
Lesotho Horseshoe Bat
Rhinolophus cervenyi

2025 Red list status
Endangered
Regional Population Trend
Unknown
Change compared
to 2016
New Assessment
Overview
Rhinolophus cervenyi -Benda et al. 2024
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – cervenyi
Common Names: Lesotho Horseshoe Bat, Drakensberg Horseshoe Bat (English)
Synonyms: Rhinolophus clivosus sensu lato K. Andersen, 1905 Lynch and Watson 1990: 532; Lynch 1994: 190; Taylor 1998: 35; Taylor 2005: 338; Monadjem et al. 2010: 188, 556; Benda and Vallo 2012: 93; Monadjem et al. 2020: 216, 659.
Taxonomic Note: This species was recently described from Lesotho based on mitochondrial and nuclear DNA markers together with morphological characters from museum specimens (Benda et al. 2024). Taylor et al (2024) provided further baculum and echolocation peak frequency characters to demarcate this species from all other horseshoe bats and extended its distribution from Lesotho to the Drakensberg Mountains of the Free State, KwaZulu-Natal and Mpumalanga provinces. Due to mitochondrial DNA introgression between this species and R. damarensis, and the similarity in size of R. cervenyi and R. clivosus (=acrotis), this new species was previously confused with either of these two species (Jacobs et al. 2013, Dool et al. 2016, Demos et al. 2019).
Red List Status: EN – Endangered C2a(i), (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.J.1, Howard, A.1 & da Silva, J.M.2
Reviewers: Smith, C.3 & Bastian, A.4.
Institutions: 1University of the Free State, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust, 4University of KwaZulu-Natal
Assessment Rationale
This horseshoe bat is restricted to montane grasslands with less than 2,500 mature individuals known from the few, isolated localities where it’s been recorded (Taylor et al. 2024). It roosts in small colonies with less than 250 individuals recorded in each subpopulation and is currently under threat from habitat degradation and roost disturbances in Lesotho and South Africa. The Drakensberg horseshoe bat is endemic to the assessment region with most localities recorded in the Maloti-Drakensberg Mountain range and associated foothills. The extent of occurrence is 102,009 km2 and the area of occupancy is 112 km2.
Regional population effects: Endemic to South Africa and Lesotho.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: New Assessment
Red List Index
Red List Index: New Assessment
Recommended citation: Taylor PJ, Howard A & da Silva JM. 2025. A conservation assessment of Rhinolophus cervenyi. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
It occurs at higher elevations, generally above 1,800m in the Maloti-Drakensberg Mountains of Lesotho and adjacent regions on the Free State, KwaZulu-Natal and Mpumalanga provinces (Howard et al. 2023; Benda et al. 2024; Taylor et al. 2024).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 1,275 m (Mondajem et al 2024)
Elevation Upper Limit (in metres above sea level): 3,010 m (Mondajem et al 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
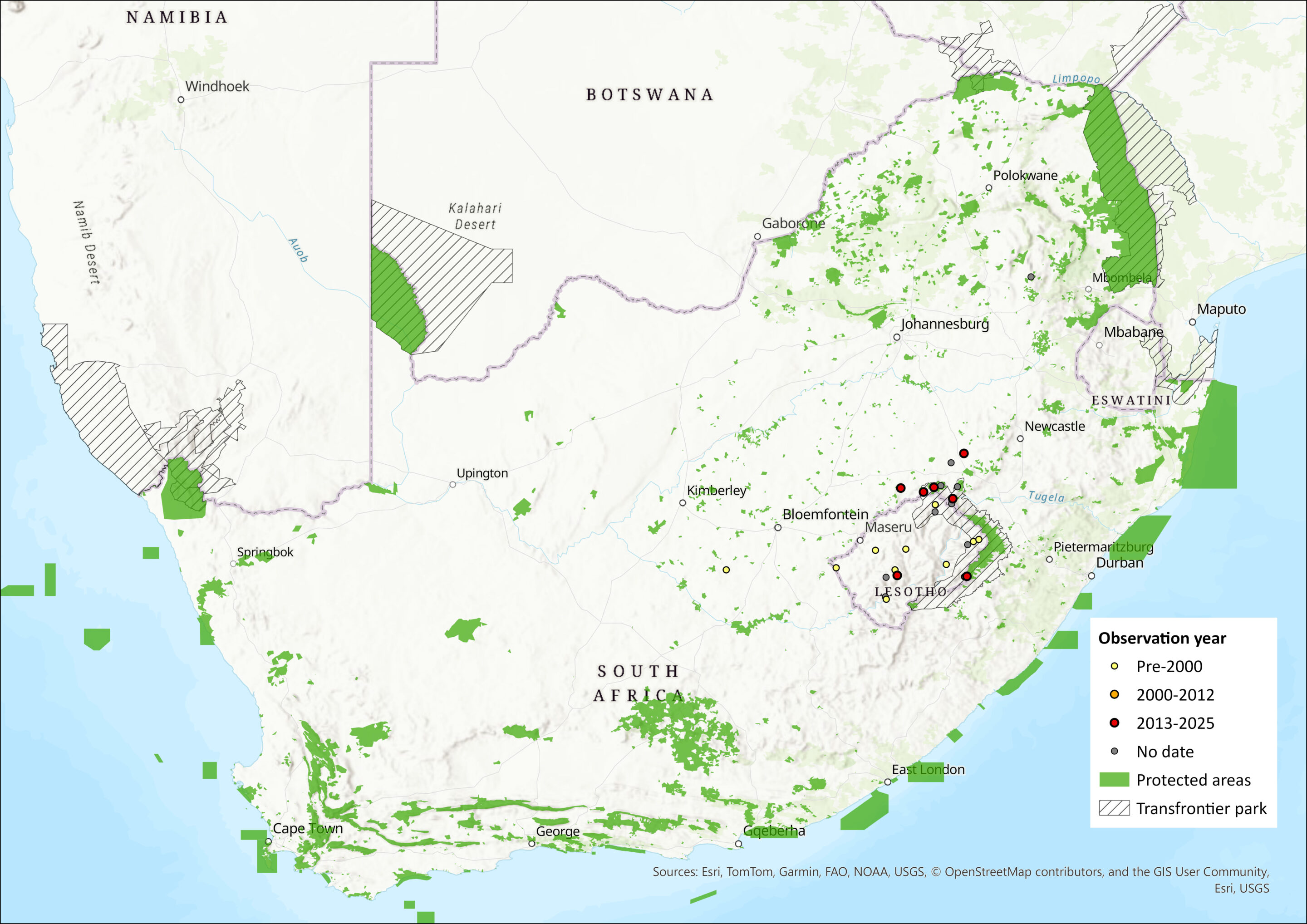 Figure 1. Distribution records for Lesotho Horseshoe Bat (Rhinolophus cervenyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Lesotho Horseshoe Bat (Rhinolophus cervenyi) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Lesotho | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
A study by Taylor et al. (2024) reported minimal range changes for this species under future climate models by suggesting the high-elevation mountains provide a potential refugia for temperate montane species. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (become a growing concern as heat waves and maximum temperatures are expected to increase over much of South Africa (Adams & Hayes 2021; Mbokodo et al. 2020).
Population information
The average colony size is largely unknown, but this species is known to be colonial. The type specimen was collected with 12 others in a cave at Sehlabathebe National Park in Lesotho. Several colonies were sampled in Lesotho (Benda et al., 2024), and one colony of approximately 15 individuals was located at Schaapplaats Farm in the eastern Free State.
Current population trend: Unknown
Continuing decline in mature individuals: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
Earlier phylogenetic studies based on mitochondrial markers showed this species to be introgressed with R. damarensis; however, recent taxonomic evidence incorporating phylogenetic comparisons using nuclear markers has confirmed this be a separate species (Benda et al. 2o24).
No population genetic study has been conducted on the species, however, given the distance between known colonies and localities, it is expected that each represents isolated subpopulations, numbering 18 in total. This will need to be confirmed through a comprehensive population genetic study.
While a confident measure of the species’ population size is unavailable, approximately 12 mature individuals have been counted per colony. Considering the difficulty in locating roosts of this species, these numbers are most probably underestimates, with numbers possibly closer to 50-60 individuals per colony, totalling ~1000 individuals for the species. Based on these estimates, each colony is thought to have a drastically low effective population size (Ne) – 5-18 individuals (based on an Ne/Nc ratio of 0.1-0.3).
Habitats and ecology
Lesotho Horseshoe Bat is endemic to the Maloti-Drakensberg Mountain range and foothills and has mostly sampled from caves in open grassland habitats. Rhinolophus species have intermediate wing loading with low aspect ratios as clutter foragers thus require dense and intact vegetation (Monadjem et al. 2020).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides which has negative effects for human health (Frank 2024).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 4.4 Grassland -> Temperate | |||
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | – |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Suspected to move altitudinally with seasons
Congregatory: Small colonies
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
National Commercial Value: No
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The main threats known to this species is the disturbance of roosting sites in caves and sandstone overhangs which are used as kraals for livestock and as campsites. Overgrazing and land use degradation are widespread threats across the species range, especially in Lesotho. Clutter-edge bats such as Rhinolophus are sensitive to vegetation changes and anthropogenic changes.
Conservation
This species has been recorded in the Maloti-Drakensberg Transfrontier Park – including the Ukuhlamba Drakenberg World Heritage Site and Sehlabathebe National Park in Lesotho. However, much of the high montane and alpine habitat in Lesotho is unprotected and under increasing anthropogenic change especially from overgrazing, settlement, mining, impoundments and from disturbance of cave roosts, e.g. for religious purposes.
Recommendations for land managers and practitioners:
- Identify and protect important roost sites for this species
- Restore and protect wetlands and grasslands across the species range to ensure adequate foraging habitat.
- Reduce pesticide use in agricultural landscapes.
Research priorities:
- Further field and acoustic surveys are required to more accurately delimit the distribution of this species.
- Ecological studies are needed to determine the dietary, reproductive and spatial ecology of R. cerveyni.
- Substantiate the genetic species delimitation in SA by including non-MT DNA markers.
- Conduct a population genetic analysis to understand the gene flow between subpopulations.
Encouraged citizen actions:
- Minimise disturbance at caves when visiting. Do not disturb bats if found roosting in exposed overhangs on hiking trails
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Benda, P., Uvizl, M., Eiseb, S. J., & Avenant, N. L. 2024. On the systematic position of the horseshoe bats (Mammalia: Chiroptera) from Lesotho. Mammalia, 88, 239–258. https://doi. org/10.1515/mammalia-2023-0119
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Demos, T.C., Webala, P.W., Goodman, S.M., Peterhans, J.C.K., Bartonjo, M., and Patterson, B.D. 2019. Molecular phylogenetics of the African horseshoe bats (Chiroptera: Rhinolophidae): expanded geographic and taxonomic sampling of the Afrotropics. BMC Evol. Biol. 19: 1–14.
Dool, S.L., Puechmaille, S.J., Foley, N.M., Allegrini, B., Bastian, A., Mutumi, G.L., Maluleke, T.G., Odendaal, L.J., Teeling, E.C., and Jacobs, D.S. 2016. Nuclear introns outperform mitochondrial DNA in interspecific phylogenetic reconstructions: lessons from horseshoe bats (Rhinolophidae: Chiroptera). Mol. Phylogenet. Evol. 97: 196–212.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Frank, E.G. 2024. The economic impacts of ecosystem disruptions: Costs from substituting biological pest control. Science 385,0344.
Jacobs, D.S., Babiker, H., Bastian, A., Kearney, T., van Eeden, R. and Bishop, J.M. 2013. Phenotypic convergence in genetically distinct lineages of a Rhinolophus species complex (Mammalia, Chiroptera). PloS one 8: e82614.
Mbokodo, I., Bopape, M.J., Chikoore, H., Engelbrecht, F. and Nethengwe, N. 2020. Heatwaves in the future warmer climate of South Africa. Atmosphere, 11(7), p.712.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Taylor, P. J., Kearney, T. C., Clark, V. R., Howard, A., Mdluli, M. V., Markotter, W. Geldenhuys, M., Richards, L. R., Rakotoarivelo, A. R., Watson, J., Balona, J., and Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate-driven diversification and a buffer against future climate change in bats, Global Change Biology, 30:e17344, https://doi.org/10.1111/gcb.17344.
