Lesser Grey-brown Musk Shrew
Crocidura silacea

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Crocidura silacea – Thomas, 1895
ANIMALIA – CHORDATA – MAMMALIA – EULIPOTYPHLA – SORICIDAE – Crocidura – silacea
Common Names: Lesser Grey-brown Musk Shrew, Peters’ Musk Shrew (English), Peters se Skeerbek (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
Although there has been some controversy over the validity of this species (Ellerman et al. 1953; Heim de Balsac & Meester 1977), subsequent morphometric studies have supported its status as a species (Meester et al. 1986; Taylor et al. 1994; Taylor & Contrafatto 1996).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessor: Russo, I.M.1 & da Silva, J.2
Reviewer: Patel, T.3
Institutions: 1Cardiff University, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors: Taylor, P., Baxter, R. & Monadjem, A.,
Previous Reviewer: Child, M.F.
Previous Contributors: Avery, M., MacFadyen, D., Avenant, N., Wilson, B. & Palmer, G.
Assessment Rationale
This species is widely distributed within the assessment region and occurs in many protected areas, including the Kruger National Park, and can be locally common in some areas (for example, Maputaland, KwaZulu-Natal Province). Although it occurs in multiple habitat types across its range, it has not been recorded from agricultural or modified habitats. Thus, the species relies on intact ecosystems and ongoing habitat loss and degradation of grasslands, woodlands and wetlands is a threat to this species. The loss of moist grasslands through climate change is an emerging threat that should be monitored closely as it may push this species into a threatened category. The species is currently listed as Least Concern as there is no evidence for net decline. Further surveys and research should focus on vetting existing museum records as many have been misidentified as C. cyanea and vice versa, leading to inaccuracies in the distribution map. Key interventions include protected area expansion of moist grassland habitats, as well as incentivising landowners to sustain natural vegetation around wetlands and to keep livestock or wildlife at ecological carrying capacity to avoid overgrazing.
Regional population effects: No significant rescue effects are possible as, although habitats are presumably connected across regions in some areas, this species is too small to disperse over long distances.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Russo IM & da Silva JM. 2025. A conservation assessment of Crocidura silacea. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
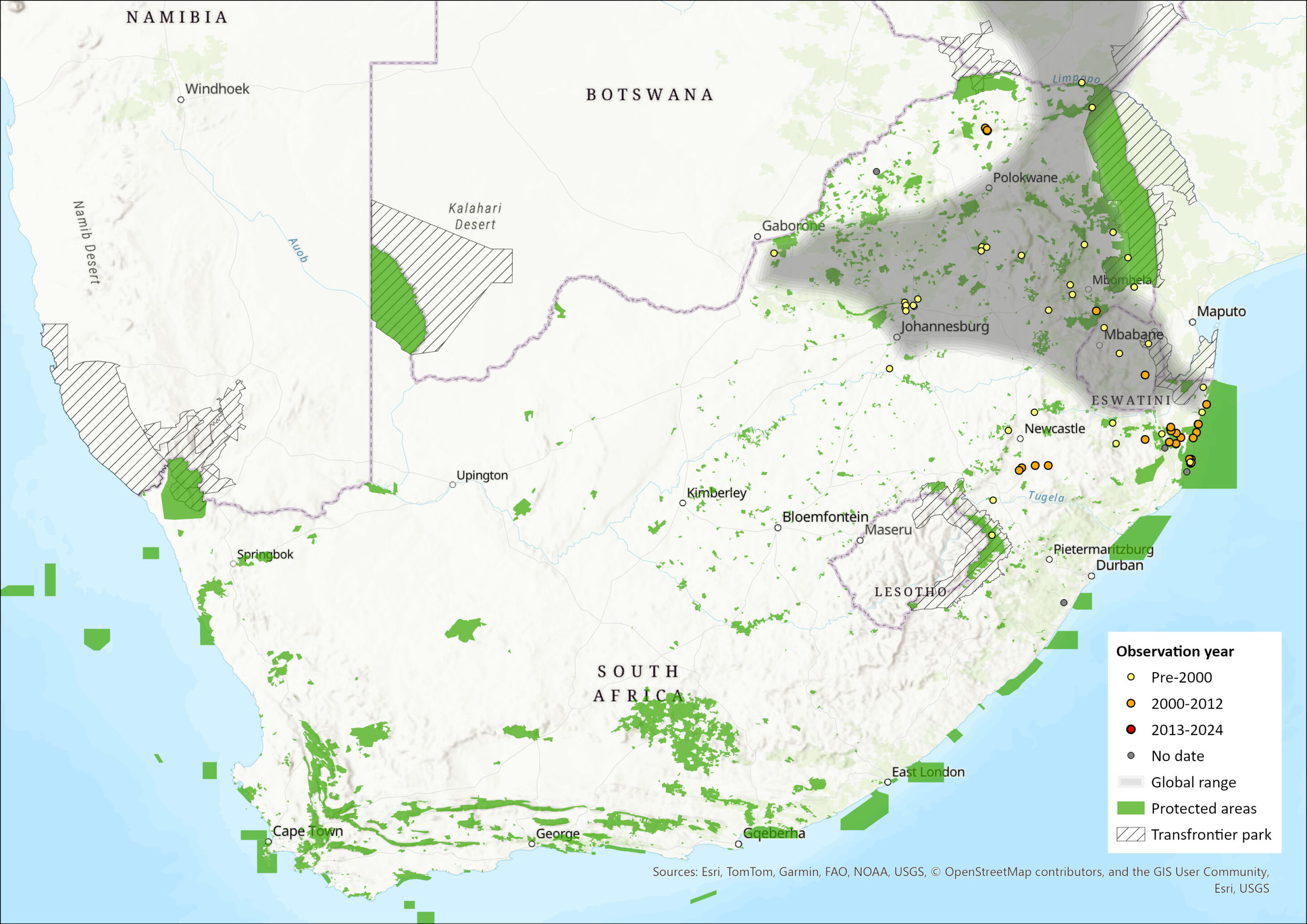
This southern African species is present in Zimbabwe, southern Mozambique, South Africa and Eswatini, and might be present in parts of Lesotho, Botswana, southern Malawi, Zambia and Angola, but requires confirmation from new field surveys. Within the assessment region, they are widely distributed in the Limpopo (Rautenbach 1982), Gauteng and Mpumalanga Provinces, with a scattered but wide distribution in the KwaZulu-Natal Province as far south as Vernon Crookes Nature Reserve (Skinner and Chimimba 2005). This species was once known in Eswatini from just two specimens (Monadjem 1998), but further field studies have confirmed a wider distribution (e.g., Avenant and Kuyler 2002). This species is very similar, and almost indistinguishable (Taylor and Contrafatto 1996), from C. cyanea but is more restricted in distribution. Existing museum records need to be exhaustively vetted as there may be errors in the distribution maps for both these species.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Lesser Grey-brown Musk Shrew (Crocidura silacea) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Presence Uncertain | Native | – | – |
| Botswana | Presence Uncertain | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Lesotho | Possibly Extant | Native | – | – |
| Malawi | Presence Uncertain | Native | – | – |
| Mozambique | Possibly Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Presence Uncertain | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
The loss of moist grasslands through climate change is an emerging threat that should be monitored closely as it may push this species into a threatened category.
Population Information
This species can be common or abundant in suitable habitats, for example, in Maputaland, the northern part of the KwaZulu-Natal Province (P. Taylor unpubl. data), however, in Mkhuze Game Reserve, KwaZulu-Natal Province, this species was the least abundant shrew sampled where C. fuscomurina and C. hirta, represented 73% of all captures (Delcros et al. 2014). Similarly, at Phinda Private Game Reserve, KwaZulu-Natal Province, it was only more abundant than S. infinitesimus with C. fuscomurina and C. hirta as the most abundant species (Rautenbach et al. 2014).
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Unknown | – | – |
Current population trend: Declining, based on ongoing habitat loss.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: Yes. Does not occur in transformed landscapes and has poor dispersal ability.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While the species has been investigated in a phylogenetic context (Willows-Munro & Conradee 2011), no population genetics study has been conducted on this species within the assessment region. While it is possible the species exists as a single metapopulation in the region, isolated subpopulations may exist (e.g., animals in KwaZulu-Natal maybe isolated from Limpopo, Mpumalanga and Gauteng animals). Further molecular studies are needed to investigate this. Given the absence of density and population size estimates, it is not possible to quantify effective population size (Ne).
Habitats and ecology
This species occurs in montane evergreen forest, savannah woodland, bushveld, grassland and coastal forest, and has been collected from under trees, in old timber and under stones (Skinner and Chimimba 2005). In Maputaland, it prefers sand forest. In Mkhuze, Kube Yini and Phinda Game Reserves, it was found in Lebombo wooded grassland, Spirostachys africana woodland, Acacia woodland, Terminalia sericea woodland, Combretum molle woodland on red sand and sand forest (Delcros et al. 2014). In Eswatini, specimens have been collected from savannah woodland (Monadjem 1998), and in tall grassland, rocky thicket, short rocky forest, and low open rocky woodland in the Maguga Dam area (Avenant and Kuyler 2002). The species thus exists in a wide range of habitats, however, it has not been captured on agricultural or transformed landscapes and so rely on intact environments.
Ecosystem and cultural services: An important prey species (e.g., Avery et al. 2002).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.7. Grassland -> Grassland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 6. Rocky areas (eg. inland cliffs, mountain peaks) | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The main threat to shrews is the loss or degradation of moist, productive areas such as wetlands and rank grasslands within suitable habitat. The two main drivers behind this are abstraction of surface water and draining of wetlands through industrial and residential expansion, and overgrazing of moist grasslands, which leads to the loss of ground cover and decreases small mammal diversity and abundance (Bowland and Perrin 1989, 1993). Suppression of natural ecosystem processes, such as severe fire, can also lead to habitat degradation through bush encroachment or loss of plant diversity through alien invasives, and is suspected to be increasing with human settlement expansion. There are also clear overlaps and synergistic effects between these threats. We infer a continuing population decline based on loss of natural habitat.
Current habitat trend: Although widespread, remaining habitat patches are in decline. For example, there was a 19.7% loss of natural habitat in KwaZulu-Natal Province from 1994 to 2008, with an average loss of 1.2% per annum (Jewitt et al. 2015). Similarly, between 2000 and 2013, there has been a 5.6% and 1.1% rate of urban and rural expansion in KwaZulu-Natal Province respectively (GeoTerraImage 2015). If this rate of loss continues into the future, there will be an estimated 12% loss of habitat over 10 years. Additionally, it may be part of a suite of species that will display a general decline with grassland and fynbos contraction due to climate change (Taylor et al. 2016). Because of their high metabolism, low dispersal capacity and short life spans, climate change will reduce the amount of suitable habitat available.
Conservation
This species is found in several protected areas across its range, including the Kruger National Park. The main interventions for this species are protecting and restoring suitable habitat, such as moist grassland and fynbos patches. Biodiversity stewardship schemes should be promoted to conserve such patches. Protecting these habitats may create dispersal corridors between patches. At the local scale, landowners and managers should be educated, encouraged and incentivised to conserve the habitats on which shrews and small mammals depend. Retaining ground cover is the most important management tool to increase small mammal diversity and abundance. This can be achieved through lowering grazing pressure (Bowland and Perrin 1989), or by maintaining a buffer strip of natural vegetation around wetlands (Driver et al. 2012). Small mammal diversity and abundance is also higher in more complex or heterogeneous landscapes, where periodic burning is an important tool to achieve this (Bowland and Perrin 1993). Removing alien vegetation from watersheds, watercourses and wetlands is also an important intervention to improve flow and water quality, and thus habitat quality for shrews. Education and awareness campaigns should be employed to teach landowners and local communities about the importance of conserving wetlands and moist grasslands.
Recommendations for land managers and practitioners:
- Landowners and communities should be incentivised to stock livestock or wildlife at ecological carrying capacity to avoid overgrazing and to maintain a buffer of natural vegetation around wetlands.
- Enforce regulations on developments that potentially impact on the habitat integrity of grasslands and wetlands.
Research priorities:
- Additional field surveys are needed to clarify and confirm the distribution of this species.
- The effects of climate change on species distribution and abundance should be modelled.
- Museum records must be confirmed to refine the distribution map.
Encouraged citizen actions:
- Citizens are requested to submit any shrews killed by cats or drowned in pools to a museum or a provincial conservation authority for identification, thereby enhancing our knowledge of shrew distribution (carcasses can be placed in a ziplock bag and frozen with the locality recorded).
Bibliography
Avenant NL, Kuyler P. 2002. Small mammal diversity in the Maguga Dam inundation area, Swaziland. South African Journal of Wildlife Research 32: 101-108.
Avery, D.M., Avery, G. and Roberts, A. 2002. A contribution from barn owl pellets to known micromammalian distributions in KwaZulu-Natal, South Africa. African Zoology 37: 131-140.
Bowland, A.E. and Perrin, M.R. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54: 251–260.
Bowland, J.M. and Perrin, M.E. 1993. Wetlands as reservoirs of small-mammal populations in the Natal Drakensberg. South African Journal of Wildlife Research 23: 39–43.
Delcros, G., Taylor, P.J., and Schoeman, M.C. 2015. Ecological correlates of small mammal assemblage structure at different spatial scales in the savannah biome of South Africa. Mammalia 79(1): 1-14.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758-1951: A reclassification. Trustees of the British Museum (Nat. Hist.), London, UK.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Heim de Balsac, H. and Meester, J. 1977. Order Insectivora. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-29. Smithsonian Institution Press, Washington, D. C., USA.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum , Pretoria, South Africa.
Monadjem A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor PJ, Contrafatto G. 1996. Mandible shape and size in three species of small musk shrews (Crocidura Wagler, 1832) from southern Africa. Mammalia 60: 753-766.
Taylor PJ, Nengovhela A, Linden J, Baxter RM. 216. Past, present, and future distribution of Afromontane rodents (Muridae: Otomys) reflect climate-change predicted biome changes. Mammalia 80: 359-375.
Taylor, P.J., Richardson, E.J., Meester, J. and Wingate, L. 1994. New distribution records for six small mammal species in Natal, with notes on their taxonomy and ecology. Durban Museum Novitates 19: 59-66.