Pangolin

2025 Red list status
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Smutsia temminckii – (Smuts, 1832)
ANIMALIA – CHORDATA – MAMMALIA – PHOLIDOTA – MANIDAE – Smutsia – temminckii
Common Names: Temminck’s Pangolin, Temminck’s Ground Pangolin, Ground Pangolin, Cape Pangolin, Steppe Pangolin, Scaly Anteater, South African Pangolin (English), Ietermagog, Ystermagog (Afrikaans), Inkakha (isiNdebele), iMfinyezi (isiZulu, siSwati), Kgaga (Sepedi, Sotho, Tswana), Khwara (Venda), Ngaka (Chichewa, Chewa, Nyanja), xiKhwarhu (Tsonga), Hambakubvu, Haka (Shona), Thagadu (Pedi), Shikwaru (Shagaan), Pangolin terrestre du Cap (French), Pangolin de Temminck (French), Pangolín del Cabo (Spanish; Castilian)
Synonyms: Manis temminckii Smuts, 1832
Taxonomic Note:
Historically included in the genus Manis (with Smutsia usually considered a subgenus) and referred to Phataginus by Grubb et al. (1998), but here included in the genus Smutsia, along with the Giant Pangolin (S. gigantea) following Gaudin et al. (2009). This grouping is supported by subsequent genetic analyses (du Toit et al. 2017; Gaubert et al. 2018; Heighton et al. 2023). The preferred common name (as accepted by the IUCN Species Survival Commission Pangolin Specialist Group) is Temminck’s Pangolin. No subspecies are recognised.
Red List Status: VU – Vulnerable, A4cd (IUCN version 3.1)
Assessment Information
Assessors: Pietersen, D.W.,1 Jansen, R.2, Panaino, W.3, Swart, J.,4 da Silva, J.M.,5 & Power, R.J.6
Reviewers: Heighton, S.7 & Atkinson, H.8
Previous contributors: Kotze, A., Selier, J., Hoffman, T.S., Hoffmann, M., Williams, V.L. & Child, M.
Institutions:
1Endangered Wildlife Trust & Mammal Research Institute, 2Department of Environmental, Water and Earth Sciences, Tshwane University of Technology, 3School of Animal, Plant and Environmental Sciences, University of the Witwatersrand & Brain Function Research Group, School of Physiology, University of the Witwatersrand, 4Welgevonden Game Reserve, 5South African National Biodiversity Institute, 6Directorate of Biodiversity Management, Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, 7IUCN Species Survival Commission Pangolin Specialist Group & Zoological Society of London, 8Peace Parks Foundation
Assessment Rationale
Temminck’s Pangolin is widely distributed across the savannah regions of northern and eastern South Africa, occurring in both protected areas and untransformed habitat outside of protected areas. This species is severely threatened by electrified fences, with an estimated 377–1,028 individuals electrocuted / year on game farm fences alone (Pietersen et al. 2016a), and the actual number probably approaching 2,000 individuals per year when electrified livestock fences are included (DW Pietersen, unpubl. data December 2023). Illegal harvesting is the next biggest threat, with statistics for the last decade indicating that about 50 Temminck’s Pangolins are rescued from the illegal wildlife trade in South Africa annually (R. Jansen, unpubl. data March 2024). At least some of these individuals are illegally imported from neighbouring countries while not all local poaching cases are detected, and it is estimated that ~100 individuals are poached annually in South Africa. Road collisions have been estimated to kill 280 individuals per year (Pietersen et al. 2016a), but this is almost certainly an over-estimate. In the Northern Cape province, a few individuals die annually as a result of being caught in illegal gin traps. The Extent of Occurrence has remained relatively stable over the past 39 years (1985–2024). Previous reports of this species going locally extinct in the Eastern Cape and Free State provinces (Pietersen et al. 2016a) were erroneous, and as a result, habitat loss was overestimated in the previous Regional Red List assessment. This species never occurred in the Eastern Cape province, while it always only marginally occurred in the northern Free State province (and still does; Pietersen et al. 2021). The subpopulation in KwaZulu-Natal province has decreased in the past three decades, presumably due to overexploitation. Data from the past 30 years (1990–2020) suggests that about 23% of the remaining natural habitat within this species’ range has been lost during this time period (Department of Forestry, Fisheries and the Environment 2024), primarily as a result of mining, agriculture and urban expansion.
This is a long-lived species with low reproductive output that is likely affected by the loss of mature individuals. In addition to the above threats, climate change also appears to be having a negative impact on the species, particularly in the far-western portion of its geographic range, with anecdotal evidence supporting published studies (Panaino et al. 2023) that Temminck’s Pangolin numbers have drastically declined in the Kalahari region following the 2014–2020 drought. With droughts predicted to increase in frequency and intensity with climate change (Bernstein et al. 2007; Davis-Reddy et al. 2020), this threat is likely to increase in the coming decades and may also affect a greater portion of this species’ range than currently assumed. The combined effects of mortalities on electrified fences, levels of illegal harvest, and more frequent and prolonged droughts, are believed to be resulting in a declining population size, with this decline believed to reach or exceed 30% over the next 27 years (three generations). Thus, we list this species as Vulnerable under Criterion A4cd.
Regional population effects: Dispersal probably occurs across borders in both directions, based on mitochondrial DNA analyses (du Toit 2014, 2021), but it is not suspected to be enough to increase the size of the local subpopulation. With the dramatic decline in the Asian pangolin populations and increased difficulties in securing pangolins in the Asian range states, trade has increasingly shifted to Africa (Challender & Hywood 2012; Heinrich et al. 2017; Ingram et al. 2018; Tinsman et al. 2023). Although other African countries are bearing the brunt of this increased trade at present, it is possible that this illicit trade will increasingly affect the South African subpopulation as well.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change.
Red List Index
Red List Index: (Not specified)
Recommended citation: Pietersen DW, Jansen R, Panaino W, Swart J, da Silva JM & Power RJ. 2025. A conservation assessment of Smutsia temminckii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
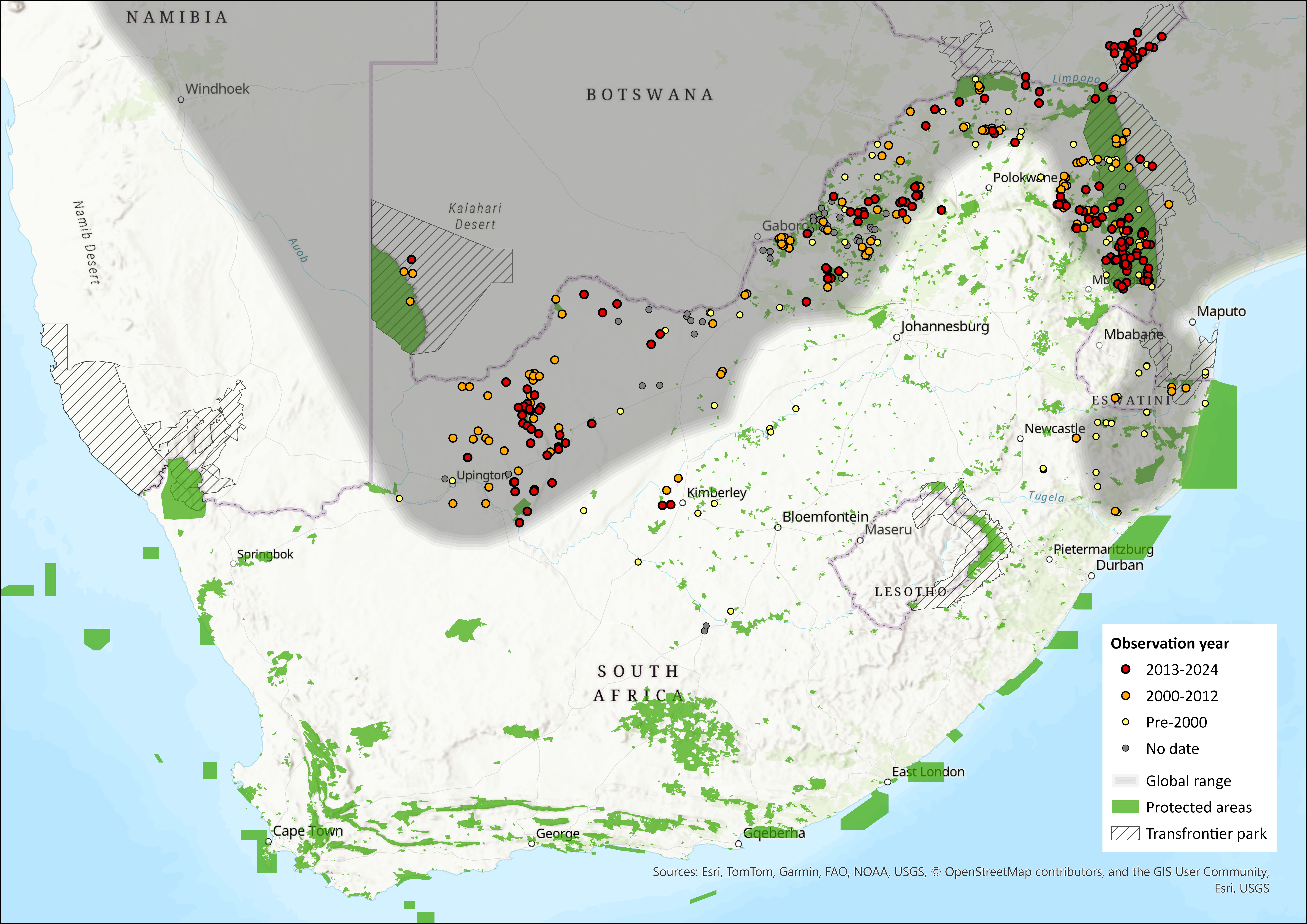
Regional Distribution and occurrence
Geographic Range
This is the most widespread of the four African pangolin species, occurring from the northern parts of South Africa northwards through East Africa, reaching its northern limits in northeastern Chad (Malbrant 1952) and central Sudan (Sweeney 1956, 1974; Pietersen et al. 2020), although its northern limits are not well defined.
Within the assessment region, it occurs in the Northern Cape, North West, Free State, Limpopo, Mpumalanga and KwaZulu-Natal provinces (Pietersen et al. 2021). However, the Free State province is unlikely to have ever been a stronghold for this species, and it was likely always restricted to protrusions of Kalahari thornveld and shrub bushveld in the north of this province (Lynch 1983; Pietersen et al. 2021). Although this species was historically recorded from the Eastern Cape province (Lloyd & Millar 1983; Lynch 1989), these records refer to misidentifications or confusion with the vernacular names of other myrmecophagous species (Stuart 1980, Pietersen et al. 2021). In KwaZulu-Natal province, the subpopulation appears to be very small and currently restricted to the extreme north (Kyle 2000; Pietersen et al. 2016a). Historically, KwaZulu-Natal province almost certainly had a larger subpopulation, but it is believed that persecution for muthi (traditional medicine), and to a lesser extent habitat loss, has resulted in a population decline (Kyle 2000; Ngwenya 2001; Pietersen et al. 2014a).
Records from Eswatini are scant, despite the eastern portion of the country harbouring apparently suitable habitat. There are records from neighbouring regions of South Africa, however, and individuals almost certainly disperse into eSwatini if there is not a resident subpopulation there. There are no known major barriers or discontinuities in its South African range, and even the Limpopo River does not appear to act as a barrier to this species (du Toit 2014; 2021).
The extent of occurrence (EOO) in South Africa and eSwatini is approximately 644,121 km2, with a high confidence level, and is based on the known distribution of accepted recent records combined with distribution modelling (Pietersen et al. 2021).
Map Status
Figure 1. Distribution records for Pangolini (Smutsia temminckii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality | ||||||||
| Angola | Extant | Native | – | – | ||||||||
| Botswana | Extant | Native | – | – | ||||||||
| Burundi | Extant | Native | – | – | ||||||||
| Central African Republic | Extant | Native | – | – | ||||||||
| Chad | Extant | Native | – | – | ||||||||
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | – | ||||||||
| Eswatini | Possibly Extinct | Native | – | – | ||||||||
| Ethiopia | Presence Uncertain | Native | – | – | ||||||||
| Kenya | Extant | Native | – | – | ||||||||
| Malawi | Extant | Native | – | – | ||||||||
| Mozambique | Extant | Native | – | – | ||||||||
| Namibia | Extant | Native | – | – | ||||||||
| Rwanda | Presence Uncertain | Native | – | – | ||||||||
| South Africa | Extant | Native | – | – | ||||||||
| South Sudan | Extant | Native | – | – | ||||||||
| Sudan | Extant | Native | – | – | ||||||||
| Tanzania, United Republic of | Extant | Native | – | – | ||||||||
| Uganda | Extant | Native | – | – | ||||||||
| Zambia | Extant | Native | – | – | ||||||||
| Zimbabwe | Extant | Native | – | – | ||||||||
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Recent studies indicate that Temminck’s Pangolin is negatively impacted by climate change through increased heat burdens and concomitant decreases in prey availability resulting in nutritional stress (Panaino et al. 2023). Pangolin scales have poor thermoregulatory abilities (McNab 1984; Heath & Hammel 1986; Weber et al. 1986; Pietersen 2013; Panaino et al. 2023), thus reducing their ability to buffer against extreme environmental temperatures. High mortality rates and overall subpopulation reductions were noted in the more arid western portions of this species’ geographical range in South Africa (W. Panaino, pers. obs. 2023, M. van Niekerk, pers. comm. July 2023), as well as in Namibia (K. Prediger, unpubl. data) following a prolonged drought (2014–2020). With the frequency and intensity of droughts predicted to increase as global temperatures rise (Bernstein et al. 2007; Davis-Reddy et al. 2020), climate change may pose a significant additional threat to this species, at least in the more arid portions of its geographical range. Intact ecosystems were associated with lower drought-induced mortality rates of Temminck’s Pangolin than degraded ecosystems (W. Panaino, pers. obs. 2023), suggesting that the former could be more resilient and may better buffer the impacts of climate change.
Population information
This species is widely distributed in northern South Africa, although now largely confined to protected areas and well-managed livestock and wildlife farms (Swart 1996; Pietersen et al. 2016a, 2021). Given their predominantly nocturnal and secretive nature, their abundance is probably underestimated. The total density in the greater Kruger National Park has been estimated at 24 individuals / 100 km² and 12 reproductively active individuals / 100 km², while total density in western Zimbabwe has been estimated at 11 individuals / 100 km2 (Swart 2013; Pietersen et al. 2014b). A study in the Kalahari region near the western-most extent of this species’ range estimated a total density of 23–31 individuals / 100 km2 and a breeding density of 16 individuals / 100 km2 (Pietersen et al. 2014b).
The South African subpopulation is inferred and suspected to be declining, predominantly owing to the combined effects of electrocution on electrified fences, illegal harvesting, habitat conversion and the impacts of climate change in portions of its geographical range. Despite the low prevalence of this species in urban traditional medicine markets (Whiting et al. 2011) and the slow turnover rates of these specimens (R. Jansen., unpubl. data), rural communities have reported an overall decrease in the abundance of this species (Baiyewu et al. 2018), supporting the inferred subpopulation decline.
Female Temminck’s Pangolin become reproductively mature in their second year but are unlikely to reproduce before they are 3–4 years old (Pietersen et al. 2014b). It is believed that males reproduce for the first time when they are 6–7 years old, although they probably reach sexual maturity before this (Pietersen et al. 2014b). Gestation lasts 105–140 days (van Ee 1966; Swart 2013; Pietersen et al. 2020). Females may only produce or successfully raise a single offspring every second year (Pietersen et al. 2020). The pup starts riding on the mother’s tail when about one month old, and becomes independent from 3–4 months (Swart 2013; Pietersen et al. 2020). Pups are suspected to initially suffer high mortality rates due to starvation and hypothermia (Pietersen 2013), and possibly disease. Additionally, young animals (up to c. 1–2 years old) fall prey to a variety of predators as their scales are still comparatively soft and their smaller size makes them more susceptible to predation. Young, dispersing individuals are also likely to encounter a greater number and variety of natural and anthropogenic threats, and are thus suspected to suffer comparatively higher mortality rates than adults (Pietersen et al. 2014a). Because males become reproductively active later than females and thus cover larger distances before establishing a home range (Pietersen et al. 2014b), males face a greater risk from anthropogenic threats than do females (Pietersen et al. 2014a).
There is a paucity of data on the longevity of any pangolin species in the wild, making estimates of generation length difficult. Longevity estimates are based on data from captive individuals (Hoyt 1987), combined with field observations (DW Pietersen, unpubl. data). Based on available growth rates, field observations, the relatively late onset of the start of reproduction, the slow reproductive rate, and longevity of the sympatric Aardvark (Orycteropus afer), which has a similar ecology and life history (Skinner & Chimimba 2005), Temminck’s Pangolin is expected to be long-lived, surviving for 20 years or more in the wild.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | Inferred | Inferred and suspected based on the high mortality rates on electrified fences, levels of illegal harvest, habitat loss and the impacts of climate change which is affecting a portion of the subpopulation. |
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Reference mitogenomes and complete nuclear genomes are available for all eight extant pangolin species, including for Temminck’s Pangolin (du Toit et al. 2017; Gaubert et al. 2018; Heighton et al. 2023). Genomic data has assisted in providing a better understanding of the evolution of pangolins (Choo et al. 2016; Heighton et al. 2023), which will be essential for future conservation genetic studies delineating genetic conservation units, identifying adaptive potential in the face of global future change, and origin tracing of traded individuals (du Toit et al. 2017, Hu et al. 2020). With that said, species-level identification of unknown scales has proven reliable for forensic purposes at a broad scale (Mwale et al. 2017; Tinsman et al. 2023). A preliminary study using mitochondrial DNA and short tandem repeats (microsatellites) detected no intra-specific geographic structure within this species’ southern African range (du Toit 2021), suggesting that there may still be gene flow between sampling sites and that the southern African subpopulation may be a continuous metapopulation. However, morphometric differences have been detected between pangolins from arid and mesic areas (Pietersen et al. 2020), suggesting some level of differentiation within the species between these two ecological areas (see Power & Olivier 2019). Moreover, the sampling in the du Toit (2021) study was limited, and the provenance of some samples unclear, indicating that caution should be taken when interpreting these findings.
With pangolin genomes having been sequenced (e.g., Gaubert et al. 2018; Heighton et al. 2023, Yan et al. 2023), studies re-examining the genetic structure within this species would be valuable to ascertain whether the genetic markers used to date (i.e., in du Toit 2021) are robust enough to detect fine-scale population structure. Single nucleotide polymorphism markers sampled from across the genome might be better able to detect whether there are adaptive/functional associations to the observed morphological differences, which would have implications for managing confiscated individuals (Heighton et al. 2023). Indeed, recent molecular work using next-generation sequencing is showing some genetic differences between different pangolin subpopulations in South Africa (S. Heighton, unpubl. data). Until such time as the South African subpopulation is studied in detail with suitably sensitive genetic markers, a precautionary approach should be applied and confiscated pangolins should be released as close to their origin as is feasible.
Habitats and ecology
A predominantly solitary, terrestrial species that occurs in arid and mesic savannah, woodlands and floodplains at lower elevations, with annual rainfall between 250 and 1,400 mm (Coulson 1989; Heath & Coulson 1997; Skinner & Chimimba 2005; Swart, 2013; Pietersen et al. 2016a). They are largely absent from Karroid regions, tropical forests, Highveld grassland and coastal regions (Pietersen et al. 2021). The range is believed to be largely determined by habitat and the presence and abundance of ant and termite prey species. It inhabits well-managed livestock and wildlife farms and other areas with natural vegetation, but is absent from areas under crop farming.
This species is predominantly nocturnal, with individuals spending the day in suitable earthen burrows dug by other species, caves or in piles of plant debris (Jacobsen et al. 1991; Heath & Coulson 1997; Pietersen et al. 2014b). Young animals are more diurnal than adults, while all age classes may become more diurnal during winter and times of food shortages.
Temminck’s Pangolin is bipedal, walking on its hind legs with the front limbs and tail held off the ground and acting as counterweights. However, when they climb steep terrain (such as steep sand dunes) or when clambering over boulders in rocky terrain, they will use all four limbs and may additionally use their tails to push themselves up or as a support when clambering down steep surfaces. They occasionally also climb onto fallen stumps and the lower portions of trees to access prey. They are entirely myrmecophagous and only eat a small number of specific ant and termite genera (Jacobsen et al. 1991; Swart 1996; Richer et al. 1997; Swart et al. 1999; Pietersen et al. 2016b; Panaino et al. 2022; Pietersen & Robertson 2023). They are largely water independent but will drink from free-standing water when it is available (Stuart 1980; Pietersen et al. 2020). Pups start dispersing when about one year old (Pietersen et al. 2014b).
In the Kalahari, home ranges of adults averaged 6.5 ± 5.9 km², while juveniles had average home ranges of 6.1 ± 4.0 km² (Pietersen et al. 2014b). In eastern South Africa, home ranges averaged 1.3–7.9 km² (van Aarde et al. 1990), while in Zimbabwe home ranges averaged 0.2–23.4 km2 (Heath & Coulson 1997).
Ecosystem and cultural services: Pangolins feed exclusively on ants and termites (Jacobsen et al. 1991; Swart 1996; Richer et al. 1997; Swart et al. 1999; Pietersen et al. 2016b; Panaino et al. 2022; Pietersen & Robertson 2023) and are thus important predators of these invertebrate taxa. It is estimated that a single pangolin consumes millions of ants and termites each year (Tang et al. 2012). Pangolin body parts are used to treat and cure numerous ailments and are still regularly prescribed and highly sought after by traditional healers within rural South African tribal communities (Bräutigam et al. 1994; Baiyewu et al. 2018).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | No |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | Yes |
| 2.2. Savanna -> Moist | – | Suitable | Yes |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | No |
Life History
| Generation Length | Justification | Data Quality |
| 9 | – | – |
Systems
System: Terrestrial
General Use and Trade Information
Temminck’s Pangolin is used at the local, national and international level for food, medicine and cultural ceremonies. The scales are used at local and international levels for ornaments and talismans (Kyle 2000; Manwa & Ndamba 2011; Baiyewu et al. 2018).
In South Africa, the pangolin is a revered animal and is not referred to lightly or for amusement. The amaZulu believe that seeing a pangolin indicates that there will be a drought, and the only way to prevent the drought is by killing the animal (Kyle 2000). The VhaVenda and Batswana tribes have special songs that they sing when the animal is sighted, and whoever initially spots the creature is accorded special treatment by the chief and tribesmen. Temminck’s Pangolin is generally believed to have connections to thunder and lightning strikes, and body parts are primarily used in physical and spiritual fortification processes to guard against evil, in love charms, and in cleansing rituals (Baiyewu et al. 2018).
Local trade levels have been steadily increasing year on year and more evidence has arisen of syndicated involvement as prices and demand continue to increase (R. Jansen, unpubl. data). International trade has been increasing across Africa (Challender & Hywood 2012; Heinrich et al. 2017; Ingram et al. 2018; Tinsman et al. 2023) and may affect the local subpopulation in the future as well. Data from the past decade indicate that around 50 Temminck’s Pangolin are rescued from the illegal wildlife trade in South Africa annually (R. Jansen, unpubl. data), although at least some of these individuals originate in neighbouring countries and are smuggled into South Africa. At least some local poaching cases undoubtedly go undetected, however, and it is estimated that the annual illegal trade in South Africa is approximately 100 individuals.
With increased convictions and extensive jail terms of up to 10 years being imposed on convicted persons in South Africa, it has become increasingly difficult to apprehend suspects involved in the illegal trade of Temminck’s Pangolin. Both the judiciary and the police service have become more familiar in dealing with pangolin-related cases and it is now considered a priority crime in the country, with operations often mandated to the Directorate for Priority Crimes Investigation (HAWKS).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Bushmeat and traditional medicine | Yes | Domestic and international trade |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 3. Medicine – human & veterinary | true | true | true | – |
| 10. Wearing apparel, accessories | true | true | true | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Pangolins cannot be maintained or successfully bred in captivity at a commercially viable scale (Challender et al. 2019).
Threats
Temminck’s Pangolin occur in numerous national parks, nature reserves and other protected areas within the assessment region, including greater Limpopo Transfrontier Park and Kgalagadi Transfrontier Park, and is protected by national law.
Major threats to this species within the assessment region are:
- Electrified fences pose the greatest threat to this species nationally, with recorded mortality rates of 3–9 individuals / 100 km of electrified fence / year (Beck 2009; Pietersen et al. 2014a; Pietersen 2022). This translates to an estimated 377–1,028 individuals electrocuted on game farm fences annually (Pietersen et al. 2016a). This estimate does not take into account electrified livestock fences, despite there being an increasing tendency for livestock farmers to place electrified strands on their livestock fences to control the movements of livestock predators (Beck 2009; Pietersen et al. 2014a; Pietersen 2022). Thus, the actual levels of mortality are likely to be higher than these figures, with an industry-wide estimate (informed from decades of experience) of around 2,000 mortalities per year.
- Habitat Loss. Land use change data between 1990 and 2020 suggest that around 23% of the remaining natural vegetation within Temminck’s Pangolin range has been lost during this time period (Department of Forestry, Fisheries and the Environment 2024), primarily as a result of mining, agriculture and urban expansion. Although some habitat loss is ongoing, it is suspected that the rate of loss has decreased in recent years, although overall habitat loss and degradation should be continuously monitored, as should the impacts of this on the species.
- Exploitation of the species for the local traditional medicine (muthi) trade is known to be occurring in South Africa (Baiyewu et al. 2018). Harvesting rates have not been quantified, but research suggests that the turnover in traditional medicine markets is low, despite this species often being present in urban muthi markets (Whiting et al. 2011). At least some of the carcasses in the muthi trade likely originate from electrified fence or road mortalities. However, many rural communities, particularly in North West, Limpopo, Mpumalanga and KwaZulu-Natal provinces, have reported decreased abundance of Temminck’s Pangolin (Baiyewu et al. 2018), suggesting that exploitation of this species is a significant threat and that only a small minority of harvested individuals make it to the urban muthi This is supported by the increasing number of criminal cases involving Temminck’s Pangolin in South Africa.
Minor threats to this species within the assessment region are:
- There is a growing demand for African pangolins in the Asian markets owing to increasing affluence in Asian countries (Challender 2011), and sophisticated international networks (Heinrich et al. 2016), which is resulting in an increased poaching rate in Africa (Challender & Hywood 2012; Heinrich et al. 2017; Ingram et al. 2018; Tinsman et al. 2023). Although some information has been received of local harvesters selling pangolins to Asian nationals, this trade is believed to be limited at present. Considering the increasing trade elsewhere in Africa, however, we caution that both local and international trade should be closely monitored to detect any sudden change in trade dynamics that could indicate a shift of syndicates to southern Africa.
- Road mortalities. Pietersen et al. (2016a) estimated that there were 280 Temminck’s Pangolin deaths on roads every year. This is almost certainly an overestimate, however, due to the relatively small section of road that was monitored, the fairly short duration of the study, and this monitoring only taking place in a single ecotype of this species’ range. It is suspected that fewer than 100 individuals are killed on roads annually in South Africa. The exact extent of this threat is difficult to quantify, however, due to persons removing carcasses from roads for muthi and for the novelty factor (for example, having animals mounted for display).
- Gin traps. A minor, localised threat is individuals being caught in gin traps. This threat is restricted to areas where farming with small livestock and Temminck’s Pangolin distributions overlap (Pietersen et al. 2014a) and is largely restricted to the western extent of this species’ geographical range. If found in time, trapped individuals can usually be released unharmed. However, due to these traps being checked infrequently, a large proportion of the individuals caught die of exposure, while others are severely injured and die as a result of their injuries or have to be euthanized.
- Climate change. Increasing daytime temperatures are resulting in increased heat loads on Temminck’s Pangolin while simultaneously resulting in decreased food availability (Panaino et al. 2023). Pangolin scales are very poor thermal insulators (McNab 1984; Heath & Hammel 1986; Weber et al. 1986; Pietersen 2013; Panaino et al. 2023), thus resulting in very little buffering from these higher daytime temperatures. This emerging threat appears to be largely restricted to the western portions of this species’ geographical range in South Africa, however further studies are required to investigate the potential impacts and extent of this threat. Intact ecosystems appear to be more adept at buffering the effects of climate change (Panaino et al. 2023).
Conservation
Several actions can maintain or improve the conservation status of Temminck’s Pangolin in South Africa, and inform future conservation assessments for this species:
- The exact magnitude of fence electrocutions should be quantified, and interventions to reduce or prevent these electrocutions should be urgently sought. Long-term field data suggest that electrified wires placed at a minimum height of 300 mm above ground level reduce mortalities by up to 90%. This is an easy and effective conservation measure, although this will require changes in provincial legislation in some instances. Conservancy formation should be incentivised to reduce the need for internal electrified fences (sensu Lindsey et al. 2009; Pietersen 2022).
- Extensive strides have been made to inform the judiciary (state prosecutors and regional magistrates) and law enforcement officials in South Africa regarding the conservation status of Temminck’s Pangolin, and several severe incarceration judgements have been handed to perpetrators in recent years. This stems from providing species specialist statements in the docket and testimony in aggravation of sentencing in the courts around the country, in addition to advising on intelligence operations. This should be further expanded nationally, particularly to rural and often isolated magisterial jurisdictions where suspects are more frequently apprehended, such as rural regions of Limpopo province.
- The number of Temminck’s Pangolin that are being retrieved during police operations, together with the suspect’s information, is being recorded, but monitoring of long-term trends is required to detect any changes in trade dynamics that could signal an increase in illegal local offtake. The development and/or implementation of standard reporting and information sharing tools for law enforcement and custom officials, such as the Trade in Wildlife Information eXchange (TWIX) system, could ensure standardised reporting and that the recorded information is securely stored and shared between relevant local and international law enforcement agencies.
- Trade tracing and/or forensic tools should be developed to determine the source locations of confiscated Temminck’s Pangolin to inform both where to release rehabilitated individuals and to identify potential poaching hotspots.
- National and regional action plans that are created in unison with key government stakeholders should be developed and implemented.
- Cross-border partnerships in counter wildlife trafficking should be developed, and used to address the international trade in Temminck’s Pangolin.
Recommendations for land managers and practitioners:
- Temminck’s Pangolin sightings should be continually collected to monitor this species’ distribution and detect any range shifts caused by climate change or illicit trade dynamics.
- No captive-breeding or other ex situ management practices are required at present, nor is it likely to be a viable (or required) avenue in the foreseeable future. This species’ specialised diet and high mortality rate in captivity also preclude any large-scale ex-situ conservation programme.
- Alternative fence designs that maintain their effectiveness but reduce Temminck’s Pangolin mortalities should be developed and trialled. Once an effective solution to electrified fence mortalities is found, this should be adopted across this species’ geographical range.
Research priorities:
- Projects should be initiated to investigate subpopulation densities across this species’ geographical range, and to monitor subpopulation densities and distribution.
- The number of Temminck’s Pangolin killed on electrified fences each year needs to be quantified more accurately across this species’ range, and the effectiveness of interventions that can be applied to modify existing electrified fence structures should be tested.
- The Area of Occupancy needs to be accurately determined through further field surveys and ecological niche models. This, combined with further estimates of density across its range, will enable more accurate calculations of subpopulation size as a baseline for quantifying subpopulation trends.
- Quantifying the scale and impact of harvesting pressure from international and local trade, and relating this to its impact on the subpopulation, is a priority.
- Studies should be undertaken to assess what impact, if any, releasing rehabilitated pangolins into areas already containing natural subpopulations is having on the resident subpopulation. This should cover ecological, genetic and potential disease introduction aspects.
- A better understanding of the longevity of this species in the wild would help to inform a population viability analysis.
- Once the above data have been obtained or generated, a population viability analysis should be performed.
Encouraged citizen actions:
- Members of the public can assist with monitoring the distribution and relative abundance of Temminck’s Pangolin by submitting their sightings to online citizen science platforms such as iNaturalist or via the Pangolert portal on the Pangolin.Africa website.
- Members of the public should report any suspicious activity related to the trade of pangolins to their nearest South African Police Service Stock Theft and Endangered Species Unit or via the Pangolert portal.
- Citizens with game farms should use any fence modifications within legal specifications that are likely to reduce Temminck’s Pangolin mortalities, and could also report the number of Temminck’s Pangolin that are annually electrocuted on their farms to assist with quantifying this threat.
Bibliography
Baiyewu, A.O., Boakye, M.K., Kotze, A., Dalton, D.L. and Jansen, R. 2018. Ethnozoological survey of the traditional uses of Temminck’s Ground Pangolin (Smutsia temminckii) within South African tribal communities. Journal of Society & Animals 26(3): 306–325.
Beck, A. 2009. Electric fence induced mortality in South Africa. M.Sc. Thesis, University of the Witwatersrand, Johannesburg, South Africa.
Bernstein, L., Bosch, P., Canziani, O., Chen, Z., Christ, R., Davidson, O., Hare, W., Huq, S., Karoly, D., Kattsov, V., Kundzewicz, Z., Liu, J., Lohmann, U., Manning, M., Matsuno, T., Menne, B., Metz, B. Mirza, M., Nicholls, N., Nurse, L., Pachauri, R., Palutikof, J., Parry, M., Qin, D., Ravindranath, N., Reisinger, A., Ren, J., Riahi, K., Rosenzweig, C., Rusticucci, M., Schneider, S., Sokona, Y., Solomon, S., Stott, P., Stouffer, R., Sugiyama, T., Swart, R., Tirpak, D., Vogel, C. and Yohe, G. 2007. Climate Change 2007: Synthesis Report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. IPCC, Geneva, Switzerland, 104 pp.
Bräutigam, A., Howes, J., Humphreys, T. and Hutton, J. 1994. Recent information on the status and utilization of African pangolins. TRAFFIC Bulletin 15: 15–22.
Challender, D.W.S. 2011. Asian pangolins: Increasing affluence driving hunting pressure. TRAFFIC Bulletin 23(3): 92–93.
Challender, D.W.S. and Hywood, L. 2012. African pangolins under increased pressure from poaching and intercontinental trade. TRAFFIC Bulletin 24(2): 53–55.
Challender, D.W.S, ‘T Sas-Rolfes, M., Ades, G.W.J., Chin, J.S.C., Sun, N.C.M., Chong, J.L., Connelly, E., Hywood, L., Luz, S., Mohapatra, R.K., De Ornellas, P., Parker, K., Pietersen, D.W., Robertson, S.I., Semiadi, G., Shaw, D., Shepherd, C.R., Thomson, P., Wang, Y., Wicker, L., Wu, S.B. & Nash, H.C. 2019. Evaluating the feasibility of pangolin farming and its potential conservation impact. Global Ecology and Conservation 20: e00714.
Choo, S.W., Rayko, M., Tan, T.K., Hari, R., Komissarov, A., Wee, W.Y., Yurchenko, A.A., Kliver, S., Tamazian, G., Antunes, A. and Wilson, R.K., 2016. Pangolin genomes and the evolution of mammalian scales and immunity. Genome Research 26(10): 1312-1322.
Coulson, I. 1989. The Pangolin (Manis temmincki Smuts, 1835) in Zimbabwe. African Journal of Ecology 27: 149–155.
Davis-Reddy, C.L., Hilgart, A., Hlanane, K., Pienaar, M., Wilson, H. and Chiloane, L. 2020. South African Risk and Vulnerability Atlas. 3rd edition. Pretoria: SAEON. Available at sarva.saeon.ac.za. Department of Forestry, Fisheries and the Environment. 2024. South Africa National Land Cover Change Assessment Dataset 1990-2020.
du Toit Z. 2014. Population genetic structure of the Ground Pangolin based on mitochondrial genomes. M.Sc. Thesis. University of the Free State, Bloemfontein, South Africa.
du Toit, Z. 2021. Linking geographic origin and mitogenomes of the Ground Pangolin in southern Africa. Ph.D. Thesis. University of the Free State, Bloemfontein, South Africa.
du Toit, Z., du Plessis, M., Dalton, D.L., Jansen, R., Grobler, P.J. and Kotzé, A. 2017. Mitochondrial genomes of African pangolins and insights into evolutionary patterns and phylogeny of the family Manidae. BMC Genomics 18(1): 1–13.
Gaubert, P., Antunes, A., Meng, H., Miao, L., Peigné, S., Justy, F., Njiokou, F., Dufour, S., Danquah, E., Alahakoon, J., Verheyen, E., Stanley, W.T., O’Brien, S.J., Johnson, W.E. and Luo, S.-J. 2018. The complete phylogeny of pangolins: scaling up resources for the molecular tracing of the most trafficked mammals on Earth. Journal of Heredity 109(4): 347–359.
Gaudin, T.J., Emry, R.J. and Wible, J.R. 2009. The phylogeny of living and extinct pangolins (Mammalia, Pholidota) and associated taxa: a morphology based analysis. Journal of Mammalian Evolution 16: 235–305.
Grubb, P., Jones, T.S., Davies, A.G., Edberg, E., Starin, E.D. and Hill, J.E. 1998. Mammals of Ghana, Sierra Leone and The Gambia. Trendrine Press, Zennor, St Ives, Cornwall, UK.
Heath, M.E. and Coulson, I.M. 1997. Home range size and distribution in a wild population of Cape Pangolins, Manis temminckii, in north‐west Zimbabwe. African Journal of Ecology 35(2): 94–109.
Heath, M.E. and Hammel, H.T. 1986. Body temperature and rate of O2 consumption in Chinese Pangolins. American Journal of Physiology – Regulatory, Integrative and Comparative Physiology 250: 377–382.
Heighton, S.P., Allio, R., Murienne, J., Salmona, J., Meng, H., Scornavacca, C., Bastos, A.D.S., Njiokou, F., Pietersen, D.W., Tilak, M.-K. Luo, S.-J., Delsuc, F. and Gaubert, P. 2023. Pangolin genomes offer key insights and resources for the world’s most trafficked wild mammals. Molecular Biology and Evolution 40(10): msad190.
Heinrich, S., Wittmann, T.A., Prowse, T.A.A., Ross, J.V., Delean, S., Shepherd, C.R. and Cassey, P. 2016. Where did all the pangolins go? International CITES trade in pangolin species. Global Ecology and Conservation 8: 241–253.
Heinrich, S., Wittman, T.A., Ross, J.V., Shepherd, C.R., Challender, D.W.S., Cassey, P. 2017. The global trafficking of pangolins: a comprehensive summary of seizures and trafficking routes from 2010–2015. TRAFFIC, Southeast Asia Regional Office, Petaling Jaya, Selangor, Malaysia.
Hoyt, R. 1987. Pangolins: past, present and future. Proceedings of the AAZPA National Conference: 107–134.
Hu, J.Y., Hao, Z.Q., Frantz, L., Wu, S.F., Chen, W., Jiang, Y.F., Wu, H., Kuang, W.M., Li, H., Zhang, Y.P. and Yu, L., 2020. Genomic consequences of population decline in critically endangered pangolins and their demographic histories. National Science Review 7(4): 798-814.
Ingram, D.J., Coad, L., Abernethy, K.A., Maisels, F., Stokes, E.J., Bobo, K.S., Breuer, T., Gandiwa, E., Ghiurghi, A., Greengrass, E., Holmern, T., Kamgaing, T.O.W., Ndong Obiang, A.-M., Poulsen, J.R., Schleicher, J., Nielsen, M.R., Solly, H., Vath, C.L., Waltert, M., Whitham, C.E.L., Wilkie, D.S. and Scharlemann, J.P.W. 2018. Assessing Africa-wide pangolin exploitation by scaling local data. Conservation Letters 11 (2): e12389.
Jacobsen, N.H.G., Newbery, R.E., de Wet, M.J., Viljoen, P.C. and Pietersen, E. 1991. A contribution of the ecology of the Steppe Pangolin Manis temminckii in the Transvaal. Zeitschrift für Säugetierkunde 56(2): 94–100.
Kyle, R. 2000. Some notes on the occurrence and conservation status of Manis temminckii, the Pangolin, in Maputaland, KwaZulu-Natal. Koedoe 43: 97–98.
Lindsey, P.A., Romanach, S.S. and Davies-Mostert, H.T. 2009. The importance of conservancies for enhancing the value of game ranch land for large mammal conservation in southern Africa. Journal of Zoology 277: 99–105.
Lloyd, P.H. and Millar, J.C.G. 1983. A questionnaire survey (1969–1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1–49.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1–218.
Lynch CD. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1–116.
Malbrant, R., 1952. Fauna du centre Africain Français (Maniferes et Oiseaux), second ed. Paul Lechevalier, Paris.
Manwa, L. and Ndamba, G.T. 2011. The language of dress among the subcultural group of the Dzimbabwe people in Masvingo, Zimbabwe. Journal of Emerging Trends in Educational Research and Policy Studies 2(6): 436–442.
McNab, B.K. 1984. Physiological convergence amongst ant-eating and termite-eating mammals. Journal of Zoology (London) 203: 485–510.
Mwale, M., Dalton, D.L., Jansen, R., de Bruyn, M., Pietersen, D., Mokgokong, P.S. and Kotzé, A. 2017. Forensic application of DNA barcoding for identification of illegally traded African pangolin scales. Genome 60(3): 272–284.
Ngwenya MP. 2001. Implications of the medicinal animal trade for nature conservation in KwaZulu-Natal, Report No. NA/124/04. Ezemvelo KZN Wildlife.
Panaino, W., Parrini, F., Kamerman, P.R., Hetem, R.S., Meyer, L.C.R., Smith, D., van Dyk, G. and Fuller, A. 2023. Temminck’s Pangolins relax the precision of body temperature regulation when resources are scarce in a semi-arid environment. Conservation Physiology 11(1): coad068.
Panaino, W., Parrini, F., Phakoago, M.V., Smith, D., van Dyk, G. and Fuller, A. 2022. Do seasonal dietary shifts by Temminck’s Pangolins compensate for winter resource scarcity in a semi-arid environment? Journal of Arid Environments 197: 104676.
Pietersen, D.W. 2013. Behavioural ecology and conservation biology of Ground Pangolins Smutsia temminckii in the Kalahari Desert. MSc Thesis. Department of Zoology and Entomology, University of Pretoria, Pretoria, South Africa.
Pietersen, D.W. 2022. Body size, defensive behaviour, and season influence mortality probability in wildlife interactions with electrified fences. African Journal of Wildlife Research 52: 172–184.
Pietersen, D.W. and Robertson, M.P., 2023. Dietary niche breadth and overlap of four sympatric southern African myrmecophagous mammal species, as inferred from the literature. African Zoology 58(2): 29-38.
Pietersen, D., Jansen, R., Swart, J. and Kotze, A. 2016a. A conservation assessment of Smutsia temminckii. In Child, M.F., Roxburgh, L., Do Linh San, E., Raimondo, D. & Davies-Mostert, H.T. (Eds.). The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Pietersen, D.W., Fisher, J.T., Glennon, K.L., Murray, K.A. and Parrini, F. 2021. Distribution of Temminck’s Pangolin (Smutsia temminckii) in South Africa, with evaluation of questionable historical and contemporary occurrence records. African Journal of Ecology 59: 597–604.
Pietersen, D.W., Jansen, R., Swart, J., Panaino, W., Kotze, A., Rankin, P. and Nebe, B. 2020. Temminck’s Pangolin Smutsia temminckii (Smuts, 1832). In: Challender, D.W.S., Nash, H.C. and Waterman, C. Pangolins: Science, Society and Conservation, pp. 175–193. Academic Press, London, United Kingdom.
Pietersen, D.W., McKechnie, A.E. and Jansen, R. 2014a. A review of the anthropogenic threats faced by Temminck’s Ground Pangolin, Smutsia temminckii, in southern Africa. South African Journal of Wildlife Research 44: 167–178.
Pietersen, D.W., McKechnie, A.E and Jansen, R. 2014b. Home range, habitat selection and activity patterns of an arid-zone population of Temminck’s Ground Pangolins, Smutsia temminckii. African Zoology 49: 265–276.
Pietersen, D.W., Symes, C.T., Woodborne, S., McKechnie, A.E. and Jansen, R. 2016b. Diet and prey selectivity of the specialist myrmecophage, Temminck’s Ground Pangolin. Journal of Zoology 298: 198–208.
Power, R.J. and Olivier, P. 2019. Zoogeography of a South African province: a framework for management. African Journal of Ecology 57(2): 198–211.
Richer, R., Coulson, I.M. and Heath, M.E. 1997. Foraging behaviour and ecology of the Cape Pangolin (Manis temminckii) in north-western Zimbabwe. African Journal of Ecology 35: 361–369.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stuart, C.T. 1980. The distribution and status of Manis temminckii Pholidota Manidae. Säugetierkundliche Mitteilungen 28: 123–129.
Swart, J.M., Richardson, P.R.K. and Ferguson, J.W.H. 1999. Ecological factors affecting the feeding behaviour of pangolins (Manis temminckii). Journal of Zoology 247: 281–292.
Swart, J.M. 1996. Foraging behaviour of the Cape Pangolin Manis temminckii in the Sabie Sand Wildtuin. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Swart, J.M. 2013. Smutsia temminckii. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume 5: Carnivores, Pangolins, Equids, Rhinoceroses, Bloomsbury Publishing, London, United Kingdom.
Sweeney, R.C.H. 1956. Some notes on the feeding habits of the Ground Pangolin, Smutsia temminckii (Smuts). Annals and Magazine of Natural History 9(108): 893–896.
Sweeney, R.C.H. 1974. Naturalist in the Sudan. Taplinger Publishing Co., New York, United States of America.
Tang, S.Y., Duan, W.W., Huang, X.G., Zhu, K.M., Li, W. and Yao, Y. 2012. The resources, status and development prospect of artificial breeding of Manis pentadactyla Linnaeus. Journal of Hunan University of Science and Technology 39: 75–77. (In Chinese).
Tinsman, J.C., Gruppi, C., Bossu, C.M., Prigge, T.L., Harrigan, R.J., Zaunbrecher, V., Koepfli, K.P., LeBreton, M., Njabo, K., Wenda, C., Xing, S., Abernethy, K., Ades, G., Akeredolu, E., Andrew, I.B., Barrett, T.A., Bernáthová, I., Bolfíková, B.C., Diffo, J.L., Fopa, G.D., Ebong, L.E., Godwill, I., Pambo, A.F.K., Labuschagne, K., Mbekem, J.N., Momboua, B.R., Moumbolou, C.K.M., Ntie, S., Rose-Jeffreys, E., Simo, F.T., Sundar, K., Swiacká, M., Takuo, J.M., Talla, V.N.K., Tamoufe, U., Dingle, C., Ruegg, K., Bonebrake, T.C. and Smith, T.B. 2023. Genomic analyses reveal poaching hotspots and illegal trade in pangolins from Africa to Asia. Science 382(6676): 1282–1286.
van Aarde, R.J., Richardson, P.R.K. and Pietersen, E. 1990. Report on the behavioural ecology of the Cape Pangolin (Manis temminckii). Internal Report. Mammal Research Institute, University of Pretoria, Pretoria, South Africa.
van Ee, C.A. 1966. A note on breeding the Cape Pangolin, Manis temminckii at Bloemfontein. International Zoo Yearbook 6: 163–164.
Weber, R.E., Heath, M.E. and White, F.N. 1986. Oxygen binding functions of blood and hemoglobin from the Chinese Pangolin, Manis pentadactyla: possible implications of burrowing and low body temperature. Respiration Physiology 64: 103–112.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J. 2011. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Journal of Zoology 284: 84–96.
Yan, D., Luo, X., Tang, J., Xu, S., Huang, K., Wang, X., Feng, T., Que, T., Jia, M., Guo, X., ur Rehman, S., Li, Z., Yang, Y., Li, K., Cui, K., Ruan, J. and Liu, Q. 2023. High-quality genomes of pangolins: insights into the molecular basis of scale formation and adaption to myrmecophagous diet. Molecular Biology and Evolution 40(1): msac262.