Klipspringer
Oreotragus oreotragus

2025 Red list status
Least Concern
Regional Population Trend
Stable
change compared
to 2016
No Change
Overview
Oreotragus oreotragus – (Zimmermann, 1783)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Oreotragus – oreotragus
Common Names: Klipspringer (English & Afrikaans), Klipbokkie (Afrikaans), Igogo (Ndebele, Zulu), Kololo (Sepedi, Setswana), Kome (Sepedi, Sesotho), Mokabaowane, Mokabaeyane (Setswana), Ngururu (Shona), Ligoka, Inyamatane, Ligoga (Swati), Ngululu, Xemi (Tsonga) Klippspringer (German), Oréotrague (French)
Synonyms: Antilope oreotragus Zimmermann, 1783
Taxonomic Note:
Eleven subspecies of Klipspringer were described by Ansell (1972). In southern Africa, the nominate O. o. oreotragus occurs within the Northern, Western and Eastern Cape provinces, probably as far east as the Free State, while O. o. transvaalensis, is located in eastern South Africa, particularly the Limpopo, North West, Mpumalanga and the KwaZulu-Natal provinces of South Africa, and it may extend into southern Mozambique. O. o. tyleri occurs in Angola and Namibia, while O. o. stevensoni is found in Zimbabwe and Botswana (Ansell 1972). The precise geographical limits of these subspecies’ distributions remains unclear. Further taxonomic resolution is needed.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Patel, T.1 & da Silva, J.2
Reviewer: Druce, D.3
Institutions: 1Endangered Wildlife Trust; 2South African National Biodiversity Institute; 3Welgevonden Game Reserve
Previous Assessors: Birss, C., Peel, M., Power, J. & Relton, C.
Previous Reviewer: Child, M.F.
Previous Contributors: Stalmans, M. & Avenant, N.
Assessment Rationale
This is a widely distributed species with no signs of significant habitat loss, as its habitats are inaccessible and remote. There are many subpopulations in formally protected areas and private land and the overall population within the assessment region is assumed to be stable as there is no evidence for decline. However, anecdotal reports suggest local declines from illegal hunting. This threat should be quantified to assess its severity. Recent records are available across a range of protected areas throughout its range, and it can occur at densities of 0.01–0.3 individuals / km². It should continue to survive in substantial numbers in extensive, inaccessible areas outside of protected areas, where habitats such as koppies, cliffs and hillsides preside. Thus, this species remains Least Concern.
Regional population effects: There are numerous routes for dispersal into the assessment region. For example, along the Lebombo Mountains through Mozambique into northeastern KwaZulu-Natal, across the Limpopo in the Mapungubwe area in northern Limpopo, as well as across the Richtersveld in the Northern Cape linking the mountain chains along the Namibian escarpment. The habitat for this species is thus mostly continuous along mountain ranges. Rescue effects are assumed to be possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Patel T & da Silva JM. 2025. A conservation assessment of Oreotragus oreotragus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has a widespread, yet patchy distribution from the southwestern limits of South Africa to northeast Africa, including southern Sudan, Eritrea, Somalia, Uganda, Rwanda, Kenya, Tanzania, Zambia, Zimbabwe, Malawi and the Ethiopian Highlands, where they occur at up to 4,380 m asl (Yalden et al. 1996; Skinner & Chimimba 2005). Additionally, their range extends along the southwest of Africa through Namibia and the southwestern regions of Angola. Isolated populations of Klipspringer are located in the Central African Republic, Nigeria and the Democratic Republic of the Congo (East 1999; Nicholas 2004; Roberts 2013). In all likelihood, the Klipspringer is now extinct in Burundi. In Mozambique, anecdotal reports suggest they occur east of the Limpopo River north of Mabalane along the main north-south road but have not recently been seen in Gorongosa National Park (M. Stalmans pers. comm. 2016).
Within the assessment region, they occur throughout most montane regions of South Africa and are largely present within their former range. However, historical evidence suggests a range reduction in certain areas, such as in regions of the Eastern Cape Province (Lloyd & Millar 1983; Lynch 1989), Free State Province (Lynch 1983; Avenant 1997, 2000), and Lesotho, where Lynch (1994) predicted them to occur, but repeated field surveys between 1996 and 2014 have not detected them (N. Avenant unpubl. data), the closest record being Cathedral Peak on the KwaZulu-Natal side of the Drakensberg Mountains. Corroborating this, 95–100% of respondents (cattle herders in Lesotho) from questionnaire data stated that Klipspringer were no longer found in their areas (Avenant 2004). There have also been no recent records (2016-current) of the species in Lesotho.
Their range is extensive across the Limpopo Province, extending into the northern and eastern regions of both the North West and northeastern Mpumalanga provinces respectively and into the Maluti Mountains (du Plessis 1969). Within KwaZulu-Natal, they occur above 2,400 m asl in the Drakensberg, along the escarpment from Giant’s Castle northwards, and have a scattered, isolated distribution across small areas of the remainder of the province, including Ithala and uMkhuze Game Reserves, Weenen Nature Reserve, as well as on a number of isolated private farms. Similarly, this species has a patchy distribution in the Free State with isolated historical records at Boshoff, Bethlehem, Colesberg, Philipolis and Leribe in Lesotho (near Ficksburg, Free State Province) (du Plessis 1969; Skinner & Chimimba 2005; Boshoff & Kerley 2013). Their range is widespread across the mountainous regions of the Western Cape, extending to the Middelburg, Graaff-Reinet, Grahamstown and Uitenhage regions of the Eastern Cape (Skinner & Chimimba 2005; Skead 2007) and into the Northern Cape to Namibia and along the Orange River to Upington, with patchy distribution in the Carnavon and Beaufort West regions (Estes 1991; Skinner & Chimimba 2005; Skead 2011). While they naturally occur in the bushveld of the Magaliesberg and Swartruggens rocky hills of North West Province (Power 2014), records from the southern parts of the province are considered introductions.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
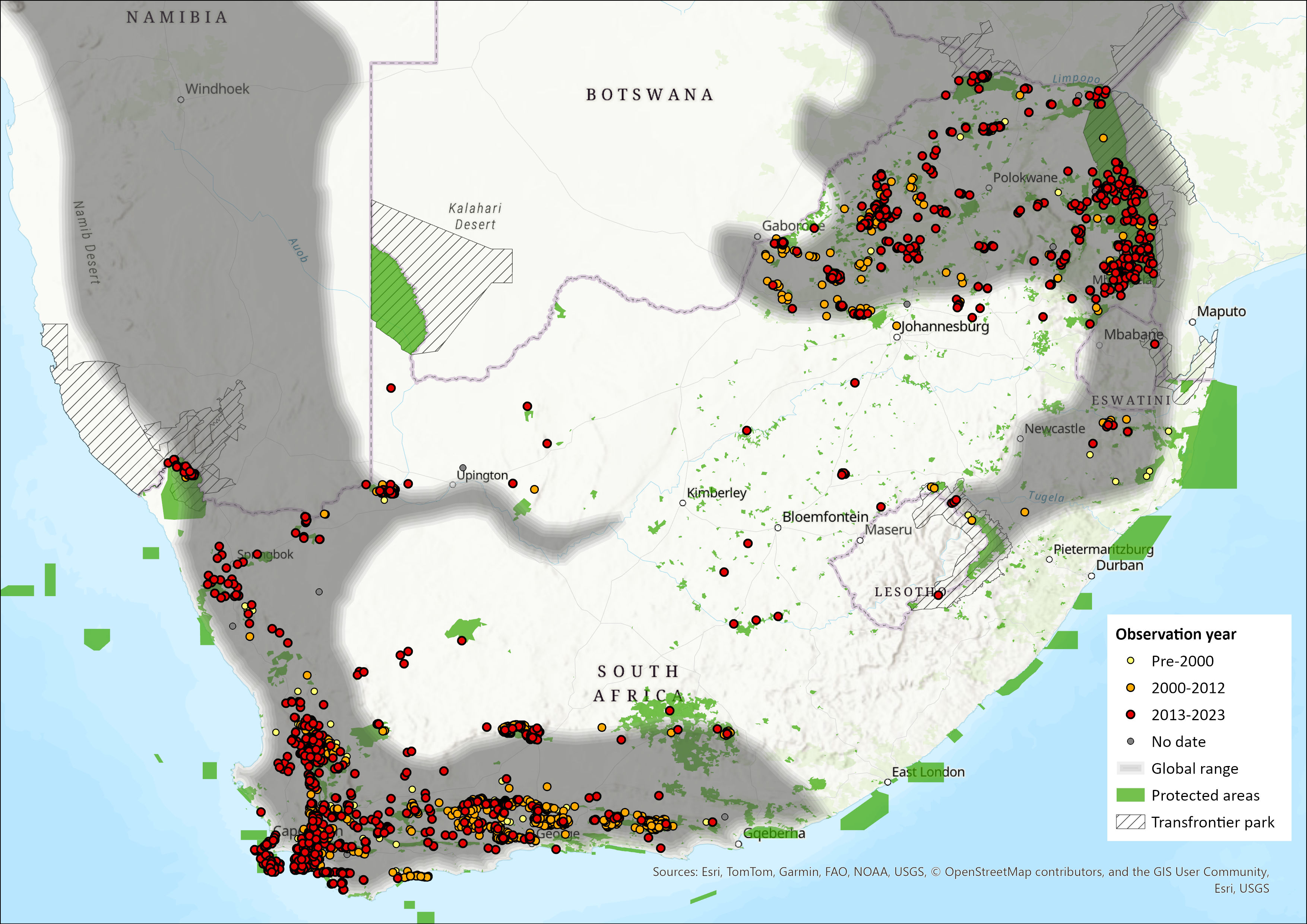
Map
Click on map to view/enlarge
Figure 1. Distribution records for Klipspringer (Oreotragus oreotragus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Possibly Extinct | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Presence Uncertain | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change leading to the loss of forage resources may become an increasing threat to this species, especially in the western areas of the assessment region, due to an increase in the frequency and severity of drought (Erasmus et al. 2002), as well as range contraction and alteration of forage resources (Midgley et al. 2002). However, the mountainous regions in which this species inhabits may be refuges from climate change. Further research is needed to assess the net effect.
Population information
This species is limited to rocky habitats, which is more than often discontinuous across the environment, leading to average population densities of between 0.01–0.3 per km² in protected areas, where it is considered common, but restricted to suitable habitats. However, within continuous regions of suitable habitat, Klipspringer densities can reach fairly high population densities. For example, across a 9.6 km² area of escarpment in Ethiopia, Klipspringer densities reached 10–14 individuals / km². East (1999) described population densities of between 0.15 and 0.30 individuals / km² in Lilongwe (Malawi), the Karoo, Mountain Zebra and Royal Natal National Parks, as well as Giant’s Castle Game Reserve. The density in the Welgevonden Game Reserve in the Limpopo Province is 0.04 individual / km2 but this is considered an underestimate, and the densities are in all probability much higher (D. Druce pers comm. 2025). Densities in the Lydenburg area, Mpumalanga Province reach 0.3 individuals / km² (M. Peel unpubl. data). These regions are considered to contain fairly extensive areas of Klipspringer habitat. A conservative global population estimate of 42,000 individuals was projected by East (1999). Overall, the population is considered stable in protected areas and on private lands, but is likely to be decreasing in regions where small, disconnected populations are threatened by unregulated hunting and competition for resources with livestock. Provincial protected areas in the Western Cape Province do not have trend data for the species but the subpopulations in the Boland Mountain Complex are considered to be declining (C. Birss unpubl. data). However, Klipspringer are considered to persist with stable populations throughout the rest of their range in the Western Cape Province, including on private land (C. Birss pers. comm. 2016).
Within the assessment region, this species is well protected and its habitat is largely continuous. Thus, we suspect that there are over 10,000 mature individuals within the assessment region (data from 2016 assessment). Additionally, the conversion from livestock to wildlife ranching and game farming may be conserving and connecting the koppies that this species inhabits, so its net population trend may well be increasing, but this remains to be quantified. Finally, rocky habitats are unlikely to be transformed for expanding agriculture and settlements, though the quarrying of rocky outcrops for granite mining is a conceivable threat should this proliferate.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Current population trend: Stable
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A phylogeographic study incorporating nine of the 11 subspecies of Klipspringer to investigate the phylogenetic and population genetic associations within the species found no genetic basis for subspecies classifications (Le Roex, 2008). High levels of genetic variation were observed for the species as a whole, likely suggesting large historical population sizes. While support was not given to subspecies designations, genetically distinct geographic structure was uncovered, clustering individuals into two major groups, a southwestern African group and an eastern and northern group. These two groups can be considered Evolutionarily Significant Units (ESUs). Further substructure was observed within the southwestern African group as a result of shifting climatic conditions in the late Pliocene/early Pleistocene, after which secondary contact occurred and Klipspringer moved upwards from southern Africa to colonise the east (Le Roex, 2008). Animals within the assessment region fall within both ESUs: individuals from the Western Cape and Northern Cape fall within the southwestern group; while animals from Gauteng and KwaZulu-Natal (and stretching all the way to Ethiopia) fall within the eastern/northern group.
More fine-scale population genetic studies would be beneficial to gain further insight into additional, more current, substructuring and the effective population sizes (Ne) of each distinct cluster. However, Ne may be inferred from the estimated population size for the region, which exceeds 10,000. If this is crudely divided into the two ESUs, each would contain over 5,000 mature individuals. Applying an Ne/Nc conversion ratio of 0.1-0.3, the Ne for each population would be approximately 500-1,500 individuals.
Habitats and ecology
This species is closely associated with steep rocky and mountainous habitats, including granite outcrops, koppies and gorges with rocky embankments where it can find refuge from predators and an adequate supply of food (Estes 1991). The African Rift Valley and the southern African escarpment support vast areas of appropriate habitat and form the core parts of its pan-African distribution. They are often found at the base of rocky outcrops (Wilson & Child 1965; Rautenbach 1982; M. Peel pers. obs. 2016) and may occasionally move away from these outcrops to browse and have been known to traverse up to 10 km between rocky habitats (Norton 1997), which is suggestive of their dispersal capabilities. There is a subpopulation within the suburbs of Nelspruit, Mpumalanga Province (M. Peel pers. obs. 2016).
The Klipspringer is highly adapted to rocky habitats and is able to move with efficiency and agility over loose rocky terrain, and low-structured granite boulders. This maneuverability is due to its body size, and the anatomy of its foot structure (posterior rotation of the proximal inter-phalangeal joint), which enables the Klipspringer to walk on the very tips of its hooves, wearing them down into a cylindrical shape, and thus providing efficient grip on rocky surfaces (Norton 1980). The Klipspringer’s insulated coat enables it to withstand extreme cold and heat and it is able live at high and low elevations and in areas of high and low rainfall and therefore is considered to have a very adaptable diet (Estes 1991). Home range sizes are known to vary with rainfall (Norton 1980), with home range size contracting with increased rainfall. Dunbar (1978) noted a territory of 8 ha in a high rainfall area (1,300 mm), while Norton (1980) found territory sizes of 49 ha and 15 ha in Springbok (160 mm) and Gamkaberg Nature Reserves, near Oudtshoorn (400 mm), respectively. It is almost exclusively a concentrate browser, Klipspringer will only feed on grass under certain conditions (Dunbar & Dunbar 1974) and are highly selective of particular plant parts (Norton 1984). For example, Norton (1984) described Klipspringer feeding on the growing shoots, fruits and flowers of a variety of shrubs and herbs, but when availability decreases, they will browse on the leaves and steams of these species (Norton 1984). Wilson and Child (1965), who assessed the 74 samples of Klipspringer stomach contents in Zambia revealed that 90% comprised of browse material, including leaves, berries, seedpods, fruit and flowers; with seasonal preferences for plants such as Euphoria tirucalli and Vellozia equisetifolia.
Klipspringers are small ungulates, with adult females (13 kg) slightly larger than males (10 kg) (Skinner & Chimimba 2005). Often, males and females will form long-lasting pair bonds, and both will exhibit sedentary, territorial behaviour (Norton 1997), although small, temporary groups of up to six individuals may assemble in favourable feeding sites (Skinner & Smithers 1990), this is more the exception. When foraging, one of the adults will feed, while the others remains vigilant, watching for both terrestrial and aerial predators. Males spend more time in anti-predator vigilance behaviour, compared to females, which enables females to concentrate a greater proportion of their energy on reproduction (Norton 1980). When disturbed, the vigilant adult produces a warning alarm call to allow the other adult and young time to retreat a short distance to the safety of rocky, sheltered habitats, before turning to face the source of threat (Norton 1997). This behaviour allows for the conservation of energy, in the case of a false alarm, or if the predator is not hunting (Norton 1980).
Rocky, mountainous habitats usually experience enhanced climatic extremes compared to lowland areas, due to altitude, aspect, wind exposure, terrestrial radiation and shelter. Thus, Klipspringer have evolved a remarkably thick and coarse pelage, with quill-like hairs, which functions as an effective insulator both against the cold and radiated heat, as well as to prevent water loss. Additionally, Klipspringer are able to survive independently from water, though they will drink when it is available, often from nearby streams or waterholes, or alternatively from temporary pools held in rocky depressions.
Norton (1980) noted that in South Africa, Klipspringers are seasonal breeders, with a peak in births between July and September, following a gestation period of approximately 210 days. For the first 2–3 months of life, young Klipspringer are hidden in dense vegetation away from predators (Norton 1980). Young are usually weaned after four or five months, then are subsequently forced away from their mothers into new territories (Norton 1980).
Ecosystem and cultural services: Due to its unique adaptations for living in rocky habitats, and charismatic nature among the antelope species, this species is touted as a flagship species for the conservation of rocky habitats within the mountainous regions of Namaqualand and the Waterberg in Limpopo Province.
This small antelope species forms a valuable prey component of a number of terrestrial and aerial predators, such as Leopard (Panthera pardus), Caracal (Caracal caracal), and to a lesser extent Chacma Baboons (Papio ursinus), Verreaux’s Eagles (Aquila verreauxii) and Martial Eagles (Polemaetus bellicosus).
The fur of this species was once used by early settlers as late as the 19th century for use in the stuffing of saddles.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
2.1. Savanna -> Savanna – Dry |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist |
– |
Suitable |
– |
|
3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude |
– |
Suitable |
– |
|
3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation |
– |
Suitable |
– |
|
6. Rocky areas (eg. inland cliffs, mountain peaks) |
– |
Marginal |
– |
|
8.1. Desert -> Desert – Hot |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Historically this species was hunted for its meat, and coarse pelage (which was often used as a stuffing in saddles) (Norton 1980), leading to its eradication in a number of regions across the assessment region, for example the Cape Peninsula in the 1930s.
Klipspringer are locally used at a subsistence level for food and they have national commercial value for trophy hunting and live animal auctions. These uses, as long as they are well managed, are not expected to cause population declines as they are not utilised to a great extent. Klipspringer are advertised as a cryptic and sought after member of the “Tiny Ten” for hunting purposes, which includes other small antelope, namely the species of duiker (Philantomba monticola, Sylvicapra grimmia, Cephalophus natalensis), grysbok (Raphicerus spp.), Steenbok (Raphicerus campestris), Suni (Neotragus moschatus) and Oribi (Ourebia ourebi), and as a bait-species for hunting Leopard. Klipspringer are fairly easy to hunt, due to their habit of utilising high vantage points for predator surveillance.
Wildlife ranching and the private sector may have generally had a positive effect on this species as it has been widely reintroduced onto private properties within its natural distribution range, although there are also indications that they may also have been introduced to areas beyond their known range (Power 2014). Regulation of translocation is required to prevent mixing of ecotypes. Small-scale translocation is encouraged between areas that may have been connected by an area of similar habitat type, within or adjacent to the bioregion of source, through which Klipspringer would have been able to move freely prior to anthropogenic partitioning of the landscape (Norton 1980). However, translocation of individuals into discretely distributed regions is discouraged, as this may interfere with the natural variability of the species, and its specialised adaptations to local environmental conditions (Norton 1980).
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Venison, skins and live animal trade in game auctions. |
Yes |
Venison (including bait for leopard hunts), trophies and live sales |
National Commercial Value: Yes
International Commercial Value: Yes
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
|
15. Sport hunting/specimen collecting |
– |
– |
true |
– |
Is there harvest from captive/cultivated sources of this species? No
Harvest Trend Comments: Venison (including bait for leopard hunts), trophies and live sales
Threats
No major threats have been identified for this species across its range. However, outside of protected areas, subsistence hunting occurs at a low intensity. For example, localised poaching in the Boland Mountains of the Western Cape are suspected to be responsible for significant declines in that population (C. Birss unpubl. data). Similar local declines may also be occurring in areas of high human settlement. Small, isolated subpopulations are most vulnerable to hunting, and newborn lambs are vulnerable to predation by domestic dogs. These threats seem to be especially severe in Lesotho, where hunting for bushmeat, skins and traditional medicine may have virtually eradicated the species (Avenant 2004; N. Avenant unpubl. data). Resource competition with domestic goats may affect adult Klipspringers, and thus many of subpopulations may have been eradicated from areas in or nearby to settlements.
Current habitat trend: Stable. Much of their habitat is rocky and inaccessible, thus is under no threat of extensive transformation. However, habitat fragmentation and transformation between suitable habitat may limit the dispersal of young Klipspringer into new territories. Hills and koppies are flagged as important areas in provincially mandated Conservation Assessments (for example, Biodiversity Conservation Assessments).
Conservation
This species occurs commonly within a number of formally protected areas within the assessment region. It occurs in lesser numbers in a large number of unprotected areas throughout its range which contain smaller, isolated areas of suitable habitat. Very large numbers are thought to survive on private farmland in Namibia, with strong potential for dispersal into South Africa – given that the Orange River can be forded. Klipspringer offer comparatively little competition to domestic livestock, and are thus commonly reintroduced onto private farmland. Although no direct conservation interventions are necessary at present, reintroductions into areas of its former natural range will help to restore a functioning population.
For example, during the 1930s, Klipspringer were substantially overexploited in the Western Cape, leading to their local extinction in the Cape Peninsula. In 1999, Table Mountain National Park (TMNP), Cape Nature and SANParks managed the successful reintroduction of 19 Klipspringer into the Cape of Good Hope section of TMNP, and later reintroductions were conducted in 2003 and 2005 where 9 and 10 additional animals were translocated directly onto Table Mountain, respectively. The individuals were sourced from three subpopulations at separate localities in the Western Cape. Routine Klipspringer counts take place across TMNP and populations are currently stable (Ferreira et al. 2010).
However, translocations should be considered carefully. Reintroduction of Klipspringer depends significantly on habitat suitability, as Klipspringer depend on the presence of rocky substrate, which provides them security from predators (Norton 1980) and vantage points. Norton (1980) recommends that equal numbers of male and female are translocated, due to their monogamous behaviour. Considering that this species has adapted so specifically to rocky terrain it is unlikely that they would be able to acclimate to non-rocky areas, even if they are not threatened by predators. The availability of suitable vegetation coverage and edible shrubs within the rocky habitat are also essential for successful Klipspringer populations. Under stressful conditions this species may utilise trees and grasses, but these do not make up their dietary preferences. Furthermore, this species has an extensive distribution, which exhibits clinal variation throughout its range and, as stated in the taxonomy notes, two subspecies are recognised in South Africa. In species exhibiting eco-typical variation across their range, ecotypes may be endemic and the maintenance of genetic diversity could be threatened by injudicious mixing of ecotypes (Birss & Palmer 2012). In the Western Cape Province, translocations of these species are managed and regulated through permits with specific consideration of the potential impact on the maintenance of genetic diversity. As such, facilitated movement of this species should be limited to within or adjacent to their bioregional boundaries.
Recommendations for land managers and practitioners:
- Develop translocation regulations, which ensures the maintenance of both genetic viability and local adaptations.
- Provide incentives for landowners to create conservancies where the benefits of this species are shared.
Research priorities:
- Current population trend estimates and distribution.
- Research into the management sustainability of this species on protected areas and wildlife ranches, and its species-specific value in the wildlife economy.
- Research on the effect of climate change on this species.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalistt and MammalMAP), especially outside protected areas.
- Landowners should ensure that disturbance of this species and its young is kept to a minimum, particularly with regards to domestic dogs.
Bibliography
Ansell, W.F.H. 1972. Part 2, 15 Family Artiodactyla. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-84. Smithsonian Institution Press, Washington, DC, USA.
Avenant NL. 1997. Mammals recorded in the QwaQwa National Park (1994-1995). Koedoe 40: 31-40.
Avenant NL. 2000. Terrestrial small-mammal diversity in Korannaberg Conservancy, Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 16: 69-82.
Avenant NL. 2004. Conserving Mountain Biodiversity in Southern Lesotho: Mammal report. United Nations Development Plan, Lesotho.
Birss C, Palmer NG. 2012. Mammals in: Western Cape State of Biodiversity 2012. CapeNature Scientific Services, Stellenbosch.
Boshoff AF, Kerley GIH. 2013. Historical incidence of the larger mammals in the Free State Province (South Africa) and Lesotho. Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Dunbar RI, Dunbar EP. 1974. Social organisation and ecology of the Klipspringer (Oreotragus oreotragus) in Ethiopia. Zeitschrift für Tierpsychologie 35: 481-493.
Dunbar, R. I. M. 1978. Competition and niche separation in a high altitude herbivore community in Ethiopia. East African Wildlife Journal 16: 183-199.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Erasmus, B.F.N., van Jaarsveld, A.S., Chown, S.L., Kshatriya, M. and Wessels, K.J. 2002. Vulnerability of South African animal taxa to climate change. Global Change Biology 8: 679-693.
Estes, R. 1991. The Behaviour Guide to African Mammals. University of California Press, Berkeley, USA.
Ferreira S, Gaylard, A, Greaver, C, Herbst M. 2010. Animal abundances in Parks 2009/2010. Scientific Services, SANParks, Skukuza, South Africa.
Le Roex, N. 2008. Phylogeographic analysis reveals strong geographical structuring in the klipspringer, Oreotragus oreotragus. University of Cape Town.
Lloyd PH, Millar JCG. 1983. A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Lynch CD. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Midgley GF, Hannah L, Millar D, Rutherford MC, Powrie LW. 2002. Assessing the vulnerability of species richness to anthropogenic climate change in a biodiversity hotspot. Global Ecology and Biogeography 11: 445-451.
Nicholas, A. 2004. An update on the status of important large mammal species in Gashaka Gumti National Park, Nigeria. Antelope Survey Update 9: 40-42.
Norton P. 1997. Klipspringer Oreotragus oreotragus. In: G. Mills and L. Hes (eds), The Complete Book of southern African Mammals, pp. 264. Struik, Winchester, Cape Town.
Norton PM. 1980. The habitat and feeding ecology of the Klipspringer, Oreotragus oreotragus (Zimmermann, 1983) in two areas of the Cape Province. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Norton, PM. 1984. Food selection by klipspringers in two areas of the Cape Province. South African Journal of Wildlife Research 14: 33-41.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Roberts, S. C. 2013. Oreotragus oreotragus. In: J. S. Kingdon and M. Hoffmann (eds), The Mammals of Africa, pp. 470-476. Bloomsbury, London, UK.
Skead, C.J. 2007. Historical Incidence of the Larger Land Mammals in the broader Eastern Cape.In: Boshoff AF, Kerley GIH, Lloyd PH (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University., Port Elizabeth, South Africa.
Skead, C.J. 2011. Historical incidence of the larger land mammals in the broader Western and Northern Cape provinces.In: Boshoff A.F., Kerley G.I.H, Lloyd P. (ed.), Centre for African Conservation Ecology, Nelson Mandela Metropolitan University, Port Elizabeth.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Skinner, J.D. and Smithers, R.H.N. 1990. The mammals of the southern African subregion. 2nd edition. University of Pretoria, Pretoria, South Africa.
Wilson VJ, Child G. 1965. Notes on klipspringer from tsetse fly control areas in Eastern Zambia. Arnoldia 1: 1-9.
Yalden, D.W., Largen, M.J., Kock, D. and Hillman, J.C. 1996. Catalogue of the mammals of Ethiopia and Eritrea. 7. Revised checklist, zoogeography and conservation. Tropical Zoology 9: 73-164.
du Plessis, SF. 1969. The past and present distribution of the Perissodactyla and Artiodactyla in southern Africa. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa