Namaqua Dune Mole-rat
Bathyergus janetta

2025 Red list status
Endangered
Regional Population Trend
Unknown
Change compared
to 2016
Uplisted
Overview
Bathyergus janetta – Thomas & Schwann, 1904
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – BATHYERGIDAE – Bathyergus – janetta
Common Names: Namaqua Dune Mole-rat, Namaqua Dune Blesmol (English), Namakwa-duinmol, Namakwalandse Duinmol (Afrikaans)
Synonyms: Bathyergus inselbergensis, Bathyergus plowsi
Taxonomic Note: Although previously regarded as only subspecifically diverse from the Cape Dune Mole-rat, B. suillus Ellerman et al. 1953), the Namaqua Dune Mole-rat is regarded as a distinct species (De Graaff 1981). Although no formal subspecies are recognised, there is considerable geographical variation in size and colour that may indicate coastal (smaller and paler) and inland (larger and black with silvery flanks) subspecies (Jarvis 2013). A recent genomic study found paraphyly of B. suillus and B. janetta (Sumbera et al. 2024), confirming previous research by Visser et al. (2014). Despite this, B. janetta is being retained as a separate species based on morphological and distributional differences.
Red List Status: EN – Endangered A4(a) (IUCN version 3.1)
Assessment Information
Assessors: Bennett, N.C.1 & da Silva, J.M.2
Reviewer: Patel, T.3
Institutions: 1University of Pretoria, South Africa,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors & Reviewers: Bennett, N.C., Maree, S. & Jarvis, J.
Previous Contributors: Relton, C., Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
The Namaqua Dune Mole-rat is now listed as Endangered due to the threat of continued habitat degradation of three isolated subpopulations. There is evidence for population decline despite the species occupying habitats that are highly protected and restricted from the public. However, the presumed lack of dispersal and increasing habitat degradation as well change in climate with reduced rainfall remains the major concerns for subpopulations of this species. This is especially true along the coastal regions, where the threat of diamond mining is yet to be quantified as well as inland populations where rainfall has failed and geophytes on which the mole-rats feed have disappeared. If the mines are rehabilitated, the species may still not be able to recolonize these sites. Resultantly, this species should be reassessed when data on the impact of mining become available. Small livestock farming around Kamieskroon has severely threatened the species due to overgrazing.
Regional population effects: Due to disjunctions in the distribution, dispersal is impossible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Climate change and habitat degradation are the two major drivers of the reduction in numbers of this species.
Red List Index
Red List Index: Uplisted
Recommended Citation: Bennett NC & da Silva JM. 2025. A conservation assessment of Bathyergus janetta. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
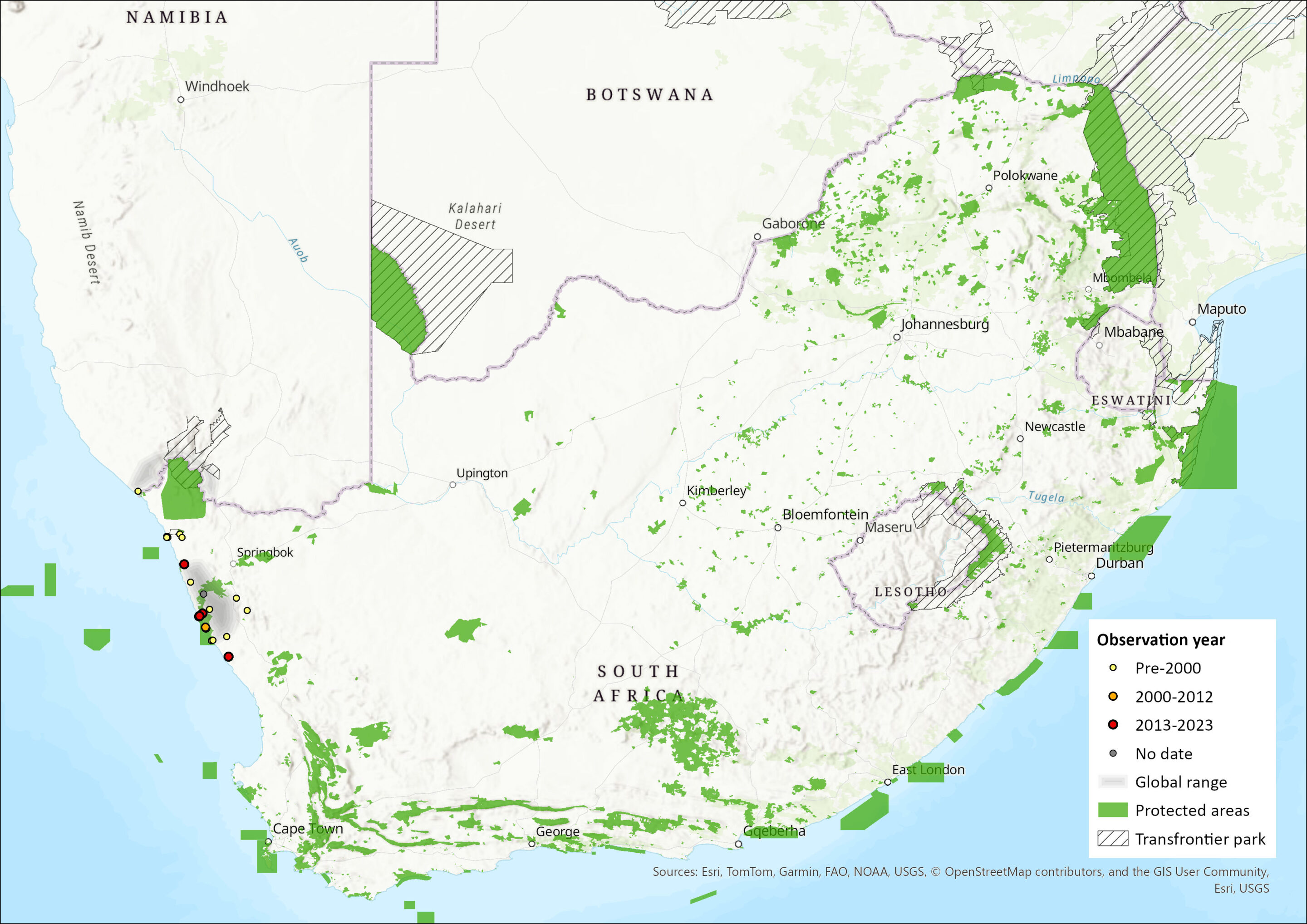
This species ranges from the north-western regions of the Northern Cape (near Rondawel) northwards along the coast to about 90 km into southern Namibia (Faulkes et al. 2004, Jarvis 2013). Inland, it extends from Springbok and Kamieskroon to Ezelfontein in the Kamiesberg Mountains (Roberts 1951, De Graaff 1981, Jarvis 2013). It is restricted primarily to sandy soils in coastal dunes. A number of isolated populations have been identified in the following areas of South Africa: Alexander Bay on the Orange River, from Port Nolloth to Groenrivier, from Steinkop to Kamieskroon, and in the red dunes of Klawer. Although this species rarely occurs above an altitude of 300 m, in the Kamiesberg it has been found at heights of 1,350 m asl (Skinner and Chimimba 2005). North of the Orange River, in the Sperrgebiet (Diamond mining area), Namibia, it occurs on seepage areas and inselbergs (isolated hills), where precipitation is higher than lower lying areas. Much of its distribution in Namaqualand overlaps with Cryptomys hottentotus, and towards Groenriviersmond and Rondawel, in the south of its range, it occurs sympatrically with B. suillus (Skinner and Chimimba 2005, Jarvis 2013). The estimated extent of occurrence within the assessment region was 10,234 km2. A recent expedition to Alexander Bay and Port Nolloth has revealed local extinctions (R. Sumbera pers comm.).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 300
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Namaqua Dune Mole-rat (Bathyergus janetta) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate Change and overgrazing by small livestock farmers have reduced the numbers to 2 individuals/km2 (N. Bennett Pers Obs.), uplisting the species to Endangered.
Population information
Namaqua Dune Mole-rats are found commonly within their range. However, their habitat is highly fragmented, and their distribution is restricted. Their patchy distribution is especially prominent in the most arid parts of the range where they are often associated with seepage lines where geophytes are found (Jarvis 2013). Their population density increases further inland where rainfall is higher, with up to 44 individuals / km² recorded (Herbst et al. 2004, Jarvis 2013).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | – |
Extreme fluctuations in the number of subpopulations: Yes
Continuing decline in number of subpopulations: Yes
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Minimum 20
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 100 years: There is a risk of extinction in the next 100 years due to climate change
Population genetics
While a recent molecular study found B. janetta to be paraphyletic with B. suillus (Sumbera et al. 2024), it is likely the mitochondrial marker used (cytB) was unable to detect the recent divergence between the species. Extensive morphological and distributional differences warrant its species status. Three subpopulations are currently known (Steinkop, Kamieskroon, toward Groenrivier), while two subpopulations (Alexander Bay, Port Nolloth) are thought to be extirpated. No estimates of population size are available for the separate subpopulations or the species as a whole.
Based on this information, only one of the Convention of Biological Diversity’s Global Biodiversity Framework’s genetic indicators can be calculated – the proportion of populations maintained. With 3 of 5 subpopulations remaining, the indicator receives a value of 0.6.
A fine scale genomic study is recommended to uncover the relationships within this species and between its sister taxa, B. suillus, and to obtain estimates of effective population size.
Habitats and ecology
Along the coast, this species is located primarily on sand dunes, while inland they make use of loam and consolidated alluvial soils, where there are geophytes and succulent vegetation (Jarvis 2013). They are considered endemic to the Succulent Karoo biome of southern Africa (Mugo et al. 1995). Average rainfall in this area is usually less than 400 mm / annum but has declined in the last 6 years. Like Georychus capensis, this species is typically solitary and subterranean, living in burrow systems up to 00.25-0.45m below the soil surface. They are seasonal breeders (Herbst et al. 2004) and following a gestation period of 52 days, B. janetta gives birth to between two and seven young. Their home range ranges from 398 m2 to 12,438 m2 (Herbst 2002). They generally consume subterranean storage organs such as succulent roots and bulbs, and occasionally pull plants, such as daisies and herbs, from the surface down into their burrows to feed on underground (Jarvis 2013).
Ecosystem and cultural services: Similar to other Mole-rats (Cryptomys hottentotus and Georychus capensis), the Namaqua Dune Mole-rat is an important eco-engineer and plays a role in modifying soil properties and increasing the humic content of the sands in which it occurs (Hagenah and Bennett 2013) Alexander bay. Burrowing activities by mole-rats may also enhance infiltration and the water holding capacity of soil (Hagenah and Bennett 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: 8 years
Age at Maturity: Female or unspecified: 1.5 years
Age at Maturity: Male: 1.5 years
Size at Maturity (in cms): Female: 28.3cm, 439g
Size at Maturity (in cms): Male: 27.3 cm, 330g
Longevity: 8-10 years
Average Reproductive Age: 1.5 to 2 years
Maximum Size (in cms): 700g
Size at Birth (in cms): 15.4g
Gestation Time: 50 to 60 days
Reproductive Periodicity: One to two litters per annum
Average Annual Fecundity or Litter Size: 2 (1-4)
Natural Mortality: Black-footed Cat and jackal
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Dispersal above ground to mate and juveniles disperse to set up new systems
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: No trade
National Commercial Value: No value
International Commercial Value: None
End Use: None
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: None
Threats
Climate change has already been seen to affect the species and could further present an increasing threat by reducing habitat quality and availability, as the geophytes on which these animals feed could dry out and disappear (Hoffman et al. 2009). Overgrazing by goats and sheep reduces the quality of above ground vegetation on which the species forages. Habitat loss through diamond mining threatens the subpopulations along the coastal belts of southern Namibia and the north-western parts of South Africa. Fortunately, the general public is entirely prohibited from entering these regions, therefore limiting the threats of human disturbance. However, data are needed on the impact of diamond mining on this species.
Conservation
Although this species lacks formal protection in many areas, it occurs within the Namaqua National Park (South Africa), the |Ai-|Ais/Richtersveld Transfrontier Park (South Africa and Namibia), and Sperrgebiet (Namibia), which have extremely low levels of human disturbance and contain key habitats for this species. Although locally abundant, subpopulations of this species are fragmented, and reintroductions are not possible. No current conservation interventions are in place for this species. Data on the impact of diamond mining should be collected before a conservation strategy can be formulated.
Recommendations for land managers and practitioners:
- Livestock farmers should reduce stocking rates to conserve ground cover.
Research priorities:
- Investigating the current extent of distribution and number of subpopulations.
- Long-term monitoring of population dynamics to establish baseline information.
- Determining the impact of diamond mining.
- A fine scale genomic study is recommended to uncover the relationships within this species and between its sister taxa, B. suillus, and to obtain estimates of effective population size.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas.
Bibliography
De Graaff, G. 1981. The Rodents of Southern Africa. Butterworths, Durban, South Africa.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758 to 1951: A Reclassification. British Museum (Natural History), London, UK.
Faulkes CG, Verheyen E, Verheyen W, Jarvis JUM, Bennett NC. 2004. Phylogeographical patterns of genetic divergence and speciation in African mole-rats (Family: Bathyergidae). Ecology 13: 613–629.
Hagenah N, Bennett NC. 2013. Mole rats act as ecosystem engineers within a biodiversity hotspot, the Cape Fynbos. Journal of Zoology 289: 19–26.
Herbst M, Jarvis JUM, Bennett NC. 2004. A field assessment of reproductive seasonality in the threatened wild Namaqua dune mole-rat (Bathyergus janetta). Journal of Zoology 263: 259–268.
Herbst M. 2002. The biology and reproductive ecology of the Namaqua dune mole-rat, Bathyergus janetta from the Northern Cape Province, South Africa. M.Sc. Thesis. University of Pretoria.
Herbst, M., Jarvis, J.U.M. & Bennett, N.C. 2004. A field assessment of reproductive seasonality in the Red data listed wild Namaqua dune mole-rat, Bathyergus janetta. Journal of Zoology. 263: 259-268.
Hoffman MT, Carrick PJ, Gillson L, West AG. 2009. Drought, climate change and vegetation response in the succulent karoo, South Africa. South African Journal of Science 105: 54–60.
Jarvis JUM. 2013. Bathyergus janetta Namaqua Dune Mole-rat. Pages 644–646in D. C. D. Happold, editor.Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Mugo DN, Lombard AT, Bronner GN, Gelderblom CM. 1995. Distribution and protection of endemic or threatened rodents, lagomorphs and macrosceledids in South Africa. South African Journal of Zoology 30: 115–126.
Roberts A. 1951. The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Šumbera R., Uhrová, M., Montoya-Sanhuesa G., Bryjová, A., Bennett, N.C & Mikula O. 2024. Genetic diversity of the largest African mole-rat genus, Bathyergus. One, two or four species? Molecular Phylogenetics and Evolution 199: 108157 1-17. https://doi.org/10.1016/j.ympev.2024.108157