
Lesser Yellow House Bat
Scotophilus viridis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Scotophilus viridis – (Peters, 1852)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – VESPERTILIONIDAE – Scotophilus – viridis
Common Names: Lesser Yellow House Bat, Green House Bat, Greenish Yellow Bat (English), Klein Geel Dakvlermuis (Afrikaans)
Synonyms: Nycticejus viridis (W. C. H. Peters, 1851); S. borbonicus viridis Koopman 1994
Taxonomic Note:
This genus is in urgent need of taxonomic revision (Monadjem et al. 2020), particularly the relationship between Scotophilus leucogaster and S. viridis. There is evidence that S. viridis is a species complex, at least in East and West Africa (Trujillo et al. 2009; Vallo et al. 2013). However, the situation in southern Africa is unclear. Currently, we recognise S. viridis as a valid species following Robbins et al. (1985), Simmons (2005) and Jacobs et al. (2006). Meester et al. (1986) recognised two subspecies in the subregion: S. b. damarensis (Thomas 1906), which was found from northeastern Namibia and S. b. viridis (Peters 1852) from the remainder of the species’ range.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Richardson, E.1 & da Silva, J.M.2
Reviewers: Smith, C.3 & Richards, L.R.4
Institutions: 1Independent Consultant at Richardson & Peplow Environmental, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust, 4Durban Natural Science Museum
Previous Assessors and Reviewers: Richards, L.R., Cohen, L., Jacobs, D., MacEwan, K., Monadjem, A., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Child, M.F. & Raimondo, D.
Assessment Rationale
Listed as Least Concern in view of its wide distribution (estimated extent of occurrence is 201,346 km2) within the northeastern part of the assessment region, its occurrence in several protected areas (including Kruger National Park and iSimangaliso Wetland Park), its ability to utilise artificial habitats, and because there are no major identified threats that could cause widespread population decline. The conservation and protection of large trees used for roosting sites, such as Leadwood (Combretum imberbe) and Mopane (Colophospermum mopane) trees, is a recommended management practice for sustaining this species.
Regional population effects: Habitat between regions is contiguous and its range is likely more extensive in neighbouring countries that currently documented. Thus, we infer rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No Change
Red List Index
Red List Index: No change
Recommended citation: Richardson E & da Silva JM. 2025. A conservation assessment of Scotophilus viridis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is widespread in sub-Saharan Africa, ranging from Senegal and The Gambia in West Africa, to northern Cameroon and Central African Republic, from there into Sudan and Ethiopia, and then south through much of East Africa and southeastern Africa, occurring as far south as eastern South Africa and Eswatini (Skinner & Chimimba 2005). Within the assessment region, they are restricted to eastern South Africa and Eswatini, extending from northern KwaZulu-Natal to Venetia Limpopo Nature Reserve and the Kruger National Park, and into Mozambique. It is under-sampled and may be shown to be more widespread in the subtropical eastern and northern parts of the region. (Monadjem et al. 2010b, 2020). Ecological niche models predict a wider distribution in the Eastern Cape province of South Africa, the northeastern parts of the country and the southern reaches of Mozambique (Monadjem et al. 2010b). The estimated extent of occurrence within the assessment region is 201,346 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 3 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): 1,508 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A)
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
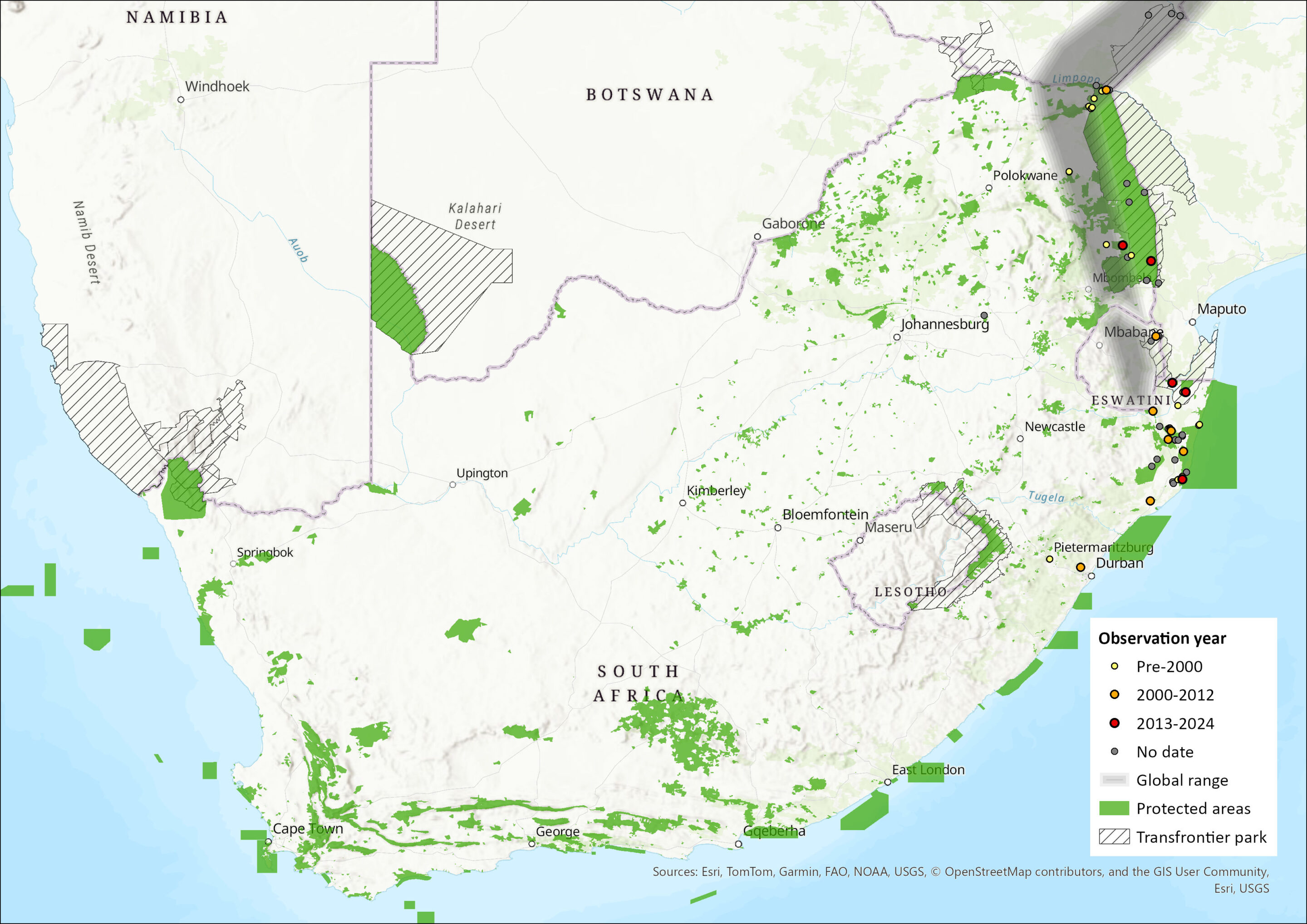 Figure 1. Distribution records for Lesser Yellow House Bat (Scotophilus viridis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Lesser Yellow House Bat (Scotophilus viridis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
Nothing is known about the potential impact of climate change on this species, but globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020). Bats may also be more susceptible to extreme weather events than to climate shift (see, for example, Matthew et al. 2020).
Bats of the closely related S. dinganii have been recorded leaving their roof roosts and hanging exposed on the exterior walls of houses during extremely hot days (Bats KZN data), suggesting that this species also may be affected by heat waves and increasing maximum temperatures. Babiker Salata (2012) demonstrated that species distribution is strongly influenced by the mean temperature of the coldest quarter. These bats appear to require regions where this temperature remains above 19 °C. Considering their preference for habitats that are wetter than those occupied by S. leucogaster (Van Cakenberghe & Happold 2013), combined with their lower thermal tolerance and limited ability to concentrate urine (relative to similar species), it is likely that rising temperatures, coupled with increasingly unpredictable rainfall patterns and more frequent droughts, will pose a significant future threat to this species.
Population Information
This is a reasonably common species (ACR 2024), although its population size has not been studied in detail. It is not well represented in museums, with only 41 records examined in Monadjem et al. (2020). Where it does occur, it is often more abundant than S. dinganii; for example, in Eswatini and Mozambique (A. Monadjem unpubl. data).
Radio-tracked individuals from Eswatini were found to roost in loose groups of 4-5 individuals Combretum imberbe and Acacia nigrescens trees (Monadjem et al. 2010a). A colony of 6-7 individuals were found roosting in a bat box in Ndumo Game Reserve (Durban Natural Science Museum specimen records). Elsewhere in southern Africa, they have been reported roosting in low numbers in the hollow of trees (Skinner and Chimimba 2005).
Current population trend: Stable
Continuing decline in mature individuals? There is currently insufficient information on population size and trends to make an accurate determination.
Extreme fluctuations in the number of subpopulations: There is currently a lack of population-level genetic studies, making it difficult to accurately determine the number and structure of subpopulations.
Continuing decline in number of subpopulations: Uncertain
All individuals in one subpopulation: There is currently a lack of population-level genetic studies, making it difficult to accurately determine the number and structure of subpopulations.
Number of mature individuals in largest subpopulation: Uncertain
Number of Subpopulations: Uncertain
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: unlikely due to the species’ widespread distribution and presumed connectivity across its range.
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: p unlikely due to the species’ widespread distribution and presumed connectivity across its range.
Probability of extinction in the wild within 100 years: unlikely due to the species’ widespread distribution and presumed connectivity across its range.
Population Genetics
No population genetic study has been conducted on the species; however, given its morphology, it is expected to disperse within the assessment region. As such, the species is expected to exist as a single subpopulation. While the species is considered reasonably common, no estimates of mature individuals are available to suggest an effective population size.
Habitats and ecology
The Lesser Yellow House Bat is considered a savannah woodland species and has been recorded from both dry and moist wooded and bushveld habitats, typically in areas with a mean annual rainfall of > 500 mm (Skinner & Chimimba 2005). It is restricted to low-lying, hot savannahs and avoids open habitats such as grasslands (Monadjem et al. 2010b), possibly due to the absence of roosting sites. In the assessment region, the species is recorded from the Dry Highveld Grassland, Central Bushveld, Mopane Bioregion, Lowveld, Indian Ocean Coastal Belt, Drakensberg Grassland, Sub-Escarpment Savanna and Sub-Escarpment Grassland.
This is not a well-studied species, with very little known about their habits. It roosts in small colonies in a variety of shelters (Monadjem et al. 2010a, 2020), such as buildings and hollow trees (Cotterill & Giddings 1987). Roofs of houses are often selected as roosting sites (Jacobs et al. 2007; Jacobs & Barclay 2009). In Kruger National Park, it was found to favour hollow Mopane Trees found along the sides and tops of ridges (Fenton et al. 1985). In Eswatini, they were recorded roosting predominantly in Leadwood and Knob Thorn (Acacia nigrescens) trees (Monadjem et al. 2010a). Roost switching appears to be common in the species, with individuals sometime changing tree hole roosts daily; they also move between day and night roost within their preferred home range (Fenton et al. 1985; Fenton & Rautenbach 1986).
Similar to most other southern African insectivorous species, they are primarily active between 19:00 and 22:00 (Cotterill & Giddings, 1987), with little to no activity occurring outside this period (Skinner and Chimimba 2005).
Like other house bats, the Lesser Yellow House Bat is insectivorous (Skinner & Chimimba 2005), existing on a diet of mainly Coleoptera, Lepidoptera and Hemiptera (Monadjem et al. 2020). They forage by slow-hawking and are characterised by fast and agile flight that is marked by diving, swooping and swerving (Van Cakenberghe and Happold 2013).
They do not become torpid at temperatures ≥21 °C (Van Cakenberghe and Happold 2013) and have a fairly low mean maximum urine concentration ability (3233 mOsmol/kg; Happold & Happold 1988).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species which destroy crops (Boyles et al. 2011; Kunz et al. 2011) and thus sustaining a healthy population of insectivorous bats can decrease the need for pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Unknown | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Unknown | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Sexes combined: mean forearm length = 4.72 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): maximum forearm length = 5.17 cm (Monadjem et al. 2020)
Size at Birth (in cms): Unknown
Gestation Time: Uncertain
Reproductive Periodicity: Exhibits seasonal monoestry, with females giving in the austral summer months (Van Cakenberghe and Happold 2013).
Average Annual Fecundity or Litter Size: Females usually give birth to twins (Van Cakenberghe and Happold 2013).
Natural Mortality: Unknown
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Reported to move 100 – 1000 m between roosts within their preferred home range (Fenton & Rautenbach 1986), while radio-tracked individuals travelled up to 4 km roosts (Monadjem et al. 2010a).
Congregatory: They are known to roost in small colonies and utilize a wide variety of roosting sites, although they exhibit a preference for tree hollows.
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that the species is traded or utilised in any form.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: The extent of their occurrence in agroecosystems and their potential role in providing insect pest suppression services remains uncertain due to the lack of available data.
International Commercial Value: Uncertain
End Use: None reported
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: N/A
Threats
There are no known major threats to this species. However, the loss of large trees used for roosting from fuelwood harvesting and agricultural expansion may result in local declines. Key roosting trees, such as C. imberbe, are slow-growing and vulnerable to be over-utilised for fuelwood (sensu Wessels et al. 2013).
Conservation
In the assessment region, the species is recorded from several protected areas, including Kruger National Park, Venetia Limpopo Nature Reserve, Hans Merensky Nature Reserve, Ndumo Game Reserve, Tembe Elephant Park, iSimangaliso Wetland Park, Pongolapoort Nature Reserve, Hluhluwe-iMfolozi Park and Mlawula Nature Reserve in Eswatini. No direct conservation interventions are necessary for this species, but it will benefit from protection of key roost sites, particularly large trees such as Combretum imberbe, Colophospermum mopane and Acacia nigrescens (Fenton et al. 1985; Monadjem et al. 20202). The conservation of such roosting trees may be critical to the continued persistence of cavity-nesting insectivorous bats in African savannahs (Monadjem et al. 2010a).
Recommendations for land managers and practitioners:
- The conservation and protection of large trees used for roosting sites is a recommended management practice for this species.
- Reduce pesticide use in agricultural landscapes.
- Retain ‘snag’ trees as these may serve as important roosting site for both bats and birds.
Research priorities:
- Enhanced sampling from a broader geographical area using both molecular and morphological datasets (Monadjem et al. 2020), are recommended to address taxonomic uncertainty in the genus Scotophilus.
- Population-level genetic studies are essential to estimate effective population size and to gain a deeper understanding of genetic relatedness among populations in southern Africa.
- Targeted surveys to determine population size, trend and distribution of additional colonies.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
- Deposit deceased specimen material with a recognised museum or biorepository.
Bibliography
ACR. 2024. African Chiroptera Report. 2024. AfricanBats NPC, Pretoria. i – xv; 1 – 1397.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Babiker Salata, H.A. 2012. Environmental factors influencing the distribution of bats (Chiroptera) in South Africa. Ph.D. Thesis. University of Cape Town, South Africa.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Cotterill, F. P. D. and Giddings, S. R. 1987. Observations on the activity patterns of the lesser house bat, Scotophilus viridis. South African Journal of Zoology 22(2): 175.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fenton, M.B., Brigham, R.M., Mills, A.M. and Rautenbach, I.L. 1985. The roosting and foraging areas of Epomophorus wahlbergi (Pteropodidae) and Scotophilus viridis (Vespertilionidae) in Kruger National Park, South Africa. Journal of Mammalogy 66: 461–468.
Fenton, M.B. and Rautenbach, I.L. 1986. A comparison of the roosting and foraging behaviour of three species of African insectivorous bats (Rhinolophidae, Vespertilionidae, and Molossidae). Canadian Journal of Zoology. 64(12):2860-7.
Happold, D.C. and Happold, M.J. 1988. Renal form and function in relation to the ecology of bats (Chiroptera) from Malawi, Central Africa. Journal of Zoology 215(4):629-55.
Jacobs, D.S. and Barclay, R.M. 2009. Niche differentiation in two sympatric sibling bat species, Scotophilus dinganii and Scotophilus mhlanganii. Journal of Mammalogy 90: 879–887.
Jacobs, D.S., Eick, G.N., Schoeman, M.C. and Matthee, C.A. 2006. Cryptic species in an insectivorous bat, Scotophilus dinganii. Journal of Mammalogy 87: 161–170.
Jacobs, D.S., Kelly, E.J., Mason, M. and Stoffberg, S. 2007. Thermoregulation in two free-ranging subtropical insectivorous bat species: Scotophilus species (Vespertilionidae). Canadian Journal of Zoology 85: 883–890.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N, O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Matthew, M. et al. 2022. Estimating flying-fox mortality associated with abandonments of pups and extreme heat events during the austral summer of 2019–20. Pacific Conservation Biology 28, 124-139
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Raabe, T., Dickerson, B., Silvy, N. and McCleery, R. 2010a. Roost use by two sympatric species of Scotophilus in a natural environment. South African Journal of Wildlife Research 40: 73– 76.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman, M.C. 2020. Bats of Southern and Central Africa: A Biogeographic and Taxonomic Synthesis. 2nd Edition. University of the Witwatersrand Press, Johannesburg, South Africa.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data 11(1):1309.
Munyati, C. and Kabanda, T.A. 2009. Using multitemporal Landsat TM imagery to establish land use pressure induced trends in forest and woodland cover in sections of the Soutpansberg Mountains of Venda region, Limpopo Province, South Africa. Regional Environmental Change 9: 41–56.
Robbins, C.B., Vree F, de. and Cakenberghe, V van. 1985. A systematic revision of the African bat genus Scotophilus (Vespertilionidae). Musee Royal de l’Afrique Centrale. Sciences Zoologiques (Tervuren, Belgium) 246: 51–84.
Simmons, N.B. 2005. Order Chiroptera . Johns Hopkins University Press, Baltimore, USA.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Trujillo, R.G., Patton, J.C., Schlitter, D.A. and Bickham, J.W. 2009. Molecular phylogenetics of the bat genus Scotophilus (Chiroptera: Vespertilionidae): perspectives from paternally and maternally inherited genomes. Journal of Mammalogy 90: 548–560.
Vallo, P., Benda, P., Červeny, J. and Koubek, P. 2013. Conflicting mitochondrial and nuclear paraphyly in small-sized
Van Cakenberghe, V. and Happold, M. 2013. Scotophilus viridis Green House Bat, pp 682-684. In: Happold M, Happold DCD (eds) Mammals of Africa: Volume IV Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London.
Wessels, K.J., Colgan, M.S., Erasmus, B.F.N., Asner, G.P., Twine, W.C., Mathieu, R., Aardt, J.A.N., van, Fisher, J.T. and Smit, I.P.J. 2013. Unsustainable fuelwood extraction from South African savannas. Environmental Research Letters 8: 14007.
