
Natal Long-fingered Bat
Miniopterus natalensis

2025 Red list status
Near threatened
Regional Population Trend
Declining
Change compared
to 2016
Uplisted
Overview
Miniopterus natalensis – (A. Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MINIOPTERIDAE – Miniopterus – natalensis
Common Names: Natal Long-fingered Bat, Natal Clinging Bat (English)
Synonyms: Miniopterus natalensis ssp. arenarius Heller, 1912
Taxonomic Note:
The currently recognised Miniopterus natalensis was previously included as a subspecies of M. schreibersii, but the latter is now restricted to North Africa and Europe (Appleton et al. 2004), with M. natalensis occurring in South Africa (Miller-Butterworth et al. 2005). Since the recognition of M. villiersi in West Africa (Fahr et al. 2006), and M. mossambicus from Mozambique, Malawi and the east of Zimbabwe (Monadjem et al. 2013) as separate species, and until specimens from the rest of the sub-region are re-examined, M. natalensis is considered potentially endemic or near endemic to the southern Africa.
Red List Status: NT – Near Threatened A4c (IUCN version 3.1)
Assessment Information
Assessors: Lötter, C.1, Richardson, E.2, Balona, J.3 & da Silva, J.M.4
Reviewers: de Villiers, M.5
Institutions: 1Inkululeko Wildlife Services (Pty) Ltd, 2Independent Consultant at Richardson & Peplow Environmental, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5CapeNature
Previous Assessors and Reviewers: MacEwan, K., Richards, L., Cohen, L., Jacobs, D., Monadjem, A., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Raimondo, D. & Child, M.F.
Assessment Rationale
In view of the several taxonomic revisions of Miniopterus in recent years M. natalensis is now likely endemic or near endemic to the assessment region. It is probably experiencing declines due to loss of roost sites in caves and old mine tunnels, loss of foraging habitat due to conversion of natural areas for agriculture and urbanisation, and mortalities from collisions with wind turbines. The Natal Long-fingered Bat is the third most frequently killed bat species at operational wind farms in South Africa (Aronson 2022), and the numbers of proposed and operational wind farms are increasing rapidly in the country. Wind farms are permanent structures and those which are located near important roost sites, or which intercept known or unknown migration routes of this species (such as between the De Hoop and Montagu caves) could cause high numbers of fatalities, disrupt migration, and thus pose a major threat to this species. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to established colonies of this species in old mine tunnels. Therefore, systematic monitoring of colony sizes and trends is needed, and the status of this species must be reviewed every few years or more often. On this basis, the species has been uplisted from Least Concern to Near Threatened A4c.
Regional population effects: Although this is a migratory species and thus probably constitutes one population in the subregion, it is quite likely also endemic or near endemic to the southern Africa.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Uplisted based on A4c. It is inferred that the population has already been reduced by 30% based on habitat loss due to wind farms. It is suspected that this decline will be ongoing based on a decline in habitat quality from wind farms and the closure of old mine tunnels.
Red List Index
Red List Index: Uplisted.
Recommended citation: Lötter C, Richardson E, Balona J & da Silva JM. 2025. A conservation assessment of Miniopterus natalensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species occurs widely within southern Africa, but with more records in the southern and eastern parts than in the arid west (Monadjem et al. 2020). It has been recorded from the southern tip of South Africa east and northwards through much of the country and neighbouring Lesotho and Eswatini, occurring in all nine South African provinces. Calls of this species have been recorded near Alexander Bay (Inkululeko Wildlife Services unpubl. data). It also occurs widely in Namibia and southern and western Angola but appears absent from much of the Kalahari (Monadjem et al. 2020). The type specimen is from Durban, South Africa (BM 1848.6.12.19) (Monadjem et al. 2020). Because of frequent misidentification between this species and other Miniopterus species, there is a need to carefully review the distribution of Miniopterus natalensis (ACR 2015). The extent of occurrence (EOO) is calculated as 1,405,662 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
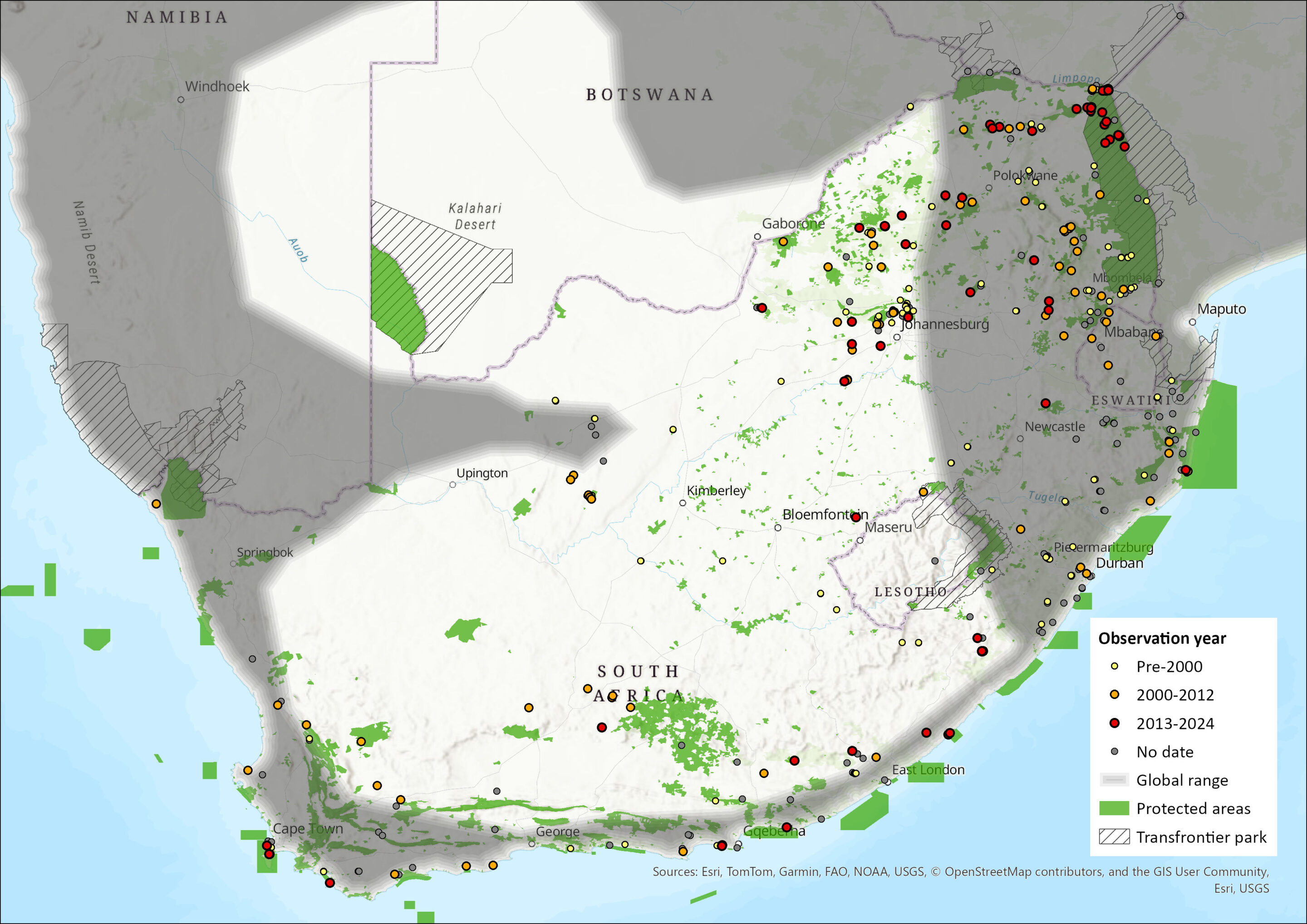 Figure 1. Distribution records for Natal Long-fingered Bat (Miniopterus natalensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Natal Long-fingered Bat (Miniopterus natalensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Pretorius et al. (2020a) used five years (2014–18) of echolocation data to investigate the migration phenology of the Natal Long-fingered Bat in relation to climate (photoperiod, minimum temperature, dew point, rainfall, barometric pressure, humidity and maximum wind speed) at a maternity cave in South Africa. Photoperiod had the greatest effect on the magnitude of phenological patterns in the activity of this species across all seasons. Although spring (September – November) arrival at the maternity site was variable across years, summer departure dates did not differ, resulting in a shorter breeding period in the 2017–18 sample year. During the 2016–17 sample year, activity of this species was significantly lower than in other years, which coincided with El Niño-induced drought conditions which likely impacted resources and led to a reduction in activity and population size. These findings suggest that the Natal Long-fingered Bat may be vulnerable to phenological mismatch and population decline, which supports the need for systematic population monitoring of this species.
Population information
A common and widespread species. It roosts in small colonies but can also be found in colonies of more than 2,500 animals, with one colony, the De Hoop Guano Cave in the Western Cape, exceeding 200,000 in certain seasons (McDonald et al. 1990). There is also a colony at the Gatkop Caves in Limpopo Province, which in 1978 was estimated to be 150,000 individuals (Van der Merwe, 1979b). At Koegelbeen Cave near Grikwastad in the Northern Cape, betweeen 70,000 and 100,000 individuals, roughly half of which were pups, were estimated (Hermann and Balona unpubl. data). In the Highveld caves, numbers vary from a few individuals up to ca. 4,000 (van der Merwe 1973). This species is very well represented in museums, with over 800 specimens examined in Monadjem et al. (2020).
Current population trend: Declining
Continuing decline in mature individuals? (Not specified)
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: Approximately 260,000 individuals have been reported for the colony inhabiting De Hoop Guano Cave (McDonald et al. 1990).
Number of Subpopulations: Pretorius et al. (2020b) reported that M. natalensis has been recorded from at least 37 caves in South Africa, but the current status of each of these colonies is not known.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
According to a phylogeographic study based on one nuclear marker, the Natal Long-fingered Bat within the assessment region forms part of a southern African clade comprising individuals from Zimbabwe, Namibia, and Eswatini (Monadjem et al. 2019). A population genomic study incorporating more fine-scale nuclear markers might uncover genetic structure, but based on the current genetic and ecomorphological evidence, the species is thought to be a single population within the assessment region.
Given the estimated population sizes in particular colonies, the effective population size for the species is thought to greatly exceed the minimum threshold of 500 (i.e., translating to a census size of 1,600- 5,000 mature individuals assuming a conversion ratio of Ne/Nc of 0.1-0.3).
Habitats and ecology
It is predominantly a temperate or subtropical species with the core of its distribution in the savannahs and grasslands of southern Africa (Monadjem et al. 2020), and has been recorded from semi-desert, dry and moist savannah, and Mediterranean-type shrubby vegetation. It is also associated with trees and orchards surrounding artificial wetlands (Sirami et al. 2013). It is generally a cave roosting species also found in similar habitats such as disused mine tunnels. The availability of suitable roosting sites may be more critical in determining its presence in an area than the surrounding vegetation (Monadjem et al. 2020). Smaller groups will also utilise crevice-type habitats. It utilises separate caves as winter hibernacula, migration stop-overs, and summer maternity roosts (van der Merwe 1975, Bats KZN data), with hibernacula generally being cooler and at higher altitudes, which may be an important consideration for wind farm planning on the South African Highveld, for example (Pretorius et al. 2020b). Females typically migrate seasonally between these caves, which may be separated by up to 260 km (Miller-Butterworth et al. 2003). Females congregate at maternity roosts where each one gives birth to a single young (Monadjem et al. 2020). This species holds the longevity record for a southern Africa bat of 13 years (van der Merwe 1989).
It is an open-air and clutter-edge forager, feeding on a variety of aerial prey including Diptera, Hemiptera, Coleoptera, Lepidoptera, Hymenoptera, and Isoptera (Moyo and Jacobs 2020; Jacobs 1999; Schoeman & Jacobs 2003; Miller-Butterworth et al. 2005).
Ecosystem and cultural services: As with all insect-eating bats, this species plays an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011, McDonald et al. 1990). McDonald et al. (1990) showed the M. natalensis in the De Hoop cave to eat between 1.1 and 2.9 g of insects per bat per night, leading to an estimated total insect consumption by the bats in the De Hoop cave to be over 100 t per year. Bats often prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
| 8.1. Desert -> Desert – Hot | – | Marginal | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: One banded individual was recovered after 13 years (van der Merwe 1989)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Females give birth in October-December (Monadjem et al. 2020)
Average Annual Fecundity or Litter Size: Litter size is 1 offspring (Monadjem et al. 2020), although it is not known if every female gives birth every year
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Females typically migrate seasonally between caves, which may be separated by up to 150km (Monadjem et al. 2020). Pretorius et al. (2020b) predicted a shortest migration least-cost path (LCP) of 101.68km between Doornhoek and Mission Rocks, and a longest migration LCP distance of 394.37 km between De Hoop and Steenkampskraal Mine.
In studies on the movements of the closely related M. schreibersii, Garin et al. (2008) found (from a total of 3,014 ringed and 198 recaptured bats) that distances between capture sites in the Iberian Peninsula averaged 77 km (range 0–258 km) and varied seasonally. In India it was found that the population of a given area tended to be centered in one large cave, but that individuals spent part of their time in secondary roosts within a 70 km radius (Nowak 1997). Similarly, van Harten et al. (2022) found in Australia that individuals of the related Miniopterus orianae bassanii moved 72 km between caves multiple times over short time periods, including on successive nights, which more than doubled the previous distance used to inform management buffer zones.
Congregatory: Yes (refer to the Population subsection).
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Within the assessment region, large areas are being developed for renewable energy facilities including wind farms. Wind turbines pose a major threat to this species since the life span of wind turbines is an estimated 20-25 years (Mortensen 2013), and there is an overlap between the species’ known and modelled distribution and predicted migration routes, and that of existing and planned wind farms (Pretorius et al. 2020b). Furthermore, of all bat fatalities reported by Aronson (2022) from data that were collated from 25 operational wind farms in South Africa for the period 2011-2020, Miniopterus natalensis was the third most prevalent. More recent data indicate that Miniopterus natalensis remains the third most frequently killed bat species at operational wind farms in South Africa, and long-term passive acoustic bat call activity data from certain wind farm sites suggest that local populations may be declining (Inkululeko Wildlife Services unpubl. data). Its high wing-loading and aspect ratio (Norberg & Rayner 1987; Jacobs 1999; Schoeman & Jacobs 2003, 2008; Miller-Butterworth et al. 2005) suggests that this bat is a fast flyer in relatively open spaces and, because it is migratory (van der Merwe 1975), means it is prone to flying in the rotor sweep zone of wind turbines both while foraging and commuting during migration. Since diligent bat fatality monitoring is not happening at all operational wind farms in South Africa, and since not all-important colonies of this species are being monitored, the impact of wind farms on this species cannot yet be quantified. However, the impact is inferred to be significant based on the rapidly growing number of wind farms and the often large, estimated numbers of actual bat fatalities at these facilities (Inkululeko Wildlife Service and SABAA unpubl. data).
The tendency of this species to roost in a few localised sites places further increases the risk of colony declines. Genetic research indicates a high degree of philopatry in this species (Miller-Butterworth 2003), which means that should a colony be lost or destroyed, it may not be repopulated from other areas, potentially leading to local extinction.
In parts of its range, it is locally threatened by habitat loss resulting from conversion of land due to agriculture and urbanisation, incidental poisoning with insecticides (and associated loss of prey base), and the disturbance of roosting and maternity caves by tourist activities and religious ceremonies (ACR 2015). Of particular concern is the possible loss of colonies which have colonized old mine tunnels from: i) recommissioning of old mines (C. Lötter pers. comm.); or ii) closure (sealing) of old mine tunnels, inter alia, to prevent their exploitation by illegal miners, which the South African government is undertaking at a national level (Government Notice 446, 21 May 2021).At one of the most important sites, Gatkop Caves in Limpopo Province, the colony may be indirectly detrimentally impacted by habitat loss, blasting, light pollution, and forms of disturbance from expanding mining activity in the area. Deliberate closure of mines or caves could also disrupt migration routes.
Conservation
The species has been recorded in more than 15 protected areas. However, of 92 caves that were identified in South Africa by Pretorius et al. (2020b), 50 were reported to contain bats and 37 (40%) were recorded to harbour M. natalensis, of which only 9% are currently located inside protected areas. The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association and other provincial conservation organisations with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as M. natalensis. Systematic monitoring of known colony sizes and trends is needed, and the status of this species must be reviewed every few years or more often. There is a need to identify and protect additional important roost sites (especially maternity caves) and to gain a better understanding of their migratory routes.
Of 12 least-cost migration routes for this species, which were predicted by Pretorius et al. (2020b), most intersect potential threat risk elements such as wind farms. Development of new wind farms must strictly avoid interception of known, predicted, or suspected migration routes of this species, and encroachment into the prescribed buffers around confirmed and potential roosts of M. natalensis (see MacEwan et al. 2020). Wind farm development should not be permitted within a 40 km or larger radial zone around the largest roosts of this species (comprising >20 000 individuals). Although this buffer size is twice the minimum buffer of 20 km for roosts of >2 000 bats recommended by MacEwan et al. (2020), 40 km is less than half of the shortest predicted migration path of 101.68km for this species (Pretorius et al. 2020b). The impact of wind farms should also be carefully monitored to determine if there are related population declines. Conservation planning and engagement with the wind energy industry will be needed in future to mitigate subpopulation loss related to wind farm development.
Turbine-fatalities of this species should be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as: i) ultrasound to deter bats (Weaver et al. 2020); ii) blanket curtailment of turbines at low wind speeds during seasons and hours of the night when this species is most active (Berthinussen et al. 2010; Arnett et al. 2011; Hayes et al. 2019); iii) acoustic smart curtailment of turbines in response to real-time bat activity (https://www.wildlifeacoustics.com/smart-system) ; and/or optimised smart curtailment of turbines during predicted periods of high bat activity based statistical modelling (https://west-inc.com/wp-content/uploads/2024/04/Optimized-Smart-Curtailment-SOQ_FINAL.pdf). As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical.
Recommendations for land managers and practitioners:
- The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association and other provincial conservation organisations with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as M. natalensis.
- Development of wind farms must strictly avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species. In particular, wind farm development should not be permitted within a 40 km or larger radial zone around the largest roosts of this species (comprising >20 000 individuals). There should be a revision of the bat and wind energy guidelines, specifically buffers around very large roosts.
- Turbine-fatalities of this species must be reduced with interventions such as ultrasound to deter bats and curtailing turbines at low wind speeds.
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Closure of mines and caves should only be done using bat-compatible methods.
- Better post-construction compliance monitoring at WEFs.
Research priorities:
- A meta-analysis of all recorded M. natalensis fatalities at wind farms in South Africa to date, to assess the cumulative impact of this industry on this species, and to identify any spatial, temporal, or turbine-specific trends, which may assist in devising effective fatality mitigation for future implementation.
- Research to assess the efficacy of different mitigation methods to reduce bat mortality at wind farms.
- Wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species throughout southern Africa.
- Research in identifying key migratory routes.
- Molecular research to resolve the species complex.
- Better determine the range of this species when compared to that of Miniopterus schreibersii (ACR 2015).
- Subpopulation sizes and trends at important roosts, and population estimate.
- Physiological effects of agrochemicals
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Deposit any dead specimens at local museums or Bat Interest Groups.
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Appleton BR, McKenzie JA, Christidis L. 2004. Molecular systematics and biogeography of the bent-wing bat complex Miniopterus schreibersii (Kuhl, 1817) (Chiroptera: Vespertilionidae). Molecular Phylogenetics and Evolution 31: 431– 439.
Arnett EB, Huso MM, Schirmacher MR, Hayes JP. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Aronson, J., Sowler, S. and MacEwan, K. 2018. Mitigation guidance for bats at wind energy facilities in South Africa. Edition 2. South African Bat Assessment Association, South Africa.Baerwald EF, Edworthy J, Holder M, Barclay RM. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Driver A, Sink KJ, Nel JN, Holness S, van Niekerk L, Daniels F, Jonas Z, Majiedt PA, Harris L, Maze K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fahr J, Djossa BA, Vierhaus H. 2006. Rapid survey of bats (Chiroptera) in the Forêt Classée du Pic de Fon, Guinea. Conservation Internationa, Washington, DC, USA.
Jacobs DS. 1999. Intraspecific variation in wingspan and echolocation call flexibility might explain the use of different habitats by the insectivorous bat, Miniopterus schreibersii (Vespertilionidae: Miniopterinae). Acta Chiropterologica 1: 93–103.
Hayes, M.A., Hooton, L.A., Gilland, K.L., Grandgent, C., Smith, R.L., Lindsay, S.R., Collins, J.D., Schumacher, S.M., Rabie, P.A., Gruver, J.C., and Goodrich-Mahoney, J. 2019. A smart curtailment approach for reducing bat fatalities and curtailment time at wind energy facilities. Ecological Applications, 0(0), 2019, e01881.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
McDonald, J.T., Rautenbach, I.L., and Nel, J.A.J. 1990. Roosting requirements and behaviour of five bat species at De Hoop Guano Cave, southern Cape Province of South Africa. S. Afr. J. Wildl. Res. 20(4): 157-161
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
Miller-Butterworth CM, Eick G, Jacobs DS, Schoeman MC, Harley EH. 2005. Genetic and phenotypic differences between South African long-fingered bats, with a global miniopterine phylogeny. Journal of Mammalogy 86: 1121–1135.
Miller-Butterworth CM, Jacobs DS, Harley EH. 2003. Strong population substructure is correlated with morphology and ecology in a migratory bat. Nature 424: 187–191.
Monadjem, A., Goodman, S., Stanley, W.T., Appleton, B. 2013. A cryptic new species of Miniopterus from south-eastern Africa based on molecular and morphological characters. Zootaxa 3746 (1): 123–142
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Mortensen, N. G. 2013. Planning and development of wind farms: Wind Resources Assessment and Siting. Technical University of Denmark, Denmark.
Moyo, S. and Jacobs, D.S. 2020. Faecal analyses and alimentary tracers reveal the foraging ecology of two sympatric bats. PLoS ONE 15(1): e0227743. https://doi.org/10.1371/journal. pone.0227743
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Pretorius, M., Broders, H., Seamark, E. and Keith, M. 2020a. Climatic correlates of migrant Natal long-fingered bat (Miniopterus natalensis) phenology in north-eastern South Africa. Wildlife Research 47: 404-414.
Pretorius, M., Broders, H. and Keith, M. 2020b. Threat analysis of modelled potential migratory routes for Miniopterus natalensis in South Africa. Austral Ecology 45: 1110-1122.
Rosevear, D.R. 1965. The Bats of West Africa. British Museum, London, UK.
Schoeman, C. M., Jacobs, D. S. 2003. Support for the allotonic frequency hypothesis in an insectivorous bat community. Oecologia 134: 154–162.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defences on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS one 3(11): e3715.
Sirami C., Jacobs D.S., Cumming G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
van der Merwe M. 1975. Preliminary study on the annual movements of the Natal clinging bat. South African Journal of Science 71: 237–241.
van der Merwe M. 1989. Longevity in Schreibers’ long-fingered bat. South African Journal of Wildlife Research 19: 87–89.
Weaver, S.P., Hein, C.D., Simpson, T.R., Evans, J.W., and Castro-Arellano, I. Ultrasonic acoustic deterrents significantly reduce bat fatalities at wind turbines. Global Ecology and Conservation 24: e01099
