
Lesser Long-fingered Bat
Miniopterus fraterculus

2025 Red list status
Near threatened
Regional Population Trend
Declining
Change compared
to 2016
Uplisted
Overview
Miniopterus fraterculus – Thomas & Schwann, 1906
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MINIOPTERIDAE – Miniopterus – fraterculus
Common Names: Lesser Long-fingered Bat, Lesser bent-winged bat (English), Klein Grotvlermuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
The currently recognised Miniopterus fraterculus is possibly a complex of morphologically similar species (Monadjem et al. 2013). Although Miniopterus fraterculus is morphologically very similar to M. natalensis, they are genetically distinct and have different echolocation calls (Miller-Butterworth et al. 2005). The specimens from Malawi and northern Mozambique fall within the size range of M. fraterculus, but have recently been shown to be a new species, M. mossambicus (Monadjem et al. 2013). Similarly, the population from Madagascar has been shown to be specifically distinct from M. fraterculus (Goodman et al. 2007), possibly rendering M. fraterculus a South African and Eswatini endemic. No subspecies are recognised (Skinner & Chimimba 2005).
Red List Status: NT – Near Threatened A4c (IUCN version 3.1)
Assessment Information
Assessors: Richardson, E.1 & da Silva, J.M.2
Reviewers: Richards, L.R.3 & Naidoo, T.3
Institutions: 1Independent Consultant at Richardson & Peplow Environmental, 2South African National Biodiversity Institute, 3Durban Natural Science Museum
Previous Assessors and Reviewers: MacEwan, K., Schoeman, C., Monadjem, A., Cohen, L., Jacobs, D., Richards, L, Sethusa, T. & Taylor, P.
Previous Contributors: Roxburgh, L., Raimondo, D. & Child, M.F.
Assessment Rationale
Although further research is needed, this species is now considered endemic to South Africa and Eswatini (Monadjem et al. 2013) so there is no rescue effect from neighbouring countries. Previously reasonably widespread in the eastern forests of South Africa, fraterculus is threatened by loss of contiguous forests, loss of foraging habitat due to agriculture and urbanisation, loss of roosting habitats and migration stop-overs due to mine and cave closures, increasing effects of droughts caused by the ENSO fluctuations, and potential losses due to wind farm deaths. These threats are not expected to lessen in the future and will continue to impact the species. Found in the eastern and northern parts of the assessment region with an extent of occurrence of 530,350 km2 and in more than 35 locations. Based on the threats that are projected to continue, the species has been uplisted from Least Concern to Near Threatened A4c.
Regional population effects: There is no rescue effect from neighbouring countries.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: Uplisted. Taxonomic changes to the genus Miniopterus mean that fraterculus is now thought to be endemic to South Africa and Eswatini (Monadjem et al. 2013). Loss of forests, loss of roosting habitats, loss of foraging areas, increased droughts caused by ENSO fluctuations, deliberate closure of roosting caves, and potential wind farm deaths have impacted, and will continue to impact species numbers.
Red List Index
Red List Index: Uplisted
Recommended citation: Richardson E & da Silva JM. 2025. A conservation assessment of Miniopterus fraterculus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Although formerly thought to be present in Central Africa, East Africa and southern Africa in discrete ranges (ACR 2015), the largest of which stretches down southeastern Africa, from Malawi, Mozambique and Zimbabwe, down to South Africa (where it extends along the south coast), recent molecular work suggests that it may be endemic to the assessment region (Miller-Butterworth et al. 2005; Goodman et al. 2007; Monadjem et al. 2013).
Within the assessment region, it occurs in the eastern parts of South Africa and in Eswatini (Monadjem et al. 2020), having been recorded from Knysna in the Western Cape, east and north along the coast to southern and western KwaZulu-Natal, in western Eswatini and north along the Drakensberg to Limpopo and Mpumalanga provinces. Although historically recorded from Gauteng there are no recent records from this province. Indeed, these outliers from Krugersdrop (three specimens collected in 1944 and deposited in the Amathole Museum) belong to M. fraterculus on the basis of skull length but there is still the possibility that these specimens have been misidentified (Monadjem et al. 2010). Further vetting of museum records is necessary to delimit distribution more accurately.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): 2200
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
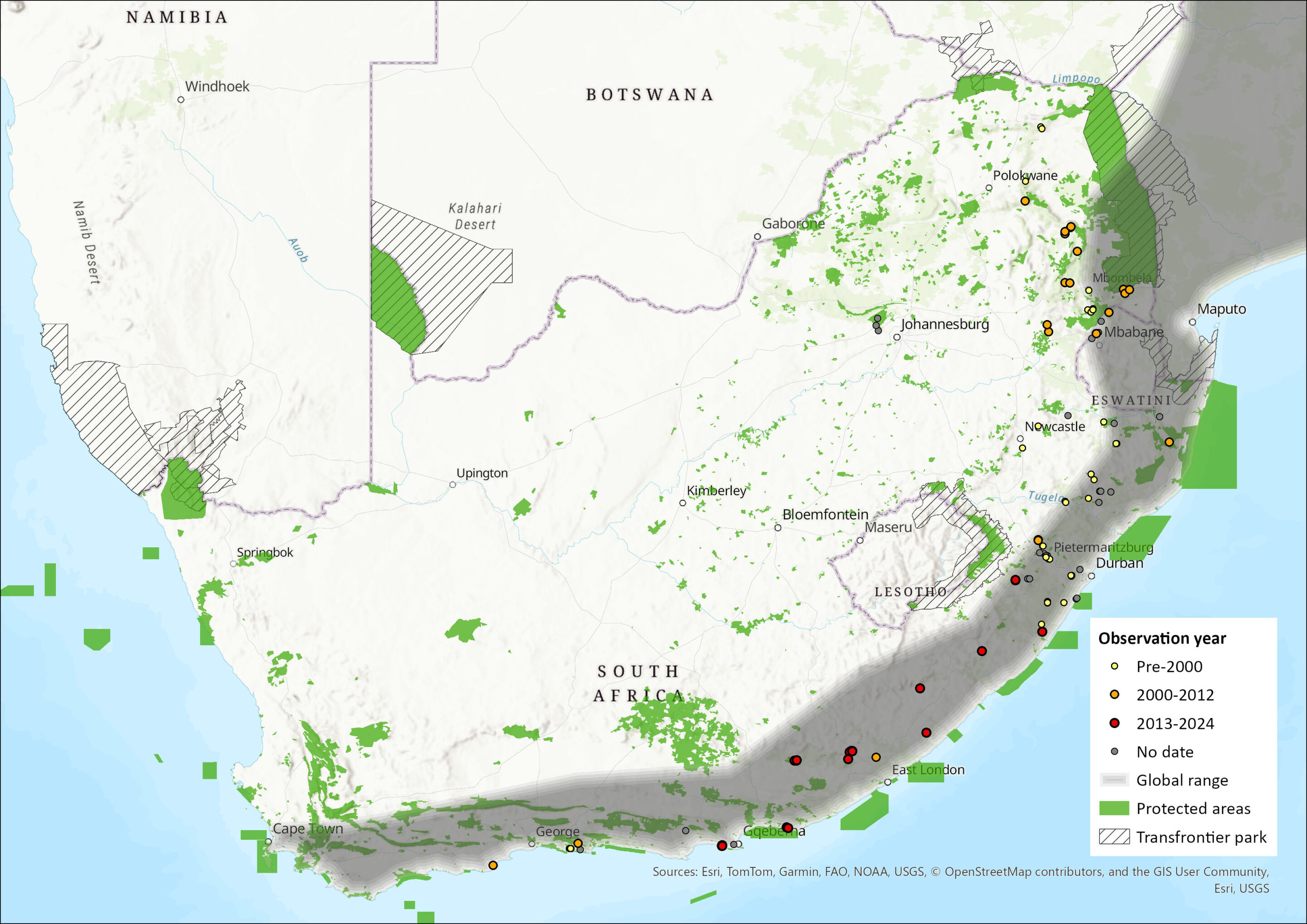 Figure 1. Distribution records for Lesser Long-fingered Bat (Miniopterus fraterculus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Lesser Long-fingered Bat (Miniopterus fraterculus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Presence Uncertain | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Pretorius et al. (2020) found that El Nino-induced drought conditions likely impacted resources and led to a reduction in activity and population size of Miniopterus natalensis. It is probable that fraterculus is similarly vulnerable to this phenological mismatch and increasing droughts will negatively affect the population size.
Population information
The species is widespread but uncommonly recorded and is not well represented in museums, with 55 specimens examined in Monadjem et al. (2020). Like M. inflatus, it occurs alongside M. natalensis but congregates in far smaller numbers (Taylor 2000). Although Friedmann and Daly (2004) listed this species as Near Threatened C1 based on presumed small population but uncertainty of continuing decline, there is no evidence that the population is fewer than 10,000 mature individuals. Subpopulation estimates are needed.
Current population trend: Assumed to be declining.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Continuing decline in mature individuals: Unknown, possible but not confirmed.
Number of subpopulations: Unknown, but the species is found in more than 35 locations.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Morphological similarities between M. frateculus and M. natalensis created much taxonomic confusion, however, a phylogenetic study using a single marker (CytB) found consideration genetic differentiation between the two species (Miller-Butterworth et al. 2005).
To further examine the genetic relationships within and among South Africa’s long-fingered bats, a more comprehensive study was undertaken using two mitochondrial markers – CytB and mitochondrial control region (Moir et al. 2020). With respect to M. frateculus, the study found high haplotypic diversity across the species range due to a high proportion of unique haplotypes, yet no geographical structure (population differentiation) among sampling localities, suggesting the species is a single metapopulation, at least historically. Additionally, the study quantified estimates of effective population size (Ne) dating from 100,000 years based on Bayesian Skyline plots (Moir et al. 2020). Based on the genetic dataset used, there has been an increase in Ne over time, and while the results in the study approximate current Ne at approximately 600,000 individuals (~CI: 300,000 – 4,000,000), caution must be taken in extrapolating contemporary Ne based on analyses designed to measure historical Ne. Broad-based mitochondrial markers, which are generally accepted as most useful for inferring patterns (e.g., phylogenies), are generally insufficient for inferring processes shaping population history due to influences from selection, sex-biased dispersal, mutation rates and sampling (Zink and Barrowclough 2008; Edwards and Bensch 2009; Schmidt & Garroway 2021); and hence. may not accurately quantify present genetic structure and abundance (see Heller et al. 2013).
Habitats and ecology
Miniopterus fraterculus is predominantly a temperate species with the core of its distribution in the montane grasslands of the South African escarpment (Monadjem et al. 2010). It is cave-dependent and hence the availability of suitable roosting sites is a critical factor in determining its distribution, but it occurs in a wide range of habitats from drier savannah bushveld to moister mistbelt and coastal forest habitats. Most localities in KwaZulu-Natal and the Eastern Cape seem associated with major river valleys and dense forested habitats (L Richards, pers com). Suitable cover can take the form of caves, overhangs, and unused mine and railway tunnels (Taylor 1998). For example, in KwaZulu-Natal, it has been found in damp sandstone caves, a solution cave of poorly consolidated glacio-fluvial boulder clay, a rocky overhang over a forest stream, a rock fissure, a railway tunnel as well as in unused mine adits (Taylor 1998). This species probably uses separate caves as winter hibernacula and summer maternity roosts, as does M. natalensis (Monadjem et al. 2010). It is a clutter-edge forager, feeding on a variety of aerial prey including Lepidoptera, Diptera, Hemiptera and Coleoptera (Miller-Butterworth et al. 2005). This is the smallest of the three species of long-fingered bats occurring in the assessment region (Stoffberg et al. 2004; Skinner & Chimimba 2005).
Ecosystem and cultural services: As this species is insectivorous, it may play an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Often, bats prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can result a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.4. Forest -> Forest – Temperate | – | Suitable | – |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | – |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
| 18. Unknown | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
This species is threatened by disturbances to roost sites (maternity and winter roosts) as a result of religious and traditional ceremonies by local communities, as well as tourist activities, carried out in caves in South Africa. The tendency of this species to roost in a few localised sites places it at risk of future population declines (Friedmann & Daly 2004). Extensive transformation of natural habitat, particularly in KwaZulu-Natal (Jewitt et al. 2015), is likely to be causing declines where the insect prey base is depleted as a result of loss of native vegetation or the use of pesticides. Fragmentation of forests, particularly in the Eastern Cape, might limit the ability of these bats to move freely to forage and migrate.
Wind farms pose an emerging threat as Miniopterus species are prone to collisions with turbine blades, particularly when wind farm sites encroach into primary habitat for these species.
Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to colonies of this species.
Conservation
Known from some protected areas in the assessment region, including large parks such as Kruger National Park, iSimangaliso Wetland Park and Serala Wilderness Area. However, to mitigate mortalities from turbine collisions on wind farms, interventions such as using ultrasound to deter bats and curtailing turbines at low wind speeds could be employed (Baerwald et al. 2009; Berthinussen et al. 2010; Arnett et al. 2011).
Recommendations for land managers and practitioners:
- Data sharing by wind farm managers into a national database, to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts, is needed.
- Closures of mines and caves should only be done using bat-compatible methods.
Research priorities:
- Systematic monitoring to identify key roost sites and delimit geographical distribution more accurately.
- Understanding the foraging and habitat needs of the species, especially minimum size of forest patches needed.
- Subpopulation estimates are needed to determine overall population size.
- Monitoring mortalities linked with wind farm operations and assessing impact on populations.
- Molecular analysis and vetting of museum records to delimit distribution more accurately and to resolve taxonomic status.
Encouraged citizen actions:
- Limit disturbance to roost sites.
- Deposit dead specimens with the Durban Natural Science Museum.
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Arnett EB, Huso MM, Schirmacher MR, Hayes JP. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Baerwald EF, Edworthy J, Holder M, Barclay RM. 2009. A large-scale mitigation experiment to reduce bat fatalities at wind energy facilities. Journal of Wildlife Management 73: 1077–1081.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Boyles JG, Cryan PM, McCracken GF, Kunz TH. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Driver A, Sink KJ, Nel JN, Holness S, van Niekerk L, Daniels F, Jonas Z, Majiedt PA, Harris L, Maze K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Edwards. S. & Bensch, S. 2009. Looking forwards or looking backwards in avian phylogeography? A comment on Zink and Barrowclough 2008. Mol Ecol 18:2930–2933.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
Goodman, S. M., Ryan, K. E., Maminirina, C. P., Fahr, J., Christidis, L. and Appleton, B. 2007. Specific status of populations on Madagascar referred to Miniopterus fraterculus (Chiroptera: Vespertilionidae), with description of a new species. Journal of Mammalogy 88: 1216-1229.
Jewitt D, Goodman PS, Erasmus BFN, O’Connor TG, Witkowski ETF. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kunz TH, Braun de Torrez E, Bauer D, Lobova T, Fleming TH. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Miller-Butterworth CM, Eick G, Jacobs DS, Schoeman MC, Harley EH. 2005. Genetic and phenotypic differences between South African long-fingered bats, with a global Miniopterine phylogeny. Journal of Mammalogy 86: 1121–1135.
Monadjem, A., Goodman, S.M., Stanley, W.T. and Appleton, B. 2013. A cryptic new species of Miniopterus from south-eastern Africa based on molecular and morphological characters. Zootaxa 3746(1): 123-142.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Pretorius, M., Broders, H., Seamark, E. and Keith, M. 2020. Climatic correlates of migrant Natal long-fingered bat (Miniopterus natalensis) phenology in north-eastern South Africa. Wildlife Research 47: 404-414.
Schmidt, C. and Garroway, C.J. 2021. The conservation utility of mitochondrial genetic diversity in macrogenetic research. Conservation Genetics, 22(3), pp.323-327.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Stoffberg, S., Jacobs, D.S. and Miller-Butterworth, C.M. 2004. Field identification of two morphologically similar bats, Miniopterus schreibersii natalensis and Miniopterus fraterculus (Chiroptera: Vespertilionidae). African Zoology 39: 47–53.
Taylor, P. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Zink, R.M. and Barrowclough, G.F. 2008. Mitochondrial DNA under siege in avian phylogeography. Molecular ecology, 17(9), pp.2107-2121.
