Oribi
Ourebia ourebi

2025 Red list status
Endangered
Regional Population Trend
Declining
change compared
to 2016
No Change
Overview
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Ourebia – ourebi
Common Names: Oribi (English), Oorbietjie (Afrikaans), Insinza (Ndebele), Phuduhudu-kgamane (Setswana), iWula (Zulu) Bleichbockchen (German), Oorbietjie (Afrikaans), Ourébi (French)
Synonyms: Antilope ourebi Zimmermann, 1783
Taxonomic Note:
The common name may be derived from the Khoikhoi name orabi (Skinner & Chimimba 2005).
Apparent regional differences in body size and colouration have led to 13 Oribi subspecies being suggested (Ansell 1972). However, to date no genetic work has been done to confirm whether these different populations are in fact different subspecies (Brashares & Arcese 2013). Nevertheless, the latest look at these different populations suggests 12 possible subspecies (Brashares & Arcese 2013). These include: O. o. ourebi, O. o. hastata, O. o. rutila, O. o. cottoni, O. o. masakensis, O. o. aequatoria, O. o. kenyae, O. o. haggardi, O. o. gallarum, O. o. montana, O. o. gosling and O. o. quadriscopa. Of these, O. o. haggardi, which is found in Kenya and Somalia, is the only population geographically isolated from the rest of the subspecies (Brashares & Arcese 2013). Sadly, O. o. keniae from Mount Kenya is now extinct (Hillman et al. 1988).
Generally, three subspecies are recognised in southern Africa (Skinner & Chimimba 2005; Brashares & Arcese 2013) with O. o. ourebi (Zimmermann, 1783) occurring in South Africa, and southern and central Mozambique. A genetic study has found that there is a high genetic variability within the South African Oribi population, and thus the population is healthy (van Vuuren et al. 2017). Moreover, the genetic evidence indicates that all Oribi in South Africa belong to a single (panmictic) population. Thus, suggestions of geographic genetic differences north and south of the Tugela River are unfounded.
| Red List Status |
| EN – Endangered, C2a(ii) (IUCN version 3.1) |
Assessment Information
Assessors: Shrader, AM.1 & da Silva, J.2
Reviewer: Patel, T.3
Contributor: Brassine, A.4
Institutions: 1Department of Zoology& Entomology, University of Pretoria, South Africa; 2South African National Biodiversity Institute; 3Endangered Wildlife Trust; 4Independent Professional Natural Scientist
Previous Assessors: Shrader, A.M., Little, I., Coverdale, B. & Patel, T.
Previous Reviewers: Downs, C., Child, M.F. & Mallon, D.P.
Previous Contributors: Rushworth, I., Ricketts, D., Currie, J. & Marchant, A.
Assessment Rationale
This charismatic subspecies is patchily distributed in grasslands in the eastern half of the country, requiring both short grass for food and long grass for food and shelter (Stears and Shrader 2015). Based on protected area data and survey returns from private landowners across the country, there were a minimum estimated total of 1,859–2,169 mature individuals (assuming a 60–70% mature population structure) at the time of the previous assessment (2016). The minimum estimate of the total number of mature individuals was likely an underestimate, due to unreturned surveys, but not significantly so as the Oribi Working Group obtained a good return rate in their 2013 survey.
The largest subpopulation occurring in Maloti-Drakensberg Transfrontier Park, KwaZulu-Natal Province, has in the past been considered a single subpopulation of around 400 individuals. Yet, numbers declined from 496 to 375 between 2010 and 2015. Moreover, spatial data suggests that this subpopulation is more likely a combination of four separate subpopulations where individuals are unlikely to move between subpopulations. Thus, it is unlikely that any subpopulation has > 250 mature individuals.
Overall, the national population in 2016 was estimated to have declined by c. 13% over three generations (1996–2014), using data from a sample of formally protected areas (N = 14) across its range with sufficient long-term data (data submitted by provincial reserves). Corroborating this, survey data from private lands (N = 74) in KwaZulu-Natal Province found that between 1999 and 2013, 27% of sites were increasing, 36% were declining and 11% were stable (Patel 2015). Thus, there was a continuing decline in the number of mature individuals within the assessment region. Correlating with the observed decline were data indicating intensifying threats, especially from illegal hunting, which increased from 73 reports in 2001 to 113 reports in 2013. Illegal hunting with dogs has evolved from small-scale into large, organised gambling syndicates that are thought to be significantly more destructive to biodiversity due to the unselective nature of these hunts. This threat, combined with ongoing grassland habitat loss, fragmentation and bad grassland management is likely to further threaten Oribi in the future.
Given the estimated continuing decline in the population, an estimated mature population size of < 2,500 mature individuals, and the largest subpopulation having < 250 mature individuals with these also declining, we list the Oribi as Endangered C2a(i). According to the Oribi Working Group, a survey of population counts on private lands has not been carried out for several years and as majority of Oribi are found on private land, there is no indication of recent Oribi numbers in the assessment region. Therefore, this species retains its threatened listing until recent data and population estimates are determined. However, if the number of mature individuals in the larger subpopulations increase to > 250 individuals, then the species would qualify for Vulnerable C2a(i) and thus should be reassessed. Key interventions include combatting illegal hunting through education and enforcement; implementing a metapopulation strategy; and continuing to promote stewardship activities in grassland areas, using Oribi as a key flagship subspecies.
Regional population effects: The subspecies’ range is not continuous across southern Africa (Skinner and Chimimba 2005, Brashares and Arcese 2013). There is no known immigration or dispersal from outside the region and hence no rescue effect.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Shrader AM & da Silva JM. 2025. A conservation assessment of Ourebia ourebi. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
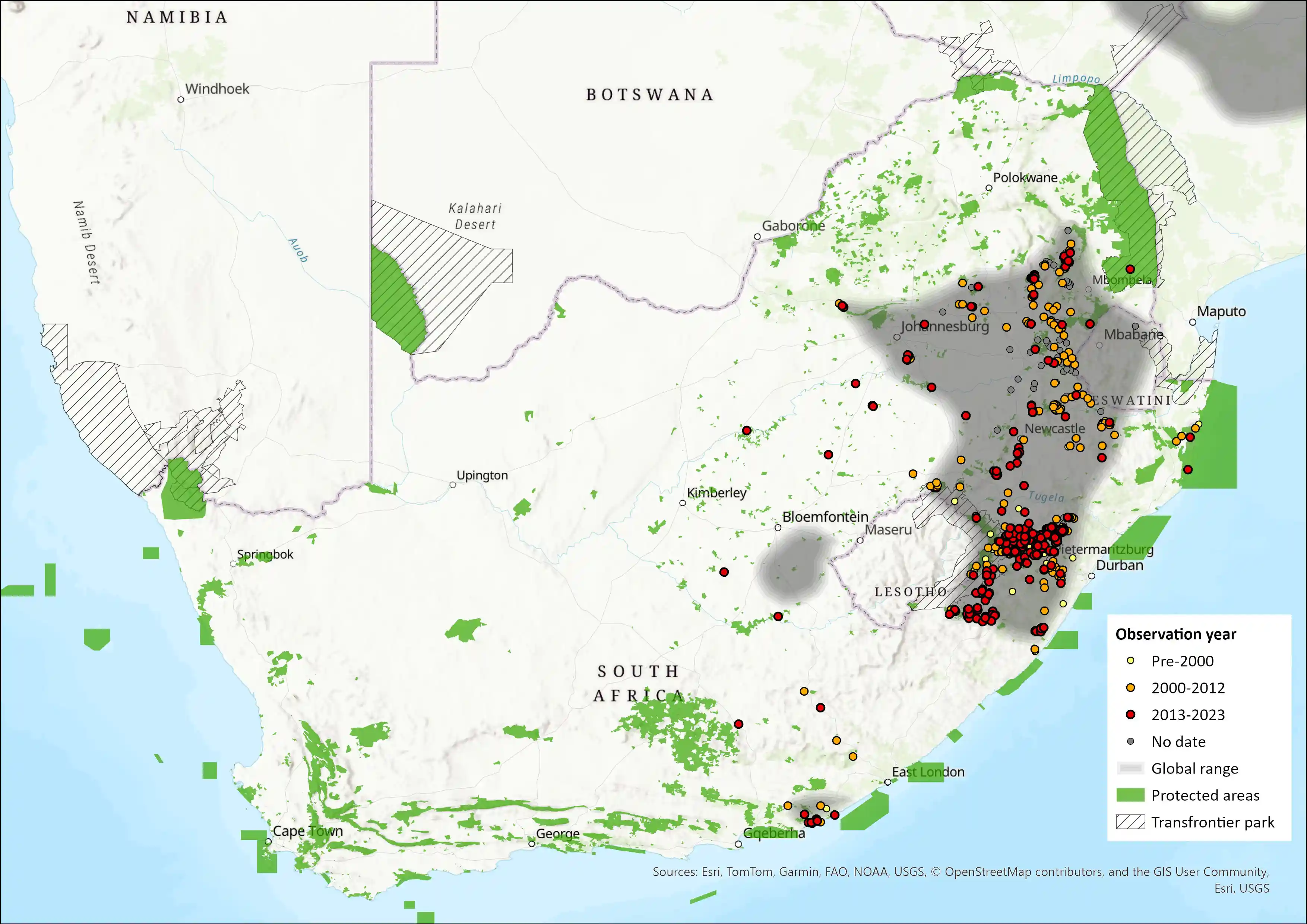
Regional Distribution and occurrence
Across Africa, Oribi have a patchy distribution ranging from Senegal to Ethiopia and Eritrea and south through eastern and western Africa to Angola and the Eastern Cape of South Africa (East 1999, Carpaneto and Fusari 2000, Fischer and Linsenmair 2001, Goldspink et al. 2002, Tekalign and Bekele 2011, Brashares and Arcese 2013, Djagoun et al. 2013, Wilfred and MacColl 2014). The Oribi still occurs widely within its former range, but its distribution is becoming increasingly fragmented as it is gradually eliminated from moderately to densely settled areas, and with changing land uses (Everett et al. 1991, Rowe-Rowe et al. 1992, Wilfred and MacColl 2014).
The subspecies O. o. ourebi (Zimmermann 1783) occurs throughout the southern part of its African range (i.e., South Africa, and central and southern Mozambique), while O. o. hastata (Peters 1852) is found in northern Mozambique, and eastern and southeastern Zimbabwe, while O. o. rutila (Blaine 1922) occurs in northeastern Botswana, northwestern Zimbabwe, and northeastern Namibia (Skinner and Chimimba 2005). In South Africa, their current range is probably similar to their historical range, occurring extensively in grasslands in Mpumalanga, Eastern Cape, and KwaZulu-Natal provinces (see Howard and Marchant 1984 for information on distribution in KwaZulu-Natal). They also occur in southern and northeastern Free State, southern Limpopo (Skinner and Chimimba 2005, Little and Magwaza 2014), as well as on formally protected land in North West Province (Kgaswane Nature Reserve), although it is unclear whether this area represented part of their historical range (Power 2014). They have also been discovered near Parys in the Vredefort Dome Granite Grasslands, an area where they formerly occurred (Power 2014). They are far more widely distributed in the Eastern Cape Province (for example, the former Transkei) than available data suggest, and future assessments should collate data from this region to make the distribution map more accurate. They may marginally occur in Lesotho (Lynch 1994), as habitat is connected with the Maloti-Drakensberg Transfrontier Park. They also occur in the highveld and Lubombo regions of Eswatini (Monadjem 1998). Introductions have predominantly been within the distributional range (Ezemvelo KwaZulu-Natal Wildlife unpubl. data). However, a few have been introduced into out of range areas such as Rust de Winter in southern Limpopo and Orkney and Brits in the North West (Marchant 1996).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): NA
Depth Upper Limit (in metres below sea level): NA
Depth Zone: NA
Map
Figure 1. Distribution records for Oribi (Ourebia ourebi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Possibly Extinct | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: NA
FAO Area Occurrence
FAO Marine Areas: NA
Climate change
For African savannas, the suggested impacts of climate change include increasing temperatures (Hulme et al. 2001), changes in rainfall patterns (Hulme et al. 2001, Dore 2005, Ayanlade et al. 2018), and bush encroachment (Wu et al. 2016). Changes in temperature and rainfall patterns may increase the likelihood of extreme droughts (Ayanlade et al. 2018) that could negatively affect food resources for Oribi populations. While foraging, Oribi tend to avoid feeding in wooded areas (Stears and Shrader 2015) due to an increased risk of attack from ambush predators. As a result, increases in woody vegetation from bush encroachment would likely reduce the availability of preferred open grasslands. An increase in woody habitats would either result in range shifts for Oribi as they try and avoid wooded areas, or they would be forced to feed more in these woody patches where their feeding efficiency would be lower due to an increase in time spent on anti-predator behaviours (Stears and Shrader 2015). However, the impact of these changes on Oribi populations is unknown.
Population information
Oribi density depends on veld management and range quality (Skinner and Chimimba 2005, Stears 2015), and, at seven study sites at midland elevation in KwaZulu-Natal, density ranged from 4–18 animals / km2 (Everett et al. 1991). Similarly, outside of the assessment region, Oribi can be locally common in suitable habitats at densities of 2–10 animals / km², but have been recorded at densities up to 45 animals / km² in exceptionally productive tropical grasslands and treeless floodplains (Brashares and Arcese 2013 and references therein). However, densities estimated from ground counts range from 0.1–0.4 animals / km² in areas where the species is uncommon or depleted (East 1999).
The total minimum count for 2013–2015, based on both protected area game count records and survey returns from private landowners from across its range, was 3,098 individuals. This yielded a minimum observed total of 1,859–2,169 mature individuals (assuming a 60–70% mature population structure). We assume this proportion of adults in the population as adult Oribi tend to be solitary, move in male-female breeding pairs, or in groups comprised of a single male and one or two females plus their offspring (Skinner and Chimimba 2005, Humphrey 2006). Moreover, as males tend to breed with one female (Skinner and Chimimba 2005) young most likely make up around one third (c. 33%) of the population.
Population estimates are confounded by inconsistent survey returns due to the difficulties of communicating with all landowners throughout the Oribi’s range (Patel et al. 2021a). For example, from 2001 to 2014, the Oribi population was estimated to have declined by 30% (i.e., ~ 700 individuals). However, these estimates were highly correlated with the number of returned surveys (Patel et al. 2021a). For example, in 2007, the population was estimated to have declined to ~1,100 individuals (including young) (Patel et al. 2021a). However, this was likely due to poor survey returns (Patel et al. 2021a). In 2011, a greater survey effort was made resulting in a population estimate of ~1500 individuals (44 of which were juveniles; Patel et al. 2021a). In 2014, even greater focus was put into the survey, resulting in a population estimate of ~1,600 (Patel et al. 2021a), emphasising the need for survey returns to obtain accurate population counts.
Most of the population (63%) exists on private land and can be considered wild and free-roaming (Oribi Working Group unpubl. data). Intensive captive breeding has been unsuccessful due to spatial requirements associated with male territoriality. While most sites contain few (< 50) individuals), there are some that have between 100 and 200 individuals. For example, both Chelmsford Nature Reserve and Maloti-Drakensberg Transfrontier Park had relatively large robust sizes, 222 (in 2012, Ezemvelo KwaZulu-Natal Wildlife unpubl. data) and 416 individuals (in 2013; Krüger and van der Westhuizen 2014), respectively. However, the number in the Maloti-Drakensberg Transfrontier Park is more likely a combination of four separate sites pertaining to Kamberg, Highmoor, Giant’s Castle and Garden Castle) between which movement of individuals is unlikely (Ezemvelo KZN Wildlife unpubl. data). However, numbers in the Maloti-Drakensberg have declined from 496 to 375 individuals between 2010 and 2015 (Ezemvelo KZN Wildlife unpubl. Data). The number of individuals in Chelmsford Nature Reserve declined to 96 in 2013, although this might be partially due to an Oribi capture operation that had taken place that year (removing 15 individuals to iSimangaliso Wetland Park) as noise and disturbance of the capture operation may have caused them to disperse onto neighbouring farms (P. Ngwenya pers. comm. 2016). Overall, then, no site is likely to consist of >250 mature individuals.
While most sites were suspected to be stable (N= 152), more were declining (N = 20) than increasing (N= 10) and 16 had uncertain trends (Little and Magwaza 2014). However, these data are a rough indication rather than a robust sample because most of the landowners or managers did not feel confident enough to indicate their population trends (Oribi Working Group unpubl. data). Corroborating this, an independent study by Patel (2015) found that, between 1999 and 2013, of the 74% of sites on private land in KwaZulu-Natal Province, 36% were increasing, 49% were decreasing and 15% were stable (similar declines on private land were noted in Marchant 2000). Of the remaining 26% of oribi sites in formally protected areas 42% were increasing, 38% were decreasing and 19% were stable (Patel et al. 2021a). For example, there was only one individual counted in Golden Gate Highlands National Park in 2010 (Ferreira et al. 2013). Overall, 37% of all sites were found to be increasing, 46% decreasing and 17% stable.
Generation length has been calculated to range between 3.5 and 6 years using the equation G = FR + z * RL (IUCN Standards and Petitions Subcommittee 2014). This estimate is wide due to the poor life history data available for the species. For example, we only have data on age of first reproduction (~10 months for females; Cade 1966, Adamczak 1999) and age of last reproduction (8–13 years; Mentis 1972). Reproductive period (RL) was determined by subtracting age at first reproduction (FR) from age of last reproduction, resulting in a range of breeding periods of 7–12 years. As we do not know the ratio between survivorship and fecundity, z is set at 0.5. As age of first reproduction (FR) is less than a year, we have inserted 0 into the formula. Using these values, the formula generates a range for the generation of between 3.5 years (G = 0 + 0.5 * 7) and 6 years (G = 0 + 0.5 * 12). The upper estimate of 6 years is similar to the 5.9 years calculated by Pacifici et al. (2013). Rounding up the lower estimate yielded a generation length of 4–6 years, which translated to a three-generation window of 2002–2014 or 1996–2014. Analysing a sample of formally protected areas across the Oribi’s range (N = 14) that had adequate long-term data over the time period, revealed a population reduction of c. 13% (1996–2014) or a population increase of c. 6% (2002–2014).
Population Information
Current population trend: Declining, based on available long-term data.
Continuing decline in mature individuals: Yes, based on survey returns concerning snaring and illegal hunting with domestic dogs.
Number of mature individuals in population: 1,859-2,169
Number of mature individuals in largest subpopulation: 222 in Chelmsford Nature Reserve, KZN (2012 count).
Number of subpopulations: At least 231
Severely fragmented: Yes, most subpopulations confined to fenced areas (predominantly standard livestock fencing, thus permeable for Oribi). However, due to distances between subpopulations, active translocation is required. Habitat is further fragmented through afforestation, agriculture, game fencing, residential and commercial development and poor livestock farming.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Van Vuuren et al. (2017) explored the phylogeography of Oribi in South Africa. Specifically, using two mitochondrial gene regions and one nuclear intron they investigated the phylogenetic relationship of the South Africa subspecies (O. o. ourebi) against the geographically closest oribi subspecies (O. o. hastata), and found the two subspecies to be substantially divergent (> 13% sequence divergence in the mitochondrial cytochrome b gene region), validating the South African subspecies as a separate evolutionarily significant unit (ESU). However, within the South African ESU, high genetic diversity and no geographically-based population genetic structure was detected, likely due to historic translocations, suggesting Oribi in South Africa are a single metapopulation and should be managed as such.
Based on minimum estimates of census (survey) counts (1,859–2,169 mature individuals) from 2015, and applying an Nc/Ne conversion ratio, the minimum effective population size (Ne) for this metapopulation is estimated at 186-650 individuals. This estimate cradles the Ne 500 threshold. A population genomic study is highly recommended incorporating samples across the assessment region, and into neighbouring countries to get a more confident estimate of Ne and the population structure within this subspecies/ESU.
Habitats and ecology
Oribi inhabit savannah woodlands, floodplains, and other open grasslands, from around sea level to about 2,200 (Mpumalanga Province). They reach their highest density on floodplains and moist tropical grasslands, especially in association with large grazers. They prefer open grassland in good condition containing a mosaic of both short grass for feeding and long grass for feeding and shelter (Rowe-Rowe 1994, Perrin and Everett 1999, Stears 2015). However, within these grasslands they avoid feeding within and close to woodland patches even if these patches are small (for example, 2–6 m in diameter; Stears and Shrader 2015). Within grasslands, they are selective feeders that focus primarily on green leaves and thus maintain high quality intake year-round. For example, they have been found to select patches of Themeda triandra grass (Shackleton and Walker 1985). Grass makes up most of their diet, with only a minor intake of forbs recorded during the wet season (Reilly et al. 1990, Stears 2015). Key grass species include, Themeda triandra, Hyparrhenia hirta, Panicum natalense, and Andropogon chinensis (Viljoen 1982, Shackleton and Walker 1985, Everett et al. 1992, Stears 2015). Within the assessment region, they are thus primarily found in the Grassland Biome, in vegetation types such as Northern KwaZulu-Natal Moist Grassland, Income Sandy Grassland, and Midlands Mistbelt Grassland (Mucina and Rutherford 2006). However, to a lesser degree, they may also be found in the more open, grass-dominated habitats of the Savannah Biome (for example, Northern Zululand Sourveld).
After burns, Oribi focus their foraging on the high-quality green flush on the burnt areas (Oliver et al. 1978, Everett et al. 1991, Stears 2015). By feeding on the green flush found on these burns, Oribi are able to increase both their crude protein and metabolisable energy intake (i.e., nutritional intake) during the nutritionally limited dry season (Stears 2015). This highlights the importance of regrowth on fire breaks and burnt grasslands as a key food resource during the winter months (Shackleton and Walker 1985, Marchant et al. 2005, Stears 2015). They also use artificially managed or altered habitat such as hayfields and grasslands used by cattle (Rowe-Rowe 1994, Perrin and Everett 1999, Skinner and Chimimba 2005). They normally consist of solitary adults, adult pairs, or groups of 1–6 comprising a male and 1–2 females and their offspring (Skinner and Chimimba 2005 and references therein). Home ranges of adult males in KwaZulu-Natal range from 0.05 to 0.47 km2 (Skinner and Chimimba 2005). The lower estimate compares to estimated home range size of 0.03 km2 in Mpumalanga (Viljoen 1982).
Ecosystem and cultural services: The Oribi is a flagship species for highlighting the value of grasslands and threats to them and is often employed in extending stewardship practices. In South Africa, 60% of grasslands have been irreversibly transformed, with only 2.4% being conserved (Carbutt and Martindale 2014). It is important to conserve the remaining natural grasslands.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.6. Grassland -> Grassland – Subtropical/Tropical Seasonally Wet/Flooded | – | Suitable | – |
Life History
Generation Length: between 3.5 and 6 years
Age at maturity: Female or unspecified: 10 months
Age at Maturity: Male: 10-14 months
Size at Maturity (in cms): Female: Total length: 102.8 cm, Shoulder height: 59.1 cm
Size at Maturity (in cms): Male: Total length: 102.3 cm, Shoulder height: 57.8 cm
Longevity: 8-12 years in the wild, 12-14 years in captivity
Average Reproductive Age: ~10 months for females
Maximum Size (in cms): Total length: 115.0 cm, Shoulder height: 63.5 cm both measures from males
Size at Birth (in cms): 2.3 kg, 37.5 cm shoulder height
Gestation Time: 210 days
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: single fawn
Natural Mortality: More pronounced in winter when their lower body condition makes them susceptible to climatic stress and disease.
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Adult males and associated females and calves move within territories. Some family groups may move between seasonal home ranges in pursuit of high-quality food. Individuals may make short excursions out of these home ranges.
Congregatory: Can be found as solitary adults, in male-female pairs, or in small groups comprising a male and one or two females and their offspring. However, groups may periodically join up to form temporary groups of up to 12 individuals.
Systems
System: Terrestrial
General Use and Trade Information
Trade is fairly well controlled. Introductions and translocations to new areas are generally from sites found in areas likely to be transformed rendering habitat no longer suitable, and translocations are performed under the auspices of the local conservation authorities (Patel 2015). However, 80% of the reintroduced populations are declining (Patel et al. 2021b). As such, these reintroductions have not been an effective conservation tool. Nevertheless, the translocations were necessary as they focused on threatened individuals (Patel et al. 2021b). The larger protected sites, however, are also sometimes used to found new sites (Grey-Ross et al. 2009, Patel 2015). These translocations are carried out under permit of the provincial nature conservation agency. Few subpopulations are kept under captive circumstances within their natural range (< 5% of properties) (Grey-Ross et al. 2009).
A small number of mature males are hunted each year. For example, at least eight mature rams were permitted for trophy hunting during the course of 2013 (Ezemvelo KwaZulu-Natal Wildlife unpubl. data). However, as not all permits are utilised, the total number hunted was likely less. As a result, they comprise a very small proportion of the total population and thus the removal can be considered sustainable. Future trends are unlikely to change. Hunting permits for mature rams in KwaZulu-Natal are done in terms of the National Environmental Management: Biodiversity Act, (No. 10 of 2004) and the Natal Nature Conservation Ordinance (No. 15 of 1974).
Change in the extent and quality of habitat is primarily due to land transformation and poor farming practices (Carbutt and Martindale 2014).
| Subsistence | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Illegal bushmeat hunting | Yes | Trophy hunting. Live sales. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 15. Sport hunting/specimen collecting | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Trophy hunting, illegal bushmeat hunting, organised illegal hunts (gambling)
Threats
In Africa, the species has been eliminated from substantial parts of its former range by the spread of agricultural settlement, livestock and increased illegal hunting. For example, in the Comoé National Park in Côte d’Ivoire, Oribi experienced a decline of around 92% between 1978 and 1998 primarily due to illegal hunting (poaching) (Fischer and Linsenmair 2001). There are also increased levels of illegal hunting in other parts of Africa (Carpaneto and Fusari 2000, Goldspink et al. 2002, Wilfred and MacColl 2014), including the assessment region (Little and Magwaza 2014). The numbers in most provincial sites have declined due to illegal hunting (for example, uncontrolled hunting with dogs) and poor land use, which has resulted in few remaining viable sites. Within the assessment region, the threats are as follows:
- Habitat destruction (loss and fragmentation): Grasslands are lost to commercial forestry activities, intensive commercial farming, grassland degradation due to overstocking, poor fire management, erosion, and mining (Little et al. 2013, Carbutt and Martindale 2014). As a grassland specialist that does not occur elsewhere, the loss of grasslands on flat to undulating terrain is a threat to its survival. There is also an emergence of unresolved land claims and changes in ownership, which may reduce potential suitable habitat via changes in land use and/or degradation through a lack of active management.
- Over-utilisation due to illegal hunting: Illegal hunting with dogs is considered the major current and intensifying threat across the country (Marchant 2000, Little and Magwaza 2014). For example, there was an approximate threefold increase (from 25 to 113) in the reported number of incidents from 2011 to 2013 (Little and Magwaza 2014). This level of removal is resulting in the decline and, in extreme instances, local extinctions on private land. In most cases, landowners are powerless to stop these hunts due to the likelihood of retribution by hunters. As a result, these illegal hunts are being conducted at unsustainable levels, both for subsistence and gambling (Grey-Ross et al. 2010, Little and Magwaza 2014). Furthermore, some recent illegal dog hunting has evolved into large organised gambling syndicates which are considerably more destructive than the local sport or food hunting of the past (Little and Magwaza 2014). Given increased unemployment, low minimum wage and removal of ration provision, the frequency of illegal hunting is likely to increase (Grey-Ross et al. 2010). Incidental trapping with snares also poses a severe threat. This threat mainly affects non-protected areas, especially small populations on private lands susceptible to edge effects, but may increasingly affect protected areas (Wittemyer et al. 2008).
- Inappropriate management: In many areas where populations are present, current farm management practices (for example, fences, poor burning practices, poor veld management, domestic dogs) do not allow coexistence at optimal levels with other livestock and game. Moreover, changes in management practices due to land claims may further decrease appropriate/available habitat (Carbutt and Martindale 2014).
- Poor law enforcement: South Africa currently has advanced environmental legislation. However, the enforcement of this legislation has been poor because of budgetary constraints and focuses on more charismatic species such as rhino (Diceros bicornis and Ceratotherium simim simum), and continues to affect grassland-dependent species. Although the Oribi is formally protected in the provinces where it occurs, the lack of funds may result in insufficient law enforcement and no coordinated national conservation effort. The growing gambling industry which uses dogs to hunt indigenous wildlife seems to have strong political backing and efforts to address this have frequently been thwarted with “cultural rights” being offered as a justification. This, along with widespread ignorance among police officers across the range about the illegality of dog hunting has made it very difficult to address. Furthermore, most Oribi occur on private farmland where law enforcement is difficult.
- Lack of awareness: The lack of awareness of the status, threats, and legal repercussions of killing Oribi prevents effective implementation of interventions (Grey-Ross et al. 2010, Little and Magwaza 2014). Concurrently, the lack of understanding of the value of grasslands in general is hindering conservation progress. Grassland ecosystems are currently the most important and yet the most underrated and highly degraded ecosystem in South Africa (Carbutt and Martindale 2014). A far better understanding and appreciation of grasslands is required, which will benefit grassland-dependent species.
- Lack of coordination/cooperative management: a coordinated national approach to Oribi conservation is required to avoid duplicated efforts and wasted funding. At present, this is being done through the Oribi Working Group, but broader involvement is required.
Current habitat trend: Declining in area and quality. Their preferred grassland habitat is highly fragmented and much habitat has been lost to afforestation in KwaZulu-Natal and Mpumalanga, including conversion to pasture for intensive livestock farming, agricultural conversion, housing and commercial development. In KwaZulu-Natal Province alone there was a 20.4% loss of natural habitat from 1994 to 2011, with an average loss of 1.2% per annum (Jewitt et al. 2015). Worryingly, in just six years (2005–2011), 7.6% (7,217 km2) of natural habitat was lost (1.3% per annum), due primarily to agriculture (5.2% increase; 4,962 km2), but also plantations, built environments and settlements, mines and dams (Jewitt et al. 2015). Similarly, rural settlements have expanded in Oribi core provinces by 1–39% between 2000 and 2013 (GeoTerraImage 2015), which is inferred to be increasing rates of illegal hunting. Additionally, poor veld management, burning regimes and frequency of burning may affect habitat quality. Shifts in grazing practices and land use also contribute to changes in quality (for example, increased stocking rate can reduce grass cover and thus habitat quality). Moreover, higher stocking rates of domestic animals (for example, cattle) can lead to direct and delayed competition with Oribi during the dry season, resulting in reduced nutritional intake (Stears 2015). In certain protected areas, competition from other large herbivores such as Blesbok (Damaliscus pygargus phillipsi) may also reduce habitat quality. There is however, a possibility that bulk grazers may facilitate Oribi by stimulating high quality grass regrowth (Skinner & Chimimba 2005; Stears 2015). One potential threat that has not been explored is a potential increased woody cover due to higher levels of CO2 as a result of global climate change (Curtis & Wang 1998). If this takes place, it would reduce available grasslands for Oribi as they do not like to feed in or near woodland patches (Stears & Shrader 2015).
Conservation
Oribi occur in several protected areas where human population densities are low, such as Golden Gate Highlands National Park (but see Ferreira et al. 2013) and Maloti-Drakensberg Transfrontier Park. Conservationists must urgently combat the ongoing illegal hunting and emerging threat of organised dog hunting as a gambling practice through education and enforcement (Grey-Ross et al. 2010); and through substitution of the activity through alternative recreational activities, such as dog racing (chasing electronic rabbits; A. Marchant pers. comm. 2016). Stewardship is essential for conservation too, with incentives to maintain individuals on private property, particularly as a flagship species for the dwindling grassland biome (Carbutt and Martindale 2014). Improved education and awareness around the legal implications and consequences of illegal hunting with dogs within local communities and law enforcement officials is also needed.
Current actions within the assessment region include habitat management, monitoring, public awareness, intensive public education programmes and South African Police Service (SAPS) training to address illegal hunting issues, Oribi Custodian Programme, revision of burning regimes in areas (for example, fire breaks and use of mosaic burning), summer mowing of grass, use of cattle to generate heterogeneity of grass height, and strengthening of the annual Oribi census to increase landowner participation across the different provinces (Marchant et al. 2005, Coverdale et al. 2006).
Such interventions should be continued and combined with the movement of populations where detrimental habitat change is inevitable. Founder subpopulations could be stocked without any other Oribi present at release sites (Bothma et al. 2010). Moreover, prior to release it is important that specific criteria be considered to determine whether the release site is suitable (see Pérez et al. 2012). For Oribi, key factors include initial population size, the amount of suitable habitat, and the stocking rates of other grazers (Patel 2015).
Translocation of animals from unsuitable habitats should follow a metapopulation plan, with suitable reintroduction areas being identified from areas within the Oribis’ natural distribution range. Conservationists must also comment on and attempt to prevent applications for development based on Oribi presence and/or habitat suitability for Oribi conservation. Finally, the formal proclamation of key habitats through the biodiversity stewardship schemes should continue.
Recommendations for land managers and practitioners: The Mission of the Oribi Working Group is to promote the long-term survival of Oribi in their natural grassland habitat through initiating and coordinating provincial conservation programmes. These include: education and awareness, habitat conservation, research and monitoring, subpopulation management, database management and forming partnerships with stakeholders. The Oribi Working Group consists of a coalition of members including private landowners.
- There is an Oribi Conservation Plan (Marchant et al. 2005) and an Oribi population and habitat viability assessment document (Coverdale et al. 2006), which should be used by landowners to assess and improve habitat quality for Oribi subpopulations. A metapopulation plan should be developed to guide translocations. Currently, the population is not managed as a metapopulation, however, there have been 10 translocations over the past 13 years, which were not very successful (Patel et al. 2021b).
- The major data deficiency is the inconsistency of the survey effort and difficulty of actually counting this species, which prevents accurate estimates of long-term trends for the population (Patel et al. 2021a). The Oribi Working Group coordinated national annual surveys that included both provincial protected areas and private landowners. This, however, relied on private landowners to voluntarily conduct their own counts and then submit their data to the Oribi Working Group. A survey has not been conducted in recent years, which makes it difficult to determine long-term trends or obtain recent population estimates. More effort needs to be put into these surveys going forward. Within KwaZulu-Natal, sites are monitored within state protected areas and the species has been included as part of the annual CITES species survey. Further monitoring priorities and sites should be established.
- The Oribi Working Group was developing programmes and projects to address management and conservation issues.
Research priorities:
- Population dynamics studies, especially impacts of illegal hunting. A study was completed that focussed on the population dynamics of Oribi in KwaZulu-Natal (Patel et al. 2021a), particularly investigating translocation success (Patel et al. 2021b. However, a further assessment of the regional population is needed. There was also a project that investigated the foraging ecology and habitat use and whether cattle compete with or facilitate Oribi (Stears 2015, Stears and Shrader 2015, Steer and Shader 2020). Further studies are needed on Oribi movement, dispersal (particularly of young males) and habitat use in response to burning and mowing.
- Competition between Oribi and short grass grazers such as Blesbok and Black Wildebeest, and domestic animals such as sheep.
- Success of prior translocations and the effects of a translocation policy that limits impacts on core subpopulations and ensures successful establishment/survival in new location. To date, no studies have been conducted to explore the impact that the removal of individuals has on the wild, free-ranging core populations (for example, Chelmsford Nature Reserve). However, one study looked into the success of the 10 translocations that have taken place since 2004 in KwaZulu-Natal (Patel et al.2021b).
- While a phylogeographic study found the Oribi in South Africa to be a distinct ESU, no geographic structure within South Africa was found, indicating the species exists as a metapopulation within the assessment region (van Vurren et al. 2017). A fine-scale population genomic study could be informative to further investigate this structure and quantify the genetic health (genetic diversity and effective population size) of the population.
- Impact of predation on fragmented populations.
- Role of grassland corridors, and how they should be designed (for example, what should the minimum width and maximum length of a corridor be to ensure it will be used since they do not appear to undertake long distance movements).
- Life history data needed for life table population modelling (e.g., age-specific mortality and fecundity, calf-survival) to better estimate mature individual population size and population trends.
Encouraged citizen actions:
- All illegal hunting activity or any hunting with dogs needs to be reported in order to determine trends in this threat and result in mitigation and follow up reporting to the chairman of the Oribi Working Group is required.
- Citizens and landowners participate each year in the annual census. Greater and more consistent participation would improve understanding of population trends and improve conservation efforts.
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas, to increase the accuracy of the distribution map.
- Landowners should form conservancies to create more suitable available and connected habitat, as well as to co-manage the threat of illegal dog hunting.
Bibliography
Adamczak VG. 1999. Variation in the mating system of Oribi, Ourebia ourebi. Ph.D Thesis. University of Liverpool, Liverpool, UK.
Ansell, W.F.H. 1972. Part 2, 15 Family Artiodactyla. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-84. Smithsonian Institution Press, Washington, DC, USA.
Ayanlade, A. Radeny, M., Morton, J.F. and Muchaba, T. 2018. Rainfall variability and drought characteristics in two ago-climatic zones: An assessment of climate change challenges in Africa. Science of the Total Environment 630: 728-737.
Bothma J du P, Van Rooyen N, Du Toit JG. 2010. Antelope and other smaller herbivores. In: J. du P. Bothma and J. G. Du Toit (eds), Game Ranch Management 5th edition, pp. 210–245. Van Schaik Publishers, Pretoria, South Africa.
Brashares, J.S. and Arcese, P. 2013. Ourebia ourebi Oribi. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume VI: Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer and Bovids, Bloomsbury Publishing, London.
Cade CE. 1966. A note on the behaviour of the Kenyan Oribi Ourebia ourebi in captivity. International Zoo Yearbook 6: 205.
Carbutt C, Martindale G. 2014. Temperate indigenous grassland gains in South Africa: Lessons being learned in a developing country. PARKS 20: 101-121.
Carpaneto GM, Fusari A. 2000. Subsistence hunting and bushmeat exploitation in central-western Tanzania. Biodiversity & Conservation 9: 1571-1585.
Coverdale B, Daly B, Friedman Y, Lemmer F, Marchant A, McCann K, Rushworth I, Wakelin J. 2006. Oribi antelope (Ourebia ourebi) population and habitat viability assessment workshop report. Conservation Breeding Specialist Group (SSC / IUCN) / CBSG Southern Africa. Endangered Wildlife Trust, Johannesburg.
Curtis PS, Wang X. 1998. A meta-analysis of elevated CO2 effects on woody plant mass, form, and physiology. Oecologia 113: 299-313.
Djagoun C, Codron D, Sealy J, Mensah GA, Sinsin B. 2013. Stable carbon isotope analysis of the diets of West African bovids in Pendjari Biosphere Reserve, Northern Benin. South African Journal of Wildlife Research 43: 33-43.
Dore, M.H.I. 2005. Climate change and changes in global precipitation patterns: What do we know? Environment International 31: 1167-1181.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Everett PS, Perrin MR, Rowe-Rowe DT. 1991. Responses by oribi to different range management practices in Natal. South African Journal of Wildlife Research 21: 114-118.
Everett PS, Perrin MR, Rowe-Rowe DT. 1992. Diet of Oribi on farmland in Natal. South African Journal of Wildlife Research 22: 7-10.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Fischer, F. and Linsenmair, K. E. 2001. Decreases in ungulate population densities. Examples from the Comoe National Park, Ivory Coast. Biological Conservation 101: 131-135.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Goldspink CR, Holland RK, Sweet G, Stewart L. 2002. A note on group sizes of oribi (Ourebia ourebi, Zimmermann, 1783) from two contrasting sites in Zambia, with and without predation. African Journal of Ecology 40: 372-378.
Grey-Ross R, Downs CT, Kirkman K. 2009a. Using housing estates as conservation tools: A case study in KwaZulu-Natal, South Africa. Applied Geography 29: 371-376.
Grey-Ross R, Downs CT, Kirkman K. 2009b. Reintroduction failure of captive-bred oribi (Ourebia ourebi). South African Journal of Wildlife Research 39: 34-38.
Grey-Ross R, Downs CT, Kirkman K. 2009c. Is use of translocation for the conservation of subpopulations of oribi Ourebia ourebi (Zimmermann) effective? A case study. African Journal of Ecology 47: 409-415.
Grey-Ross R, Downs CT, Kirkman K. 2010. An assessment of illegal hunting on farmland in KwaZulu-Natal, South Africa: implications for oribi (Ourebia ourebi) conservation. South African Journal of Wildlife Research 40: 43-52.
Hillman, J. C., Cunningham van Someren, G. R., Gakahu, C. G. and East, R. 1988. Chapter 8: Kenya. In: R. East (ed.), Antelopes: Global Survey and Regional Action Plan. Part 1. East and Northeast Africa, pp. 41-53. IUCN, Gland, Switzerland.
Howard PC, Marchant AN. 1984. The distribution and status of some large mammals on private land in Natal. Lammergeyer 34: 1-58.
Hulme, M., Doherty, R., Ngara, T., New, M. and Lister, D. 2001. African climate change: 1900-2100. Climate Research 17: 145-168.
Humphrey G. 2006. The social structure and spatial distribution of Oribi (Ourebia ourebi, Zimmermann, 1783) on Kasouga farm in the Eastern Cape Province of South Africa. M.Sc. Thesis. Department of Zoology and Entomology, Rhodes University, Grahamstown, South Africa.
IUCN Standards and Petitions Subcommittee. 2014. Guidelines for Using the IUCN Red List Categories and Criteria. Version 11.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Krüger S, van der Westhuizen R. 2014. Report on the 2013 Oribi Survey in the Maloti Drakensberg Park World Heritage Site. Ezemvelo KZN Wildlife, unpublished report, 7pp.
Little I, Magwaza JF. 2014. Annual oribi survey report 2013. Oribi Working Group and Endangered Wildlife Trust.
Little, I.T.; Hockey, P.A.R.; Jansen, R. 2013. A burning issue: Fire overrides grazing as a disturbance driver for South African grassland bird and arthropod assemblage structure and diversity. Biological Conservation 158: 258-270.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Marchant A, Rushworth I, McCann K. 2005. Oribi (Ourebia ourebi) conservation plan. Oribi Working Group unpublished Report.
Marchant AN. 1996. Survival of six antelope species bought at wildlife auctions. Lammergeyer 44: 31-38.
Marchant AN. 2000. The status of oribi (Ourebia ourebi) on private land and nature reserves in KwaZulu-Natal, South Africa since 1981. Lammergeyer 46: 70-74.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Oliver MDN, Short NRM, Hanks J. 1978. Population ecology of Oribi, grey rhebuck and mountain reedbuck in Highmoor State Forest Land. South African Journal of Wildlife Research 8: 95-105.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Patel, T. 2015. Population dynamics and relocation success of the oribi antelope (Ourebia ourebi) in KwaZulu-Natal, South Africa. M.Sc. Thesis. University of KwaZulu-Natal, Pietermaritzburg, South Africa.
Patel, T., Stears, K., Little, I.T. and Shrader, A.M. 2021a. Assessing the performance of oribi antelope populations at multiple scales: the limitations of citizen-led oribi conservation. African Journal of Wildlife Research 51: 127-135.
Patel, T., Stears, K., Little, I.T. and Shrader, A.M. 2021b. Evaluating oribi translocations for conservation: the importance of translocation guidelines and herbivore stocking rates. African Journal of Wildlife Research 51: 144-148.
Perrin MR, Everett PS. 1999. Habitat use by oribis at midlands elevations in KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 29: 10-14.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Pérez I, Anadón JD, Díaz M, Nicola GG, Tella JL, Giménez A. 2012. What is wrong with current translocations? A review and a decision-making proposal. Frontiers in Ecology and the Environment 10: 494-501.
Reilly BK, Theron GK, Bothma J du P. 1990. Food preferences of oribi Ourebia ourebi in the Golden Gate Highlands National Park. Koedoe 33: 55-61.
Rowe-Rowe DT, Everett PS, Perrin MR. 1992. Group sizes of oribis in different habitats. South African Journal of Zoology 27: 140-143.
Rowe-Rowe, D. T. 1994. The ungulates of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Shackleton C, Walker BH. 1985. Habitat and dietary species selection by oribi antelope at Mount Sheba Nature Reserve. South African Journal of Wildlife Research 15: 49-53.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stears K. 2015. Key factors driving the foraging ecology of oribi: Fear, cattle and the quality and quantity of food. Ph.D Thesis. University of KwaZulu-Natal, Durban, South Africa.
Stears K, Shrader AM. 2015. Increases in food availability can tempt oribi antelope into taking greater risks at both large and small spatial scales. Animal Behaviour 108: 155-164.
Stears K. and Shrader AM 2020. Coexistence between wildlife and livestock is contingent on cattle density and season but not differences in body size. PLoS One 15: e0236895
Tekalign W, Bekele A. 2011. Population Status, Foraging and Diurnal Activity Patterns of Oribi (Ourebia ourebi) in Senkele Swayne’s Hartebeest Sanctuary, Ethiopia. Ethiopian Journal of Science 34: 29-38.
van Vuuren, B. J., Rushworth, I., & Montgelard, C. (2017). Phylogeography of oribi antelope in South Africa: evolutionary versus anthropogenic panmixia. African Zoology, 52(4), 189–197. https://doi.org/10.1080/15627020.2017.1386077
Viljoen PC. 1982. Die ekologie van die oorbietjie Ourebia ourebi ourebi. Finale verslag. Transvaalse Provinsiale Administrasie. Afdeling Natuurbewaaring.
Wilfred P, MacColl A. 2014. Legal subsistence hunting trends in the Ugalla ecosystem of western Tanzania. European Journal of Wildlife Research 60: 371-376.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
Wu, M., Schurgers, G., Rummukainen, M., Smith, B., Samuelsson, P., Jansson, C., Siltberg, J. and May, W. 2016. Vegetation-climate feedbacks modulate rainfall patterns in Africa under future climate change. Earth System Dynamics 7: 627-647.