
Laephotis cf. namibensis
Laephotis cf. namibensis

2025 Red list status
Vulnerable
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Laephotis cf. namibensis – Taylor, Strydom, Richards, Markotter, Toussaint, Kearney, Cotterill, Howard, Weier, Keith, Neef, Mamba, Magugula & Monadjem, 2022
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Laephotis – namibensis
Common Names: No available common name (previously included under Namib Long-eared Bat)
Synonyms: Laephotis namibensis Setzer, 1971
Taxonomic Note:
Taxonomic uncertainty characterises this taxon. Although originally placed within Laephotis namibensis Setzer, 1971, and previously thought to be conspecific with Laephotis wintoni (Monadjem et al. 2020), recent molecular phylogenetic studies using mitochondrial DNA have shown genetic structuring between the Western Cape population in South Africa and Namibian individuals, hereafter referred to as L. cf. namibensis, pending formal naming of this species (Taylor et al. 2022; 2024). There appears to be morphological distinction between the two mitochondrial lineages, albeit based on limited skull samples of South African individuals.
Laephotis namibensis sensu stricto is considered an arid-occurring species, whereas L. cf. namibensis is associated with temperate, fynbos habitat of the Cederberg Wilderness Area (Taylor et al. 2024).
Red List Status: VU – Vulnerable D1 (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1, Naidoo, T.1 & da Silva. J.M.2
Reviewers: Taylor, P.J.3, Howard, A.3 & Laverty, T.4
Institutions: 1Durban Natural Science Museum, 2South African National Biodiversity Institute, 3University of the Free State, 4New Mexico State University
Previous Assessors and Reviewers: Jacobs, D., Cohen, L., MacEwan, K., Monadjem, A., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Relton, C., Raimondo, D. & Child, M.F.
Assessment Rationale
This South African endemic taxon is only known from a single, isolated population found in the Cederberg Wilderness Area of the Western Cape Province of South Africa. It roosts in rock faces that are unlikely to be transformed yet remain difficult to access and hence to study the population. Because no information is available on population size and it is not abundant from elsewhere in its range, we assume there are fewer than 1,000 mature individuals in the assessment region and hence list it as Vulnerable D1. It could qualify for Endangered, however, there seems to be no major threats to the Cederberg population (Jacobs et al. 2005; Sirami et al. 2013). Further field studies are needed to monitor the status of the Cederberg individuals, as well as to identify additional subpopulations and to estimate population sizes and trends. Validation of acoustic data gathered from additional localities in the Western Cape province of South Africa (Alguhas, Franschhoek, Malmesbury, Riviersonderend and Wellington; see Sirami et al. 2013), is urgently needed. The EOO for this species based on verified records is 7.27 km2.
Regional population effects: Individuals have only been captured from a single locality within the assessment region. Furthermore, although it has intermediate wing loading (Jacobs et al. 2005; Schoeman & Jacobs 2008), and thus is possibly capable of effective dispersal, it is uncertain whether there are any other suitable roost sites or colonies within the immediate vicinity of the Cederberg Wilderness Area (although see Geographic Range below concerning acoustic activity recorded elsewhere).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR, Naidoo T & da Silva JM. 2025. A conservation assessment of Laephotis cf. namibensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Endemic to South Africa, this taxon is restricted to the temperate Cape Floristic Biome of the assessment region (Taylor et al. 2022, 2024). This little-known species has been recorded from a single area in the Western Cape, South Africa (Algeria in the Cederberg Mountains) (Skinner & Chimimba 2005; Monadjem et al. 2020), where it has been recorded regularly (for example, Jacobs et al. 2005; Sirami et al. 2013). Recent acoustic survey data suggest this species is more widely distributed across the Western Cape than current capture data suggests, with activity recorded in the Cederberg, Agulhas, Franschhoek, Malmesbury, Riviersonderend and Wellington areas (Sirami et al. 2013). However, these data need validation (see Russo & Voight 2016), and thus a conservative approach is adopted in reporting the taxon from only a single locality where animals were netted.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 425m
Elevation Upper Limit (in metres above sea level): 1695m
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
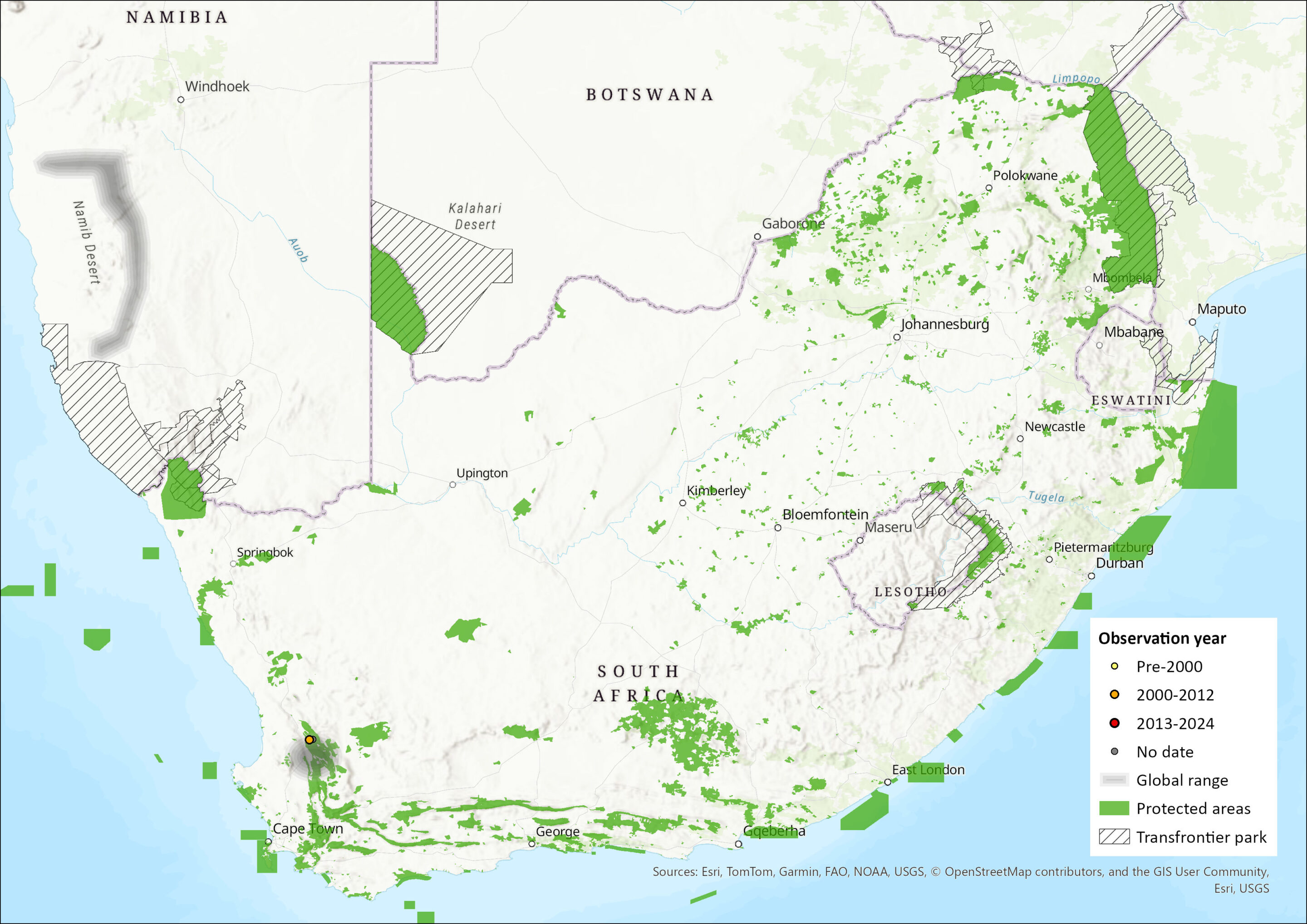 Figure 1. Distribution records for Laephotis cf. namibensis within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Laephotis cf. namibensis within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Most studies on the effects of climate change on bats are based on predictive species distribution modelling. However, the limited occurrence records for L. cf. namibensis do not support predictive distribution modelling under future climate models, hence it is difficult to establish the possible impacts of climate change on the taxon. In general, there is a here is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability, with increasing global aridity, which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020). The mountainous Cederberg range is generally classified as semi-arid receiving <400 mm rainfall on average, however precipitation may increase to >1000 mm per annum in some years (Schulze 1997; Quick & Eckardt 2015). Increased temperatures and increased precipitation seasonality associated with climate change, are likely to impact the region and its fauna.
Population information
Its abundance within the assessment region is unknown but is regularly caught in mist nets over water in the Cederberg (for example, Jacobs et al. 2005; Sirami et al. 2013). It is suspected that the population numbers <1,000 individuals.
Current population trend: Unknown
Continuing decline in mature individuals? Cannot be determined as population trends remain poorly understood
Number of mature individuals in population: Suspected to be <1,000
Number of mature individuals in largest subpopulation: Unknown
All individuals in one subpopulation: Presumed to be.
Extreme fluctuations in the number of subpopulations: Cannot be determined as population trends remain poorly understood
Continuing decline in number of subpopulations: Cannot be determined as population trends remain poorly understood
Number of mature individuals in largest subpopulation: Unknown
Severely fragmented: Yes, this species is known from only one locality in the assessment region and is seemingly isolated from those populations from the Namib Desert.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Cannot be determined
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic studies have yet been conducted on the species, although the pronounced geographic differentiation between animals in South Africa and their nearest neighbours in Namibia suggests limited geneflow between these regions. This spatial isolation most likely represents two genetically distinct subpopulations (Taylor et al. 2022). A fine-scale genomic study across the species’ range is recommended.
Habitats and ecology
In the assessment region, the species is recorded from the temperate Northwest Fynbos bioregion, with individuals most often found in close proximity to water in mountainous areas (Monadjem et al. 2020; Kearney 2013). Individuals from Algeria Forest were mist-netted as they flew along a water course, close to the water surface (ACR 2024). Similarly, they were found to be active near trees and orchards surrounding wetlands as well as over wetlands (Sirami et al. 2013).
Radio tracking data from the Western Cape shows that L. cf. namibensis makes use of narrow crevices in vertical rock faces as diurnal roosting sites (Jacobs et al. 2005). A specimen has also been taken from a roost under exfoliating rock (Skinner & Chimimba 2005). This small, insectivorous species has intermediate wing loading and is considered a slow manoeuvrable flyer, that often flies low over water surfaces or the ground, and close to vegetation (Jacobs et al. 2005; Monadjem et al. 2020). It is a clutter-edge forager, with Lepidoptera and Coleoptera constituting large portions of its diet, and Hemiptera and Tricoptera to a lesser extent (Jacobs et al. 2005; Schoeman 2006).
Within the assessment region, a pregnant female was caught in November 2002, and three lactating females were caught in November 2004 (Jacobs et al. 2005). A post-lactating female was collected towards the end of January (Seamark & Brand 2005).
Ecosystem and cultural services: Insectivorous bats are important regulators of insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats may feed on arthropods that damage crops, and thus agricultural areas with bats often require less pesticides which may also be detrimental to human health (Frank 2024; Kunz et al. 2011)
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.8 Mediterranean-type shrubby vegetation | Suitable | ||
| 1.9 Subtropical/Tropical moist montane | Suitable |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Forearm length range = 4.0 – 4.2 cm (Jacobs et al. 2005)
Size at Maturity (in cms): Male: Forearm length = 3.8 cm (Jacobs et al. 2005)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Unknown
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Possibly exhibits seasonal monoestry, with partuition coinciding with the austral summer months (Jacobs et al. 2005; Seamark & Brand 2005)
Average Annual Fecundity or Litter Size: Unknown
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: No available information, however, it appears to make use of waterways and water sources.
Congregatory: Yes, reported to roost collectively in rocky crevices (Jacobs et al. 2005).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: None reported
National Commercial Value: May contribute to insect population control and thus have agroeconomic value.
International Commercial Value: May contribute to insect population control and thus have agroeconomic value in respect of exported products.
End Use: None reported
Is there harvest from captive/cultivated sources of this species? None reported
Harvest Trend Comments: N/A
Threats
Agricultural expansion in the Western Cape may be reducing the prey base of this species by removing natural vegetation and through the use of pesticides. Additionally, climate change has been identified as an increasing global threat to other bat species (Sherwin et al. 2013), and may similarly impact L. cf. namibensis. The Fynbos biome is projected to experience a loss of area of between 51% and 65% by 2050 depending on the climate change scenario (Midgley et al. 2002).
Conservation
The species is recorded from the protected Cederberg Wilderness Area within the Western Cape region of South Africa. No specific interventions can be implemented until further studies into the distribution and general natural history of this species are completed. This species would benefit from the long-term protection of additional key roost sites when identified, as well as holistic land management that reduces pesticide use and conserves buffer strips of natural vegetation.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes and maintain buffer strips of natural vegetation.
Research priorities:
- Enhanced molecular DNA studies, inclusive of population-level genetics, to determine gene flow and the effective population size (Ne) within the assessment region.
- Surveys, combining physical capture and acoustic monitoring, to determine geographical distribution and identify key roost sites.
- Systematic monitoring to measure population size and trends of the Cederberg population.
- Field-based validation of acoustic data gathered from additional localities in the Western Cape province of South Africa (Alguhas, Franschhoek, Malmesbury, Riviersonderend and Wellington; see Sirami et al. 2013), is urgently needed.
- Research into the potential agroeconomic contribution of the species in relation to insect pest control, as it may forage in orchards (see Sirami et al. 2013)
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, AfriBats on iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A., 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S., 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Frank, E.G. 2024. The economic impacts of ecosystem disruptions: Costs from substituting biological pest control. Science 385,0344.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
Jacobs DS, Barclay RM, Schoeman MC. 2005. Foraging and roosting ecology of a rare insectivorous bat species, Laephotis wintoni (Thomas, 1901), Vespertilionidae. Acta Chiropterologica 7: 101–109.
Kearney T. 2013. Laephotis botswanae Botswanan Long-eared Bat. Pages 581–582 in Happold M, Happold D, editors. Mammals of Africa. Volume IV. Hedgehogs, shrews and bats. Bloomsbury Publishing, London, UK.
Kearney TC, Seamark EC. 2005. Morphometric analysis of cranial and external characters of Laephotis Thomas, 1901 (Mammalia: Chiroptera: Vespertilionidae) from southern Africa. Annals of the Transvaal Museum 42: 71–87.
Kearney TC, Volleth M, Contrafatto G, Taylor PJ. 2002. Systematic implications of chromosome GTG-band and bacula morphology for southern African Eptesicus and Pipistrellus and several other species of Vespertilioninae (Chiroptera: Vespertilionidae). Acta Chiropterologica 4: 55–76.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Midgley GF, Hannah L, Millar D, Rutherford MC, Powrie LW. 2002. Assessing the vulnerability of species richness to anthropogenic climate change in a biodiversity hotspot. Global Ecology and Biogeography 11: 445-451.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Peterson RL. 1973. The first known female of the African Longeared Bat Laephotis wintoni (Vespertilionidae: Chiroptera. Canadian Journal of Zoology 51: 601–603.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N., 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Quick, L.J. and Eckardt, F.D., 2015. The Cederberg: a rugged sandstone topography. In Landscapes and landforms of South Africa (pp. 85-93). Cham: Springer International Publishing.
Schoeman MC, Jacobs DS. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Schoeman MC. 2006. The relative influence of competition and coevolution on the community structure of insectivorous bats in southern Africa. Ph.D. Thesis. University of Cape Town.
Schulze RE. 1997. Atlas of agro-hydrology and agro-climatology, WRC Report TT82/96. Water Resource Commission, Pretoria
Seamark EC, Brand M. 2005. Bat survey in the Cederberg Wilderness Area, Western Cape, South Africa (28 January– 3 February 1999). African Bat Conservation News 3: 7–9.
Sherwin HA, Montgomery WI, Lundy MG. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Sirami C., Jacobs D.S., Cumming G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
