Littledale’s Whistling Rat
Parotomys littledalei

2025 Red list status
Near threatened
Regional Population
Trend
Stable
Change compared
to 2016
No Change
Overview
Parotomys littledalei – Thomas, 1918
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Parotomys – littledalei
Common Names: Littledale’s Whistling Rat (English)
Synonyms: molopensis, namibensis (Jackson 2013)
Taxonomic Note: Molecular data suggest that Parotomys should be synonymised with Otomys (Taylor et al. 2011, 2014), but the genera are recognised as distinct pending a final phylogenetic analysis of species relationships within the Otomyinae (Monadjem et al. 2015). This species is similar to P. brantsii but distinguishable, both through dental morphology (un-grooved upper incisors) and whistle frequency and length (Le Roux et al. 2002; Monadjem et al. 2015).
Red List Status: NT – Near Threatened
Assessment Information
Assessor: Schradin, C.1,2 & da Silva, J.M.3
Reviewer: Pillay, N.2
Institutions:1Institut pluridisciplinaire Hubert Curien,2University of the Witwatersrand,3South African National Biodiversity Institute
Previous Assessors & Reviewers: Kerley, G. & Child, M.
Previous Contributors: Roxburgh, L., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Mondajem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed, under a precautionary risk tolerance, as Near Threatened B2b(iii,iv)+c(iii). It became locally extinct in Goegap Nature Reserve after a severe drought in 2003, retuning in low numbers in 2014 with the population having recovered only in 2022. Thus, this species might be especially vulnerable to an increase in intensity and duration of droughts because of climate change. Additionally, habitat degradation from overgrazing of rangelands may threaten this species because it is more reliant on plant cover than P. brantsii. However, it also has a wide distribution within the assessments region and occurs in several protected areas (including Goegap, Richtersveld and Augrabies National Parks). Ongoing protected area expansion in the area should sustain resilient subpopulations across the range. We recommend more research and long-term monitoring of subpopulation trends, geographic distribution, effects of climate change and threat level. Vulnerability to increased drought periods are of special research emphasis. This species probably qualifies for Least Concern, but further subpopulation trends and area of occupancy estimates are needed to demonstrate this. It should be reassessed following new data.
Regional population effects: Possible through dispersal from Namibia across contiguous and largely unfragmented habitat, although the Orange River represents a barrier to dispersal from Namibia. Dispersal ability might be low, as indicated by the long absence from Goegap Nature Reserve after the 2003 drought. Dispersal might depend on connected areas of shrub patches.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Schradin C & da Silva JM. 2025. A conservation assessment of Parotomys littledalei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
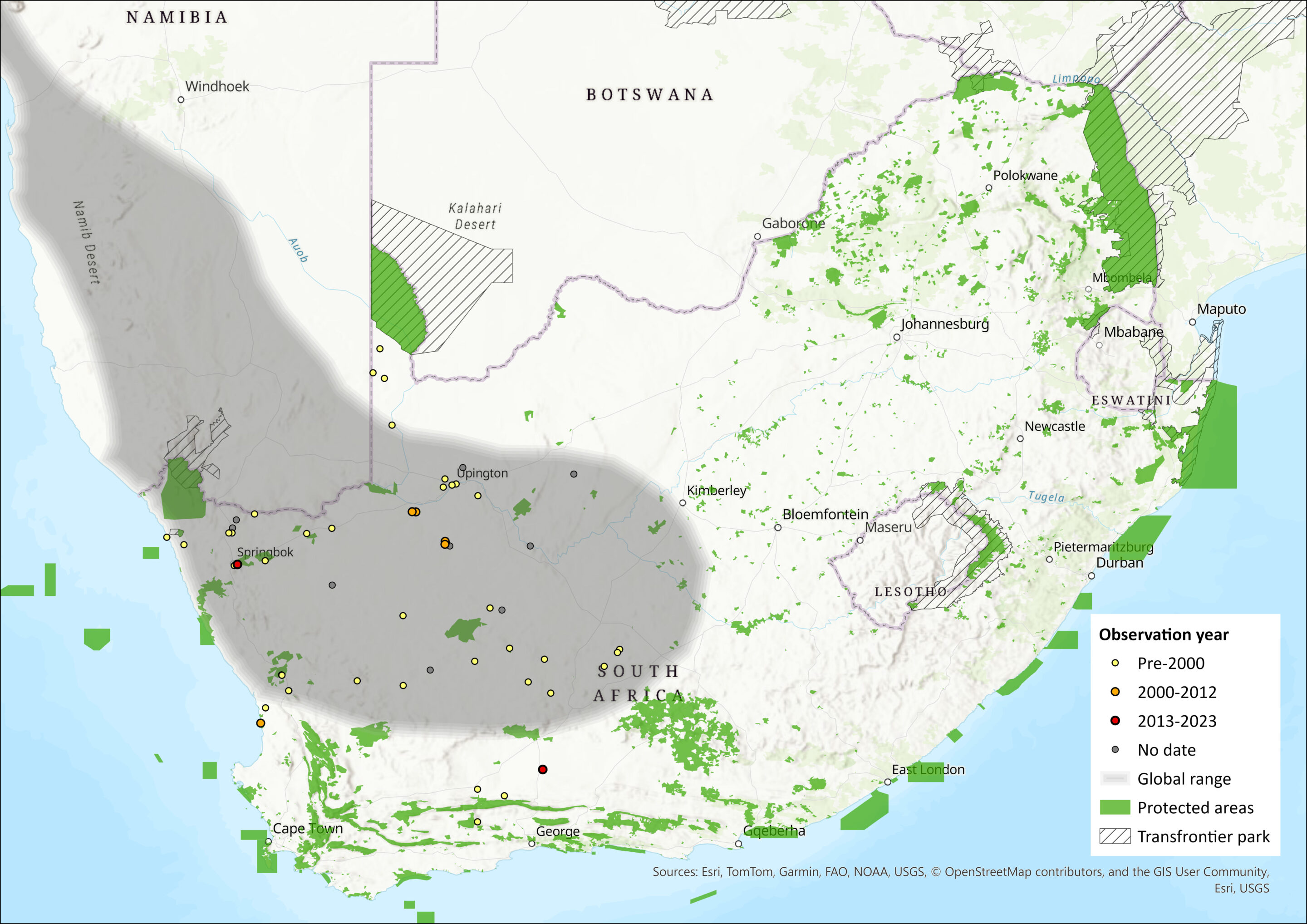
This species has a narrow distribution in the driest parts of southern Africa, from the western regions of South Africa north into Namibia and mostly along a narrow strip of desert (Monadjem et al. 2015). It occurs in the South-West Africa Biotic Zone (Namib Desert and Karoo regions) (Jackson 2013) and is absent from the central Namib Desert. It has not been recorded from Angola. Museum records need vetting to improve the accuracy of its distribution map. It is suspected to have an area of occupancy of < 2,000 km2 due to its patchy distribution and vulnerability to local extinctions. However, this should be more accurately quantified in future assessments.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 1500
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Littledale’s Whistling Rat (Parotomys littledalei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
An increase in the frequency and intensity of droughts due to climate change would increase extinction risk. Because the species is mainly folivorous, feeding largely on succulent shrubs, it might be especially vulnerable to month long droughts without green plant material, because it cannot alter its diet. It is not known to hoard food.
Population information
Relatively common in suitable habitats (although caught seldomly in small mammal traps) and is rarely seen but frequently heard (Jackson 2013). It undergoes population irruptions in response to environmental conditions and has a patchy distribution, linked to the distribution of deep sandy soils. Thus, detecting a population trend is difficult and long-term, systematic monitoring is necessary. The presence of P. littledalei in an area is usually indicated by a complex warren system under bushes (Coetzee & Jackson 1999; Jackson 2000). Population data from Goegap Nature Reserve, Northern Cape Province, indicate that it is more prone to local extinction than P. brantsii, where a density of 15 individuals / ha was recorded in 2001 and subsequently was not recorded until 2014 at a density of 1 individual / ha, with the population recovering to 12 individuals /ha in 2022 (C. Schradin unpubl. data). At the same site, P. brantsii exhibited densities of 2–10 individuals / ha remaining stable for a longer period until 2016, but then becoming locally extinct in 2017, with indication of returning in 2024 with individual/ ha a (C. Schradin unpubl. data).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
No population genetic study has been undertaken on this species, which would be beneficial to understand whether the small, fragmented colonies are actually part of one or more metapopulations or whether substantial genetic structure exists within the species.
The combination of small patchy colonies and and bottleneck events could suggest limited genetic diversity which could worsen from increases in the frequency and intensity of droughts due to climate change. This may be further exacerbated by the species propensity to inbreed; however, this may be regulated cyclical inbreeding and outbreeding, depending on population size and dispersal opportunities. Whether such a breeding pattern is adaptive for living in small, isolated populations needs to be verified.
Due to the limited information on the potential population genetic structure and diversity within the species, neither of the Convention on Biological Diversity’s genetic diversity indicators can be quantified at this time.
Habitats and ecology
This diurnal species occurs in shrubland and is more dependent on ground cover than P. brantsii (Jackson 2000). They avoid open habitats. It is not known if the species can persist in disturbed or modified habitats, but it does occur in rangelands. It has a patchy habitat distribution, reflecting forage availability and the need for deep soils. It occurs specifically in coastal hummocks, sand dunes and gravel plains of the Namib Desert, as well as dry riverine systems, extending inland from the coastal plains of Namibia (Coetzee & Jackson 1999; Jackson 2013).
Littledale’s Whistling Rat is herbivorous only, feeding on fresh plant material, including annuals, succulent perennials, non-succulent perennials, and grasses (Jackson 2013). It depends on plant leaves and succulents as food and cannot switch to seeds or other resources. Thus, it depends on the presence of green foliage and became locally extinct in Goegap Nature Reserve in 2003 due to a prolonged drought that removed all green plant material (C. Schradin unpubl. data).
Burrows are constructed below bushes and linked together through surface pathways that also link to foraging areas and contain several nest chambers filled with shredded vegetation (Jackson 2013). Its large kidneys (1.4% of body mass compared to 0.6% of P. brantsii) enable the most concentrated production of urine of any southern African otomyine species (Jackson et al. 2004). This helps to avoid water loss. It emits a high-pitched alarm whistle when disturbed by predators (Le Roux et al. 2002; Jackson 2013). Under artificial conditions, P. littledalei females prefer to inbreed, leading to a male bias in litter composition (Pillay 2002). Whether such inbreeding is adaptive for living in small, isolated populations must be tested.
Ecosystem and cultural services: Desmet and Cowling (1999) suggested that the nutrient-rich soil patches around P. brantsii burrows may facilitate the re-vegetation of mine dumps, which might similarly apply to P. littledalei. This should be interpreted with some caution as the rats require forage and would only recolonise areas where there is at least some plant cover.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
3.4. Shrubland -> Shrubland – Temperate |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
8.2. Desert -> Desert – Temperate |
– |
Suitable |
– |
Life History
Generation Length: minimum 90 days
Age at maturity: female or unspecified: 36 – 49 days
Age at Maturity: Male: 42 – 56 days
Size at Maturity (in cms): Female: About 140 cm
Size at Maturity (in cms): Male: About 153 cm
Longevity: (Not specified)
Average Reproductive Age: 45 days
Maximum Size (in cms): About 165 cm
Size at Birth (in cms): head body length = 63 mm
Gestation Time: 41-49 days
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? From the plants on which they feed
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
This is a species that should be flagged as being potentially threatened due to projected aridification because of climate change (Boko et al. 2007). Increased frequency and duration of drought conditions may affect the forage resources on which it depends. This response to global change may be exacerbated by the sensitivity of this rodent to high ambient temperatures (du Plessis et al. 1989), which may limit foraging behaviour under hotter conditions. For example, it became locally extinct in Goegap Nature Reserve after a severe drought in 2003 and returned in low numbers in 2014 (C. Schradin unpubl. data).
Similarly, overgrazing in some areas reduces habitat quality. This species may be more threatened by overgrazing than P. brantsii because it is more restricted to areas of adequate cover (Jackson 2000). Whereas grazing should be encouraged to decrease bush encroachment (see Habitats and Ecology), overgrazing should be avoided, especially in the more open vegetation types. As such, the proliferation of wildlife ranching should be monitored for its potential negative impacts as overgrazing may impact key vegetation types that this species requires. However, more research is needed to understand the net effects of local overgrazing on this species. For example, a recent study found that, contrary to prediction, grass cover has increased, and dwarf shrub cover has decreased in the Nama and Succulent Karoo, which is attributed to a general decrease in stocking rate in the area (Masubelele et al. 2014).
Conservation
It occurs commonly but patchily in protected areas across the range; for example, Richtersveld and Augrabies National Parks and Goegap Nature Reserve. Systematic monitoring is required to assess population trends and dynamics. The species would benefit from continued protected area expansion to enable it to track shifting habitats caused by climate change. For example, WWF South Africa is purchasing Knersvlakte to increase the size of a reserve, and, in 2014, the Goegap Nature Reserve was increased in size, from 150 km2 to around 250 km2, by including several neighbouring farms. Removing ground cover and green plant material through overgrazing will negatively affect this species and thus rangelands should be managed holistically by lowering stocking rates and conserving buffer zones.
Recommendations for land managers and practitioners:
- Land managers should decrease stocking rates to conserve key resource areas.
- Long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
Research priorities:
- Effects of overgrazing and climate change on key resource area quality and distribution and its impact on subpopulation trends.
- Effect of extended drought periods of population extinction over a larger geographic scale.
- Long distance dispersal ability (to re-colonise habitat where the species became locally extinct), which includes assessing the extent of fragmentation of subpopulations.
Encouraged citizen actions:
- Inform local farmers and guest farms about the concerns regarding the vulnerability of this species to droughts to get information on subpopulation trends.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
Boko M, Niang I, Nyong A, Vogel C, Githeko A, Medany M, Osman-Elasha B, Tabo R, Yanda P. 2007. Africa. Climate Change 2007: Impacts, Adaptation and Vulnerability. In: M. L. Parry, O. F. Canziani, J. P. Palutikof, P. J. van der Linden, and C. E. Hanson (eds), Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, pp. 433–467. Cambridge University Press, Cambridge, UK.
Coetzee CG, Jackson TP. 1999. The comparative behaviour and ecology of the two species of Parotomys (Mammalia, Rodentia, Otomyinae) found in the arid areas of southern Africa. Journal of the Namibian Science Society 47: 87–75.
Desmet PG, Cowling RM. 1999. Patch creation by fossorial rodents: a key process in the revegetation of phytotoxic arid soils. Journal of Arid Environments 43: 35–45.
Jackson TP, Bennett NC, Spinks AC. 2004. Is the distribution of the arid-occurring otomyine rodents of southern Africa related to physiological adaptation or refuge type? Journal of Zoology 264: 1–10.
Jackson TP. 2000. Adaptation to living in an open arid environment: lessons from the burrow structure of the two southern African whistling rats, Parotomys brantsii and Parotomys littledalei. Journal of Arid Environments 46: 345–355.
Jackson TP. 2013. Parotomys littledaleiLittledale’s Whistling Rat. In: Happold DCD (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, Bloomsbury Publishing, London, UK.
Le Roux A, Jackson TP, Cherry MI. 2002. Differences in alarm vocalizations of sympatric populations of the whistling rats, Parotomys brantsiiand P. littledalei. Journal of Zoology 257: 189–194.
Masubelele ML, Hoffman MT, Bond WJ, Gambiza J. 2014. A 50 year study shows grass cover has increased in shrublands of semi-arid South Africa. Journal of Arid Environments 104: 43-51.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Pillay N. 2002. Inbreeding in Littledale’s whistling rat Parotomys littledalei. Journal of Experimental Zoology 293: 171–178.Pillay N. 2002. Reproduction and postnatal development in Littledale’s whistling rat Parotomys littledalei (Muridae, Otomyinae). Mammalia 66: 71-82.
Pillay N. and Rymer T. 2017. Preference for outbreeding in inbred Littledale’s whistling rats Parotomys littledalei. Evolutionary Biology 44: 21-30.
Taylor PJ, Maree S, Cotterill FPD, Missoup AD, Nicolas V, Denys C. 2014. Peripatric speciation across a Neogene volcanic archipelago: molecular and morphological evidence for a Pleistocene radiation of laminate-toothed rats (Otomys: Rodentia) across equatorial Africa. Biological Journal of the Linnean Society 113: 320–344.
Taylor, P.J., Lavrenchenko, L.A., Carleton, M.D., Verheyen, E., Bennett, N.C., Oosthuizen, C.J. and Maree, S. 2011. Specific limits and emerging diversity patterns in East African populations of laminate-toothed rates, genus Otomys (Muridae: Murinae: otomyini): revision of the Otomys typus complex. Zootaxa 3024: 1-66.
du Plessis A, Erasmus T, Kerley GIH. 1989. Thermoregulatory patterns of two sympatric rodents: Otomys unisulcatus and Parotomys brantsii. Comparative Biochemistry and Physiology, Series A.