Large Grey Mongoose
Herpestes ichneumon

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Herpestes ichneumon – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – HERPESTIDAE – Herpestes – ichneumon
Common Names: Large Grey Mongoose, Egyptian Mongoose, Ichneumon, Ichneumon mongoose (English), Grootgrysmuishond (Afrikaans), Mosêlamotlhaka (Tswana), Inhlangala (Zulu)
Synonyms: Viverra ichneumon Linnaeus, 1758
Taxonomic Note:
Meester et al. (1986) recognised two subspecies of Herpestes ichneumon in southern Africa; namely H. i. cafer (Gmelin 1788) from Hermanus in the Western Cape along the coast to KwaZulu-Natal, and into Mpumalanga, eastern Eswatini, Mozambique and Zimbabwe; and H. i. mababiensis Roberts 1932, which is restricted to northern Botswana.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Do Linh San, E.1, Streicher J.P.2,3,4 & da Silva, J.M.2
Reviewer: Power, R.J.5
Institutions: 1Sol Plaatje University; 2South African National Biodiversity Institute, 3BirdLife South Africa, 4University of KwaZulu-Natal, 5North West Province’s Directorate of Biodiversity Management
Previous Assessors and Reviewers: Do Linh San, E., Emslie, K., Maddock, A.H., Perrin, M., Stuart, C., Stuart, M. & Palomares, F.
Previous Contributor: Relton, C.
Assessment Rationale
The Large Grey Mongoose is listed as Least Concern as the species is relatively common, with a very wide diet, there are no major threats, and it is present in a number of protected areas within the assessment region. This species seems to be very adaptable, occupying a large range of habitats in its wide distribution range, but it might be more specialised in the assessment region, possibly as a result of intraguild competition and predation pressure. Because this species is often associated with riparian habitats and wetlands, we therefore recommend monitoring in areas where development may be affecting water supply and/or quality, to determine potential negative impacts on this mongoose through changes in understorey vegetation (for cover) and possibly food availability.
Regional population effects: The Large Grey Mongoose can disperse across regional borders between South Africa and Mozambique, as its range is continuous across much of southeast Africa and this species is not constrained by fences. Considering that this mongoose has generally not been recorded very far inland (see Figure 1), it is, however, possible that dispersal and movements are at least partially dependent upon the presence of water corridors – and related (understorey) vegetation – such as permanent and seasonal rivers and streams, dam networks or wetlands.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Do Linh San E, Streicher JP & da Silva JM. 2025. A conservation assessment of Herpestes ichneumon. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
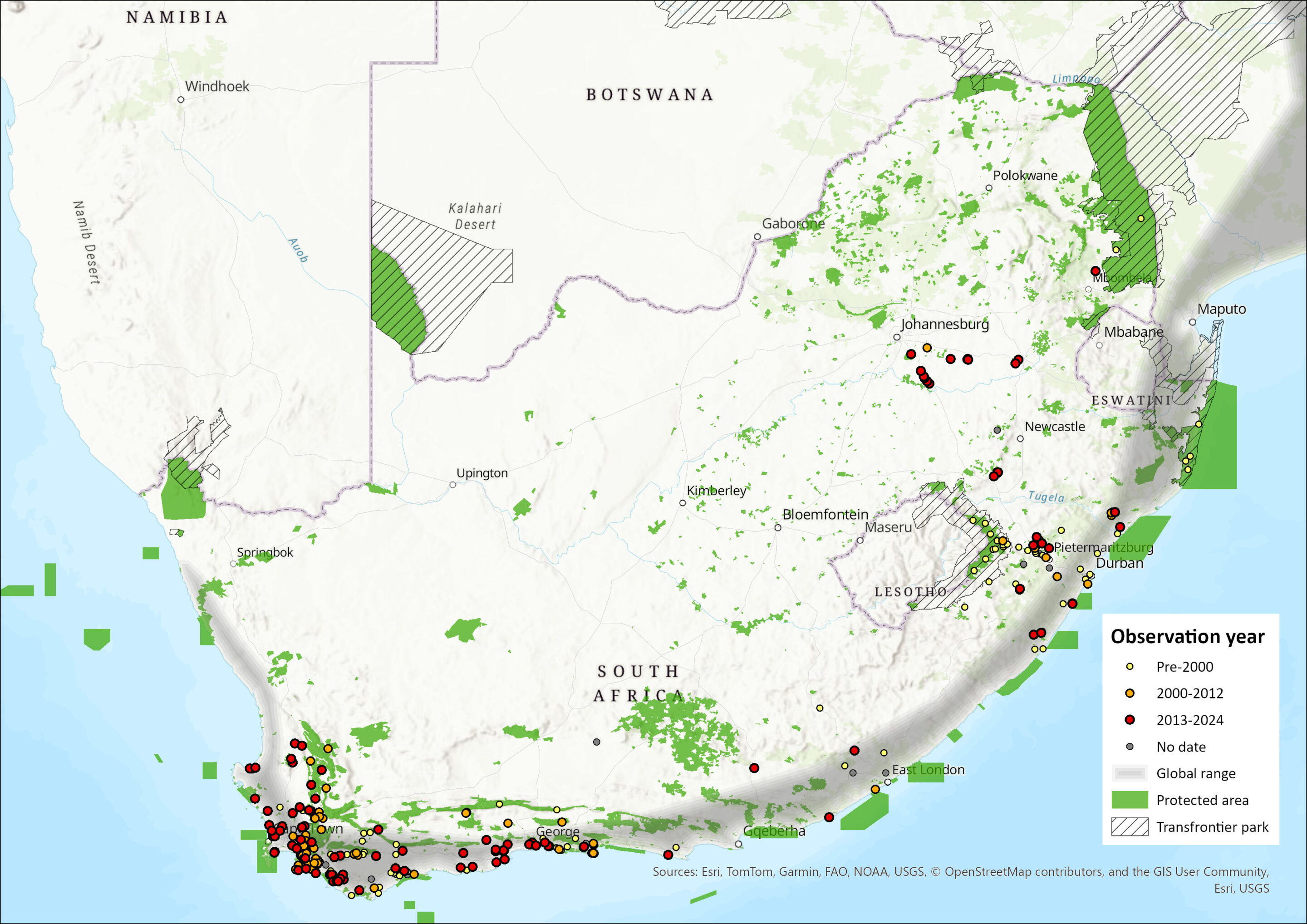
The Large Grey Mongoose is found mainly in sub-Saharan Africa, from Senegal and Gambia to East Africa, then southwards in Angola, Zambia, Malawi and Mozambique (Ngatia et al. 2021). It is absent from much of drier southern Africa, but is present in northeast Namibia (Pallett & Thomson 2022), northern Botswana, northern and eastern Zimbabwe, and all along the South African coastline (Palomares 2013). This species has not been introduced to Madagascar (Goodman 2012), contrary to what may have been suggested in some sources (e.g. Haltenorth & Diller 1980).
In the assessment region the Large Grey Mongoose is mostly present along the coast, from the Western Cape (as far north as Kleinsee) to KwaZulu-Natal through the Eastern Cape Province. There are increasingly more records from the hinterland of the Eastern Cape, Mpumalanga, eastern Gauteng and the eastern Free State (Figure 1). The species is possibly extant in eastern Lesotho (Lynch 1994). It was once recorded in the Lubombo region of Eswatini (Skinner & Chimimba 2005), but no observations were made or reported over the past two decades at least (Monadjem 1998). It is present in the extreme east of Mpumalanga Province, essentially in Kruger National Park, though few records exist there. However, the species has been observed in Marievale Bird Sanctuary, Gauteng in 2016 (V. Pretorius & M. Pretorius pers. comm. 2016), as well as camera-trapped and observed at Sasol’s Synfuels Plant in Secunda, Mpumalanga, in 2014–2016 (Swanepoel et al. 2018). These observations therefore suggest that the distribution range of the Large Grey Mongoose in the assessment region might be much larger than initially thought. Although range expansion cannot be excluded, the most parsimonious explanation is that the presence of this small carnivore has been overlooked in at least some areas due to its relatively low densities (see Population) and secretive nature. However, it is also possible that interspecific competition with other abundant and generalist small carnivores such as the Cape Grey Mongoose (Herpestes pulverulentus) and to a lesser extent the Slender Mongoose (H. sanguineus) might play a role, particularly in the Nama-Karoo and Grassland biomes, respectively. Additionally, the impact of mesopredators such as Black-backed Jackals (Lupulella mesomelas) and Caracals (Caracal caracal) on this mongoose is likely to be stronger in the more open habitats that characterise these two biomes. The importance of intraguild predation or at least predation pressure (creating a “landscape of fear”) on this species has been demonstrated in Spain where Herpestes ichneumon is rare in areas where the Iberian Lynx (Lynx pardinus) is abundant (Palomares et al. 1996). The presence of the Large Grey Mongoose in the inland biomes might be restricted to densely vegetated linear riverine habitats and pockets of wetlands – although it does not seem to be the case far inland – and where it may compete for food and safe resting sites with the Water Moongoose (Atilax paludinosus).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 3000 m (in Ethiopia)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Large Grey Mongoose (Herpestes ichneumon) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Benin | Extant | Native | – | Resident |
| Botswana | Extant | Native | – | Resident |
| Burkina Faso | Extant | Native | – | Resident |
| Burundi | Extant | Native | – | Resident |
| Cameroon | Extant | Native | – | Resident |
| Central African Republic | Extant | Native | – | Resident |
| Chad | Extant | Native | – | Resident |
| Congo | Extant | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Côte d’Ivoire | Extant | Native | – | Resident |
| Djibouti | Presence Uncertain | Native | – | Resident |
| Egypt | Extant | Native | – | Resident |
| Eritrea | Extant | Native | – | Resident |
| Eswatini | Presence Uncertain | Native | – | Resident |
| Ethiopia | Extant | Native | – | Resident |
| Gabon | Extant | Native | – | Resident |
| Gambia | Extant | Native | – | Resident |
| Ghana | Extant | Native | – | Resident |
| Guinea | Extant | Native | – | Resident |
| Guinea-Bissau | Extant | Native | – | Resident |
| Israel | Extant | Native | – | Resident |
| Jordan | Extant | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Lebanon | Extant | Native | – | Resident |
| Lesotho | Possibly Extant | Native | – | Resident |
| Liberia | Extant | Native | – | Resident |
| Libya | Extant | Native | – | Resident |
| Malawi | Extant | Native | – | Resident |
| Mali | Extant | Native | – | Resident |
| Mauritania | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | Resident |
| Niger | Extant | Native | – | Resident |
| Nigeria | Extant | Native | – | Resident |
| Portugal | Extant | Introduced | – | Resident |
| Rwanda | Extant | Native | – | Resident |
| Senegal | Extant | Native | – | Resident |
| Sierra Leone | Extant | Native | – | Resident |
| Somalia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Spain | Extant | Introduced | – | Resident |
| South Sudan | Extant | Native | – | Resident |
| Sudan | Extant | Native | – | Resident |
| Syrian Arab Republic | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Togo | Extant | Native | – | Resident |
| Türkiye | Extant | Native | – | Resident |
| Uganda | Extant | Native | – | Resident |
| Western Sahara | Extant | Native | – | Resident |
| Zambia | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
In Europe, the range and population size of the Large Grey Mongoose have increased over the past 35 years, in both Portugal and Spain, due to the reduction of this species’ natural predators (Delibes 1999), as well as land-use and climate change (Barros et al. 2015, 2016). No similar longitudinal studies have been conducted in the assessment region nor in Africa in general, so it is unclear whether land-use changes – including the direct and indirect reduction of natural predators – and climate change, has had or would have similar “positive” effects in the future. However, the presence of a wider range of predators and competitors in Africa (Caro & Stoner 2003) compared to the Iberian Peninsula suggest that the local abundance of this species is unlikely to change drastically. The situation might be different in terms of range expansion (Swanepoel et al. 2018), and the widespread use of camera-trapping in the assessment region should, sooner than later, shed light on this issue.
Population
On the African mainland, this species is widespread and locally common (Palomares 2013). Densities ranging from 0.1 (East Africa: Hendrichs 1972) to 1.2 individual(s) / km² (South Africa: Maddock 1988) have been recorded. Palomares and Delibes (1992a) estimated a density of 2 individuals / km² in optimal habitats in Spain. This is generally well below maximum densities documented for other mongoose species in the assessment region, but it is still relatively high. It is currently not possible to estimate population size precisely, but we infer that the population is currently stable based on this species’ extent of occurrence and the lack of major threats (see Threats).
Population Information
Current population trend: Unknown, but probably stable.
Continuing decline in mature individuals: Unknown, but probably not.
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: It is not currently possible to determine the extent or number of subpopulations.
Severely fragmented: No. This species seems to have a relatively continuous distribution along the South African coast.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
The sequencing of the first mitochondrial genome of Herpestes ichneumon enabled this species to be investigated in a broad phylogenetic context with others in the Feliformia suborder (Boukhoud et al. 2021), expanding on past molecular research on these species (Barros et al. 2016). Unfortunately, no population genetic study looking at individuals within the assessment region has been undertaken.
While the species is confined to riparian, lacustrine and coastal vegetation and therefore predominantly exists along the coast where habitats may be fragmented, it is also known to be common in other environments, including cultivated areas (see Habitats and Ecology). Therefore, it is possible that the species exists as a single metapopulation – with the dispersal of one individual per generation between potential subpopulations/groups. Assuming no genetically distinct group has gone extinct, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM)– would receive a value of 1.0 (all populations remaining).
While precise population sizes (e.g. census [Nc]) or effective population size (Ne) data does not exist, assumptions that it is stable within the IUCN context suggests that there may be at least 10,000 individuals within the assessment region. Even though the 10 000 value is not an actual census count, this quantity can still be used as a proxy for quantifying the GBF’s headline genetic indicator – proportion of populations with an Ne > 500. Based on the assumption that the species is likely to occur as a single metapopulation, the Ne of the species is ~1000–3000 individuals, based on a Ne/Nc conversion ratio of 0.1-0.3. Having the population above Ne 500, translates to an indicator score of 1.0 (1/1 population with Ne > 500).
Further population genetic analyses are needed to understand the genetic structure and health of this species, and to verify the indicator values estimated broadly here.
Habitats and ecology
The Large Grey Mongoose is essentially associated with mesic associations, with habitats possessing understorey vegetation in riparian, lacustrine and coastal (streams, rivers, marsh, swamps) habitats (Ben-Yaacov & Yom-Tov 1983; Palomares & Delibes 1993a; Angelici et al. 1999). It can, however, be very common locally in heavily grassed and cultivated areas, such as observed by Kingdon (1977) in East Africa and in the KwaZulu-Natal Midlands (Ramesh and Downs, 2015; Streicher et al. 2021) where individuals interestingly avoided heavily forested areas. Although they were not averse to using artificial plantations in KwaZulu-Natal (Ramesh & Downs 2015). It has also been recorded in irrigated and cultivated areas in Morocco. This mongoose generally avoids humid forests and extreme deserts (Delibes 1999), but there are exceptions. For example, in Egypt, Kasparek (1993) reported that the species inhabits desert areas far from the coast. In South Africa, the species broadly occurs in Fynbos, Grasslands, Savanna and Thicket biome vegetation types, but seems to locally prefer cover. The species was reported from Mixed Bushveld in Eswatini (Monadjem 1998), and it can be expected that woodlands will still be important.
Large Grey Mongooses are entirely terrestrial, but they are good swimmers. They can sometimes be observed foraging along pond or dam banks, and in the shallow waters, as Water Mongooses do. Their strong forelimbs and long, curved claws also make them particularly suited to digging for prey. They are opportunistic, omnivorous feeders, and their diet may vary seasonally, regionally, and even between neighbouring family groups (Palomares 1993a). Accordingly, the range of prey is wide and includes small mammals, birds, reptiles and amphibians, fish, crabs, insects and arachnids, gastropods, carrion, fungi, fruit and other plant material (Rowe-Rowe 1978; Smithers & Wilson 1979; Stuart 1983; Palomares 1993a; Angelici 2000). Small mammals such as Otomys spp., Rhabdomys pumilio and Mastomys spp. have locally been shown to dominate the diet. In Spain, young European Rabbits (Oryctolagus cuniculus) may locally constitute the main prey (Delibes 1976), while in Israel Ben-Yaacov and Yom-Tov (1993) found that they mainly fed on poultry and rats. Large prey are killed by a neck bite and small prey by a head bite (Estes 1991).
The Large Grey Mongoose is generally diurnal, with peaks of activity in the morning and late afternoon (Palomares & Delibes 1992b; Maddock & Perrin 1993; Streicher et al. unpubl. data). During the night it sleeps singly or in family groups in underground dens or dense thickets (Palomares & Delibes 1993b). The species is crepuscular in Israel (Ben-Yaacov & Yom-Tov 1983) and, according to Palomares (2013), nocturnal activity may take place where individuals are subject to human disturbance. Most of the active time is spent foraging, while travelling and social interactions only occupy a minimum of time in the day (Palomares & Delibes 1992b, 1993c). Foraging is characterised by intense prey searching. Animals walk with the nose close to the ground, inspect every small hole, frequently excavating and sniffing around bushes and shrubs, and investigating larger burrows (Palomares 2013).
This species is predominantly solitary, although it is not rare to observe groups of one to four adult individuals – up to two or three females and one male – with their young (Maddock & Perrin 1993; Ben-Yaacov & Yom-Tov 1983; Palomares & Delibes 1993d), with even an exceptional record of a pack of 14 individuals in Namibia (Shortridge 1934). Males, however, often spend most of their time alone. Very little information is available on the spatial ecology of this species in the assessment region. In Vernon Crookes Nature Reserve, KwaZulu-Natal, Maddock (1988) found home range sizes of 2.77 km² for a female and 2.59 km² for a male that were tracked for nearly a year or more. More recent studies by Streicher and colleagues (2020) in KwaZulu-Natal Midlands found a home range size of 15.99 km² for a male and an average (± S.E.) home range size of 5.76 km² ± 2.93 for four females. In residential suburbs of KwaZulu-Natal Streicher et al. (unpubl. data) found the home range of a female large grey mongoose to be 0.73 km² in size. In southwestern Spain, average home range sizes vary between 2.8 and 3.5 km² for young and adults (sex combined), respectively (Palomares 1994). There, males are territorial over their entire home range, while females are only territorial in their core areas. In the KwaZulu-Natal Midlands, however, Streicher et al. (2020) found considerable home range and core area overlap between females. Body mass plays an important role in this species’ spatial organisation and dominance relationships, with heavier females accessing richer food patches and heavier males encompassing more female home ranges in their territories (Palomares 1993b, 1994).
Large Grey Mongooses communicate both through olfaction and vocalisation. Temporary latrines – often found in open microsites near or inside thickets or next to dens – may be located within core areas (Palomares 1993c), while more permanent ones may be used to mark home ranges (Ben-Yaacov & Yom-Tov 1983). Individuals may also use perianal gland secretions to mark the ground, large stones or rock edges. Five different calls have been recorded in free-ranging individuals in Spain, and these were related to contact, alarm, aggression, intimidation and pain (Palomares 1991). The most frequently repeated is the contact call, which is a short duration call emitted repeatedly by every member of the group to maintain contact during foraging. The alarm call is a deep, sharp growl.
Little information is available on reproductive biology in the assessment region. In Africa, births have been recorded almost throughout the year (review in Palomares 2013). An average of 2.7 to 3.3 young (with a range of one to four) (Ben-Yaacov & Yom-Tov 1983; Palomares & Delibes 1992a) are born after a gestation period of 9–10 weeks. Normally females give birth to a single litter per year, but captive females breed again if they lose their cubs. Postnatal physical and behavioural development has been studied by Ben-Yaacov & Yom-Tov (1983). This species can live up to 13 years in captivity (Kingdon 1977).
Ecosystem and cultural services: Like other small carnivores, the Large Grey Mongoose may predate on pest species, such as rodents (including rats and mice in human-dominated habitats) and insects. In North Africa, this species is often protected by local people because it is valued as a predator of snakes (F. Cuzin & K. de Smet pers. comm. 2007). In fact, the ancient Egyptians kept these mongooses as pets, presumably to control rodents and snakes (Maddock 1997); hence the other name given to this species, Egyptian Mongoose. The species is suggested to be investigated in its role as a possible bioindicator for wetland status (Swanepoel et al. 2018).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.2. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent/Irregular Rivers/Streams/Creeks | – | Suitable | – |
| 5.3. Wetlands (inland) -> Wetlands (inland) – Shrub Dominated Wetlands | – | Suitable | – |
| 5.4. Wetlands (inland) -> Wetlands (inland) – Bogs, Marshes, Swamps, Fens, Peatlands | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
To our knowledge this species is not harvested or traded in a significant way in the assessment region.
In Gauteng, at the faraday market, some parts were found of the species, however, this was a low prevalence (Whiting et al. 2013). In the Eastern Cape, 15% of herbalist shops sold body parts of the species (Simelane and Kerley 1998). The species is used in the Western Cape where their skin is used in the Xhosa culture for protection against evil spirits (Nieman 2018). However, these uses are not significant and are not known to threaten the species.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Little | – | – | – |
National Commercial Value: No
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Very low likely
Threats
There are currently no major threats to the species in the assessment region. There is mention of their use in body parts trade for medicinal use (Simelane and Kerley 1998; Whiting et al. 2013; Nieman 2018), though it is unlikely that this can constitute a high threat at least at the present, though it may be seen as an emerging, hitherto, not acknowledged threat.
Like most small carnivores the Large Grey Mongoose occasionally falls victim to road traffic collisions (W. Collinson unpubl. data; see Ben-Yaacov & Yom-Tov 1983 for Israel). On farmland it might also accidentally be poisoned by carcasses set out for damage-causing predators such as Black-backed Jackals or Caracals. As this species is closely associated with riverine and wetland vegetation in a large part of its range, the loss of the corresponding habitats may potentially result in localised declines. The drainage of swamplands for conversion to arable land may, for example, constitute a local threat.
Conservation
The Large Grey Mongoose is listed on Appendix III of the Bern Convention, and Annex V of the European Union (EU) Habitats and Species Directive (Delibes 1999). In the assessment region, this species is present in many protected areas, including De Hoop Nature Reserve, Garden Route National Park, uKhahlamba Drakensberg Park, and Kruger National Park. Because it is often associated with riparian habitats and wetlands though, we recommend monitoring in areas where development may be affecting water supply and/or quality, to determine potential negative impacts. It seems the species is relatively tolerant of modified or disturbed habitats within the assessment region (Ramesh & Downs 2015).
As a precautionary measure, and in line with recommendations for other water-dependent species, water management practices, especially outside protected areas, should be carefully planned to avoid negatively impacting riverine ecosystems and wetlands. The most important interventions for this species are those that conserve watersheds and riparian valleys. There is a need to enforce the National Water Act (No. 36 of 1998) and to ensure that the tools provided for in this act – for example, ecological reserve determination and resource quality objectives – are applied to protect our freshwater ecosystems.
Recommendations for land managers and practitioners:
- As a general measure, maintain and improve the ecological integrity of river systems and wetlands (i.e. conservative stocking rates of animals in camps with stream/river frontage)
Research priorities:
- Monitoring Large Grey Mongoose subpopulation trends and measuring the impact – if any – of water quality and quantity deterioration in areas where development takes place.
- General studies on the biology and ecology of this species in different habitat types, including genetic studies
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Report illegal land- and water-use practices to the authorities.
- Protect sensitive riparian areas.
Bibliography
Angelici FM. 2000. Food habits and resource partitioning of carnivores (Herpestidae, Viverridae) in the rainforests of southeastern Nigeria: preliminary results. Revue d’Ecologie (Terre et Vie) 55: 67–76.
Angelici, F. M., Luiselli, L. and Politano, E. 1999. Distribution and habitat of selected carnivores (Herpestidae, Mustelidae, Viverridae) in the rainforests of southeastern Nigeria. Zeitschrift für Säugetierkunde 64: 116-120.
Barchan D, Kachalsky S, Neumann D, Vogel Z, Ovadia M, Kochva E, Fuchs S. 1992. How the mongoose can fight the snake: the binding site of the mongoose acetylcholine receptor. National Academy of Sciences of the United States of America 89: 7717–7721.
Barros, T., Carvalho, J., Pereira M.J.R., Ferreira, J.P. and Fonseca, C. 2015. Following the trail: factors underlying the sudden expansion of the Egyptian Mongoose (Herpestes ichneumon) in Portugal. PLoS ONE 10: e0133768.
Barros T, Ferreira E, Rocha RG, Gaubert P, Bandeira V, Souto L, Mira A, Fonseca C. 2016. Genetic signature of the northward expansion of the Egyptian mongoose Herpestes ichneumon (Herpestidae) in the Iberian Peninsula. Biological Journal of the Linnean Society 118(3): 686–697.
Ben-Yaacov R, Yom-Tov Y. 1983. On the biology of the Egyptian Mongoose, Herpestes ichneumon, in Israel. Zeitschrift für Säugetierkunde 48: 34–45.
Boukhdoud, L., Parker, L.D., McInerney, N.R., Saliba, C., Kahale, R., Maldonado, J.E. and Bou Dagher Kharrat, M. 2021. First mitochondrial genome of the Egyptian mongoose Herpestes ichneumon (Carnivora, Herpestidae). Mitochondrial DNA Part B, 6(2), pp.624-626.
Caro TM, Stoner CJ. 2003. The potential for interspecific competition among African carnivores. Biological Conservation 110(1): 67–75.
Delibes M. 1976. Datos sobre la alimentación del meloncillo, Herpestes ichneumon widdringtoni Gray, 1842, en España. Säugetierkundliche Mitteilungen 24: 38–42.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and
Estes, R.D. 1991. The Behavior Guide to African Mammals: including Hoofed Mammals, Carnivores and Primates. University of California Press, Berkeley and Los Angeles, California, USA.
Goodman, S. 2012. Les Carnivora de Madagascar. Association Vahatra, Antananarivo, Madagascar.
Haltenorth, T. and Diller, H. 1980. A field guide to the mammals of Africa including Madagascar. Collins, London, UK.
Hendrichs, H. 1972. Beobachtungen und Untersuchungen zur Ökologie und Ethologie, insbesondere zur sozialen Organisation ostafrikanischer Säugetiere. Zeitschrift für Tierpsychologie 30: 146-189.
Kasparek M. 1993. The Egyptian Mongoose, Herpestes ichneumon, in Western Egypt. Zoology in the Middle East 9: 31–32.
Kingdon, J. 1977. East African Mammals: An Atlas of Evolution in Africa. Volume IIIA (Carnivores). Academic Press, London, UK.
Maddock AH, Perrin MR. 1993. Spatial and temporal ecology of an assemblage of viverrids in Natal, South Africa. Journal of Zoology 229: 277–287.
Maddock AH. 1997. Large grey mongoose Herpestes ichneumon. In: Mills G, Hes L. (ed.), The Complete Book of Southern African Mammals., Struik, Cape Town, South Africa.
Maddock, A.H. 1988. Resource partitioning in a viverrid assemblage. Ph.D. Thesis, University of Natal.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of Southern African Mammals. Monograph number 5. Transvaal Museum, Pretoria, South Africa.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe, 41(2), pp.45-59.
Nel JL, Roux DJ, Maree G, Kleynhans CJ, Moolman J, Reyers B, Rouget M, Cowling RM. 2007. Rivers in peril inside and outside protected areas: a systematic approach to conservation assessment of river ecosystems. Diversity and Distributions 13: 341-352.
Ngatia, D.K., Webala, P.W., Mware, M.J., Butynski, T.M., de Jong, Y.A. and Ferguson, A.W. 2021. Biogeography of the Egyptian mongoose Herpestes ichneumon (Linnaeus, 1758) in Africa, with first records for Laikipia County, central Kenya. African Journal of Ecology, 59(2), pp.359-369.
Nieman, W.A. 2018. Culture, conflict and cuisine: a quantitative assessment of terrestrial vertebrate off-take at the human-wildlife interface (Doctoral dissertation, Stellenbosch: Stellenbosch University).
Pallett J, Thomson G. 2022. A conservation assessment of Large Grey Mongoose Herpestes ichneumon. In: NCE, LCMAN, MEFT (eds) 2022. Conservation Status and Red List of the Terrestrial Carnivores of Namibia. Pp 134-135. MEFT, LCMAN & NCE, Windhoek, Namibia
Palomares F, Delibes M. 1992a. Some physical and population characteristics of Egyptian mongooses (Herpestes ichneumon L., 1758) in southwestern Spain. Zeitschrift für Säugetierkunde 57: 94–99.
Palomares F, Delibes M. 1992b. Circadian activity patterns of free-ranging large gray mongooses, Herpestes ichneumon, in southwestern Spain. Journal of Mammalogy 73: 173–177.
Palomares F, Delibes M. 1993a. Key habitats for Egyptian mongooses in Doñana National Park, south-western Spain. Journal of Applied Ecology 30: 752–758.
Palomares F, Delibes M. 1993b. Resting ecology and behaviour of Egyptian mongooses (Herpestes ichneumon) in southwestern Spain. Journal of Zoology 230: 557–566.
Palomares F, Delibes M. 1993c. Determining activity types and budgets from movement speed of radio-marked mongooses. Journal of Wildlife Management 57: 164–167.
Palomares F, Delibes M. 1993d. Social organization in the Egyptian mongoose: group size, spatial behaviour and inter-individual contacts in adults. Animal Behaviour 45: 917–925.
Palomares F, Ferreras P, Fedriani JM, Delibes M. 1996. Spatial relationships between Iberian lynx and other carnivores in an area of southwestern Spain. Journal of Applied Ecology 33: 5–13.
Palomares F. 1991. Vocalizations emitted by the Egyptian mongoose, Herpestes ichneumon, living in the wild. Mammalia 55: 148–150.
Palomares F. 1993a. Opportunistic feeding of the Egyptian mongoose, Herpestes ichneumon (L.) in southwestern Spain. Revue d’Ecologie (Terre et Vie) 48(295–304).
Palomares F. 1993c. Faecal marking behaviour by free-ranging common genets Genetta genetta and Egyptian mongooses Herpestes ichneumon in southwestern Spain. Zeitschriftt für Säugetierkunde 58: 225–231.
Palomares F. 1994. Site fidelity and effects of body mass on home-range size of Egyptian mongoose. Canadian Journal of Zoology 72: 465–469.
Palomares F. 2003b. Individual variations of male mating tactics in Egyptian mongooses (Herpestes ichneumon): can body mass explain the differences? Mammalia 57: 317–324.
Palomares, F. 2013. Herpestes ichneumon Egyptian Mongoose (Ichneumon). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 306-310. Bloomsbury, London, UK.
Ramesh, T, Downs, CT. 2015. Impact of land use on occupancy and abundance of terrestrial mammals in the Drakensberg Midlands, South Africa. Journal for Nature Conservation 23: 9—18.
Rowe-Rowe DT. 1978. The small carnivores of Natal. Lammergeyer 25: 1–48.
Shortridge, G.C. 1934. The mammals of South West Africa: a biological account of the forms occurring in that region. Heinemann, London, UK.
Simelane, T.S. and Kerley, G.I.H. 1998. Conservation implications of the use of vertebrates by Xhosa traditional healers in South Africa. South African Journal of Wildlife Research-24-month delayed open access, 28(4), pp.121-126.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skowno, A.L., Poole, C.J., Raimondo, D.C., Sink, K.J., Van Deventer, H., Van Niekerk, L., Harris, L.R., Smith Adao, L.B., Tolley, K.A., Zengeya, T.A., Foden, W.B., Midgley, G.F. & Driver, A. 2019. National Biodiversity Assessment 2018: The status of South Africa’s ecosystems and biodiversity. Synthesis Report. South African National Biodiversity Institute, an entity of the Department of Environment, Forestry and Fisheries, Pretoria. pp. 1–214.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Streicher JP, Ramesh T, Downs CT. 2020. Home range and core area utilisation of three co-existing mongoose species: large grey, water and white-tailed in the fragmented landscape of the KwaZulu-Natal Midlands, South Africa. Mammalian Biology 100(3):273—283.
Stuart CT. 1983. Food of the large grey mongoose Herpestes ichneumon in the south-west Cape Province. South African Journal of Zoology 18: 401–403.
Stuart CT. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1–58.
Swanepoel LH, Loock D, Matthews WS, Emslie KW. 2018. Expanding the geographical distribution of the Egyptian Mongoose, Herpestes ichneumon (Linnaeus, 1758), in South Africa. Check List 14(4): 595–599.
Whiting, M.J., Williams, V.L. and Hibbitts, T.J. 2013. Animals traded for traditional medicine at the Faraday market in South Africa: species diversity and conservation implications. Animals in traditional folk medicine: implications for conservation, pp.421-473.