
Little Free-tailed Bat
Mops pumilus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mops pumilus – (Cretzschmar, 1826)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MOLOSSIDAE – Mops – pumilus
Common Names: Little Free-tailed Bat, Lesser Free-tailed Bat, Little Wrinkle-lipped Bat, White-bellied Free-tailed Bat (English), Klein Losstertvlermuis (Afrikaans), Kleine Bulldoggfledermaus (German)
Synonyms: Chaerephon pumila (Cretzschmar, 1826) [orth.error]; Tadarida pumila (Cretzschmar, 1826);
Taxonomic Note:
Although previously included under the genus Chaerephon (ACR 2024), this species has been reclassified under the genus Mops (Simmons and Cirranello 2020). Lamb et al. (2011) found strong support for the monophyly of the Chaerephon and Mops taxa (about 17.2 Mya), but not for either of the genera separated. Ammerman et al. (2012) also found support for a Mops – Chaerephon clade, which resulted in paraphyly of Chaerephon, and confirm again that Chaerephon is distinct from Tadarida. A further morphological study by Gregorin and Cirranello (2015) confirmed the paraphyly of Chaerephon but was unable to resolve the relationships with the genus. Simmons and Cirranello (2020) include all the Chaerephon species in the genus Mops. Taxonomic revision of this genus is required. Several species have been separated from this complex in recent years, including M. leucogaster, M. atsinanana and M. pusillus (formerly referred to as Chaerephon). Discordance between different molecular markers and genetic introgression have made it difficult to resolve the species within the mainland African complex (Taylor 2009; Naidoo et al. 2013, 2016).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Naidoo, T.1, Richards, L.R.1, Balona, J.2, van der Meulen, K.3 & da Silva, J.M.4
Reviewer: Richardson, E.J.5
Institutions: 1Durban Natural Science Museum, 2Gauteng and Northern Regions Bat Interest Group, 3University of the Free State, 4South African National Biodiversity Institute, 5Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Monadjem, A., Cohen, L., Jacobs, D., MacEwan, K., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Relton, C. & Raimondo, D.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (estimated extent of occurrence in the assessment region alone is 292,796 km2), its tolerance of a broad range of habitats including urban areas, presumed large population, large-sized colonies in parts of its range, its occurrence in several protected areas and because it is unlikely to be declining fast enough to qualify for listing in a threatened category. Savannah habitats are well protected within the assessment region, and no direct interventions are necessary.
Regional population effects: This species has a continuous distribution through the northeastern parts of South Africa into Mozambique and Zimbabwe and has a high wing-loading (Norberg & Rayner 1987; Schoeman & Jacobs 2008); thus, dispersal is likely and rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Naidoo T, Richards LR, Balona J, van der Meulen K & da Silva JM. 2025. A conservation assessment of Mops pumilus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species of Free-tailed Bat is one of the most common and widespread species in the eastern parts of the assessment region (Monadjem et al. 2020). In the rest of its range it is found in Senegal, eastwards to Yemen and southwestern Saudi Arabia, and as far south as South Africa. It has been recorded from the island of Bioko and the Annobon Islands (Equatorial Guinea), Pemba and Zanzibar (Tanzania), the Comoros Islands, Mayotte (Goodman 2007), and Madagascar (Bouchard 1998) and on the Aldabra Atoll of the Seychelles (Hutson 2004). Habitat models suggest that suitable conditions occur over much of southern Mozambique (Monadjem et al. 2020). Within the assessment region, it occurs widely in the northeastern areas, from the far-eastern Eastern Cape along the coast in KwaZulu-Natal and through Eswatini, to the Mpumalanga and Limpopo provinces.
However, there is evidence (Bats KZN data) that this species moves with the ENSO fluctuations and may not be present in all parts of this range in all years.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m
Elevation Upper Limit (in metres above sea level): Appears absent from regions greater than 1000-1500 m asl range (Monadjem et al. 2020)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
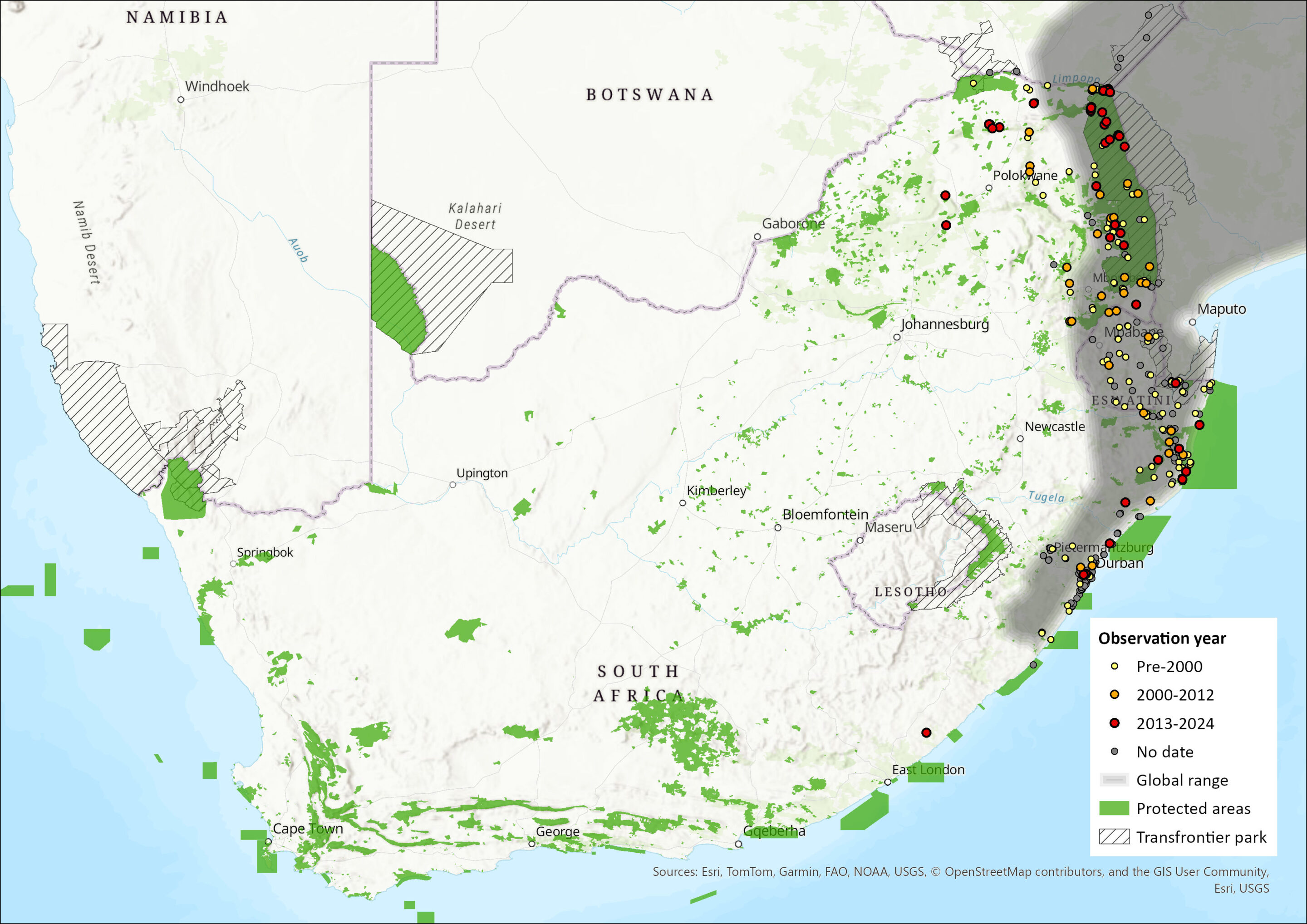 Figure 1. Distribution records for Little Free-tailed Bat (Mops pumilus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Little Free-tailed Bat (Mops pumilus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Equatorial Guinea -> Annobón | Extant | Native | – | – |
| Equatorial Guinea -> Bioko | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mayotte | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Sao Tome and Principe | Extant | Native | – | – |
| Sao Tome and Principe -> Sâo Tomé | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Seychelles | Extant | Native | – | – |
| Seychelles -> Aldabra | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
No formal study on the direct effects of climate change has taken place, however, due to the increasing temperatures predicted for the eastern region (Mbokodo et al. 2020) the energetic costs of thermoregulation is likely to increase, with dehydration becoming more common (Buffenstein et al. 1999; Vivier and van der Merwe, 2007). Roost preferences will likely shift to select for more suitable microclimates to maintain basal metabolism. Bats may be more susceptible to extreme weather events than to climate shift (see, for example, Matthew et al. 2020). Bats of this species are known to shift their home range in response to the ENSOs (Bats KZN data), suggesting that their current range might shift in the future.
Population Information
This is a widespread and abundant species (Monadjem et al. 2020), occurring in colonies ranging from about 5–20 to thousands of individuals (Taylor 1998, Mickleburgh et al. 2008). For example, colonies of up to 2,000 have been estimated to occur in the roof of a sugar mill in KwaZulu-Natal (Monadjem et al. 2020). Rautenbach (1997) suggests that this species may be one of the most common within its range in southern Africa, as it has benefited substantially from urban expansion. It is very well represented in museums, with over 1,000 specimens examined in Monadjem et al. (2020).
Population Information
Current population trend: Stable
Continuing decline in mature individuals? No suspected declines
Extreme fluctuations in the number of subpopulations: Not known
Continuing decline in number of subpopulations: None observed
All individuals in one subpopulation: See below
Number of mature individuals in largest subpopulation: Not determined
Number of Subpopulations: See below
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 100 years: Cannot be determined yet
Population Genetics
Phylogenetic studies on this species have sought to resolve the genetic relationships among M. pumilus (then Chaerephon pumilus) (Reddy 2008; Jacobs et al. 2004). While some taxonomic clarities were provided with respect to the placement of C. pumilus from Madagascar, and two major clades were identified for C. pumilus in southern Africa, more comprehensive detailed investigations are needed to uncover the contemporary population genetic structure within M. pumilus.
Interestingly, investigations into the genetic distinctiveness of the light and dark forms of the species were not able to find sufficient differences to classify each as separate species (Aspertsberger et al. 2003; Jacobs et al. 2004) however some genetic differentiation was identified and the two forms were found to have differences in echolocation frequency. This could suggest within-species differentiation (e.g., separate subpopulations). Reddy (2008) attempted to investigate this further in the two identified clades for southern Africa, which appeared to correspond to areas of differing rainfall; however, he found no association with the different colour forms and the two clades.
Discordance was found between different molecular markers and genetic introgression have made it difficult to resolve the species within the mainland African complex (Taylor 2009, Naidoo et al. 2013, 2016)
It is therefore highly recommended that a more widespread, integrative study incorporating several ecological and behavioural characteristics, be conducted to validate the two clades, as well as investigate any potential adaptive associations within the species and its potential populations.
Given that the species is considered widespread and abundant, it is expected that the effective populations sizes exceed the Ne 500 threshold; however, this requires confirmation through genetic analyses.
Habitats and ecology
The Little Free-tailed Bat occurs across a variety of habitats from semi-arid savannah in the north of its range to forested regions further south (Happold 1987). In Zimbabwe it occurs within dry mopane woodland habitats below 1,000 m asl (Skinner and Chimimba 2005). Brickwork under roofs, corrugated roofs or other crevices in buildings provide suitable roosting sites for Little Free-tailed Bats within built-up areas, but natural roost sites include cracks and crevices in rocks and trees (Skinner and Chimimba 2005, Mickleburgh et al. 2008, Monadjem et al. 2020).
The species is considered an open-air forager, feeding on Lepidoptera, Diptera, Hemiptera, Isoptera, Tricoptera, and Hymenoptera (Aldridge and Rautenbach 1987; Bohmann et al. 2011). The Little Free-tailed Bat typically exhibits summer seasonal breeding. For example, females in the Kruger National Park experienced synchronised breeding with three peaks in parturition: early November, late January and early April (van der Merwe et al. 1986). This was similar in Eswatini, with peaks in parturition estimated to occur in November, January and March (Monadjem 1998). Individuals in Malawi experienced a fourth peak in parturition in about May, but Monadjem (1998) suggests that this is likely to be limited to low latitude regions and more tropical climates. The Little Free-tailed Bat has a gestation period of approximately 60 days, and a single young of about 3.2 g is produced (Skinner and Chimimba 2005). Sexual maturity in females is reached at an age of between 5 and 12 months (Skinner and Chimimba 2005).
Ecosystem and cultural services: As this species is insectivorous and often found in large numbers, it may contribute to a large extent in controlling insect populations (Boyles et al. 2011, Kunz et al. 2011). Noer et al. (2012) found that Little Free-tailed bats in Eswatini selectively foraged over sugar cane fields instead of other habitats in the area. Bats often prey on the insect species that destroy crops (Boyles et al. 2011, Kunz et al. 2011) and the ecosystem services provided by the more common bat species need to be better understood and protected by authorities (Russo et al. 2018) Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
| 17. Other | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: Between 5 and 12 months depending on area (Skinner & Chimimba 2005)
Age at Maturity: Male: Likely 1 year
Size at Maturity (in cms): Female: Total length 8.96 cm, Forearm 3.81 cm
Size at Maturity (in cms): Male: Total length 8.85 cm, Forearm 3.76 cm
Longevity: Not known
Average Reproductive Age: One year
Maximum Size (in cms): Not known
Size at Birth (in cms): Approximately 56% (~4.90 cm) of the adult total length (Happold & Happold 1989)
Gestation Time: ~60 days (Happold 2013)
Reproductive Periodicity: Seasonal polyoestry coinciding with the austral summer and early autumn
Average Annual Fecundity or Litter Size: Typically 1 pup
Natural Mortality: Various nocturnal predators
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: May possibly shift distribution / roosting pattens during ENSOs (BatsKZN data)
Congregatory: Yes, roost can number several individuals to a few thousand individuals
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Currently, the major threat to this species is human intolerance of bats in building roofs (Bats KZN info) but with wind farms moving into the high-bat areas in the eastern and north-eastern parts of the country all free-tailed bats are at risk due to their open-air foraging habits (Baerwald et al. 2008, Cryan and Barclay 2009, South African Bat Assessment Association Pre-Construction Guidelines www.sabaa.org.za). Their range, these bats are threatened from persecution as a pest, especially when they roost in buildings (ACR 2024).
Conservation
In the assessment region, the species is recorded from the protected areas of Kruger National Park, Baobab Tree Reserve, Hans Meresky Nature Reserve, Ndumo Game Reserve, Tembe Elephant Park, iSimangaliso Wetland Park, Pongolapoort Nature Reserve, Pongola Nature Reserve, Hluhluwe-iMfolozi Game Reserve, Dlinza Forest Nature Park, Harold Johnson Nature Reserve and Oribi Gorge Nature Reserve. Although no specific conservation efforts are necessary, this species would benefit from the protection of key roost sites.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
Research priorities:
- Studies into the impact of wind farms on this species.
- Long-term monitoring studies of colonies to determine movement patterns.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2024. African Chiroptera Report 2023. Van Cakenberghe, V. and Seamark, E.C.J. (Eds.) African Chiroptera Project, Pretoria. i – xviii; 1 – 1210 pp.
Aldridge, H.D.J.N., Rautenbach, I.L. 1987 Morphology, echolocation and resource partitioning in insectivorous bats. The Journal of Animal Ecology 56: 763–778.
Ammerman, L. K., Lee, D.N., and Tipps, T.M. 2012. First molecular phylogenetic insights into the evolution of free-tailed bats in the subfamily Molossinae (Molossidae, Chiroptera). Journal of Mammalogy, 93 (1): 12 – 28. doi: http://dx.doi.org/10.1644/11-MAMM-A-103.1
Baerwald, E.F., D’Amours, G.H., Klug, B.J., and Barclay, R.M. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Bohmann, K., Monadjem, A., Lehmkuhl Noer, C., Rasmussen, M., Zeale, M.R., Clare, E., Jones, G., Willerslev, E., Gilbert, M.T. 2011. Molecular diet analysis of two African free-tailed bats (Molossidae) using high throughput sequencing. PloS one 6(6):e21441.
Bouchard, S. 1998. Chaerephon pumilus. Mammalian Species 574: 1-6.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cryan, P.M. and Barclay, R.M. 2009. Causes of bat fatalities at wind turbines: hypotheses and predictions. Journal of Mammalogy 90: 1330–1340.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria.
Goodman, S.M. 2007. Final report for the project entitled, “Survey of microchiropteran bats of islands in the western Indian Ocean”. CABS at Conservation International on 28 August 2007.
Gregorin, R. and A. Cirranello 2015 Phylogeny of Molossidae Gervais (Mammalia: Chiroptera) inferred by morphological data. Cladistics: The International Journal of the Willi Henning Society, 32 (1): 2 – 35 (for 2016). doi: 10.1111/cla.12117.
Happold, D.C.D. 1987. The Mammals of Nigeria. Oxford University Press, London, UK.’
Happold, M. 2013. Tadarida pumila Little Free-tailed Bat; pp 528-530 in Happold, M. & Happold, D.C.D. (eds) Mammals of Africa: Volume IV. Bloomsbury Publishing, London.
Hutson, A. M. 2004. The bats of Aldabra atoll, Seychelles. Phelsuma 12: 126-132.
Jacobs, D.S., Eick, G.N., Richardson, E.J. and Taylor, P.J. 2004. Genetic similarity amongst phenotypically diverse little free-tailed bats, Chaerephon pumilus. Acta Chiropterologica, 6(1), pp.13-21.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Mickleburgh, S., Hutson, A.M., Racey, P.A., Bergmans, W. and Cotterill, F.P.D. 2008. Chaerephon pumilus. Page e.T4317A67362329.
Lamb, J. M., Ralph, T.M.C., Naidoo, T., Taylor, P.J., Ratrimomanarivo,F., Stanley, W.T. and Goodman, S.M. 2011 Toward a molecular phylogeny for the Molossidae (Chiroptera) of the Afro-Malagasy region. Acta Chiropterologica, 13 (1): 1 – 16. doi: 10.3161/150811011X578589.
Mo, M., Roache, M., Davies, J., Hopper, J., Pitty, H., Foster, N., Guy, S., Parry-Jones, K., Francis, G., Koosmen, A., Colefax, L., Costello, C., Stokes, J., Curran, S., Smith, M., Daly, G., Simmons, C., Hansen, R., Prophet, D., Judge, S., Major, F., Hogarth, T., McGarry, C., Pope, L., Brend, S., Coxon, D., Baker, K., Kaye, K., Collins, L., Wallis, M., Brown, R., Roberts, L., Taylor, S., Pearson, T., Bishop, T., Dunne, P., Coutts-McClelland, K., Oliver, L., Dawe, C., Welbergen, J.A. 2022 Estimating flying-fox mortality associated with abandonments of pups and extreme heat events during the austral summer of 2019–20. Pacific Conservation Biology 28, 124-139. https://doi.org/10.1071/PC21003
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N., 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem A. 1998. Reproduction in the little free-tailed bat Chaerephon pumila (Microchiroptera: Molossidae) in Swaziland. Durban Museum Novitates 23: 39–41.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Naidoo, T., Schoeman, M.C., Goodman, S.M. et al. 2016. Discordance between mitochondrial and nuclear genetic structure in the bat Chaerephon pumilus (Chiroptera: Molossidae) from southern Africa. Mamm Biol 81, 115–122 (2016). https://doi.org/10.1016/j.mambio.2015.11.002
Naidoo, T., Schoeman, M.C., Taylor, P.J., Goodman, S.M. and Lamb, J.M. 2013. Stable Pleistocene-era populations of Chaerephon pumilus (Chiroptera: Molossidae) in southeastern Africa do not use different echolocation calls. African Zoology, 48 (1): 125 – 142. doi:10.3377/004.048.0112
Noer, C.L., Dabelsteen, T. Bohmann, K, and Monadjem, A. 2012. Molossid bats in an African agro-ecosystem select sugarcane fields as foraging habitat. African Zoology 47(1): 1-11.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Rautenbach N. 1997. Little free-tailed bat Tadarida pumila. Page 78 in Mills G, Hes L, editors. Struik Publishers, Cape Town, South Africa.
Reddy, D. 2008. Genetic analysis of Chaerephon pumilus (Chiroptera: Molossidae) from southern Africa.
Russo, D., Bosso, L., and Ancillotto, L. 2018. Novel perspectives on bat insectivory highlight the value of this ecosystem service in farmland: Research frontiers and management implications. Agriculture, Ecosystems & Environment 266: 31-38.
Rydell, J., Bach, L., Dubourg-Savage, M-J., Green, M., Rodrigues, L., and Hedenström, A. 2010. Mortality of bats at wind turbines links to nocturnal insect migration? European Journal of Wildlife Research 56: 823–827.
Schoeman, M.C., and Jacobs, D.S. 2008. The relative influence of competition and prey defenses on the phenotypic structure of insectivorous bat ensembles in southern Africa. PLoS One 3: e3715.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Simmons, N. B. and Cirranello, A.L. 2020. https://batnames.org/explore.html. Accessed: 10 May 2020. URL: https://batnames.org/explore.html
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Taylor, P.J. 1999. Problems with the identification of southern African Chaerephon (Molossidae) and the possibility of a cryptic species from South Africa and Swaziland. Acta Chiropterologica 1: 191–200.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J, Lamb, J., Reddy, D., Naidoo, T., Ratrimomanarivo, F., and Goodman, S. M. 2009. Cryptic lineages of little free-tailed bats, Chaerephon pumilus (Chiroptera: Molossidae) from southern Africa and the western Indian Ocean islands. African Zoology, 44(1), 55–70. https://doi.org/10.1080/15627020.2009.11407439
van der Merwe, M., Rautenbach, I.L. and van der Colf, W.J. 1986. Reproduction in females of the little free-tailed bat, Tadarida (Chaerephon) pumila, in the eastern Transvaal, South Africa. Journal of Reproduction and Fertility 77: 355–364.
Vivier, L, and van Der Merwe, M. 1997. Reproduction in the female Angolan free-tailed bat, Tadarida (Mops) candylura (Microchiroptera: Molossidae), in the eastern Transvaal, South Africa. Journal of Zoology (London) 243: 507–521.
