely threatened by tuna-seine fisheries. In fact, in 1972 approximately 270,000 Spotted Dolphins were incidentally killed due to accidental bycatch (Perrin et al. 1982). Gear modifications, which enabled dolphins to escape more easily aided in the decline of bycatch, though mortalities of 55,000–60,000 individuals in 1987 (Hall & Boyer 1989) and 15,000–16,000 individuals in 1992 (Hall & Lennert 1997) were recorded. Additionally, elsewhere in the world (such as off Japan), this species is directly targeted in drive fisheries; a catch of 4,184 individuals was recorded in 1978 (Miyazaki 1983).
A few individuals are known to have been entangled in the shark nets off KwaZulu-Natal (Best 2007). In addition, a low proportion (4%) of the dolphins taken in gill nets off Zanzibar were S. attenuata (Amir et al. 2005). The species has also been documented to be affected by ship strikes, although this appears to be rare (Schoeman et al. 2020).
Current habitat trend: Declining, due to increasing competition with pelagic fisheries for prey resources.
Conservation
No species-specific conservation initiatives have been identified for Stenella species within the assessment region, although S. attenuata, S. coeruleoalba and S. longirostris are all listed in Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), and are protected by national legislation under the Marine Living Resources Act (No. 18 of 1998).
Ship-based sighting surveys are recommended for the assessment region to determine general abundance, seasonality and distribution of Stenella species, and the pelagic zone of the temperate Agulhas Current should be specifically targeted. Additionally, investigations into the severity of threats and possible mitigation measures (including the use of alternative fishing gear technologies) associated with pelagic trawl, gillnet and purse-seine fisheries are required for these species. In response to the negative effects associated with fisheries bycatch of other dolphin species, Buscaino et al. (2009) suggest a collaborative response towards sustainable exploitation of oceanic resources, a decrease in the intensity of marine extraction and the establishment of protected areas.
Recommendations for managers and practitioners:
- Accurate bycatch assessments in the pelagic trawl, gillnet and purse-seine fisheries.
- Enforce regulations associated with deep water fisheries, including bycatch mitigation efforts.
- Sightings data should be recorded during systematic monitoring of other marine species.
Research priorities:
- Basic life history parameters, population size, structure and trends within the assessment region. Bycatch assessments in pelagic fisheries, including a specific reassessment of the western Indian Ocean fisheries.
- Taxonomic resolution of the Stenella genus.
- Distribution and the identification of core concentration regions of these species within South African waters.
- The impact of climate change on Spotted Dolphins.
Encouraged citizen actions:
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, e.g. wwfsa.mobi, FishMS 0794998795.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution.
- Report any stranding reports to the relevant local authorities
Bibliography
Amir, O. A., Jiddawi, N. S., and Berggren, P. 2005. The occurrence and distribution of dolphins in Zanzibar, Tanzania, with comments on the differences between two species of Tursiops. Western Indian Ocean Journal of Marine Science 4(1):85-93.
Archer, F. I. 2002. Striped dolphin Stenella coeruleoalba. In: W. F. Perrin, B. Wersig and J. G. M. Thewissen (eds), Encyclopedia of marine mammals, pp. 1201-1203. Academic Press.
Archer, F. I. and Perrin, W. F. 1999. Stenella coeruleoalba. Mammalian Species 603: 1-9.
Barlow, J. 1984. Reproductive seasonality in pelagic dolphins (Stenella spp.): implications for measuring rates. Report of the International Whaling Commission (Special Issue) 6: 191-198.
Best, P. B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town. 338pp.
Buscaino, G., Buffa, G., Sara, G., Bellante, A., Tonello, Jr. A.J., Hardt, F.A.S., Cremer, M.J., Bonanno, A., Cuttitta, A. and Mazzola, S. 2009. Pinger affects fish catch efficiency and damage to bottom gill nets related to bottlenose dolphins. Fisheries Science 75: 537-544.
Cockcroft, V. G. and Krohn, R. 1994. Passive gear fisheries of the southwestern Indian and southeastern Atlantic oceans: an assessment of their possible impact on cetaceans. Reports of the International Whaling Commission, Special Issue 15: 317-328.
DeMaster, D. P., C. W. Fowler, S. L. Perry and M. F. Richlen. 2001. Predation and competition: the impact of fisheries on marine mammal populations over the next one hundred years. Journal of Mammalogy 82(3): 641-651.
Dolar, M. L. L., Walker, W. A., Kooyman, G. L. and Perrin, W. F. 2003. Comparative feeding ecology of spinner dolphins (Stenella longirostris) and Fraser’s dolphins (Lagenodelphis hosei) in the Sulu Sea. Marine Mammal Science 19(1): 1-19.
Donahue, M. A. and Edwards, E. F. 1996. An annotated bibliography of available literature regarding cetacean interactions with tuna purse seine fisheries outside of the eastern tropical Pacific. Southwest Fisheries Science Center Administrative Report 96-20: 46 pp.
Findlay KP. 1989. The distribution of cetaceans off the coast of South Africa and South West Africa/Namibia. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Fitch JE, Brownell RL. 1968. Fish otoliths in cetacean stomachs and their importance in interpreting feeding habits. Journal of the Fisheries Board of Canada 25: 2561–2574.
Fraser FC, Noble BA. 1970. Variation of pigmentation in Meyer’s dolphin, Stenella coeruleoalba (Meyer). Investigations on Cetacea 2: 147–164.
Gambell R, Best PB, Rice DW. 1975. Report on the International Indian Ocean whale marking cruise, 24th Nov 1973 – 3rd Feb 1974. Reports of the International Whaling Commission.
Hall MA, Boyer SD. 1989. Estimates of incidental mortality of dolphins in the eastern Pacific fishery for tropical tunas in 1987. Reports of the International Whaling Commission 39: 321–322.
Hall MA, Lennert C. 1997. Incidental mortality of dolphins in the eastern Pacific Ocean tuna fishery in 1995. Report of the International Whaling Commission 47: 641–644.
Hassani, S., Antoine, L. and Ridoux, V. 1997. Diets of albacore, Thunnus alalunga, and dolphins, Delphinus delphis and Stenella coeruleoalba, caught in the northeast Atlantic albacore drift-net fishery: a progress report. Journal of Northwest Atlantic Fishery Science 22: 119-124.
Hohn AA, Scott MD. 1983. Segregation by age and sex in schools of spotted dolphins in the eastern tropical Pacific. 5th Biennial Conference on the Biology of Marine Mammals.
IUCN (International Union for Conservation of Nature). 2012. Stenella attenuata. Available at: http://www.iucnredlist.org. (Accessed: 21 February 2016).
Kasuya T, Miyazaki N, Dawbin WH. 1974. Growth and reproduction of Stenella attenuata in the Pacific coast of Japan. Scientific Reports of the Whales Research Institute, Tokyo 26: 157–226.
Kasuya T, Miyazaki N. 1982. The stock of Stenella coeruleoalba off the Pacific coast of Japan. Mammals of the Seas. FAO Fish Ser. 5 4: 21–37.
Kasuya, T. 1985. Effect of exploitation on reproductive parameters of the spotted and striped dolphins off the Pacific coast of Japan. Scientific Reports of the Whales Research Institute 36: 107-138.
Kebke A, Samarra F, Derous D. 2022 Climate change and cetacean health: impacts and future directions. Phil. Trans. R. Soc. B 377: 20210249. https://doi.org/10.1098/rstb.2021.0249
Kroese M. 1993. Age, growth and reproductive biology of striped dolphins, Stenella coeruleoalba (Meyer, 1833) off the coast of southern Africa. M.Sc. Thesis. University of Port Elizabeth.
LeDuc, R.G., Perrin, W.F. and Dizon, A.E. 1999. Phylogenetic relationships among the delphinid cetaceans based on full cytochrome b sequences. Marine Mammal Science 15: 619-648.
MacLeod, C.D. 2009. Global climate change, range changes and potential implications for the conservation of marine cetaceans: a review and synthesis. Endangered Species Research 7: 125-136.
Mitchell E. 1970. Pigmentation pattern evolution in delphinid cetaceans: an essay in adaptive coloration. Canadian Journal of Zoology 48(717–740).
Miyazaki N. 1984. Further analyses of reproduction in the striped dolphin, Stenella coeruleoalba , off the Pacific coast of Japan. Reports of the International Whaling Commission (Special Issue) 6: 343–353.
Miyazaki, N. 1983. Catch statistics of small cetaceans taken in Japanese waters. Reports of the International Whaling Commission 33: 621-631.
Moreno, I. B., Zerbini, A. N. Danilewicz, D., Santos, M. O. S., Simoes-Lopes, P. C., Lailson Brito, J. and Azevedo, A. F. 2005. Distribution and habitat characteristics of dolphins of the genus Stenella (Cetacea: Delphinidae) in the southwest Atlantic Ocean. Marine Ecology Progress Series 300: 229-240.
Myrick A, Hohn AA, Barlow J, Sloan PA. 1986. Reproductive biology of female spotted dolphins, Stenella attenuata, from the eastern tropical Pacific. Fishery Bulletin U.S. 84: 247–259.
Norris KS, Würsig B, Wells RS, Würsig M, Brownlee SM, Johnson C, Solow J. 1985. The behaviour of the Hawaiian spinner dolphin, Stenella longirostris. Fishery Bulletin U.S. 77: 821–849.
Perrin WF, Coe JM, Zweifel JR. 1976. Growth and reproduction of the spotted porpoise, Stenella attenuata, in the offshore eastern tropical Pacific. Fishery Bulletin US 74: 229–269.
Perrin WF, Henderson JR. 1984. Growth and reproductive rates in two populations of spinner dolphins, Stenella longirostris, with different histories of exploitation. Reports of the International Whaling Commission (Special issue) 6: 417–430.
Perrin WF, Würsig B, Thewissen JGM. 2002. Encyclopedia of Marine Mammals. Academic Press, San Diego, California, USA.
Perrin, W. F. and Gilpatrick, J. W. 1994. Spinner dolphin Stenella longirostris (Gray, 1828). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Volume 5: The first book of dolphins, pp. 99-128. Academic Press.
Perrin, W. F., Wilson, C. E. and Archer, F. I. 1994. Striped dolphin Stenella coeruleoalba (Meyen, 1833). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Volume 5: The first book of dolphins, pp. 129-159. Academic Press.
Perrin, W.F., Smith, T.G. and Sakagawa, G.T. 1982. Status of populations of spotted dolphin, Stenella attenuata, and spinner dolphins, Stenella longirostris, in the eastern tropical Pacific. Mammals of the Seas. FAO Fish Ser.5(4): 67–83.
Perrin, W.F., Warner, R.R., Fiscus, C.H. and Holts, D.B. 1973. Stomach contents of porpoise, Stenella spp., and yellowfin tuna, Thunnus albacares, in mixed-species aggregations. Fishery Bulletin 71: 1077-1092.
Reeves, R.R., McClellan, K. and Werner, T.B. 2013. Marine mammal bycatch in gillnet and other entangling net fisheries, 1990 to 2011. Endangered Species Research 20: 71-97.
Ross, G. J. B. 1984. The smaller cetaceans of the south east coast of southern Africa. Annals of the Cape Provincial Museums (Natural History) 15: 173-410.
Schoeman, R. P., Patterson-Abrolat, C., & Plön, S. (2020). A global review of vessel collisions with marine animals. Frontiers in Marine Science. https://doi.org/doi: 10.3389/fmars.2020.00292
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Van Waerebeek, K., Gallagher, M., Baldwin, R., Papastavrou, V. and Al-Lawati-Samira, M. 1999. Morphology and distribution of the spinner dolphin, Stenalla longirostris, rough-toothed dolphin, Steno bredanensis, and melon-headed whale, Peponocephala electra, from waters off the Sultanate of Oman. Journal of Cetacean Research and Management 1(2): 167-177.
Würtz, M. and Marrale, D. 1993. Food of striped dolphin, Stenella coeruleoalba, in the Ligurian Sea. Journal of the Marine Biological Association of the United Kingdom 73: 571-578.
