
Long-tailed Serotine Bat

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Cnephaeus hottentotus – (A. Smith, 1833)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Eptesicus – hottentotus
Common Names: Long-tailed Serotine Bat, Hottentot Serotine Bat, Hottentot Bat, Long-tailed Greater Serotine Bat, Long-tailed House Bat (English), Langstert-dakvlermuis (Afrikaans)
Synonyms: Vespertilio hottentotus A. Smith, 1833; Vespertilio hottentata A. Smith, 1833; Eptesicus hottentotus pallidior (Shortridge, 1942)
Taxonomic Note:
Based on recently published molecular studies, and on novel qualitative and quantitative morphological comparisons of representatives of Histiotus and New and Old World Eptesicus, Cláudio et al. (2023) provided evidence for restricting the name Eptesicus to the species E. fuscus and E. guadeloupensis, and allocating the remaining New World species under a new genus, keeping Histiotus as a full genus, and raising Cnephaeus to generic rank to comprise all Old World taxa currently under Eptesicus. Based on this research, Eptesicus hottentotus should now be referred to as Cnaephaeus hottentotus.
Earlier morphological and molecular research on E. hottentotus (now C. hottentotus) indicated that this species is the only member of the genus in southern Africa (Kearney et al. 2002). Within this taxon, three subspecies have previously been recognised in southern Africa (Monadjem et al. 2020): E. h. hottentotus from the Eastern and Western Cape (Meester et al. 1986), E. h. pallidor from the Northern Cape and Namibia (Meester et al. 1986), and E. h. bensoni from Zimbabwe, Mozambique and eastern South Africa (Skinner & Chimimba 2005). Further molecular studies are required.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Lotter, C1., Balona, J.2 & da Silva, J.M.3
Reviewer: Richards, L.R.4
Institutions: 1Inkululeko Wildlife Services (Pty) Ltd, 2Gauteng and Northern Regions Bat Interest Group, 3South African National Biodiversity Institute, 4Durban Natural Science Museum.
Previous Assessors and Reviewers: Watson, J, Monadjem, A., Cohen, L., Jacobs, D., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D. & Page-Nicholson, S.
Assessment Rationale
This species is listed as Least Concern in view of its wide distribution, and its occurrence in many protected areas across its range. Within the assessment region, it is presumed that more than 50 colonies occur in at least eight protected areas. However, despite being widespread, this species has a patchy or sparse occurrence, which is probably a function of its specific roosting requirements in suitable rocky terrain (Monadjem et al. 2020). Moreover, where there is suitable rocky habitat for this species, it is typically found in low abundance. Long-term passive acoustic bat call monitoring data from various proposed and operational wind farm sites in southern Africa confirms that although this species is widespread in the assessment region, it is closely associated with rocky terrain, where relatively few calls of this are typically recorded (Inkululeko Wildlife Services unpubl. data). Based on wing morphology and clutter-edge foraging behaviour, the Long-tailed Serotine Bat has been categorised as having a “Medium” risk of fatality from turbines (MacEwan et al. 2020). For these reasons, even though there are no known fatalities of this species from operational wind farms in South Africa (Doty & Martin 2013; MacEwan 2016; Aronson 2022; Inkululeko Wildlife Services, unpubl. data), the possible threat of wind energy development on the species must be closely monitored.
Regional population effects: The population is continuous in countries neighbouring the assessment region and the species has intermediate wing-loading (Schoeman & Jacobs 2008), which indicates that dispersal and rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Lötter C, Balona J & da Silva JM. 2025. A conservation assessment of Cnephaeus hottentotus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This largely southern African species occurs widely but sparsely from extreme southwestern Angola and through parts of Namibia in the west, through South Africa, Eswatini, Lesotho, Botswana, Zimbabwe, with isolated records in Zambia, central Mozambique, and southern Malawi (Kearney et al. 2008), and a single record as far north as southwestern Kenya (ACR 2024). In the assessment region, the species occurs in eight provinces of South Africa: Limpopo, Mpumalanga, KwaZulu-Natal, Free State, North West, Northern Cape, Eastern Cape and the Western Cape. The type specimen is from Uitenhage, Eastern Cape (Monadjem et al. 2020). The estimated extent of occurrence is 1,344,572 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 25 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 2430 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
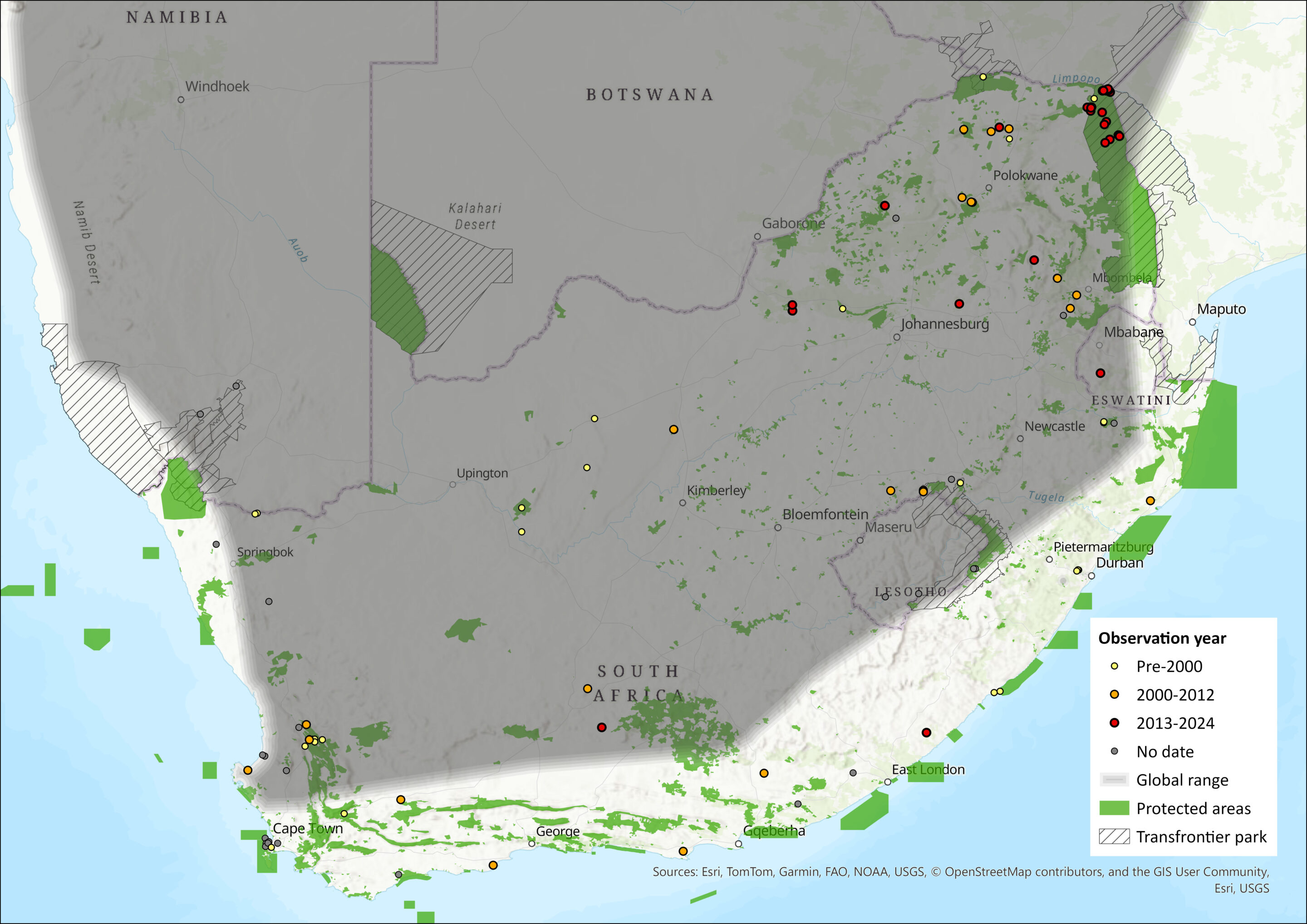 Figure 1. Distribution records for Long-tailed Serotine Bat (Cnephaeus hottentotus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Long-tailed Serotine Bat (Cnephaeus hottentotus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020). However, there has not been any recent studies and information available to predict the impacts of climate change on this species.
Population Information
The Long-tailed Serotine Bat is relatively well represented in museums, with over 60 specimens examined by Monadjem et al. (2020). This species is widely but sparsely distributed and generally has a low abundance where it does occur. It is more common in some regions of its distribution such as Zimbabwe, but seems to be rarer in South Africa. The patchy nature of its distribution is probably a function of its roosting requirements in suitable rocky terrain (Monadjem et al. 2020). Shortridge (1942) indicated this bat was not plentiful around Goodhouse in the Northern Cape of South Africa and only observed half a dozen individuals. In other instances, specimens have either been found singly or in groups of two to five (Watson 1998; ACR 2015). Small groups of two to four individuals have been reported from rock crevices (Herselman and Norton 1985) and caves (Skinner and Chimimba 2005). Long-term passive acoustic bat call monitoring data from various proposed and operational wind farm sites in southern Africa confirms that although this species is widespread in the assessment region, it is closely associated with rocky terrain, where relatively few calls of this are typically recorded (Inkululeko Wildlife Services unpubl. data).
Current population trend: Stable
Continuing decline in mature individuals? No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown, but it is presumed that more than 50 colonies occur in at least eight protected areas.
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The population genetic structure for this species is unknown, but it is thought to occur in small, potentially isolated colonies. If so, this species could harbour several genetically distinct subpopulations. Assuming individuals can travel between colonies, and hence the species exists as a single metapopulation within the assessment region, the low number of individuals (~5 animals in each of the 50 colonies, equating to ~500 animals) could be cause for concern should threats arise. A population genetic study is recommended (or at least a phylogeographic study) to better ascertain genetic connectivity within the species.
Habitats and ecology
This species is often associated with mountainous or broken rocky terrain in close proximity to water (see references in ACR 2024, and based on recent records from Schaapplaats Farm, Free State), occurring in a range of habitats including semi-arid Karoo and Kalahari, montane grasslands, marshland and well-wooded riverbanks (Skinner & Chimimba 2005). It roosts in small groups of two to five individuals in caves, mines (Skinner & Chimimba 2005) and rock crevices (Herselman & Norton 1985). A single specimen was collected hanging on the outside wall of a building in Zimbabwe (Skinner and Chimimba 2005). In Zimbabwe, it occurs in Miombo woodland in gorges and granitic hills (Cotterill 1996), while in South Africa it is usually captured or recorded with ultrasonic bat detectors near rocky outcrops, faces, and gorges (Monadjem et al. 2010; Inkululeko Wildlife Services unpubl. data). This species is a clutter-edge forager, feeding primarily on coleopteran species (Schoeman & Jacobs 2003; Schoeman 2006).
No detailed data on reproductive behaviour is currently available for the assessment region. However, a post-lactating female was netted over a river at Schaapplaats Farm by researchers from the University of the Free State in late February, suggesting that breeding likely occurs during the austral summer. This inference is further supported by observations from Zimbabwe, where heavily pregnant or lactating females were reported in mid-November (Cotterill & Happold, 2013).
Ecosystem and cultural services: As this species is insectivorous, it is likely to contribute to controlling insect populations that damage crop plants (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.4. Forest -> Forest – Temperate | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Forearm mean = 5.10 ± 0.16 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 4.88 ± 0.16 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): Female forearm length = 5.30 cm; Male forearm length = 5.20 cm
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: Seasonally monoestrous, likely giving birth between November and December (Cotterill and Happold 2013).
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: There is evidence that this species is preyed upon by Barn Owls (Tyto alba) in the Western Cape (Avery et al. 2005), and it is also known to be a prey item of Bat Hawks (Macheiramphus alcinus) (Mikula et al. 2016).
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Unknown.
Congregatory: Roosts in small groups of less than five individuals (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to indicate that this species is traded within the assessment region.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Due to the specific roosting requirements and associated sparse occurrence and small colony sizes of this species, it may be significantly impacted by disturbances that result in degradation or loss of roosting habitat and/or high mortality of individuals. Mining and renewable energy development, in particular, may post a serious threat to colonies or populations in rocky regions. Where wind farms have or will involve construction of turbines on rocky ridges, hills and mountain tops, any associated colonies of the Long-tailed Serotine could be significantly impacted or even lost by roost disturbance (from e.g. blasting or excavation activities) and/or turbine-related fatality or displacement of individuals. Based on wing morphology and clutter-edge foraging behaviour, the Long-tailed Serotine has been categorised as having a “Medium” risk of fatality from turbines (MacEwan et al. 2020). For these reasons, even though there are to date no known fatalities of this species from operational wind farms in South Africa (Doty & Martin 2013; MacEwan 2016; Aronson 2022; Inkululeko Wildlife Services, unpubl. data), the possible threat of wind energy development on the Long-tailed Serotine must be closely monitored. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) may also pose a serious threat to colonies that are roosting in old mine tunnels. Therefore, systematic monitoring of colony sizes and trends is needed, and the status of this species must be checked every few years or more often.
Conservation
The Long-tailed Serotine Bat has been recorded from several protected areas in South Africa including the Kruger National Park, Mapungubwe National Park, Magaliesberg Natural Environment, Ithala Game Reserve, Ukahlamba Drakensberg World Heritage Site, Sterkfontein Nature Reserve, Cederberg Wilderness Area and Table Mountain National Park. It is presumably also present in other protected areas within other parts of this species’ range. No direct interventions are necessary, but colonies of this species would benefit from the protection of rocky habitat types, riverine areas and surrounding natural vegetation where renewable (especially wind but also solar) energy projects are developed.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
- Protection of mountainous areas where the species is likely to roost.
- Development of wind farms must avoid encroachment into the prescribed buffers around confirmed roosts and suitable rocky habitat (e.g. ridges, gorges, cliff faces, batholiths, etc) where roosts of this species potentially occur.
- Potential future turbine-fatalities of this species must be reduced with interventions such as ultrasound to deter bats and curtailing turbines at low wind speeds.
Research priorities:
- Taxonomic resolution through an integrative approach is needed.
- Further studies are needed into the distribution of this species.
- Possible future wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species in eastern southern Africa.
- Systematic monitoring of subpopulation trends is required.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- Grounded or injured bats should be reported to the nearest bat interest group or local wildlife veterinarian.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.Avery, D.M., Avery, G. and Palmer, N.G. 2005. Micromammalian distribution and abundance in the Western Cape Province, South Africa, as evidenced by Barn owls Tyto alba (Scopoli). Journal of Natural History, 39(22), pp.2047-2071.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cláudio, V.C., Novaes, R.L.M., Gardner, A.L., Nogueira, M.R., Wilson, D.E., Maldonad, J.E., Oliveira, J.A., and Moratelli, R. 2023. Taxonomic re-evaluation of New World Eptesicus and Histiotus (Chiroptera: Vespertilionidae), with the description of a new genus. Zoologia 40: https://doi.org/10.1590/S1984-4689.v40.e22029
Cotterill, F. P. D. 1996. New distribution records of insectivorous bats of the families Nycteridae, Rhinolophidae and Vespertilionidae (Microchiroptera: Mammalia) in Zimbabwe. Arnoldia (Zim.) 10: 71–89.
Cotterill, F.P.D. and Happold, M. 2013. Eptesicus hottentotus Long-tailed serotine, pg 555-556. In: Happold, M and Happold, D.C.D (eds) Mammals of Africa: Volume IV. Blomsbury Publishing, London.
Doty AC, Martin AP. 2013. Assessment of bat and avian mortality at a pilot wind turbine at Coega, Port Elizabeth, Eastern Cape, South Africa. New Zealand Journal of Zoology 40: 75–80.Herselman, J. C. and Norton, P. M. 1985. The distribution and status of bats (Mammalia: Chiroptera) in the Cape Province. Ann. Cape. Prov. Mus. 16: 73–126.
Juste, J., Benda, P., Garcia-Mudarra, J.L. and Ibáñez, C. 2013. Phylogeny and systematics of Old World serotine bats (genus Eptesicus, Vespertilionidae, Chiroptera): an integrative approach. Zoologica Scripta 42(5): 441–457.
Kearney TC, van Cakenberghe V, Seamark EC, Ndhlovu C, Cohen L. 2008. Notes on the taxonomy and distribution of Eptesicus hottentotus (Chiroptera: Vespertilionidae) and reidentification of some museum voucher specimens. Annals of the Transvaal Museum 45: 127–134.
Kearney, T., Volleth, M., Contrafatto, G. and Taylor, P. J. 2002. Systematic implications of chromosome GTC-band and bacula morphology for Southern African Eptesicus and Pipistrellus and several other species of Vespertilioninae (Chiroptera: Vespertilionidae). Acta Chiropterologica 4: 55-76.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum, Bloemfontein 10:177-241.
MacEwan KL. 2016. Fruit bats and wind turbine fatalities in South Africa. African Bat Conservation News 42: 3–5.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. (2020). South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Mikula, P., F. Morelli, R. K. Lučan, D. N. Jones and P. Tryjanowski (2016). Bats as prey of diurnal birds: a global perspective. Mammal Review, 46 (3): 160 – 174
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2010. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.Schlitter, D. A. and Aggundey, I. R. 1986. Systematics of African bats of the genus Eptesicus (Mammalia: Vespertilionidae). 1. Taxonomic status of the large serotines of eastern and southern Africa. Cimbebasia 8: 168-174.
Schoeman, C. M., Jacobs, D. S. 2003. Support for the allotonic frequency hypothesis in an insectivorous bat community. Oecologia 134: 154–162.
Schoeman, M. C. 2006. The relative influence of competition and coevolution on the community structure of insectivorous bats in southern Africa. PhD thesis, University of Cape Town, Cape Town.
Schoeman, M.C. and Jacobs, D.S. 2008. The relative influence of competition and prey defences on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS one 3(11): e3715.
Shortridge, G.C. 1942. Field notes on the first and second expeditions of the Cape Museums mammal survey of the Cape Province, and descriptions of some new subgenera and subspecies. Annals of the South African Museum 3: 281-291.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Sowler S, Stoffberg S, MacEwan K, Aronson J, Ramalho R, Forssman K, Lötter C. 2017. South African Good Practice Guidelines for Surveying Bats at Wind Energy Facility Developments – Pre-construction. Endangered Wildlife Trust and the South African Bat Assessment Advisory Pane.
Watson, J. P. 1998. New distributional records for three microchiropteran bats (Vespertillionidae [Vespertilionidae], Rhinolophidae) from the Free State Province, South Africa. South African Journal of Wildlife Research 28(4): 127-131.
