Long-tailed Forest Shrew
Myosorex longicaudatus

2025 Red list
status
M. L. boosmani

2025 Red list
status
Myosorex longicaudatus
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Myosorex longicaudatus – Meester & Dippenaar, 1978
ANIMALIA – CHORDATA – MAMMALIA – EULIPOTYPHLA – SORICIDAE – Myosorex – longicaudatus
Common Names: Long-tailed Forest Shrew (English), Langstertbosskeerbek (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
A molecular study found this species to be genetically distinct from the other southern African Myosorex species (Willows-Munro and Matthee 2009). In the phylogeny presented, Myosorex longicaudatus is sister to a clade containing the Tanzanian species M. geata and M. kihaulei, while all the other southern African Myosorex species formed a well-supported monophyletic lineage.
This species will possibly be split into M. l. longicaudatus, the type specimen of which originates from the Knysna Forest (Meester and Dippenaar 1978), and M. l. boosmani.
The latter subpopulation, tentatively labelled a subspecies, is found in the Boosmansbos Forest (Langeberg Mountains), and differs in size and cranial configuration from subpopulations in the Knysna Forests (Dippenaar 1995). It was recorded from this locality in May 2025, some 46 years after its original discovery (CapeNature 2025). Molecular work should be undertaken to clarify the taxonomic status of the putative subspecies, as it is likely they represent two separate species.
Red List Status:
Myosorex longicaudatus EN – Endangered, B1ab(ii,iii)
M.l.boosmani, CR – Critically Endangered, B2ab(ii,iii)
Assessment Information
Assessors: Taylor, P.1 & da Silva, J.2
Reviewer: de Villiers, M.S.3
Contributors: Smith, C.4 & Patel, T.4
Institutions: 1University of the Free State, South Africa,2South African National Biodiversity Institute,3CapeNature, 4Endangered Wildlife Trust
Previous Assessors: Baxter, R., Willows-Munro, S. & Taylor, P.
Previous Reviewer: Child, M.F.
Previous Contributors: Roxburgh, L. & Hoffmann, M.
Assessment Rationale
This is an endemic species restricted to a narrow range of forest within the Western and Eastern Cape provinces. It is restricted to pristine primary habitat that has not been degraded. Ongoing urban expansion (having increased by 8% and 6% for Western and Eastern Cape provinces between 2000 and 2013 respectively), especially along the coast, is inferred to have reduced and fragmented suitable habitat. Correspondingly, Southern Coastal and Swamp forest have declined by at least 1–3% between 2000 and 2013. The current extent of occurrence (EOO) for this species may be <5,000 km², while its area of occupancy (AOO), based on remaining forest patches, is estimated to be 691 km² and 10.1 km² for M. l. longicaudatus and M. l. boosmani respectively. Furthermore, this species needs a moist microhabitat to survive, which makes if susceptible to extensive fragmentation and climate change. Climate models project that suitable Afromontane Forest will mostly disappear in the coming decades (projected 68-70% decrease between 1975 and 2050).
Although well-protected in some areas, forests are becoming degraded through over-use and continually encroached upon by residential and commercial development along the coast. Thus, due to restricted range, fragmented remaining habitat patches, and both current and projected (from climate change) loss of habitat and habitat quality, we list this species as Endangered B1ab(ii,iii). Similarly, the subpopulation in the Langeberg Mountains qualifies for Critically Endangered B2ab(ii,iii), as its effective AOO may well be slightly less than the 10.1 km² estimated. Although its habitat is protected, its vulnerability is due to the predicted effect of climate change on the only known locality. The EOO is probably an underestimate due to lack of recent systematic surveys (apart from a recent expedition to Boosmansbos Forest) and the southern aspect of the Southern Cape Fold Mountains is likely to contain suitable habitat for the species. Surveys are desperately needed to confirm its presence in these habitats. However, we take a precautionary approach as the current EOO value is well below the threshold for Endangered and it is not clear how much further the EOO could potentially be expanded. Further genetic work is also needed to determine the taxonomic status of M. l. longicaudatus and M. l. boosmani, and a re-assessment may be needed. Similarly, the ongoing habitat loss and from urban expansion and climate change must be monitored. Some of the known localities such as Boosmansbos and Diepwalle are protected, so non-climate change anthropogenic degradation is unlikely.
Key interventions include the protection of forest habitats, and the creation of corridors between patches to facilitate gene flow and allow adaptation to climate change, as well as the enforcement of regulations restricting disturbance to protected forests.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor P & da Silva JM. 2025. A conservation assessment of Myosorex longicaudatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
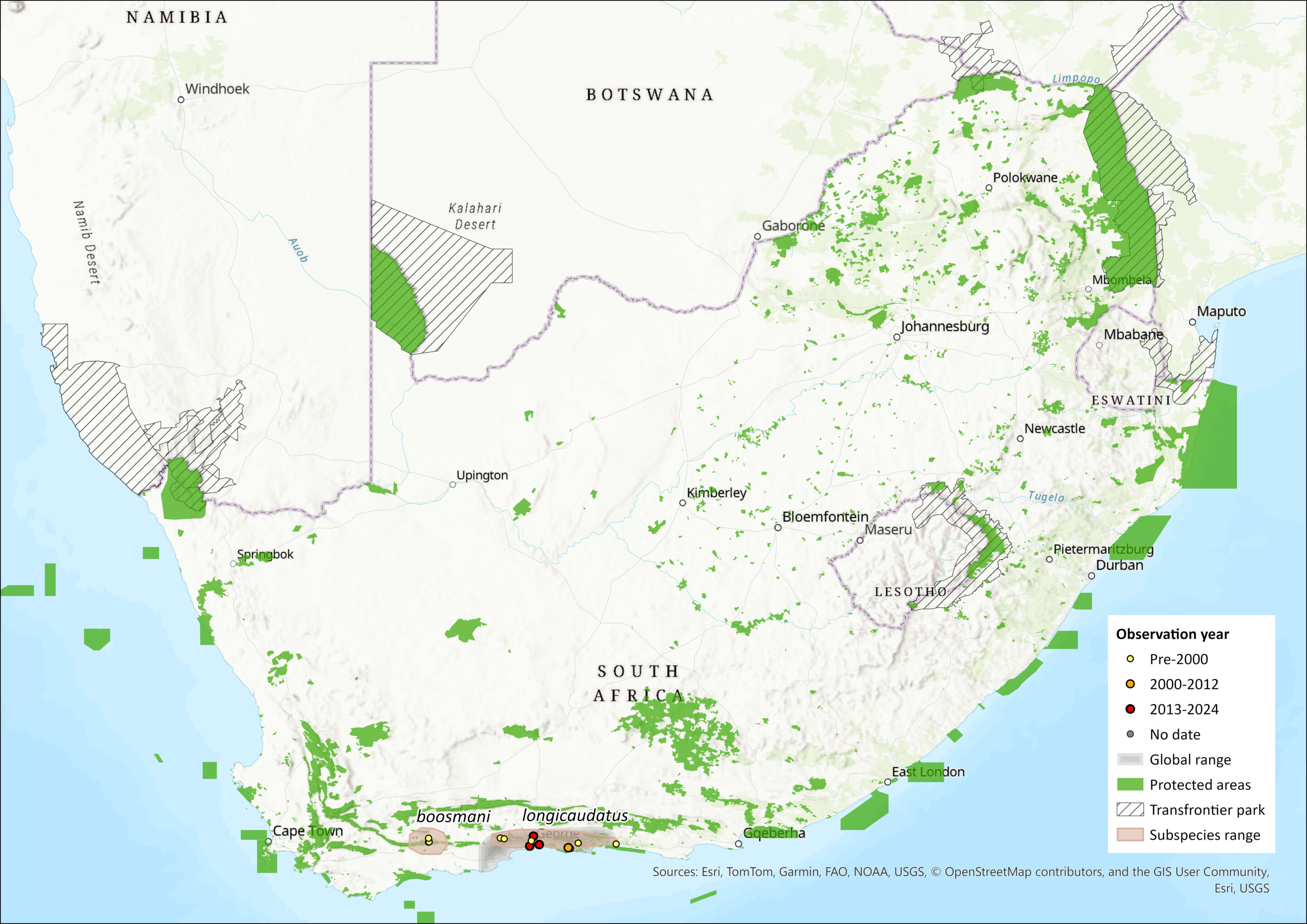
The Long-tailed Forest Shrew was only discovered in 1976 and is restricted to Afromontane evergreen forest, mostly preserved along the deep valleys and cooler south-facing slopes along the south coast of South Africa. It also occurs in fynbos in the Langeberg Mountains. It is a relic species from previous southward radiations, as reflected by its closest phylogenetic link being that of Myosorex geata – a highly localised endemic of the Uluguru Mountains in Tanzania (Willows-Munro and Matthee 2009). The remaining three South African species cluster as a monophyletic entity with strong support for a sister taxon association between M. cafer and M. sclateri (Willows-Munro and Matthee 2009).
Specifically, the species is known from the southeastern parts of the Western and Eastern Cape provinces, occurring from the Langeberg Mountains, Western Cape to the Lottering Forest, Eastern Cape. Within the Knysna Forest, it occurs in Lottering State Forest, Diepwalle State Forest, Tonnelbos Forest and the Ruitersbos State Forest (Skinner and Chimimba 2005). It generally occurs at elevations up to 2,000 m asl. The Langeberg subpopulation (M. longicaudatus boosmanii) is separated from the Knysna subpopulations by the dry Gouritz River Valley, where M. l. boosmani is unlikely to occur (Dippenaar 1995). It is sympatric with M. varius but allopatric with M. cafer (Skinner and Chimimba 2005).
The extent of occurrence (EOO) for this species is estimated to be somewhere between 2,214 and 14,123 km²; the data are uncertain, but it is possible that the true EOO is smaller than 5,000 km². Its area of occupancy (AOO) was estimated using current (2014) remaining forest patches, which corresponds to 691 km² (and 582 km² if we include only the forest area that intersects with the EOO) and 10.1 km² for M. l. longicaudatus and M. l. boosmani respectively. Myosorex l. longicaudatus is known from only four localities, and M. l. boosmani from just one locality. These localities are suspected to be isolated due to the species’ presumed inability to disperse across unsuitable habitat. We note that the EOO may be an underestimate as most data collection in the region has been ad hoc, with the last semi-systematic surveys carried out in 1970s and early 1990s by CapeNature (G. Palmer pers. comm. 2015). The most recent records of M. l. longicaudatus are two records from 2022 (iNaturalist) and records from 1995 and 1996 (CapeNature). Previously all records are from 1975 to 1981 (138 occurrences listed in GBIF; https://www.gbif.org/occurrence/search?offset=0&taxon_key=2435920). A single specimen of M. l. boosmani was located at the typic locality in May 2025 (unpublished data, CapeNature). The Southern Cape Fold Mountains, particularly the southern aspect, may have suitable potential habitat for the more common subspecies and are relatively well protected (G. Palmer pers. comm. 2015). Surveys are needed to confirm its presence there.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 2000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Long-tailed Forest Shrew (Myosorex longicaudatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> Western Cape Province | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate modelling indicated a potential reduction in area of occupancy of 68–70% between 1975 and 2050 (Taylor et al. 2017), which, assuming a linear rate of loss equates to a 9.3% loss of suitable habitat over a ten-year period.
Population information
This species is suspected to be relatively common in suitable habitat, with the highest numbers of individuals found at the forest edge (Dippenaar 1995). Subpopulation numbers may vary as one survey found no specimens after intensive trapping in an area where a previous survey had caught quite high numbers (R. Baxter unpubl. data). No density estimates are available.
Population Information
Current population trend: Declining. Inferred from ongoing forest habitat loss.
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown, but may be only five based on remaining forest patches and verified localities.
Severely fragmented: Yes. Forest habitat is fragmented by residential and industrial development along the coast.
Number of Subpopulations: 5
Justification: Unknown, but may be only five based on remaining forest patches and verified localities.
quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While the species has been investigated in a phylogenetic context, no population genetic studies have been undertaken. However, it is expected that there is no current gene flow between the two currently recognised subspecies M. l. longicaudatus and M. L. boosnami given that they are separated by inhospitable habitat. While the species is considered common in suitable habitat, the lack of density estimates, and approximate population sizes makes prevents the calculation of effective population size (Ne).
Habitats and ecology
Long-tailed Forest Shrews are found in forests, forests edges, fynbos and boggy grassland, and depend on moist microhabitats (typically above the 800 mm isohyet). It is restricted to pristine primary habitat that has not been degraded. It is nocturnal. They have longer tails than the other three species of forest shrew occurring in the assessment region (74% of the length of the head and body compared with 46–50% in the others), which suggests an arboreal lifestyle (Meester and Dippenaar 1978). There is little geographic variation in body colour (Skinner and Chimimba 2005). Stomach content analysis suggests it eats primarily insects and seeds (Skinner and Chimimba 2005).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | Resident | Suitable | Yes |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
General notes regarding trade and use of this species: (Not specified)
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The main threat to shrews is the loss of forest habitats due to water abstraction from agriculture, industry and urban sprawl. Degradation of these habitats from overgrazing and imprudent fire regimes is the second most severe threat. Shrews have a high metabolic rate and thus rely on highly productive and stable environments.
Unfettered urban expansion, especially coastal development, is the major threat to this species. Loss of forest habitat, and fragmentation of remaining habitats, is likely to be causing significant population decline through lack of connection between patches, disruption of ecological functioning, and increased exposure to human disturbances at forest edges. Fragmentation also increases its susceptibility to climate change. Climate change is considered to be the principal emerging threat to this species (Taylor et al. 2017), both due to loss of habitat and because shrews cannot tolerate extremes of temperature for long and thus their foraging time will be reduced. Because of their high metabolism, low dispersal capacity and short life spans, climate change will reduce the amount of suitable habitat available. Habitat in neighbouring areas is arid and unsuitable for this species and thus it would not be able to disperse to other areas if the climate in its current range became unsuitable. It is thought that coastal subpopulations might be at less risk than non-coastal subpopulations.
Conservation
The main intervention for this species is the protection and restoration of Afromontane forest patches and ecotonal habitat. As habitat loss from climate change will be further compounded by habitat loss from land transformation (Driver et al. 2012), a critical intervention is to increase the extent of protected area networks that connect mountainous areas to lowland or coastal habitats, thus facilitating dispersal routes along elevational gradients. This species has confirmed presences in important formally protected areas such as the Diepwalle Forest Reserve and Garden Route National Park, Ruitersbos Nature Reserve and Boosmansbos Wilderness Area. Once further surveys have been conducted to identify additional subpopulations, biodiversity stewardship agreements with private landowners should be used to connect forest patches and conserve the integrity of associated wetlands and fynbos grasslands. Protecting such habitats may create dispersal corridors between forest patches that will enable adaptation to climate change.
All forests in South Africa are protected by law, although the degree to which they are enforced may vary. Increased enforcement of forest-related transgressions should be used to minimise disturbance to existing forest patches, as well as stricter zonation on development to decrease fragmentation of remaining forests. Assisted colonisation could become imperative if climate changes continue to make existing habitat within the species’ range unsuitable.
Recommendations for land managers and practitioners:
- More accurate estimates of forest patch occupancy through extensive live-trapping to attempt to recapture individuals should be conducted through dedicated surveys by specialists and conservation authorities, thus informing spatial conservation planning.
- Enforce regulations on developments that potentially impact forests.
- Surveys should be conducted to assess population size, trends and distribution of M. l. boosmani. As most of the patches they are found on are on protected areas, fire management is necessary.
Research priorities:
- Systematic surveys in the Southern Cape Fold Mountains to determine whether the extent of occurrence extends further.
- Molecular studies to resolve the taxonomic resolution of the two putative subspecies.
Encouraged citizen actions:
- Citizens are requested to submit any shrews killed by cats or drowned in pools to a museum or a provincial conservation authority for identification, thereby enhancing our knowledge of shrew distribution (carcasses can be placed in a Ziplock™ bag and frozen with the locality recorded).
Bibliography
CapeNature. 2025. Elusive Boosmansbos long-tailed forest shrew recorded for the first time in 46 years! https://www.capenature.co.za/news/2025/elusive-boosmansbos-long-tailed-forest-shrew-recorded-for-the-first-time-in-46-years.
Dippenaar, N. J. 1995. Variation in Myosorex longicaudatus (Soricidae) in the Southern Cape Province, South Africa. Journal of Mammalogy 76: 1071-1087.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria.
IUCN. 2020. The IUCN Red List of Threatened Species. Version 2020-2. Available at: www.iucnredlist.org. (Accessed: 13 June 2020).
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa
Meester, J. and Dippenaar N.J. 1978. A new species of Myosorex from Kynsna, South Africa (Mammalia: Soricidae). Annals of the Transvaal Museum 31: 29-43.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, P. J., Ogony, L., Ogola, J., Baxter, R. M. 2017. South African mouse shrews (Myosorex) feel the heat: using species distribution models SDMs and IUCN Red List criteria to flag extinction risks due to climate change and habitat loss. Mammal Research, 62:149-162. DOI 10.1007/s13364-016-0291-z.
Willows-Munro, S. and Matthee, C.A. 2009. The evolution of the southern African members of the shrew genus Myosorex: understanding the origin and diversification of a morphologically cryptic group. Molecular Phylogenetics and Evolution 51: 394–398.