Namaqua Rock Mouse
Micaelamys namaquensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Micaelamys namaquensis – (A. Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Micaelamys – namaquensis
Common Names: Namaqua Rock Rat, Golden Rat, Namaqua Rock Mouse, Namaqua Aethomys (English), Namakwalandse Klipmuis (Afrikaans), Kondlo (Tsonga), Lekôtéé (Tswana), Fondo (Venda), Namaqua-Buschratte (German)
Synonyms: Aethomys namaquensis (A. Smith, 1834), and 20 others listed in Kesner et al. (2013)
Previously listed as Aethomys namaquensis.
Taxonomic Note: Two subgenera, namely Micaelamys and Aethomys have been recognised within the genus Aethomys. Although A. namaquensis and A. granti have traditionally been allocated to the subgenus Micaelamys, recent molecular studies reported the paraphyly of the genus (Ducroz et al. 2001; Castiglia et al. 2003; Russo et al. 2006), and the two subgenera have since been elevated to full generic rank. Therefore, the genus Micaelamys now includes M. namaquensis and M. granti. This close relationship between the species is evident from dental morphology, karyology, gross sperm and bacular morphology and cranial phenetic analysis. However, the two species can be distinguished on both morphological and chromosomal characteristics (Visser & Robinson 1986, 1987; Chimimba et al. 1999).
There are as many as 16 recognised subspecies of M. namaquensis (Roberts 1951; Skinner & Chimimba 2005), based on a limited number of samples with little or no assessment of patterns of geographic variation over the entire distributional range of the species. However, a comprehensive intraspecific morphometric study within M. namaquensis covering a wide geographic range showed that patterns of intraspecific variation suggested the recognition of four subspecies based on traditional morphology/cranial morphometrics (Chimimba 2001). Similarly, a recent molecular study identified at least eight well supported lineages based on mitochondrial DNA (mtDNA) sequences (Russo et al. 2010). This differentiation (considerable mtDNA diversity) is in contrast with the most recent taxonomic treatment, which only recognised four subspecies. Some of the mtDNA lineages broadly correspond with the distributional patterns of the previously described subspecies monticularis, namaquensis, lehocla and alborarius. This supports earlier views that this taxon may represent a species complex.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Russo, I.M.1 & da Silva, J.M.2
Reviewer: Smith, C.3
Institutions: 1Cardiff University,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors: Russo, I.M., MacFadyen, D. & Taylor, P.
Previous Reviewer: Child, M.F.
Previous Contributors: Avenant, N., Avery, M., Baxter, R., Monadjem, A., Palmer, G. & Wilson, B.
Assessment Rationale
Listed as Least Concern in view of its extremely wide distribution within the assessment region, tolerance of a broad range of habitats, including occurring commensally with human settlements, and because there are no major threats likely to cause population decline. It is the most common species recorded in rocky areas across the assessment area. While the effects of local threats such as gravel mining should be monitored, no major decline is expected. The Namaqua Rock Mouse likely comprises a species complex and, pending further research, may require reassessment.
Regional population effects: Opportunities for dispersal occur where rocky areas are continuous and are widespread along the border.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Russo IM & da Silva JM. 2025. A conservation assessment of Micaelamys namaquensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widely distributed in semi-arid to mesic savannahs of southern Africa, generally south of the Caprivi strip, in a wide variety of habitats that contain rocky outcrops (Monadjem et al. 2015). Its distribution extends marginally into the western parts of Angola and across the Zambezi into central Mozambique and southern Malawi (Monadjem et al. 2015). It has also been recorded in rocky areas at Shangani Ranch, southern Zimbabwe (D. MacFadyen unpubl. data) and rocky habitats from eastern Botswana (Russo et a. 2010).
Within the assessment region, it is considered common in all provinces within South Africa, and also occurs extensively in Eswatini and Lesotho, avoiding only parts of the coastal areas in Kwa-Zulu Natal and the Eastern Cape provinces, and central and coastal Mozambique (Skinner & Chimimba 2005). Although it is often encountered away from rocky habitats (for example, in woodlands and grasslands), even small rocky knolls can offer sufficient habitat for a population to thrive (Power 2014). Similarly, the amount of rubble or size of the rocks (or boulders) does not seem to influence its distribution, as it has been captured on hilly slopes covered in small rocks and stones (for example, the foothills of the Lubombo Mountains) or on small, isolated rocky outcrops (Monadjem 1998). It co-occurs with Elephantulus myurus in many parts of its range in the assessment area (Lancaster & Pillay 2010).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
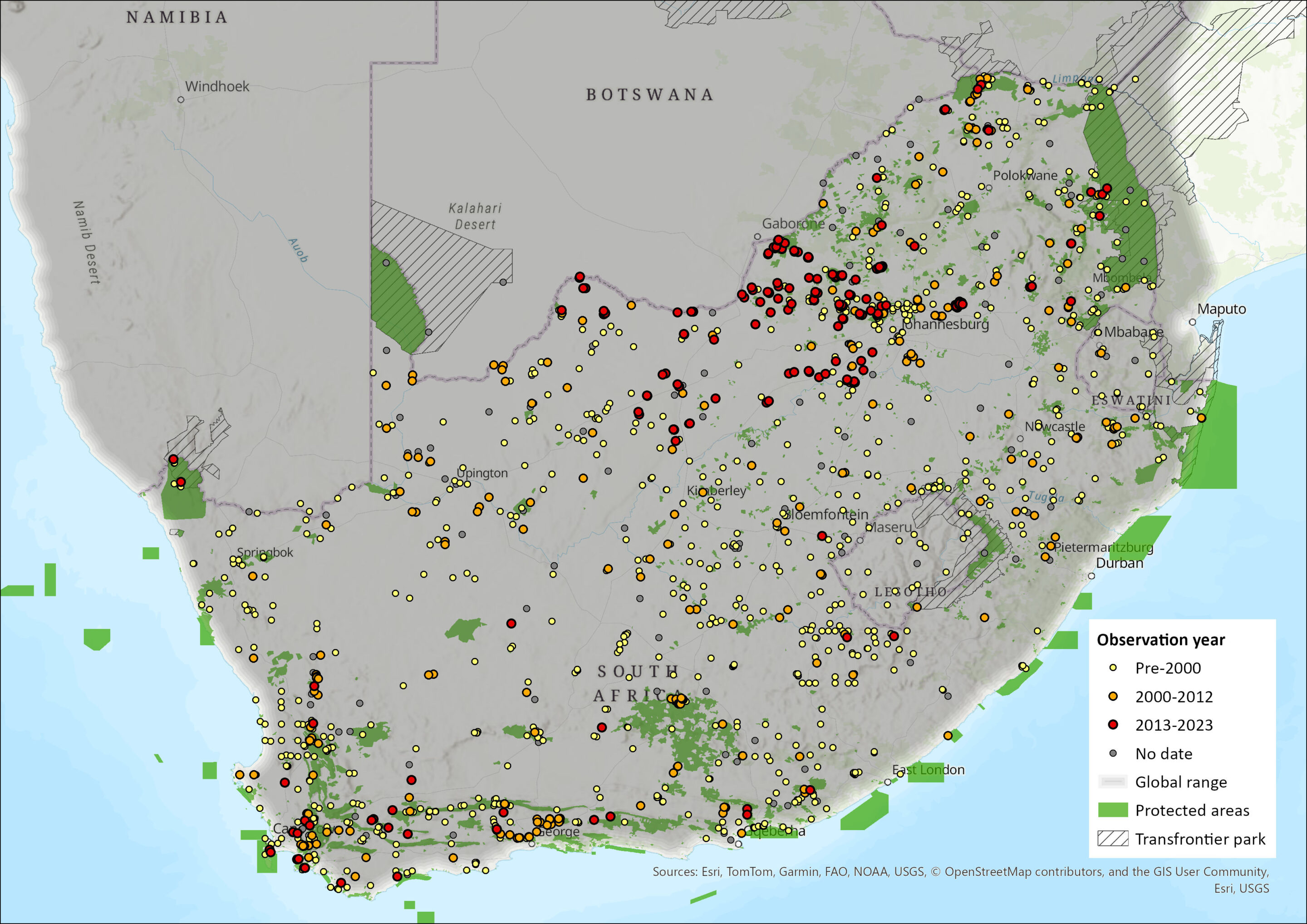
Map
Figure 1. Distribution records for Namaqua Rock Mouse (Micaelamys namaquensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
This species occurs throughout South Africa. There is a possibility that it might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). Rainfall is predicted to decrease in its range. These decreases will result in a more arid and drought-prone habitat which could affect food availability in the species’ distributional range.
Population Information
This species is widespread and especially common in highveld rocky grasslands (Kesner et al. 2013). In the western Soutpansberg of the Limpopo Province it is common at all altitudes at densities of up to 54 individuals / ha (Taylor et al. 2015). Similarly, it was regularly trapped in rocky outcrops in Tussen-die-Riviere Nature Reserve, Free State Province (Watson 2006), and it is common in the Korannaberg Mountain Range between Black Rock and Van Zylsrus in the Northern Cape Province. It was the most abundant species sampled on the Bokkeveld Plateau near Nieuwoudtville, Northern Cape Province, comprising 75% of 219 small mammal individuals (O’Farrell et al. 2008), and in Telperion/Ezemvelo Nature Reserve, Gauteng Province, comprising 59% of all samples (Fagir et al. 2014). In Eswatini, it is also closely associated with rocky outcrops where it is often the most common rodent species present (Monadjem 1998).
Russo et al. (2010) reveal at least eight genetically unique lineages (A – H) across the distributional range of the species. In addition, subclades have been identified in two of these lineages (A (A1-5) and B (B1-3)). Most lineages, or subclades within them, show a strong association with different vegetation types of southern Africa, including the Grassland and Savanna biomes, Albany Thicket, western Fynbos, Bushmanland/Upper Karoo Bioregion, Nama-Karoo, Kalahari Duneveld, Sub-Escarpment Grassland Bioregion (Grassland) and Eastern Kalahari Bushveld (Russo et al. 2010). A total of 14 subpopulations have been identified based on mitochondrial DNA.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: No
Number of mature individuals in population: > 10,000
Number of mature individuals in largest subpopulation: Unknown, but >10,000
Number of subpopulations: 14, based on mitochondrial DNA, with at least eight genetically unique lineages with a further six subclades.
Severely fragmented: Naturally
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
As noted in the Population Section, eight distinct mitochondrial DNA lineages with an additional six subclades of the Namaqua Rock Mouse have been identified that generally correspond to specific biomes or bioregions (Russo et al. 2010). Considering the species is thought to be abundant and widespread, it is possible each lineage exists as individual metapopulations. However, the molecular knowledge currently available indicates the possibility that more fine scale influences on the movements of this species may exist. Further molecular research incorporating nuclear markers is needed to identify whether these eight lineages represent Evolutionarily Significant Units, whether further subpopulation structure is present within them, and if the different subpopulations are showing differential signs of genetic health (e.g., any lineage with an effective population size less than 500, or decreased levels of heterozygosity).
Based on the available information, it is possible to quantify one of the two population genetic indicators in the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF). The complementary genetic indicator – proportion of populations maintained (PM), would receive a value of 1.0, considering each of the eight lineages/six subclades remain (14/14 populations remaining).
The number of mature individuals in the largest subpopulation is calculated at 10,000 (see Population Section). Using a Ne/Nc (Ne = effective population size, mature individuals contributing to breeding; Nc= mature individuals) conversion ratio between 0.1-0.3, yields Ne values between 1,000-3,000 individuals which is above the Ne 500 ratio. Due to the lack of effective population size or census data for each population, the GBF’s headline indicator, proportion of populations within a species with an effective population size (Ne) greater than 500, cannot be estimated for each of the populations. This is an estimate for the largest subpopulation so the Ne 500 ratio for some of the smaller subpopulations may be lower. A fine scale population genetic study is required for this species to assess the number of populations and quantify Ne for each of these populations.
Habitats and ecology
This species occupies a wide range of habitats, if they contain patches of rocky areas. It prefers areas with rocky koppies, outcrops and boulders, and shelters in rocky crevices, hollow trees or in burrows constructed under logs or dense shrubs. On Telperion Nature Reserve, Gauteng Province, it was recorded in grassy areas where only a few scattered red sandstone rocks occurred, less than 20% rock cover (MacFadyen 2014). In the Swartberg region of the Western Cape Province, it was found at low altitudes on north-facing slopes with more than 30% ground cover (Breytenbach 1982). In the Kalahari, it occurs in open shrub, open woodland and on the fringes of pans where there are calcareous outcrops (Skinner & Chimimba 2005). In the KwaZulu-Natal Province, it occupies sparse to dense bushveld and medium to tall grassland within 1 km of rocky habitat (Taylor 1998). On the Bokkeveld plateau, Northern Cape Province, it was trapped solely on dolerite ridges (O’Farrell et al. 2008). It is absent from agricultural monocultures, rockless grasslands, sodic areas and wetlands. It is commensal with humans, often found in houses and huts. Juveniles are regularly recorded in the buildings at Tswalu Kalahari Reserve in the Northern Cape Province (D. MacFadyen pers. obs.).
It is a predominantly nocturnal and communal species (Perrin 1981). It is omnivorous, feeding on seeds, green plant material and insects (Breytenbach 1982; Kerley et al. 1990; Monadjem 1997). The species has a preference for Protea caffra seeds and may consume large numbers in a relatively short period of time. On a few occasions where two individuals were captured, signs of cannibalism were recorded (MacFadyen 2014). The presence of this species is often observed by nests of large amounts of dry grass between rock cracks and between rocky overhangs. It closely resembles the Red Veld Rat (Aethomys chrysophilus), which is larger, more reddish and heavier in built. The colour of the pelage, the length of the tail and body size in M. namaquensis varies greatly over its wide distributional range (Chimimba 2001). This variation suggested that M. namaquensis may reflect either a complex of species or subspecies.
Ecosystem and cultural services: They act as seed dispersers (Bond & Breytenbach 1985) and move large amounts of vegetation between areas and are thus important in regulating the nutrient cycle of soil. They are also pollinators of certain geoflorous Protea species by consuming the nectar; for example, Protea welwitschia, which exists in undisturbed sites in the Rocky Highveld Grasslands of the Gauteng and Mpumalanga Provinces. However, there are also concerns that the abundance of this species equates to it being a reservoir for zoonotic diseases (Fagir et al. 2014).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.4. Forest -> Forest – Temperate |
– |
Suitable |
– |
|
2.1. Savanna -> Savanna – Dry |
– |
Suitable |
– |
|
3.4. Shrubland -> Shrubland – Temperate |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
4.4. Grassland -> Grassland – Temperate |
– |
Suitable |
– |
|
6. Rocky areas (eg. inland cliffs, mountain peaks) |
– |
Suitable |
– |
|
8.1. Desert -> Desert – Hot |
– |
Marginal |
– |
|
8.2. Desert -> Desert – Temperate |
– |
Marginal |
– |
|
14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land |
– |
Suitable |
– |
|
14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens |
– |
Suitable |
– |
|
14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Due to their high numbers, they are probably used as bush meat in certain rural areas, but this is unlikely to impact the population significantly.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species as it inhabits areas that are not prone to agriculture, livestock production and forestry. However, mining, especially for granite, would probably be the number one cause of local habitat destruction, especially in the northern provinces. Overall, this species is common and not declining.
Habitat trend: Stable. It is commensal with humans and able to live in modified landscapes. However, the mining sector is suspected to be expanding rapidly in Limpopo, Mpumalanga and North West provinces (NW READ 2014; Desmet & Schaller 2015), but the extent of impacts from this sector is currently poorly known (V. Egan pers. comm. 2015; Lötter 2015).
Conservation
This species is considered common in all provinces within South Africa and occurs in numerous protected areas across its range; for example, Telperion Nature Reserve (Mpumalanga), Ezemvelo Nature Reserve and Premier Game Farm (Gauteng), Venetia Limpopo Nature Reserve (Limpopo), Rooipoort Nature Reserve and Tswalu Kalahari Reserve (Northern Cape). No specific interventions are currently necessary. However, legislation regarding the protection of habitat from mining rock (especially granite) and mitigating rock crushing in the Limpopo, Mpumalanga and North West Provinces, would benefit this species. Similarly, protected area expansion through biodiversity stewardship to connect naturally fragmented rocky habitats would facilitate adaption to climate change.
Recommendations for land managers and practitioners:
This species is abundant and no management recommendations are required.
Research priorities:
- A study is needed to investigate the taxonomy of this species (the recognition of species/subspecies) based on traditional morphology, cranial morphometrics and DNA.
Encouraged citizen actions:
- Landowners and city planners can conserve natural vegetation around rocky outcrops.
- Public pressure to enforce legislation to ensure buffer areas are protected when sites are mined.
Bibliography
Bond WJ, Breytenbach GJ. 1985. Ants, rodents and seed predation in Proteaceae. South African Journal of Zoology 20: 150–154.
Breytenbach, G.J. 1982. Small mammal responses to environmental gradients in the Groot Swartberg of the Southern Cape. University of Pretoria.
Castiglia R, Corti M, Colangelo P, Annesi F, Capanna E, Verheyen W, Sichilima A, Makundi R. 2003. Chromosomal and molecular characterization of Aethomys kaiseri from Zambia and Aethomys chrysophilus from Tanzania (Rodentia, Muridae). Hereditas 139: 81–89.
Chimimba CT. 2001. Infraspecific morphometric variation in Aethomys namaquensis (Rodentia: Muridae) from southern Africa. Journal of Zoology 253: 191–210.
Chimimba, C.T., Dippenaar, N.J. and Robinson, T.J. 1999. Morphometric and morphological delineation of southern African species of Aethomys (Rodentia: Muridae). Biological Journal of the Linnean Society 67: 501–527.
Desmet PG, Schaller R. 2015. North West Biodiversity Sector Plan Technical Report. North West Department of Rural, Environment and Agricultural Development, Mahikeng, South Africa.
Ducroz JF, Volobouev V, Granjon L. 2001. An assessment of the systematics of arvicanthine rodents using mitochondrial DNA sequences: evolutionary and biogeographical implications. Journal of Mammalian Evolution 8: 173–206.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J. and Midgley, G.F., 2024. Projections of future climate change in Southern Africa and the potential for regional tipping points. In Sustainability of Southern African Ecosystems under Global Change: Science for Management and Policy Interventions (pp. 169-190). Cham: Springer International Publishing.
Fagir DM, Ueckermann EA, Horak IG, Bennett NC, Lutermann H, others. 2014. The Namaqua rock mouse (Micaelamys namaquensis) as a potential reservoir and host of arthropod vectors of diseases of medical and veterinary importance in South Africa. Parasites & Vectors 7: 366.
Kerley, G.I.H, Knight, M.H. and Erasmus, T. 1990. Small mammal microhabitat use and diet in the southern Kalahari, South Africa. S. Afr. J. Wildl. Res. 20: 123-126.
Kesner MH, Linzey AV, Chimimba CT. 2013. Aethomys namaquensis Namaqua Veld Rat (Namaqua Aethomys). In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 371–373. Bloomsbury Publishing, London, UK.
Lancaster J, Pillay N. 2010. Behavioral interactions between a coexisting rodent Micaelamys namaquensis and macroscelid Elephantulus myurus. Current Zoology 56: 395–400.
Lötter, M.C. 2015. Technical Report for the Mpumalanga Biodiversity Sector Plan – MBSP. Mpumalanga Tourism & Parks Agency, Mbombela.
Lötter, M.C., Cadman, M.J. and Lechmere-Oertel, R.G. 2014. Mpumalanga Biodiversity Sector Plan Handbook. MTPA. Nelspruit.
MacFadyen DN. 2014. The dynamics of small mammal populations in Rocky Highveld Grassland, Telperion, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Monadjem A. 1997. Stomach contents of 19 species of small mammals from Swaziland. South African Journal of Zoology 32: 23–26.
Monadjem, A. 1998. Mammals of Swaziland. The Conservation Trust of Swaziland and Big Game Parks.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
NW READ. 2014. North West Environment Outlook Report 2013. North West Provincial Government, Mahikeng, South Africa.
O’Farrell PJ, Donaldson JS, Hoffman MT, Mader AD. 2008. Small mammal diversity and density on the Bokkeveld escarpment, South Africa-implications for conservation and livestock predation: short communications. African Zoology 43: 117–124.
Perrin MR. 1981. Notes on the activity patterns of 12 species of southern African rodents and a new design of activity monitor. South African Journal of Zoology 16: 248–258.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Roberts, A. 1951. The Mammals of South Africa. Central New Agency, Johannesburg, South Africa.
Russo IM, Chimimba CT, Bloomer P. 2010. Bioregion heterogeneity correlates with extensive mitochondrial DNA diversity in the Namaqua rock mouse, Micaelamys namaquensis (Rodentia: Muridae) from southern Africa-evidence for a species complex. BMC Evolutionary Biology 10: 307.
Russo IM, Chimimba CT, Bloomer P. 2006. Mitochondrial DNA differentiation between two species of Aethomys (Rodentia: Muridae) from southern Africa. Journal of Mammalogy 87: 545– 553.
Shortridge, G.C. 1942. Field notes on the first and second expeditions of the Cape Museums mammal survey of the Cape Province, and descriptions of some new subgenera and subspecies. Annals of the South African Museum 3: 281-291.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor PJ, Munyai A, Gaigher I, Baxter R. 2015. Afromontane small mammals do not follow the hump-shaped rule: altitudinal variation in the Soutpansberg Mountains, South Africa. Journal of Tropical Ecology 31: 37-48.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Visser, D.S. and Robinson, T.J. 1986. Cytosystematics of the South African Aethomys (Rodentia: Muridae). South African Journal of Zoology 21: 264–268.
Visser, D.S. and Robinson, T.J. 1987. Systematic implications of spermatozoan and bacular morphology for the South African Aethomys. Mammalia 51: 447–454.
Watson JP. 2006. Check list of the mammals of Tussen-die-Riviere Provincial Nature Reserve, Free State Province, South Africa. Koedoe 49: 111-117.