Nyika Climbing Mouse
Poemys nyikae

2025 Red list status
Data Deficient
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Poemys nyikae – Wroughton, 1909
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Poemys- nyikae
Common Names: Nyika Climbing Mouse, Nyika African Climbing Mouse (English), Nyikae-Klimmuis (Afrikaans)
Synonyms: Dendromus nyikae (Wroughton, 1909); Dendromus longicaudatus (A. Roberts, 1913); Dendromus (Poemys) angolensis (A. Roberts, 1929); Dendromus nigrifrons angolensis (J. Eric Hill & T. D. Carter, 1941); Dendromus longicaudatus bernardi (Lundholm, 1955)
Taxonomic Note: Evolutionary relationships among taxa with the genus Dendromus have historically been complicated by morphological similarity and overlapping distributions. Previously included under Dendromus melanotis, D. nyikae was subsequently recognised as distinct by Musser and Carleton (2005). Despite this classification, the taxonomic status of the species within the assessment region and its relationship to D. melanotis, remained in question by some until a recent phylogenetic study provided conclusive evidence supporting the elevation of the taxon to species rank (Voelker et al. 2021). Molecular DNA analyses further indicated that D. melanotis and D. nyikae should be classified separately from other Dendromus species under the resurrected genus Poemys (Voelker et al. 2021). This reclassification represents a major revision of African murid taxonomy, as it not only alters generic limits but also highlights deep evolutionary divergence within the group. In addition, Voelker et al. (2021) demonstrated that Poemys comprises six distinct genetic lineages: three that cannot be assigned to any described morphological taxa (sp. indet. 12–14), two corresponding to valid species (P. pecilei and P. nyikae), and one lineage that should be removed from synonymy (P. cf. vulturnus). Members of this clade are primarily distributed from South Africa to Kenya, with one species (P. pecilei) occurring in the Republic of the Congo. These findings underscore the importance of integrative approaches combining molecular and morphological data for resolving cryptic diversity and ensuring accurate taxonomic assignments.
Red List Status: DD– Data Deficient
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.2
Reviewers: Patel, T3.
Institutions: 1Durban Natural Science Museum,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors & Reviwers: MacFadyen, D., Shoeman, C. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
This species exists as an isolated subpopulation in Limpopo Province. While it is widespread throughout the rest of its range and commonly collected in Zimbabwe, surveys have not revealed any further new specimens within the assessment region. It is possible that the few records have been misidentified with other Poemys and/or Dendromus species, particularly P. melanotis s. l. There is a lack of information on the species’ basic biology, ecology, physiology, and distribution within the assessment region. Incomplete distributional datasets remain a significant constraint for both conservation and land-use planning (Freitag et al. 1998). Further molecular work is required to confirm the taxonomic status of the species within the assessment region. For this reason, we list this species as Data Deficient until new evidence is available for reassessment.
Regional population effects: No dispersal is suspected, thus this is suspected to be an isolated population (Skinner & Chimimba 2005).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Richards LR & da Silva JM. 2025. A conservation assessment of Poemys nyikae. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
First described from the Ntchisi region in Malawi, this species occurs widely but patchily throughout southern Africa, extending into the Eastern Arc mountains of Tanzania in the north (Monadjem et al. 2015). It is found in natural moist savannah habitat and montane grasslands with dense, tall stands of grass (Rautenbach 1982; Skinner and Chimimba 2005; Happold 2013). Although widespread throughout Zimbabwe, within the assessment region it has only been collected from the Tzaneen district of the Limpopo province in South Africa (Rautenbach 1982). While recent specimens from Phinda Private Game Reserve in KwaZulu-Natal were provisionally assigned to P. nyikae (Rautenbach et al. 2014), subsequent genetic analyses revealed the specimens to be D. melanotis sensu lato (Solano et al. 2014). It has not been recorded from Eswatini or Lesotho.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): Unknown
Elevation Upper Limit (in metres above sea level): 1800 m asl (Happold and Happold 1989)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
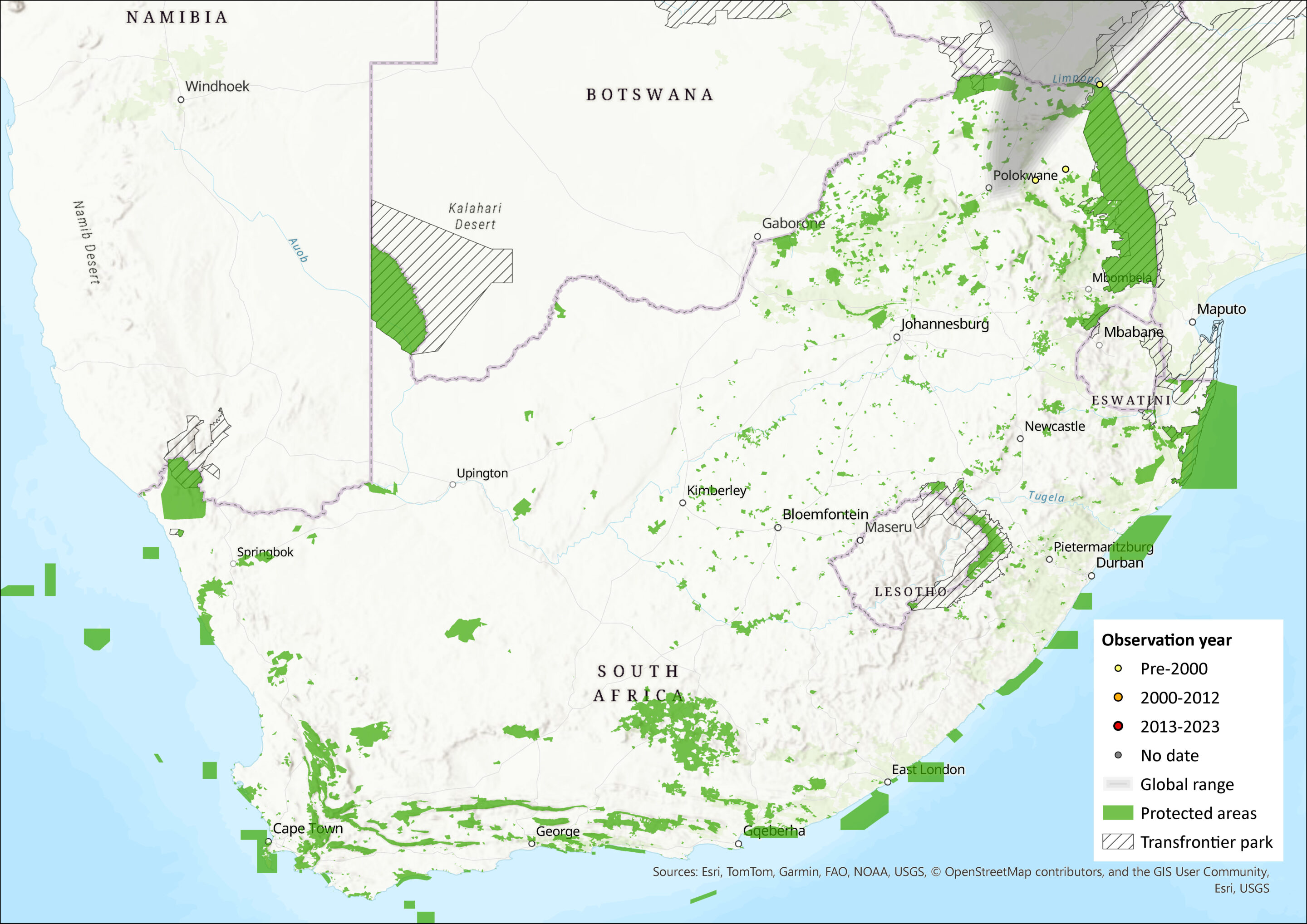
Map
Figure 1. Distribution records for Nyika Climbing Mouse (Poemys nyikae) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To date, no formal studies have examined the potential impacts of climate change, along with associated bioclimatic and environmental shifts, on the biology, ecology, or physiology of this species. The species is more commonly associated with high-altitudinal habitats, including moist savanna woodland and grassland with ample grass cover (Happold 2013). Midgeley and Thuiller (2010) predicted a 30% replacement of grassland-dominated habitats by more arid-adapted or tolerant vegetation types (e.g. thorn-scrub savanna). This may result in the displacement or range contraction of grassland-associated species, that may include P. nyikae within the assessment region. Rodent population declines tend to be more pronounced in habitats experiencing significant anthropogenic transformation or, alternatively, in high-altitude environments where the effects of climate change are likely to be intensified (Wan et al. 2022). Further research is required to assess the potential impacts of climate change on this species within the assessment region.
Population information
It is generally uncommon but can be locally abundant (Happold 2013). Population numbers appear to be positively correlated with grass height and density (Happold and Happold 2013). It is a common species in Zimbabwe but has not been recently recorded within the assessment region.
Population Information
Continuing decline in mature individuals? This cannot be determined at present due to insufficient data on the species’ population dynamics within the assessment region.
Extreme fluctuations in the number of subpopulations: The species is only known from one locality / population within the assessment region.
Continuing decline in number of subpopulations: N/A
All individuals in one subpopulation: Assumed to be as the species is only known from one locality / population within the assessment region.
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: None
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: This cannot be determined at present due to insufficient data on the species’ population dynamics within the assessment region.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: This cannot be determined at present due to insufficient data on the species’ population dynamics within the assessment region.
Probability of extinction in the wild within 100 years: This cannot be determined at present due to insufficient data on the species’ population dynamics within the assessment region.
Population genetics
While this species is thought to be more widespread throughout Africa, within the assessment region, it exists as an isolated subpopulation in Limpopo Province. Given the limited abundance data within the assessment region, the effective population size (Ne) for this subpopulation cannot be estimated. A population genetic study comparing specimens from within the assessment region and beyond should be undertaken to better understand the population structure and diversity within the species.
Habitats and ecology
Although rare, this species can be locally abundant in montane grasslands or Miombo and Mopane woodland with ample grass cover (Kingdon 1974; Rautenbach 1978 Happold and Happold 1986). Specimens from Limpopo were found in dense grass in Mopane woodland at an altitude greater than 1,000 m (Rautenbach 1982). They may also occur marginally in grassland habitats within pine plantations, and they can make use of abandoned beehives (Happold 2013). Happold and Happold 1989 demonstrated a positive correlation in population numbers and grass cover and height.
Information on the diet, reproductive biology, and general habits of this species within the assessment region remains scarce. However, data from extralimital populations suggest that it is omnivorous, primarily consuming vegetation and seeds, along with insects such as beetles (Hanney, 1965; Happold, 2013). It is an adept climber, utilizing its prehensile tail to navigate grass stalks in search of seedheads (Happold, 2013). The species constructs simple burrows consisting of a tunnel leading to a nest chamber lined with vegetation (Hanney, 1965). It is noted for its nail on the fifth hind digit instead of a claw present in most other Climbing mice species.
Ecosystem and cultural services: There are currently no known cultural services or folklores associated with this species. Similar to other small mammals, this species may play a role in regulating invertebrate numbers, seed dispersal, and nutrient cycling. Additionally, it is a valuable prey species and can comprise up to 30% of the diet of African Grass Owls (Tyto capensis) (Happold 2013).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.5. Forest -> Forest – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
2.2. Savanna -> Savanna – Moist |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Unknown |
– |
|
3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist |
– |
Unknown |
– |
|
4.5. Grassland -> Grassland – Subtropical/Tropical Dry |
– |
Suitable |
– |
Life History
Generation Length: Unknown
Age at maturity: female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms) sexes combined: Mean head and body length = 6.68 cm (Happold 2013)
Size at Maturity (in cms): Male: No available data
Longevity: No available data
Average Reproductive Age: No available data
Maximum Size (in cms): 7.80 cm (Happold 2013)
Size at Birth (in cms): No available data
Gestation Time: No available data
Reproductive Periodicity: No information is available for individuals within the assessment region, however, Kingdon (1974) reports reproductively active animals in August and January.
Average Annual Fecundity or Litter Size: The litter size within the assessment region remains unknown; however, nests containing litters of up to eight young have been documented in other areas (De Graaf 1981).
Natural Mortality: likely taken by a variety of predators, including African Grass Owls (Happold and Happold 1989).
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: N/A
Does the species have a free-living larval stage? N/A
Does the species require water for breeding? N/A
Movement Patterns
Movement Patterns: The movement patterns within the assessment region remains poorly understood. However, Happold and Happold (1989) observed a marked decline in population size following changes in grassland structure after burning in Malawi. It is uncertain whether this reduction results from post-burn mortality or from individuals relocating to avoid unsuitable habitat conditions.
Congregatory: No available data, but like other Climbing species (Dedromus and Peomys), family groups are likely to remain together in their burrow.
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
Local Livelihood: Unknown
National Commercial Value: Unknown
International Commercial Value: Unknown
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
It is unknown whether this species faces any threats within the assessment region. It would be expected that disturbances through forestry and agriculture would impact suitable habitat in this localized area in the Limpopo Province. Overgrazing and incorrect fire regimes that reduce vegetation cover may lead to habitat degradation. More research is needed to quantify the potential threats facing this species. Climate change is also expected to reduce Afromontane grassland, further jeopardising the species similar to other Afromontane species (Taylor et al. 2016). The impact of these threats on the population remains to be quantified.
Conservation
Within the assessment area, it is uncertain whether the species occurs in any protected areas as according to our knowledge, no further verified records have been documented since its discovery in the Limpopo province of South Africa. No protected area expansion is possible without further molecular and survey work to clarify its distributional range within the assessment region. Once revised, a reassessment will be needed.
It is important to first understand the status, distribution and habitat requirements within the assessment area prior to the provision of particular management recommendations.
Recommendations for land managers and practitioners:
- Landowners should maintain and/or expand corridors of grassland to offset the impacts of urbanisation and ensure movement and gene flow between populations, especially corridors of natural grassland in agricultural and mining areas.
- Employ correct land management practices to prevent overgrazing and indiscriminate burning.
- Protected areas expansion through provincial biodiversity stewardship programmes.
- Reduce over-stocking on cattle and game ranches.
Research priorities:
- This species is in urgent need of further taxonomic resolution, specifically with historical and new genetic material from South African, Zimbabwean and Malawian populations.
- Determination of population size and trends.
- Understanding local movements and dispersal mechanisms.
- Habitat modelling must be used to determine habitat requirement of this species.
- Research into the reproductive biology and physiology of the species and the potential impacts of climate change associated extreme fluctuations in temperature and precipitation seasonality on the species.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
- Deceased specimens should be submitted to the nearest accredited museum or biological repository for proper documentation and preservation.
Bibliography
De Graaff, G. 1981. The Rodents of Southern Africa: Notes on Their Identification, Distribution, Ecology and Taxonomy. Butterworths, Pretoria.
Freitag, S., Nicholls, A.O. & Van Jaarsveld, A.S. 1998. Dealing with established reserve networks and incomplete distribution data sets in conservation planning. South African Journal of Science, 94(2):79-86.
Hanney, P. 1965. The muridae of Malawi (Africa: Nyasaland). In: Proceedings of the Zoological Society of London 1965, Volume: 146(4), pp. 577-633. Blackwell Publishing Ltd, Oxford, U.K.
Happold, D.C. & Happold, M. 1989. Biogeography of montane small mammals in Malawi, Central Africa. Journal of Biogeography, 1:353-67.
Happold, D.C.D. 2013. Dendromus nyikae Nyika African Climbing Mouse. Pages 181–182 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Mammal Diversity Database. 2025. Poemys nyikae (Wroughton, 1909) Nyika African Climbing Mouse.
https://mammaldiversity.org/taxon/1003723
Midgley, G.F. and Thuiller, W., 2011. Potential responses of terrestrial biodiversity in Southern Africa to anthropogenic climate change. Regional Environmental Change, 11(Suppl 1): 127-135.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Rautenbach, A., Dickerson, T. & Schoeman, M.C. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Rautenbach, I.L. 1978. Ecological distribution of the mammals of the Transvaal (Vertebrata: Mamalia). Annals of the Transvaal Museum, 31(10):131-56.
Rautenbach, I.L. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Solano, E., Taylor, P.J., Rautenbach, A., Ropiquet, A. & Castiglia, R. 2014. Cryptic speciation and chromosomal repatterning in the South African climbing mice Dendromus (Rodentia, Nesomyidae). PloS one 9: e88799.
Taylor, P.J, Nengovhela, A., Linden, J. & Baxter, R.M. 2016. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia 80(4):359-75.
Wan, X., Yan, C., Wang, Z. and Zhang, Z. 2022. Sustained population decline of rodents is linked to accelerated climate warming and human disturbance. BMC Ecology and Evolution, 22(1), p.102.