Natal Red Duiker
Cephalophus natalensis

2025 Red list status
Near Threatened
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Cephalophus natalensis – A. Smith, 1834
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Cephalophus – natalensis
Common Names: Natal Red Duiker, Natal Duiker, Red Duiker (English), Rooiduiker (Afrikaans), Ipunzi ebovu (Ndebele), Mungulwi, Kutsoa (Sepedi), Phuthi e kgubedu (Sesotho), Umsumbi, Imphunzi, Umsumpe (Swati), Mhunti (Tsonga), Phithi, Tshipiti (Venda), Impunzi (Xhosa), Umsumpe, Umkhumbi (Zulu)
Synonyms: No Synonyms
Taxonomic Note:
Although Harvey’s Duiker (C. harveyi) has occasionally been included within C. natalensis (Grubb & Groves 2001; Grubb 2005), we consider these species distinct, following Kingdon (1982), East (1999), and Hoffman and Bowland (2013). Meester et al. (1986) listed two subspecies, including C. n. natalensis from KwaZulu-Natal (KZN), eastern Mpumalanga and southern Mozambique, and C. n. robertsi Rothschild 1906 from Mozambique and the regions north of the Limpopo River (Skinner & Chimimba 2005).
Red List Status: Near Threatened – NT, B2ab(ii,v) (IUCN version 3.1)
Assessment Information
Assessors: Ehlers Smith, Y.1 & da Silva, J. M.2
Reviewer: Shrader, AM.3
Contributor: Patel, T.4
Institutions: 1Ezemvelo KZN Wildlife, 2South African National Biodiversity Institute, 3University of Pretoria, South Africa, 4Endangered Wildlife Trust
Previous Assessors & Reviewers: Ehlers Smith, Y., Williams, S., Relton, C. & Child, MF.
Previous Contributors: Rushworth, I. & Page-Nicholson, S.
Assessment Rationale
This species is restricted to forest patches within northeastern South Africa and Eswatini. They can occur at densities as high as 1 individual / ha. In KZN, there was an estimated 3,046–4,210 individuals in protected areas alone, with the largest grouping of 1,666–2,150 individuals occurring in iSimangaliso Wetland Park (2012–2014 counts; Ezemvelo KZN Wildlife (EKZNW) unpubl. data). Recent counts indicate 2,304 individuals (EKZNW unpubl. data 2025), indicating that the population in iSimangaliso Wetland Park has remained stable since the previous assessment. While no other provincial estimates are available, they are regularly recorded on camera traps in the Soutpansberg Mountains of Limpopo and the Mariepskop forests of Mpumalanga, including on private lands outside protected areas (S. Williams unpubl. data). Reintroductions are probably a successful conservation intervention for this species, although caution should be taken when moving animals. For example, genetics are sourced from within the province for EKZNW protected areas. Reintroduced individuals from the 1980/90s are still present in areas of southern KZN and are slowly moving into adjacent farmlands (Y. Ehlers-Smith unpubl. data). The estimated area of occupancy, using remaining (South African National Land Cover, 2022) forest patches within the extent of occurrence, is 1, 149 km2. This yields a total mature population size of 11,490–57,450 animals based on minimum and maximum densities. This may be an overestimate as not all patches are suitable or will be occupied.
As long as habitat is conserved through protected area expansion and biodiversity stewardship schemes, this species should continue to be stable within protected areas. However, suitable forest habitat continues to be lost in all areas of its range. For example, 20% of woodland cover was lost from 1990 to 2006 in the Soutpansberg Mountains region due to fuelwood extraction and pine/Eucalyptus plantations; and, in just six years (2005–2011), 7.6% of KZN’s natural habitat was lost (1.3% / annum), due primarily to agriculture expansion. Poaching pressure may also cause local declines. While the area of occupancy (AOO) of 1,149 km2 may be an underestimate as it does not include thicket habitats, not all patches will be occupied due to poaching pressure. Thus, we list the species as Near Threatened B2ab(ii,v) due to inferred ongoing loss of suitable habitat and severe hunting pressure outside of protected areas. Suitable habitat is severely fragmented by land conversion and degradation. Further field surveys from across its range, especially outside protected areas, are needed to more accurately measure population size through more comprehensive density and occupancy data. Long-term monitoring should also be used to assess subpopulation trends, especially outside protected areas. This species should be reassessed as such data become available.
Regional population effects: There are discrete subpopulations in Limpopo and Mpumalanga provinces. Although the KZN subpopulation occurs in highly fragmented forest patches, there are likely to be linkages and dispersal routes with Mozambique, such that the rescue effect is possible for KZN, but its rarity in southern Mozambique means no significant rescue effects are anticipated. Continued research into the dispersal capabilities and current distribution is required to confirm this assumption.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Ehlers Smith Y & da Silva JM. 2025. A conservation assessment of Cephalophus natalensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Natal Red Duiker formerly occurred widely in coastal and riverine forests and thickets, escarpments and montane forests from southeastern Tanzania southwards to northeastern KZN in South Africa (East 1999; Hoffmann & Bowland 2013) but currently has a disjunct distribution. There are no confirmed records of this species from Zimbabwe or Zambia (IUCN SSC Antelope Specialist Group 2016). Although this species is restricted to specialised habitats of forests and dense thickets, and fragmentation of its distribution is a natural phenomenon, this species has experienced extensive range contraction because of habitat alteration (Skinner & Chimimba 2005). In Mozambique, this species has a discontinuous distribution north and south of the Zambezi River. There are occurrence records in several isolated inland areas towards the Maputo Elephant Game Reserve (Skinner & Chimimba 2005), but their current presence in southern Mozambique needs to be confirmed. Previous records of this species in Zambia and Malawi (East 1999; Skinner & Chimimba 2005) are now considered to belong to Harvey’s Duiker (Cephalophus harveyi; Hoffmann & Bowland 2013).
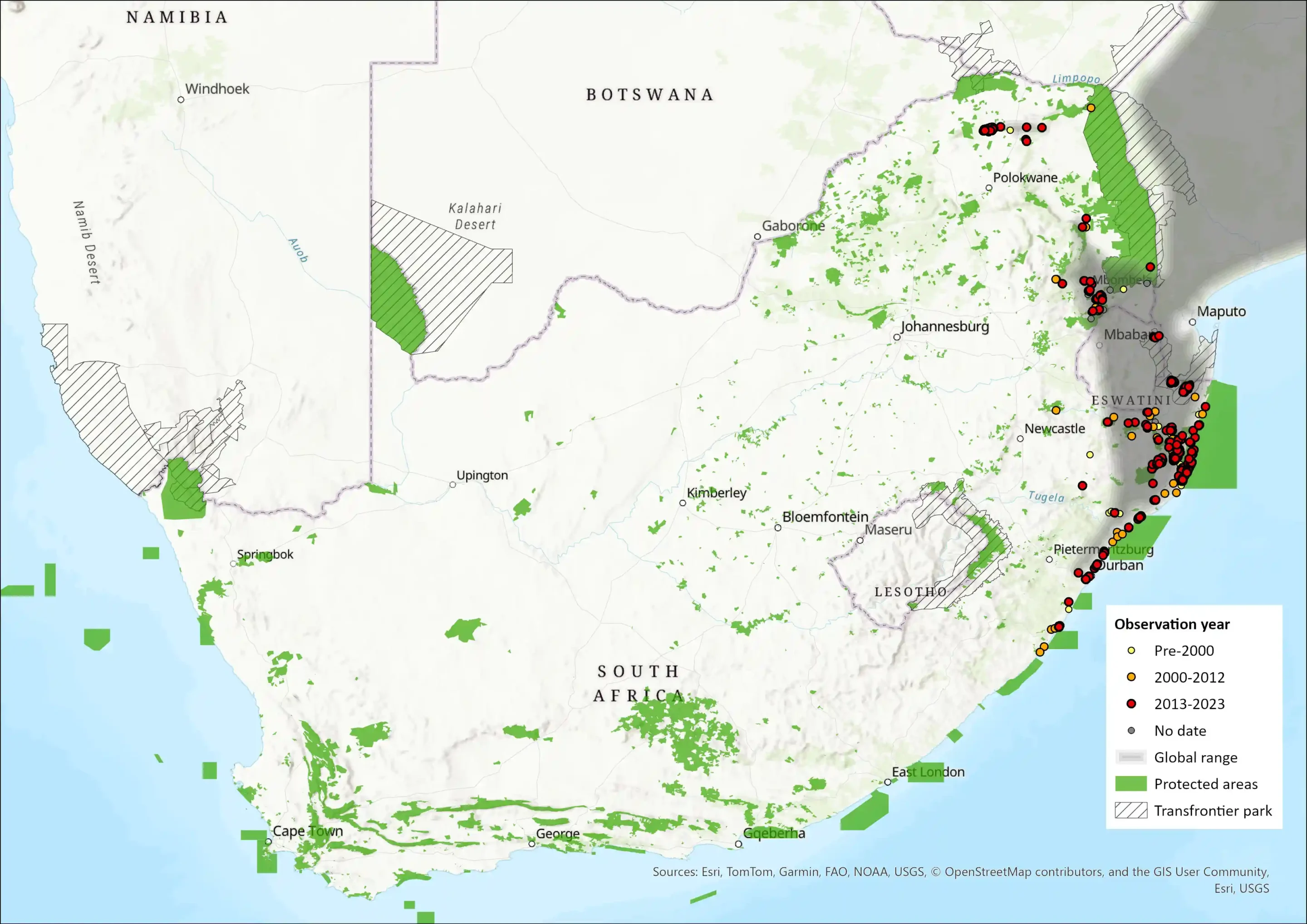
Within the assessment region, the species occurs along the eastern regions of South Africa, from the far northern stretches of KZN along the coastal areas in forest and bushveld, as well as in forest fragments in Mpumalanga and Limpopo (Soutpansberg) provinces. Although no records are available, they occur in scattered pockets of riverine thickets and forests of the lowveld region in Eswatini (Monadjem 1998). Similarly, known subpopulations in South Africa are currently very fragmented due to the patchy nature of suitable habitat. Until recent times, their distribution extended as far south as Pondoland (Eastern Cape – southern KZN) (Fitzsimons 1920; du Plessis 1969 and expanded across much larger proportions of Limpopo and Mpumalanga (Skinner & Chimimba 2005). The estimated AOO, using remaining (South African National Land Cover, 2022) forest patches within the extent of occurrence, is 1, 149 km2. This may be overestimated as not all patches are suitable or will be occupied. They do not occur in southern Pondoland Scarp forests and probably do not occur in mist belt forests (Y. Ehlers- mith unpubl. data). In KZN, they are probably restricted to lowland coastal, dune and sand forests where there is a grassland-forest mosaic where they move between patches as they often feed on the edges of forests but will ruminate in dense cover (Y. Ehlers-Smith unpubl. data). Further habitat selection data are needed to more accurately estimate AOO.
In the late 1980s, several reintroductions had been made into their former range (Bourquin & van Rensburg 1984; Bowland 1990). For example, they were reintroduced to Mpenjati Nature Reserve, as well as San Lameer Golf Estate, a farm near Upper Melville and in Umzumbe during the late 1980s/early 1990s. While recent camera-trap surveys (2014–2016; Y. Ehlers-Smith unpubl. data) failed to detect Natal Red Duiker on Mpenjati, they appear to have radiated outwards as they have been photographed at low density in nearby farms (for example, forest patches on sugar cane farms) and villages (Y. Ehlers-Smith unpubl. data). The viability of these reintroductions is currently being assessed.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Natal Red Duiker (Cephalophus natalensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
To explore the potential impacts of climate change, a comparison between the current and the future predictive niche was made using Bioclim variables, specifically for South Africa. The current MaxENT (Maximum Entropy) species distribution model indicates areas of high suitability (0.98) concentrated along the eastern coastal belt and escarpment, particularly in KwaZulu-Natal and Eastern Cape. However, the future projection incorporating future climatic conditions indicates that maximum suitability drops to 0.85, with notable shift towards inland areas and northern extent. The difference between the two models indicates a incongruity between area expansion and quality reduction.
Important climatic variables within the current niche include: (bio2 – Mean Diurnal Range 31.4%) daily temperature fluctuation is the primary limiting factor; (bio6 – Min Temperature of Coldest Month, 7.0%) indicating a cold temperature tolerance threshold; (bio17 – Precipitation of Driest Quarter, 10.0%) dry season moisture availability is a limitation. For the future niche the three largest contributors include: (bio6 -Min Temperature of Coldest Month 27.3%) increasing importance of cold limits; (bio2 – Mean Diurnal Range 14.0%) remains important but less dominant, and (bio12 -Annual Precipitation, 8.7%) overall rainfall becomes a more limiting factor.
Overall, we are predicting a range shift rather than simple contraction. The species may find newly suitable areas (possibly at higher elevations or more southern latitudes) while losing optimal core habitat. Modeling suggests habitat degradation, whereby the total area increases slightly, but the quality of that habitat decreases substantially, with fewer areas of high suitability. Overall, the results suggest increased climate vulnerability, whereby the shift from diurnal temperature range (bio2) to minimum temperature (bio6) as the primary driver indicates increasing importance of extreme cold events, suggesting potential vulnerability to climate variability. The emergence of annual and monthly precipitation variables in future models indicates water availability will become increasingly limiting, suggesting precipitation stress. The species faces a moderate climate change threat with habitat degradation rather than dramatic range loss being the primary concern under projected climate scenarios.
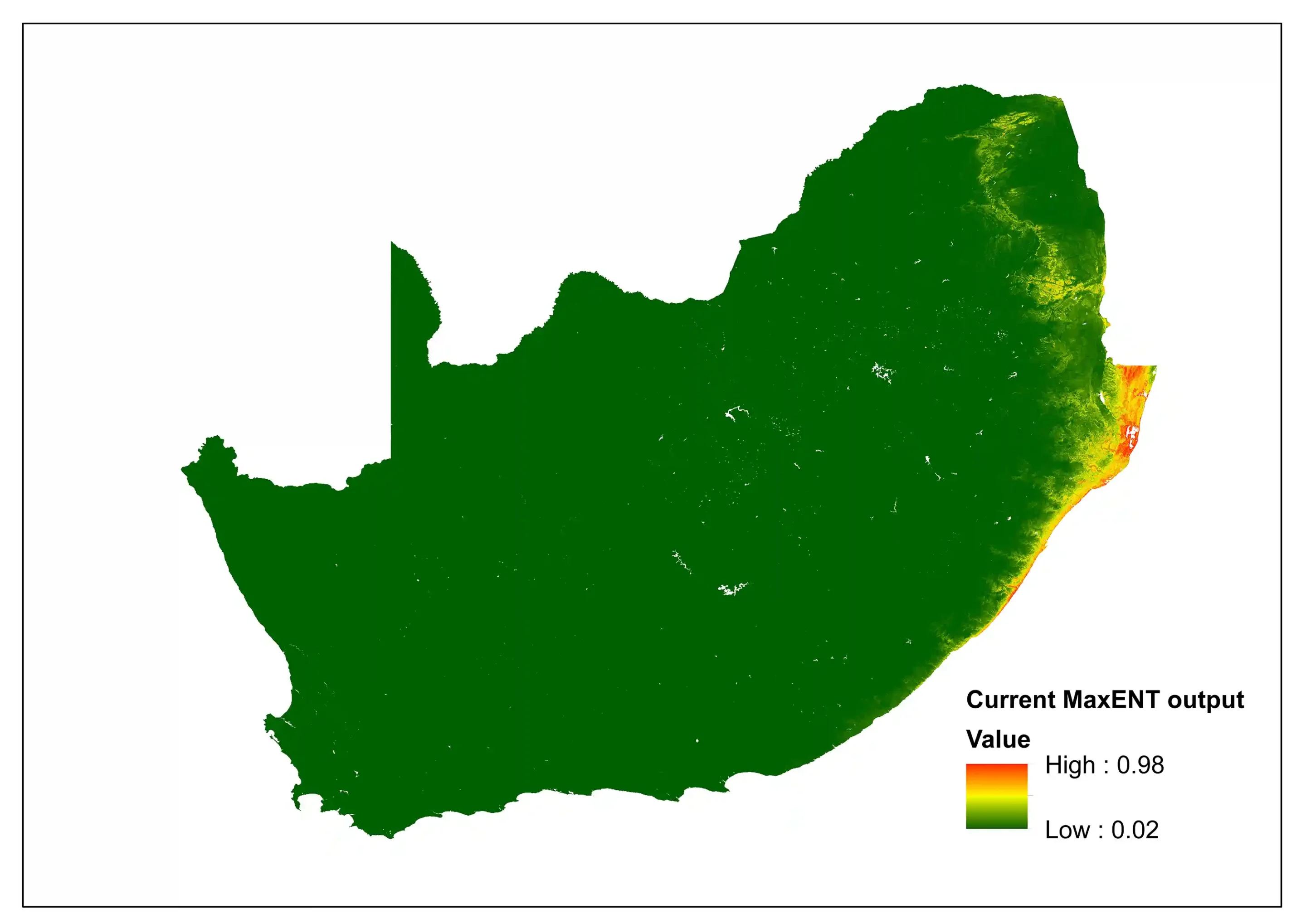 Figure 2: A MaxEnt (Maximum Entropy) species distribution map for Cephalophus natalensis based on contemporary environmental conditions. The higher the value, the higher the likely distribution of the species.
Figure 2: A MaxEnt (Maximum Entropy) species distribution map for Cephalophus natalensis based on contemporary environmental conditions. The higher the value, the higher the likely distribution of the species.
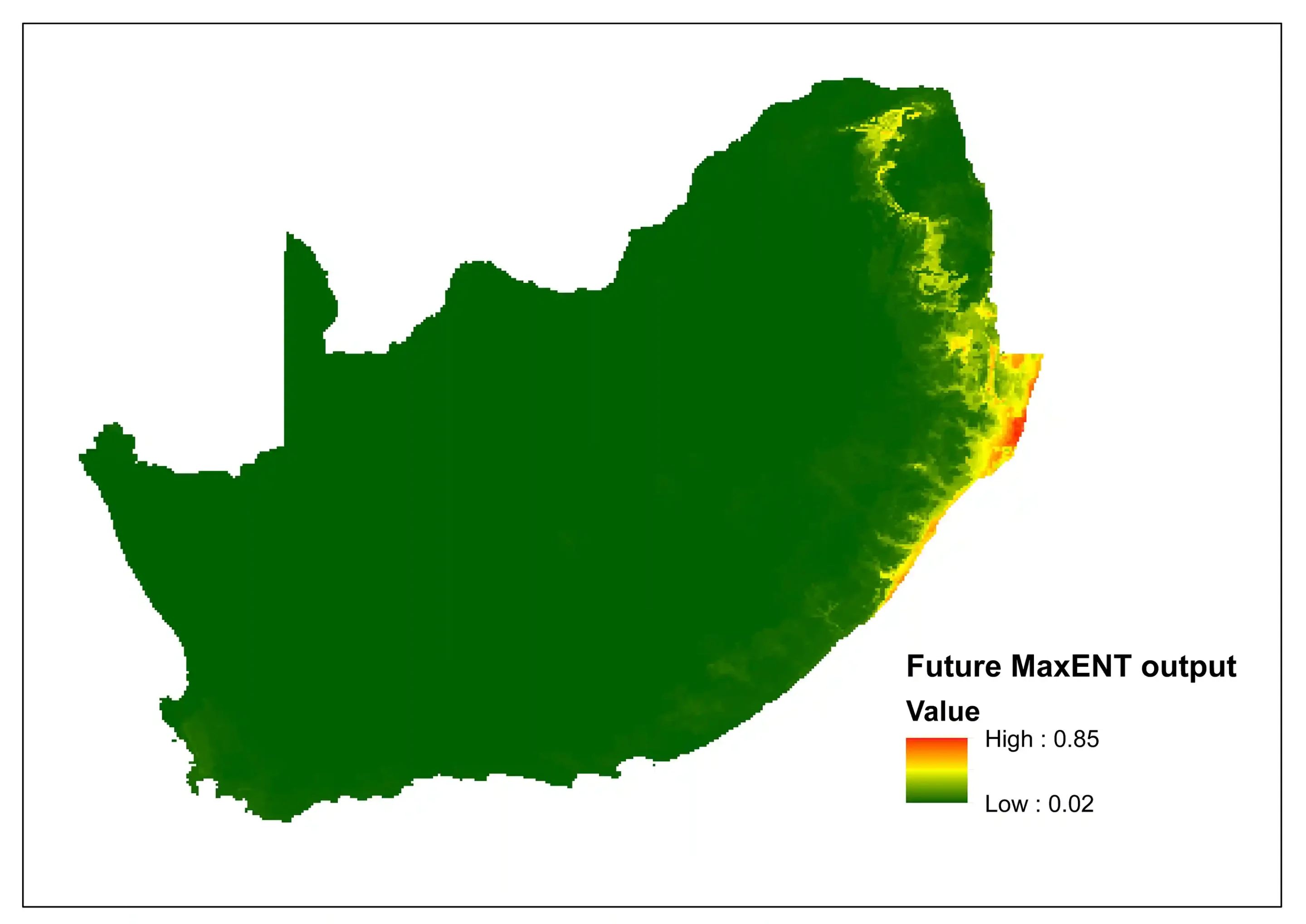 Figure 3: A MaxEnt (Maximum Entropy) species distribution map for Cephalophus natalensis based on predicted future climate scenarios. The higher the value, the higher the likely distribution of the species.
Figure 3: A MaxEnt (Maximum Entropy) species distribution map for Cephalophus natalensis based on predicted future climate scenarios. The higher the value, the higher the likely distribution of the species.
Population
The Natal Red Duiker is known to reach relatively high numbers in suitable habitats (Bowland 1997). For example, in St Lucia, KZN, this species was recorded at densities ranging from 1–2 animals / ha (Bowland 1990). However, in less favourable areas, such as smaller forest clumps, estimates of 0.2–0.4 animals / ha were recorded (Bowland 1990). Although a global population estimate of 42,000 was documented by East (1999), due to the cryptic nature of this species, this was expected to be an underestimate. Globally, the population trend of this species is believed to be declining. However, wildlife recovery across much of Mozambique is likely to include this species (East 1999; Hoffmann & Bowland 2013), although the Natal Red Duiker is still considered to be highly threatened in Mozambique, hampering potential dispersal into South Africa.
Within the assessment region, it is suspected that the population has declined significantly following large-scale decline in the habitat (through timber plantations and cane sugar crops) in the past but appears to have stabilised (Rowe-Rowe 1994). However, mining in northern KZN has had a negative impact on local populations. The population is largely confined to extensive protected areas (for example, iSimangaliso Wetland Park; Ramesh et al. 2016) and is unlikely to decline in the future within these protected areas. There are suspected to be 6–9 major subpopulations although we are missing data from several regions in KZN, Limpopo and Mpumalanga. In 2004, it was estimated that there were four major subpopulations where the iSimangaliso Wetland Park contained 1,000 animals and Hluhluwe-iMfolozi Park contained 300 animals (Friedmann & Daly 2004). An assessment conducted between 2011 and 2012 estimated at least 4,210 individuals in seven formally protected areas in KZN, with 2,150 animals in the largest subpopulation in iSimangaliso Wetland Park and 1,200 in Mkhuze Game Reserve (EKZNW unpubl. data). More recently (2013/14), there were estimated to be 3,046 animals in eight formally protected areas in KZN, the largest of which is iSimangaliso Wetland Park with 1,666 animals and 600 on Mkhuze Game Reserve (EKZNW unpubl. data). ISimangaliso Wetland Park now has 2,304 animals (EKZNW unpubl. Data 2025). Generation length is estimated as 5.2 years (Pacifici et al. 2013), yielding a three–generation period of 15.5 years (2010–2025). While no reliable long-term data are available to measure population trends over this period, estimates from 2011 and 2024 indicate that the major subpopulation in iSimangaliso Wetland Park has at least remained stable over approximately three generations. Overall, using the AOO estimate of 1,149 km2 and maximum/minimum densities (20–100 animals / km2), we estimate a population of 22,980–114,900 animals. As they mainly occur solitarily or a female with her offspring or in small groups (3–5 individuals) in loose association (Skinner & Chimimba 2005), we assume a c. 50% mature population structure, which yields 11,490–57,450 mature animals. Research to estimate population size in the southern extent of its range is underway (Y. Ehlers-Smith unpubl. data).
Individuals are still present in areas of reintroduction in southern KZN and are slowly moving into adjacent areas. For example, they were introduced in Mpenjati Nature Reserve in the late 1980s/early 1990s, but have been found on a forest patch within a sugar cane farm as they have radiated outwards (Y. Ehlers-Smith unpubl. data). In the Soutpansberg region of the Limpopo Province, this species is common along the forested southern slopes (Power 2002) and are suspected to be numerous on private lands. However, in many areas, subpopulations outside of protected areas are suspected to be declining due to ongoing habitat loss, bushmeat hunting and illegal sport hunting with dogs. More research is necessary to determine population size and trends in such areas.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | Severe hunting pressure outside of protected areas. |
Current population trend: Declining from ongoing habitat loss.
Number of mature individuals in population: 11,490–57,450
Number of individuals in largest subpopulation: There are currently 2,304 individuals present in iSimangaliso Wetland Park.
Number of subpopulations: At least 6–9 major subpopulations.
Severely fragmented: Yes. Suitable forest and thicket habitats are isolated from each due to habitat conversion and degradation.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: No
Number of subpopulations: 9
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While phylogenetic studies examining the relationships within Cephalophinae have been undertaken (van Vuuren & Robinson 2001; Johnston & Anthony 2012), there has been no fine-scale examination of the population genetic structure and diversity within this species. While it is thought to exist in fairly high numbers and exists inside and outside of protected areas, the species is a forest/thicket specialist and its habitat has been extensively altered, resulting in at least three geographically (and presumed genetically) isolated subpopulations (two subpopulations in KZN – Durban & northern coastal KZN and Southern coastal KZN – and one in Mpumalanga). These subpopulations are therefore assumed to be genetically distinct.
Because the sizes of each subpopulation are unknown and estimates for the species as a whole vary widely (e.g., between 18,000-90,000 within the assessment region), it is difficult to hazard a confident estimate of effective population size (Ne). Assuming conservatively that 18,000 animals exist, and these are distributed equally between subpopulations, Ne for each could be between 600- 1800, which would suggest each subpopulation is genetically stable/healthy. A comprehensive assessment of the population genetic structure and diversity within the assessment region is needed to verify the species’ extent of connectivity and overall genetic health, including estimate of effective population size.
Considering C. natalensis and C. harveyi are only recently differentiated and known to hybridise where they co-occur (van Vuuren & Robinson 2001; Johnston & Anthony 2012), it is recommended that the translocation of animals mimic, as much as possible in their natural movement (dispersal pathways). Accordingly, animals should only be moved to and from sites that are closest to the introduction site. Moreover, there should be no translocations of hybrid individuals outside areas where they do not naturally co-occur.
Habitats and ecology
Across their range, Natal Red Duikers occur within indigenous forests and dense thickets, including coastal, riverine, swamp and montane slope forests and forest clumps, as well as wooded ravines (Bowland 1997; Skinner & Chimimba 2005). They have been recorded at elevations of up to 200 m asl (Rowe-Rowe 1994). Natal Red Duikers occasionally wander into more open grasslands to forage, but will immediately return to the shelter of forested regions at any sign of disturbance (Bowland 1997).
Heinichen (1972) recorded predominantly shrubs, fruits and dry leaves in the diet of the Red Duiker, and noted that they frequently swallow whole fruits. Recently fallen fruits, flowers and leaves from the forest canopy constitute a large proportion of their diet, and active browsing from live vegetation occurs less commonly (Bowland 1997), although they will browse on the fine stems of shrubs growing low to the ground (Skinner & Chimimba 2005). This species is considered a concentrate selector with specialised adaptations for efficient absorption, thus does not tolerate large quantities of fibre in its diet (Faurie & Perrin 1995).
Natal Red Duikers exhibit sedentary behaviour for a considerable portion of the day, and are most active around dawn and dusk (Bowland & Perrin 1995). This sedentary behaviour is a function of their small body size and ruminant digestive system (Bowland & Perrin 1995). Although core areas of habitat remain stable, home ranges vary temporally, and Natal Red Duikers are more active in higher quality habitats (Bowland & Perrin 1995). This species is not territorial and some individual home ranges overlap by up to 100%, however, occasionally individuals exhibit temporal separation (Bowland & Perrin 1995). They are solitary, and do not form lasting associations with one another, aside from females and their young (Bowland & Perrin 1995). This species is a non-seasonal breeder, producing a single lamb following a gestation period of approximately 210 days. A calving interval of 236 days (range = 222–273 days, n = 5; Spence 1991) has been estimated, which is similar to that of the Common (Sylvicapra grimmia) and Blue Duiker (Philantomba monticola) (Skinner & Chimimba 2005).
Ecosystem and cultural services: This is a flagship species for forest habitats and is a valuable seed disperser, feeding on fallen wild fruit and potentially browsing on the lower branches of Grewia spp. They are also important prey species for Leopards (Panthera pardus), as has been documented on the southern slopes of the Soutpansberg Mountains (Power 2002).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
Life History
Generation Length: 5.2 years (Pacifici et al. 2013)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Although this species is used for trophy hunting, trade is unlikely to have any effect on the population as long as permits are well regulated. This species is targeted by international trophy hunters, as one of the “Tiny 10” small antelope. It is also hunted as bushmeat extensively throughout its range and is common in bushmeat markets (IUCN SSC Antelope Specialist Group 2016), the effects of which may cause local declines or extinctions within the assessment region. In Mozambique, this species was recognised as a preferred species for illegal bushmeat hunting (de Boer & Baquete 1998).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Illegal bushmeat hunting | Yes | International and national trophy hunting. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | – | – | – |
| 15. Sport hunting/specimen collecting | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Trophy hunting
Threats
Due to its strict habitat specialty, the Natal Red Duiker is vulnerable to habitat modification, and, as a result, has disappeared from much of its historic range. It has experienced extensive habitat loss, due to expanding development of property, subsistence agriculture and commercial timber plantations (Bowland 1997). Large proportions of their former habitat in the Limpopo and Mpumalanga provinces have been altered. In the inland tropical forest areas, large proportions of mesic natural forests have been cleared for plantations, and in the drier regions for agriculture, especially for sugar cane crops. Sand forest habitat continues to be lost west of False Bay Park, KZN, from pineapple crops (I. Rushworth pers. comm. 2016). Afforestation with exotic timber plantations and crop agriculture is continuing to reduce habitat for this species. The decline in habitat in the coastal forest of northern KZN has previously been estimated to be more than 70% (Mathias & Bourquin 1984). Expanding urban development along the KZN coastline is likely to become an increasing threat in the future, as much of this development is encroaching on suitable forest and dispersal routes of Natal Red Duiker habitats.
Additionally, this species is vulnerable to hunting pressure; as with the Blue Duiker (Philantomba monticola) hunting with dogs is a threat to all small antelopes (sensu Grey-Ross et al. 2010). Informal or bushmeat hunting has the potential to cause local declines or extinctions too. There is suspected to be severe pressure from sport hunting with dogs and bushmeat hunting outside of protected areas; for example, in the Maputaland area of KZN (I. Rushworth pers. comm. 2016).
Ineffective management of livestock and game farms where overgrazing is common does not appear to have a major detrimental impact on Natal Red Duikers, as overgrazing tends to lead to an increase in the density of woody plants (bush thickening), which is actually beneficial to forest duikers that have a preference for shelter and browse resources.
Habitat trend: Declining in area. In KZN, there was a 20.4% loss of natural habitat from 1994 to 2011, with an average loss of 1.2% per annum (Jewitt et al. 2015). Worryingly, in just six years (2005–2011), 7.6% (7,217 km2) of natural habitat was lost (1.3% per annum), due primarily to agriculture (5.2% increase; 4,962 km2), but also plantations, built environments and settlements, mines and dams (Jewitt et al. 2015). Similarly, in the Soutpansberg Mountains of Limpopo, 20% of woodland cover was lost from 1990 to 2006 due to fuelwood extraction and pine/Eucalyptus plantations (Munyati & Kabanda 2009).
Conservation
The Natal Red Duiker is present within a number of South Africa’s protected areas, including iSimangaliso Wetland Park, Hluhluwe-iMfolozi Park, Ndumo Game Reserve, Enseleni Nature Reserve, Kenneth Stainbank Nature Reserve and Tembe Elephant Park in KZN (East 1999; Hoffmann & Bowland 2013), Blyde River Nature Reserve in Mpumalanga, Happy Rest Nature Reserve and Luvhondo Private Nature Reserve in Limpopo Province (S. Williams unpubl. data). Much of the habitat alteration has already taken place and much of what habitat remains is currently protected in existing reserves. Some of the previous pressure that was exerted on the species may be reduced as some communities convert areas to conservation (for example, around Tembe Elephant Reserve). However, ongoing habitat loss and poaching pressure outside protected areas should be mitigated through further protected area expansion, both formally, such as through transfrontier areas (Smith et al. 2008); and through biodiversity stewardship programmes. Landowners should also create conservancies to sustain functioning and protected subpopulations of this species and to connect habitats, especially along the coast. As the species causes habitat degradation outside of its natural range, regulation of translocation is also required to prevent extralimital introduction. However, reintroduction into suitable areas within the natural range should be continued and documented to improve reintroduction technique.
Recommendations for land managers and practitioners:
- Continued monitoring of the subpopulations within existing conservation areas.
- Enforcement of translocation regulations.
Research priorities:
Current ongoing research by the University of KZN in collaboration with Ezemvelo KZN Wildlife includes the impacts of changing land-use on biodiversity, particularly mammals such as Natal Red Duiker, using camera trapping to investigate the metapopulation dynamics of forest mammals in the fragmented sub-tropical coastal forests of southern KZN.
- Recent population sizes and trends from KZN, Mpumalanga and Limpopo protected areas; and surveys to improve the understanding of population estimates of various subpopulations.
- Collate evidence for successful reintroductions to improve translocation success.
- Occupancy and subpopulation trends on private lands.
- Further taxonomic work is needed to investigate the status of this species relative to Harvey’s Red Duiker.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Create conservancies to connect habitat patches.
- Report illegal hunting to provincial conservation authorities.
Bibliography
Bourquin O, van Rensburg J. 1984. Vertebrates of the Vernon Crookes Nature Reserve: additional and confirming records. Lammergeyer 34: 59-61.
Bowland AE, Perrin MR. 1995. Temporal and spatial patterns in blue duikers Philatomba monticola and red duikers Cephalophus natalensis. Journal of Zoology 237: 487-498.
Bowland AE. 1997. Red duiker Cephalophus natalensis. In: Mills G, Hes L (ed.), The Complete Book of Southern African Mammals, pp. 260. Struik Publishers, Cape Town, South Africa.
Bowland, A.E. 1990. The ecology and conservation of blue duiker and red duiker in Natal. Ph.D. Thesis, University of Natal.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Faurie AS, Perrin MR. 1995. Rumen morphology and volatile fatty acid production in the blue duiker (Cephalophus monticola) and the red duiker (Cephalophus natalensis). Zeitschrift für Säugetierkunde 60: 73-84.
Fitzsimons FW. 1920. The Natural History of South Africa. Longmans, Green and Co., London, UK.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Grey-Ross R, Downs CT, Kirkman K. 2010. An assessment of illegal hunting on farmland in KwaZulu-Natal, South Africa: implications for oribi (Ourebia ourebi) conservation. South African Journal of Wildlife Research 40: 43-52.
Grubb, P. 2005. Artiodactyla. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World. A Taxonomic and Geographic Reference (3rd ed), pp. 637–722. Johns Hopkins University Press, Baltimore, USA.
Grubb, P. and Groves, C.P. 2001. Revision and Classification of the Cephalophinae. In: V.J. Wilson (ed.), Duikers of Africa: Masters of the African Floor, pp. 703-728. Chipangali Wildlife Trust, Bulawayo,Zimbabwe.
Heinichen IG. 1972. Preliminary notes on the Suni, Nesotragus moschatus and Red Duiker, Cephalophus natalensis. Zoologica Africana 7: 157-165.
Hoffmann, M. and Bowland, A.E. 2013. Cephalophus natalensis Natal Red Duiker. In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. Volume VI: Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer and Bovids, pp. 258-261. Bloomsbury, London, UK.
IUCN SSC Antelope Specialist Group. 2016. Cephalophus natalensis. The IUCN Red List of Threatened Species 2016: e.T4144A50183272. Available at: http://dx.doi.org/10.2305/IUCN.UK.2016-1.RLTS.T4144A50183272.en..
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kingdon, J. 1982. East African Mammals: An Atlas of Evolution in Africa. Academic Press, London, UK.
Mathias I, Bourquin O. 1984. Capture of blue duiker in Natal. Lammergeyer 33: 30-34.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Munyati, C. and Kabanda, T.A. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9(1): 41–56.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Power RJ. 2002. Prey selection of leopards Panthera pardus in the Soutpansberg, Limpopo Province, and the utilisation options for this population. Mammal Research Institute, University of Pretoria, Pretoria.
Ramesh T, Kalle R, Rosenlund H, Downs CT. 2016. Native habitat and protected area size matters: Preserving mammalian assemblages in the Maputaland Conservation Unit of South Africa. Forest Ecology and Management 360: 20-29.
Rowe-Rowe, D. T. 1994. The ungulates of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Spence JM. 1991. Breeding duikers at Tygerberg Zoopark. Pan-African Association of Zoological Gardens Bulletin 1: 5-7.
de Boer WF, Baquete DS. 1998. Natural resource use, crop damage and attitudes of rural people in the vicinity of the Maputo Elephant Reserve, Mozambique. Environmental Conservation 25: 208-218.
de Vos V. 1979. Extraordinary jumping ability of the Red Forest Duiker Cephalophus natalensis. Koedoe 22: 217.
du Plessis SF. 1969. The past and present geographical distribution of the Perissodactyla and Artiodactyla in southern Africa. M.Sc. dissertation. University of Pretoria, Pretoria, South Africa.