
Killer Whale
Orcinus orca

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Orcinus orca – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Orcinus – orca
Common Names: Killer Whale, Orca (English), Moordvis (Afrikaans), Epaulard, Orque (French), Espadarte (Spanish; Castilian), Orca assassina (Italian)
Synonyms: Delphinus orca Linnaeus, 1758; Orcinus glacialis (Berzin & Vladimirov, 1983); Orcinus nanus Mikhalev, Ivashin, Savusin & Zelenaya, 1981
Taxonomic Note:
The Killer Whale shows morphological, genetic, ecological, and behavioural differences among geographic regions, and there has been discussion about potential taxonomic revisions. The Committee on Taxonomy of the Society for Marine Mammalogy (www.marinemammalscience.org/species-information/list-marine-mammal-species-subspecies), which is generally regarded as the authority for marine mammal taxonomy, have recognised a single Killer Whale species, Orcinus orca (Linnaeus, 1758), and two unnamed subspecies in the eastern North Pacific (ENP), the ENP resident Killer Whale (O. o. un-named subsp.) and the ENP transient Killer Whale (O. o. un-named subsp.) also known as Bigg’s Killer Whale. The Committee noted, however, “Other forms of Killer Whales in the North Pacific, North Atlantic and Antarctic [Southern] Ocean may warrant recognition as separate subspecies or even species, but the taxonomy has not yet been fully clarified or agreed (Hoelzel et al. 2007; Morin et al. 2010; Foote et al. 2009, 2013; Moura et al. 2014a,b, 2015).” One population (a “distinct population segment”) of the ENP resident subspecies (the “southern resident” population) was listed as Endangered under the Canadian Species at Risk Act in 2003 and the U.S. Endangered Species Act in 2005.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Hoelzel A.R.1, Shabangu, F.2,3, & da Silva, J.4
Reviewer: Vermeulen, E.3
Institutions: 1Durham University, 2Department of Forestry, Fisheries and the Environment, 3The Whale Unit, University of Pretoria, 4South African National Biodiversity Institute,
Previous Assessors: Reisinger, R., de Bruyn, N., Bester, M., Findlay, K.
Previous Reviewer: Child, M.F.
Previous Contributors: Relton, C., Atkins, S., Elwen, S., Meyer, M., Oosthuizen, H. & Plön, S.
Assessment Rationale
This taxonomic unit is treated as one species in this assessment even though it may be more complicated. At least two subpopulations of Killer Whales have been recognised within South African waters, those occurring off mainland South Africa, and those associated with the region around the Prince Edward Islands. While a monitoring programme recently initiated at the Prince Edward Islands presently provides some data for that subpopulation and will provide more complete information (especially demographic parameters) in future, the South African subpopulation is almost entirely unknown. No abundance estimates are available for the entire assessment region, but there are considered to be 20–50 mature individuals occurring around the Prince Edward Islands. Population size, structure and movement of Killer Whales should be determined. There is some indication that the genetic diversity in South African waters is higher than in other regions of the world, but world-wide genetic diversity is generally low, and there is not yet any clear indication of population substructure off South Africa (Moura et al. 2014a; 2015). The acoustic repertoire of Killer Whales can be used to identify different populations as done elsewhere (e.g. Selbmann et al. 2021).
No major threats that could cause rapid decline were identified and there is no reason to believe there are range wide declines. However, the minor threat of competition with the longline fisheries, which may cause occasional, intentional deaths, was identified in both subpopulations. Interactions with longline fisheries should continue to be monitored and ideally a photographic identification catalogue should be established to determine which individuals are depredating. With increasing marine traffic in many parts of the world including the assessment region, underwater noise is a raising threat for killer whales as their acoustic behaviour and call detectability decrease with increasing underwater noise (Shabangu et al. unpublished data).
The species is wide-ranging and common, and its habitat does not seem to be fragmented. Such assumed connectivity, combined with the lack of major identified threats, means we list this species as Least Concern. If taxonomy is resolved, re-assessment at subspecies or subpopulation level may be required.
Regional population effects: The Killer Whale is an extremely wide-ranging pelagic cetacean, with a continuous distribution and no obvious barriers to dispersal, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Hoelzel AR, Shabangu F & da Silva JM. 2025. A conservation assessment of Orcinus orca. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Killer Whale is a well-known cosmopolitan species, and with the exception of humans, may be the most widespread mammal across the globe (Heyning & Dahlheim 1988; Rice 1998). In general, this species occurs most commonly within high productivity and temperate nearshore regions (Forney & Wade 2006), however they are not restricted by water temperature or depth.
The species ranges across the entire assessment region from Kosi Bay to the Orange River mouth, and around the Prince Edward Islands, across all water depths (Best 2007). Sightings from the Durban whaling grounds suggest peak occurrence in June and October, and Elephant Seal (Mirounga leonina) remains in the stomach contents of some individuals captured there indicate movement from sub-Antarctic waters (Findlay 1989; Findlay et al. 1992; Best et al. 2010), which is also suggested by satellite tracking (Reisinger et al. 2015). Together these may indicate seasonal or transient occurrence of at least some individuals. Around the Prince Edward Islands, Killer Whale calls are intermittently detected year-round with high call number in autumn through spring (Shabangu et al. 2024).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): 452
Depth Upper Limit (in metres below sea level): 0
Depth Zone: Mesopelagic
Map
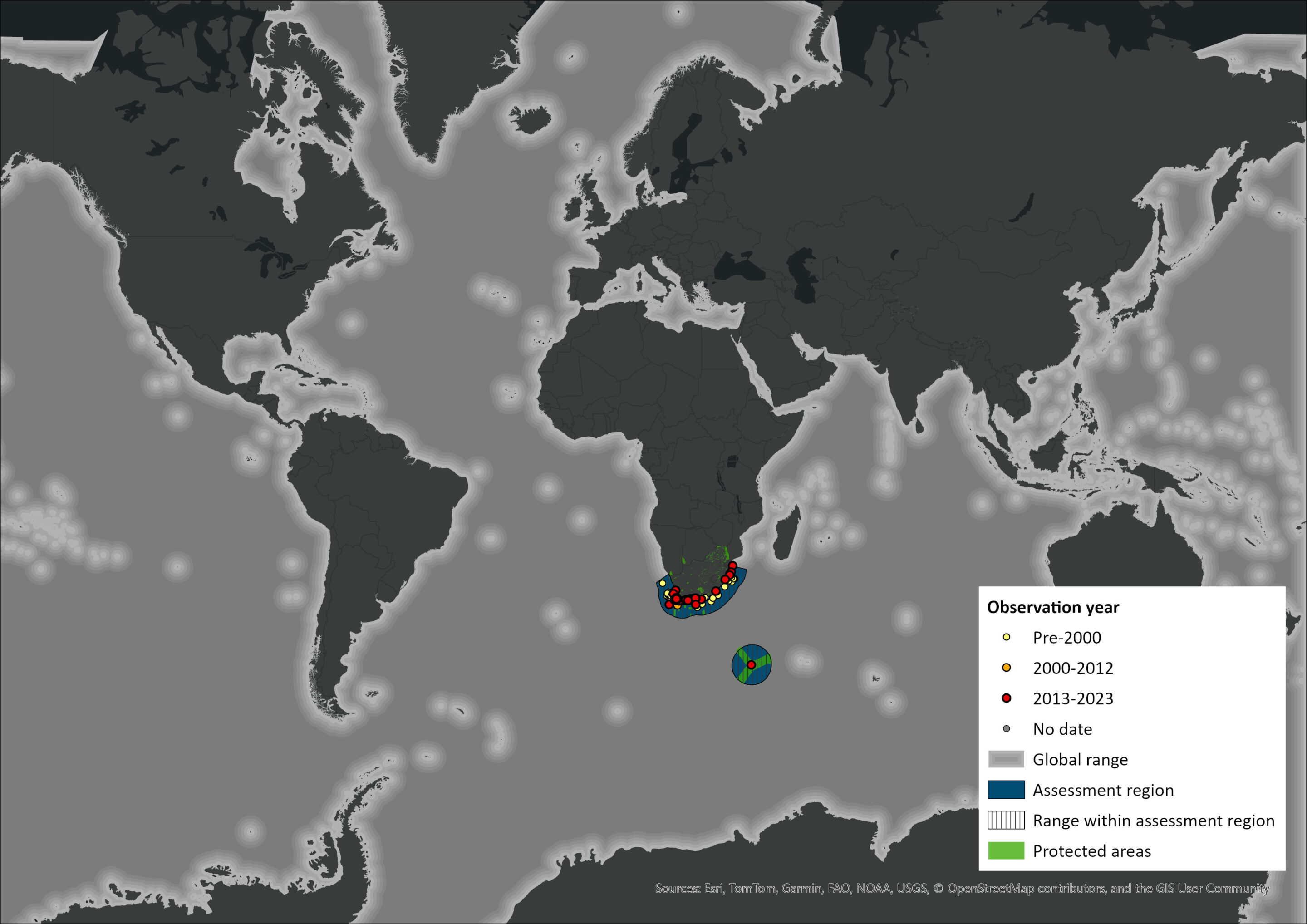 Figure 1. Distribution records for Killer Whale (Orcinus orca) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Killer Whale (Orcinus orca) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Antarctic, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | – |
| American Samoa | Extant | Native | – | – |
| Angola | Presence Uncertain | Native | – | – |
| Anguilla | Extant | Native | – | – |
| Antarctica | Extant | Native | – | – |
| Antigua and Barbuda | Extant | Native | – | – |
| Argentina | Extant | Native | – | – |
| Aruba | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Bahamas | Extant | Native | – | – |
| Bahrain | Presence Uncertain | Native | – | – |
| Bangladesh | Extant | Native | – | – |
| Barbados | Extant | Native | – | – |
| Belgium | Presence Uncertain | Native | – | – |
| Belize | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Bermuda | Extant | Native | – | – |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | – |
| Brazil | Extant | Native | – | – |
| British Indian Ocean Territory | Extant | Native | – | – |
| Brunei Darussalam | Extant | Native | – | – |
| Cabo Verde | Extant | Native | – | – |
| Cambodia | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Canada | Extant | Native | – | – |
| Canada -> Newfoundland I | Extant | Native | – | – |
| Cayman Islands | Extant | Native | – | – |
| Chile | Extant | Native | – | – |
| China | Extant | Native | – | – |
| Christmas Island | Presence Uncertain | Native | – | – |
| Cocos (Keeling) Islands | Extant | Native | – | – |
| Colombia | Extant | Native | – | – |
| Comoros | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Cook Islands | Extant | Native | – | – |
| Costa Rica | Extant | Native | – | – |
| Croatia | Presence Uncertain | Native | – | – |
| Cuba | Extant | Native | – | – |
| Curaçao | Extant | Native | – | – |
| Cyprus | Presence Uncertain | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Denmark | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Dominica | Extant | Native | – | – |
| Dominican Republic | Extant | Native | – | – |
| Ecuador | Extant | Native | – | – |
| Ecuador -> Galápagos | Extant | Native | – | – |
| Egypt | Presence Uncertain | Native | – | – |
| El Salvador | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Presence Uncertain | Native | – | – |
| Falkland Islands (Malvinas) | Extant | Native | – | – |
| Faroe Islands | Extant | Native | – | – |
| Fiji | Extant | Native | – | – |
| Finland | Presence Uncertain | Native | – | – |
| France | Extant | Native | – | – |
| French Guiana | Extant | Native | – | – |
| French Polynesia | Extant | Native | – | – |
| French Southern Territories | Extant | Native | – | – |
| French Southern Territories -> Kerguelen | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Germany | Extant | Vagrant | – | – |
| Ghana | Extant | Native | – | – |
| Gibraltar | Extant | Native | – | – |
| Greece | Presence Uncertain | Native | – | – |
| Greenland | Extant | Native | – | – |
| Grenada | Extant | Native | – | – |
| Guadeloupe | Extant | Native | – | – |
| Guam | Extant | Native | – | – |
| Guatemala | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Guyana | Extant | Native | – | – |
| Haiti | Extant | Native | – | – |
| Heard Island and McDonald Islands | Extant | Native | – | – |
| Honduras | Extant | Native | – | – |
| Hong Kong | Presence Uncertain | Native | – | – |
| Iceland | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Indonesia | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Ireland | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Italy | Extant | Native | – | – |
| Jamaica | Extant | Native | – | – |
| Japan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kiribati | Extant | Native | – | – |
| Korea, Democratic People’s Republic of | Presence Uncertain | Native | – | – |
| Korea, Republic of | Presence Uncertain | Native | – | – |
| Kuwait | Presence Uncertain | Native | – | – |
| Lebanon | Presence Uncertain | Native | – | – |
| Liberia | Extant | Native | – | – |
| Libya | Presence Uncertain | Native | – | – |
| Madagascar | Extant | Native | – | – |
| Malaysia | Extant | Native | – | – |
| Maldives | Extant | Native | – | – |
| Malta | Presence Uncertain | Native | – | – |
| Marshall Islands | Extant | Native | – | – |
| Martinique | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mayotte | Presence Uncertain | Native | – | – |
| Mexico | Extant | Native | – | – |
| Micronesia, Federated States of | Extant | Native | – | – |
| Monaco | Extant | Native | – | – |
| Montenegro | Presence Uncertain | Native | – | – |
| Montserrat | Presence Uncertain | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Myanmar | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nauru | Extant | Native | – | – |
| Netherlands | Extant | Native | – | – |
| New Caledonia | Extant | Native | – | – |
| New Zealand | Extant | Native | – | – |
| Nicaragua | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Niue | Extant | Native | – | – |
| Norfolk Island | Presence Uncertain | Native | – | – |
| Northern Mariana Islands | Extant | Native | – | – |
| Norway | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Palau | Extant | Native | – | – |
| Panama | Extant | Native | – | – |
| Papua New Guinea | Extant | Native | – | – |
| Peru | Extant | Native | – | – |
| Philippines | Extant | Native | – | – |
| Pitcairn | Extant | Native | – | – |
| Portugal | Extant | Native | – | – |
| Puerto Rico | Extant | Native | – | – |
| Qatar | Presence Uncertain | Native | – | – |
| Russian Federation | Extant | Native | – | – |
| Réunion | Presence Uncertain | Native | – | – |
| Saint Barthélemy | Extant | Native | – | – |
| Saint Helena, Ascension and Tristan da Cunha | Extant | Native | – | – |
| Saint Kitts and Nevis | Extant | Native | – | – |
| Saint Lucia | Extant | Native | – | – |
| Saint Martin (French part) | Extant | Native | – | – |
| Saint Pierre and Miquelon | Extant | Native | – | – |
| Saint Vincent and the Grenadines | Extant | Native | – | – |
| Samoa | Extant | Native | – | – |
| Sao Tome and Principe | Presence Uncertain | Native | – | – |
| Saudi Arabia | Presence Uncertain | Native | – | – |
| Senegal | Extant | Native | – | – |
| Seychelles | Presence Uncertain | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Singapore | Extant | Native | – | – |
| Sint Maarten (Dutch part) | Extant | Native | – | – |
| Solomon Islands | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Extant | Native | – | – |
| Spain | Extant | Native | – | – |
| Sri Lanka | Extant | Native | – | – |
| Sudan | Presence Uncertain | Native | – | – |
| Suriname | Extant | Native | – | – |
| Svalbard and Jan Mayen | Extant | Native | – | – |
| Sweden | Presence Uncertain | Native | – | – |
| Syrian Arab Republic | Presence Uncertain | Native | – | – |
| Taiwan, Province of China | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Thailand | Extant | Native | – | – |
| Timor-Leste | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Tokelau | Presence Uncertain | Native | – | – |
| Tonga | Extant | Native | – | – |
| Trinidad and Tobago | Extant | Native | – | – |
| Tunisia | Extant | Native | – | – |
| Turks and Caicos Islands | Presence Uncertain | Native | – | – |
| Tuvalu | Presence Uncertain | Native | – | – |
| Türkiye | Presence Uncertain | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| United Kingdom of Great Britain and Northern Ireland | Extant | Native | – | – |
| United States Minor Outlying Islands | Extant | Native | – | – |
| United States of America | Extant | Native | – | – |
| United States of America -> Aleutian Is. | Extant | Native | – | – |
| United States of America -> Hawaiian Is. | Extant | Native | – | – |
| Uruguay | Extant | Native | – | – |
| Vanuatu | Extant | Native | – | – |
| Venezuela, Bolivarian Republic of | Extant | Native | – | – |
| Viet Nam | Extant | Native | – | – |
| Virgin Islands, British | Extant | Native | – | – |
| Virgin Islands, U.S. | Extant | Native | – | – |
| Wallis and Futuna | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 18. Arctic Sea | Extant | Native | – | Resident |
| 21. Atlantic – northwest | Extant | Native | – | Resident |
| 27. Atlantic – northeast | Extant | Native | – | Resident |
| 31. Atlantic – western central | Extant | Native | – | Resident |
| 34. Atlantic – eastern central | Extant | Native | – | Resident |
| 37. Mediterranean and Black Sea | Extant | Native | – | Resident |
| 41. Atlantic – southwest | Extant | Native | – | Resident |
| 47. Atlantic – southeast | Extant | Native | – | Resident |
| 48. Atlantic – Antarctic | Extant | Native | – | Resident |
| 51. Indian Ocean – western | Extant | Native | – | Resident |
| 57. Indian Ocean – eastern | Extant | Native | – | Resident |
| 58. Indian Ocean – Antarctic | Extant | Native | – | Resident |
| 61. Pacific – northwest | Extant | Native | – | Resident |
| 67. Pacific – northeast | Extant | Native | – | Resident |
| 71. Pacific – western central | Extant | Native | – | Resident |
| 77. Pacific – eastern central | Extant | Native | – | Resident |
| 81. Pacific – southwest | Extant | Native | – | Resident |
| 87. Pacific – southeast | Extant | Native | – | Resident |
| 88. Pacific – Antarctic | Extant | Native | – | Resident |
Climate change
Environmental or climate change is a possible threat to Killer Whales since the acoustic ecology of these animals respond to changes in environmental conditions that might also influence prey availability and abundance (Shabangu et al. 2024).
Population Information
Killer Whales at the Prince Edward Islands bear some morphological resemblance to Antarctic Type B Killer Whales (and are genetically closely related; Moura et al. 2015), whilst those found in mainland waters have until recently been assumed to be Type A (Best 2007). However, strong evidence should be provided before populations are defined as or assigned to ecotypes (de Bruyn et al. 2013). Among Killer Whales stranded on the South African coast, small individuals with significant tooth wear have been designated as a new flat-toothed morphotype (Best et al. 2014). Although this is potentially reflected in the large haplotype diversity among South African Killer Whales, there is so far no clear indication of population structure (Moura et al. 2014).
There are no population estimates for mainland South Africa and there are very few sighting and stranding data available (but see Findlay et al. 1992; Best et al. 2010). However, an ongoing photo-identification study at the Prince Edward Islands has identified around 57 individuals, of which 37 were mature (Reisinger et al. 2011b; Reisinger & de Bruyn 2014).
The Prince Edward Islands population and individuals that prey on marine mammals and sharks in coastal South Africa may be considered cultural groups, where culture refers to “information or behaviour – shared by a population or subpopulation – which is acquired from conspecifics through some form of social learning” (Rendell & Whitehead 2001). In this case the specific hunting strategies used by these socially connected groups at particular locations could be considered culture. As such, they may qualify as conservation value subpopulations.
There is potentially some exchange and almost certainly some spatial overlap between the two subpopulations. Four of the 36 Killer Whales landed by whalers off Durban, had Southern Elephant Seal remains in their stomachs (Best et al. 2010) and the Prince Edward Islands (PEIs) are the nearest Southern Elephant Seal breeding colony. Among Killer Whales satellite-tracked from the PEIs, one individual travelled northwards to ~34.9°S; 37.3°E (Reisinger et al. 2015). This position is near to, but does not actually overlap, the distribution of South African sightings reported by Best et al. (2010). However, this is more likely to represent a lack of sighting (or reporting) effort offshore of South Africa. Moreover, Killer Whales are capable of travelling long distances. Yet there have been no photographic matches between the PEIs and South Africa, and Killer Whales from South Africa and the PEIs are morphologically distinct (Best 2007; Best et al. 2010); although the differences may not be apparent to untrained/inexperience observers or in poor sighting conditions at sea. The PEIs population is composed mainly of animals which show strong seasonal residence at the PEIs (Keith et al. 2001; Reisinger et al. 2011a; R. Reisinger & N. de Bruyn unpubl. data). All the PEIs animals sampled share a single haplotype (they are closely related) (Janse van Rensburg et al. 2013), and northward movements from the PEIs of satellite tracked individuals have been rapid, with individuals subsequently returning to the PEIs (Reisinger et al. 2015). This leads to the inference that the subpopulations are distinct but may overlap spatially at times.
Although these subpopulation sizes are small and thus would qualify for a threatened listing, this species is wide-ranging and its distribution is not yet known to be genetically fragmented. There is no expected continuing decline in the number of subpopulations or mature individuals. Thus, there is no reason to suspect this species is threatened within South African waters.
Current population trend: Stable
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: The subpopulations are defined as: 1) South African and 2) PEIs, but it is unknown whether these apply to the entire South African population.
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of subpopulations: 2
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Genetic diversity is known to be low for the Killer Whale worldwide (Hoelzel et al. 1998; 2002; 2007; Moura et al. 2014a), but diversity among putative populations can be high, both among and within ecotypes (Hoelzel et al. 2007; Moura et al. 2014b). In the North Pacific, genetic differentiation between ecotypes in sympatry is comparable to the genetic distance between populations of the same ecotype, either side of the ocean (Hoelzel et al. 2007; Moura et al. 2014b). The worldwide pattern of genetic diversity is different for mitochondrial DNA (mtDNA) and nuclear DNA, with the basal population being from the North Pacific for mtDNA, and from the Antarctic for nuclear DNA (Moura et al. 2015). There is evidence for a bottleneck in multiple populations around the time of the last glacial maximum, which may have affected patterns of connectivity (Moura et al. 2014a). Killer whales seen off South Africa in coastal waters tend to be transitory, though some individuals are seen repeatedly in the same areas. More data is needed to assess if there is any population structure across this range. There does appear to be some level of genetic differentiation between South Africa, Marion Island and Crozet Island (Janse van Rensburg et al. 2013; Hoelzel et al. unpublished).
While there is limited information about the population genetic structure of this species, it is assumed that no subpopulation has been lost (all are extant). Further genomic investigations are needed to quantify the effective population sizes for the species’ genetically distinct subpopulations.
Habitats and ecology
Killer Whales may occur in any marine or large estuarine habitats (although are not recorded in estuaries in the assessment region) but are most common in areas of high marine productivity, particularly at higher latitudes and near shore (Dahlheim & Heyning 1999; Forney & Wade 2006). Movements can be extensive. For instance, a Killer Whale tagged in the Canadian Arctic travelled over 5,400 km in approximately one month (Matthews et al. 2011) and Offshore Killer Whales from Alaska have moved over 4,000 km (Dahlheim et al. 2008). In the Antarctic, they readily enter areas of floe ice in search of prey (Pitman & Ensor 2003).
Globally, Killer Whales are recognised as super-predators capable of adapting their behaviour (both social and hunting) to prey species availability (Jefferson et al. 1991): more than 140 species of marine vertebrate have been reported as prey (Ford et al. 1998; Ford 2009). This includes most marine mammal species (except river dolphins and manatees), seabirds, sea turtles, many species of bony and cartilaginous fishes, and cephalopods (Dahlheim & Heyning 1999; Ford & Ellis 1999; Ford 2009). Off the South African coast, prey includes toothed whales and dolphins, baleen whales, bony and cartilaginous fishes (some depredated from longline fishing vessels), seabirds (although these may largely be non-consumptive or surplus killing) and possibly Cape Fur Seals (Arctocephalus pusilus pusilus) (Best et al. 2010). At the Prince Edward Islands prey includes seals, penguins and Patagonian Toothfish,(Dissostichus elegenoides) (depredated from longline fishing vessels) (Williams et al. 2009; Reisinger et al. 2011a). These prey are hunted using diverse foraging tactics, such as intentional beaching to catch seals onshore (Lopez & Lopez 1985) and washing seals off ice floes (Visser et al. 2008). They are known to use cooperative techniques to herd fish and to attack large prey (Dahlheim & Heyning 1999; Baird 2000). They also use echolocation clicks to hunt different kinds of prey and navigate underwater (Shabangu et al. 2024).
Despite this generally eclectic diet, at least some populations feed on a narrow range of prey (Ford et al. 1998). In the northeast Pacific, three sympatric ecotypes of Killer Whales have been described and each of these specialises in a narrow range of prey. Resident Killer Whales feed exclusively on squid and fishes (primarily salmon), Transient Killer Whales feed mainly on marine mammals (pinnipeds and cetaceans) and Offshore Killer Whales feed largely on bony and cartilaginous fishes (Ford et al. 1998; Herman et al. 2005; Dahlheim et al. 2008). The flat-toothed morphotype recently described in South African waters by Best et al. (2014) may take similar prey as the northeast Pacific Offshore Killer Whales (Ford et al. 2011); the tooth wear described in the South African animals is likely caused by predating on cartilaginous fishes, as in the northeast Pacific Offshores.
In the Southern Ocean, Type A Killer Whales are proposed to specialise on Antarctic Minke Whales (Balaenoptera bonaerensis) but have on occasion been seen to prey on Southern Elephant Seals (Mirounga leonina). Pack Ice (Large Type B) Killer Whales feed almost exclusively on seals (especially Weddell Seals [Leptonychotes weddelli]) but have been observed taking Antarctic Minke Whales on occasion. Gerlache (Small Type B) Killer Whales may feed mainly on fish, but they also take Gentoo Penguins (Pygoscelis papua) and Chinstrap Penguins (Pygoscelis antarctica). Ross Sea (Type C) Killer Whales are putative fish specialists, particularly Antarctic Toothfish (Dissostichus mawsonii). The diet of Pitman et al. (2011) “Subantarctic Killer Whales” (Type D) is unknown, but they may feed on fish, as they have been observed interacting with longline fisheries (Pitman & Ensor 2003; Pitman et al. 2007, 2011, Pitman & Durban 2010, 2012; Tixier et al. 2016). Killer Whales at the sub-Antarctic Crozet Islands (which appear most similar to Antarctic Type A Killer Whales) seem to be generalists, taking elephant seals, fur seals, penguins, large whales and fish (Guinet 1992). Off South Africa some Killer Whales are known to consume both fish and marine mammals, based on stomach contents (Best et al. 2010).
Ecosystem and cultural services: Coastal delphinids (including Killer Whales), as long-lived, long-term residents along the coast, can serve as important sentinels of the health of coastal marine ecosystems (Wells et al. 2004). As top-level predators on a wide variety of fishes and squids, they concentrate contaminants through bioaccumulation and integrate broadly across the ecosystem in terms of exposure to environmental impacts.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
| 9.10. Marine Neritic -> Marine Neritic – Estuaries | – | Suitable | Yes |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
Life History
Model based estimates of generation time are 25.7 years (Taylor et al. 2007). The most detailed information on demographic parameters is from a long term (1970) study of populations of Resident Killer Whales in the northeast Pacific (Olesiuk et al. 1990, 2005). Some key parameters from this population (1996–2004):
Mean age at first birth: 15.4 years
Mean calving interval: 5.5 years
Estimated reproductive potential: 4.5 calves
Realised calf production: 2.2 calves
Mean life expectancy (females): 30 years
Mean life expectancy (males): 19 years
Longevity (females): ~ 80 years
Estimated maximum longevity (males): ~ 40–50 years
Mean annual mortality: 3.4%
Cumulative juvenile mortality (up to recruitment age, 15.5 years): 39.2%
Population composition: juveniles ~ 47%; reproductive females ~ 24%; post-reproductive females ~ 11%; adult males ~ 18%. Based on a photographic identification study at the Prince Edward Islands (Tosh et al. 2008; Reisinger et al. 2011b; Reisinger & de Bruyn 2014).
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: Daily and Monthly
Congregatory: Yes, fewer than 20 individuals
Systems
System: Marine
General Use and Trade Information
There is no contemporary trade or use of this species in South Africa.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats to this species have been identified; however, unofficial communications indicate that deepwater longliners use dynamite to scare off Killer Whales that remove catches from the longlines. Killer Whale mortalities are expected, but numbers are unknown. Killer Whales steal hooked fish from deep-water longline fishing vessels in South African waters and around the PEIs (Tilney & Purves 1999; Kock et al. 2006; Williams et al. 2009). From 1999-2001 Killer Whales interacted with 33– 64% of longlining cruises to the PEIs (Kock et al. 2006). In South African waters from 2002–2006, Killer Whales depredated 0.5% of total catch on monitored vessels (Williams et al. 2009). Fishermen (illegally) attempt to deter these Killer Whales using explosives or firearms (Kock et al. 2006); this may obviously result in injury or death, but has not been quantified. Poncelet et al. (2010) suggested that lethal interactions with longline fisheries may be one of the causes for the declining population size and survivorship of the Crozet Killer Whale population (1964-2002). Tixier et al. (2010) report relatively constant depredation rates around the Crozet Islands, but the social transmission and spread of this behaviour among Killer Whales is likely (e.g. Fearnbach et al. 2014).
Killer Whales depredate Patagonian Toothfish from seven longline fishing vessels around the Crozet Islands, ~1,000 km east of Marion Island. Tixier (2012) estimated that Killer Whales depredated 116 t / year of Patagonian Toothfish between 2001 and 2010. Pot fishing and acoustic harassment devices (AHDs) have been used in an attempt to mitigate this interaction. Catch per unit effort during pot fishing was not economically sustainable (Gasco et al. 2010) and Killer Whales appear to become habituated to AHDs after the first exposure, which also raises concerns about the effect of the loud amplitude noise on Killer Whales’ hearing (Tixier et al. 2014b). Echolocation clicks and social calls of the PEIs Killer Whales respond to underwater noise changes (Shabangu et al. unpublished data), and those in the South African mainland probably also respond to noise as seen for other odontocetes (Shabangu et al. 2022).
Tixier et al. (2014a) showed that several variables influenced the interaction between Killer Whales and fishing vessels: short longline sets (< 5 km) and faster hauling speeds decreased losses; the depth of longline sets, the distance travelled between sets and the number of vessels operating simultaneously all decreased the probability of interaction (Tixier et al. 2014a). Tixier et al. (2010) recommended that vessels move > 40 nautical miles before fishing again and, after an interaction, lines < 5 km be used. However local factors may influence how effective such mitigation measures are (Tixier et al. 2014a).
Persistent bio-accumulating contaminants are inferred to pose a potential threat to Killer Whales that inhabit, or prey on high trophic level prey that occupy coastal zones (Cockcroft 1999), such as the individuals which prey on common dolphins in False Bay. Contaminants such as persistent organic pollutants (POPs) are ubiquitous in marine environments and since they accumulate at higher trophic levels (biomagnification), Killer Whales are particularly at risk (e.g. Ross et al. 2000). Noël et al. (2009) found relatively high concentrations of POPs in Killer Whales at the Crozet Islands, 1,000 km east of Marion Island. Although polychlorinated biphenyl (PCB) concentrations were lower than in Northern Hemisphere populations, in 70% of individuals they were still above a conservative 1.3 mg / kg marine mammal health threshold (Mos et al. 2007), demonstrating that marine mammals from even remote locations are at risk.
Current habitat trend: Stable. Killer Whales are cosmopolitan and do not show obvious habitat preferences. However, culturally specialised Killer Whale groups may be at risk from shifting prey distribution and abundance because it appears that these specialised groups may have difficulty adapting their diet and behaviour. In general, increased marine resource utilisation poses a threat to this species, either directly (squid and fish-eating Killer Whales) or indirectly (marine mammal-eating Killer Whales).
Conservation
This species is fully protected in South African waters through national legislation – Marine Living Resources Act, 1998 (Act No. 18 of 1998) and the Prince Edward Islands Act, 1948 (Act No. 43 of 1948). The species is listed in Appendix II of CITES. Mitigation of Killer Whale interaction with longline fishing vessels is considered the only major conservation intervention necessary for this species at present. Thus, continued research into the spatiotemporal patterns of longline-Killer Whale interactions is necessary, and observer protocols should be standardised to ensure the collection of valuable, unbiased data (Kock et al. 2006).
Studies on taxonomy, subpopulation structure, abundance and life history are needed for the South African and Prince Edward Islands areas. Regional subpopulations of Killer Whales can be small and highly specialised, and therefore vulnerable to over-exploitation and habitat deterioration. Several small subpopulations elsewhere in the world have already been recognised as having a high risk of extinction (e.g. Southern Resident Killer Whales; Krahn et al. 2004). Within potential subpopulations, cultural uniqueness which has high conservation value should be identified and conserved.
Photographic identification at the Prince Edward Islands is the only systematic monitoring programme for this species (e.g. Reisinger et al. 2011b; Reisinger & de Bruyn 2014). Similar, although unsystematic, photo-identification has been carried out along the South African coast for the past five years under a large-scale collaborative effort among scientists and citizen science groups alike.
Recommendations for managers and practitioners:
- Interactions between Killer Whales and longline fisheries require monitoring.
- Investigations into the development of effective depredation mitigation measures are urgently required.
- Assessment of the severity of factors that are expected to pose potential threats by virtue of distribution patterns of Killer Whales should be made.
- Implementation of best whale watching practices to avoid harassment and injury of Killer Whales.
Research priorities:
- Further population genetic analyses of South African and Prince Edward Island Killer Whale stocks to determine population identity and patterns of distribution.
- Current population size and trend estimates are urgently required.
- Foraging range and diet, including potential response of prey resources to the effects of climate change.
- Within potential subpopulations, cultural uniqueness which has high conservation value should be identified and conserved.
- Movement and foraging range studies are required to assess overlap, if any, of identified groups.
- Investigate impacts of pollutants on the health and welfare of Killer Whales.
- Research on the effects of underwater noise produced by seismic survey signals and marine traffic on Killer Whale wellbeing and ecology.
Current ongoing research projects:
- Prince Edwards Islands – Marion Island Marine Mammal Programme, Mammal Research Institute, Department of Zoology and Entomology, University of Pretoria: The Mammal Research Institute has opportunistically monitored Killer Whales at the Prince Edward Islands since the 1970s (see Condy et al. 1978), but recently (2006) dedicated Killer Whale work has been launched. The project is based on photographic identification of individuals and includes aspects such as satellite tracking, genetics and stable isotope analyses (www.marionseals.com).
- South Africa – no dedicated projects, although some opportunistic photo-identification work has been conducted and additional work has been proposed.
- South Africa, France and UK – population genomic work to better understand the evolution and population structure among South African and PEIs regions.
- Establishment of Killer Whale call catalogue in the PEIs region
Encouraged citizen actions:
- Foraging ecology research at Marion Island is progressing but impeded by the financial investment that is required for satellite telemetry. Aid in funding this aspect can be securely provided through: http:// www.givengain.com/cause/4655/.
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, e.g. wwfsa.mobi, FishMS 0794998795.
- Buy fresh produce that has been grown in pesticide free environments.
- Save electricity and fuel to mitigate CO2 emissions and hence, the rate of climate change.
- Buy local products that have not been shipped.
- Reduce boat speed in bays and harbours.
- Report sightings on MammalMAP. Sightings of Killer Whales and photographs suitable for photographic identification can be uploaded to MammalMAP or reported to the Mammal Research Institute, Faculty of Natural and Agricultural Sciences, University of Pretoria.
- When participating in whale/dolphin watching tours, ensure regulations are adhered to.
- Don’t approach or chase Killer Whales in boats.
Bibliography
Baird, R.W. 2000. The killer whale: foraging specializations and group hunting. In: J. Mann, R. C. Connor, P. L. Tyack and H. Whitehead (eds), Cetacean societies: field studies of dolphins and whales, pp. 127-153. University of Chicago Press, Chicago, USA.
Best PB, Meÿer MA, Lockyer C. 2010. Killer whales in South African waters—a review of their biology. African Journal of Marine Science 32: 171-186.
Best PB, Meÿer MA, Thornton M, Kotze PGH, Seakamela SM, Hofmeyr GJG, Wintner S, Weland CD, Steinke D. 2014. Confirmation of the occurrence of a second killer whale morphotype in South African waters. African Journal of Marine Science 36: 215-224.
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Cockcroft VG. 1999. Organochlorine levels in cetaceans from South Africa: a review. Journal of Cetacean Research and Management, Special Issue 1: 169-176.
Condy PR, van Aarde RJ, Bester MN. 1978. The seasonal occurrence and behaviour of killer whales Orcinus orca, at Marion Island. Journal of Zoology 184: 449-464.
Dahlheim, M. E. and Heyning, J. E. 1999. Killer whale Orcinus orca (Linneaus, 1758). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 6: The second book of dolphins and the porpoises, pp. 281-322. Academic Press.
Dahlheim, M. E., Schulman-Janiger, A., Black, N., Ternullo, R., Ellifrit, D., and Balcomb, K. C. 2008. Eastern temperate North Pacific offshore killer whales (Orcinus orca): occurrence, movements, and insights into feeding ecology. Marine Mammal Science 24: 719-729.
Fearnbach H, Durban JW, Ellifrit DK, Waite JM, Matkin CO, Lunsford CR, Peterson MJ, Barlow J, Wade PR. 2014. Spatial and social connectivity of fish-eating “Resident” killer whales (Orcinus orca) in the northern North Pacific. Marine Biology 161: 459-472.
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Findlay KP. 1989. The distribution of cetaceans off the coast of South Africa and South West Africa/Namibia. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Ford JK, Ellis GM, Barrett-Lennard LG, Morton AB, Palm RS, Balcomb III KC. 1998. Dietary specialization in two sympatric populations of killer whales (Orcinus orca) in coastal British Columbia and adjacent waters. Canadian Journal of Zoology 76: 1456-1471.
Ford JK, Ellis GM, Matkin CO, Wetklo MH, Barrett-Lennard LG, Withler RE. 2011. Shark predation and tooth wear in a population of northeastern Pacific killer whales. Aquatic Biology 11: 213-224.
Ford, J. K. B. 2009. Killer whale Orcinus orca. In: W. F Perrin, B. Würsig and J. G. M. Thewissen (eds), Encyclopedia of marine mammals, pp. 650-657. Academic Press, Amsterdam.
Ford, J. K. B. and Ellis, G. M. 1999. Transients: Mammal-hunting killer whales of British Columbia, Washington, and southeastern Alaska. Univ. British Columbia Press.
Forney, K. A. and Wade, P. 2006. Worldwide distribution and abundance of killer whales. In: J. A. Estes, R. L. Brownell, Jr., D. P. DeMaster, D. F. Doak and T. M. Williams (eds), Whales, Whaling and Ocean Ecosystems, pp. 145-162. University of California Press.
Gasco N, Tixier P, Guinet C. 2010. Results of trials undertaken around Crozet Island using pots to target Patagonian toothfish. Document WG-FSA-10/10. CCAMLR, Hobart, Australia.
Guinet C. 1992. Comportement de chasse des orques (Orcinus orca) autour des iles Croze. Canadian Journal of Zoology 70: 1656-1667.
Herman DP, Burrows DG, Wade PR, Durban JW, Matkin CO, LeDuc RG, Barrett-Lennard LG, Krahn MM. 2005. Feeding ecology of eastern North Pacific killer whales Orcinus orca from fatty acid, stable isotope, and organochlorine analyses of blubber biopsies. Marine Ecology Progress Series 302: 275-291.
Heyning, J. E., and Dahlheim, M. E. 1988. Orcinus orca. Mammalian Species 304: 1-9.
Hoelzel, A.R., Dahlheim. M. and Stern, S.J. 1998. Low genetic variation among killer whales (Orcinus orca) in the eastern North Pacific, and genetic differentiation between foraging specialists. J. Hered. 89:121-128
Hoelzel, A.R., Natoli, A., Dahlheim, M.E., Olavarria, C., Baird, R.W. and Black, N.A. 2002. Low worldwide genetic diversity in the killer whale (Orcinus orca): implications for demographic history. Proceedings of the Royal Society of London B: Biological Sciences 269: 1467-1473.
Hoelzel, A.R., Hey, J., Dahlheim, M.E., Nicholson, C., Burkanov, V. & Black, N. 2007. Evolution of Population Structure in a Highly Social Top Predator, the Killer Whale. Mol. Biol. Evol. 24:1407–1415
IUCN (International Union for Conservation of Nature). 2013. Orcinus orca. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org. Downloaded on 21 February 2016.
Janse van Rensburg C, de Bruyn PJN, Reisinger RR, Best PB, Thornton M, Plön S, Hoelzel AR. 2013. Molecular DNA analysis of killer whales (Orcinus orca) from sub-Antarctic Marion Island and mainland South Africa. Zoological Society of Southern Africa Symposium, 14-17 July 2013, Thsipise, South Africa.
Jefferson, T.A., Stacey, P.J., and Baird, R.W. 1991. A review of killer whale interactions with other marine mammals: predation to coexistence. Mammal Review 21: 151-180.
Keith, M., Bester, M. N., Bartlett, P. A. and Baker, D. 2001. Killer whales (Orcinus orca) at Marion Island, Southern Ocean. African Zoology 36: 163-175.
Kock K-H, Purves MG, Duhamel G. 2006. Interactions between cetacean and fisheries in the Southern Ocean. Polar Biology 29: 379-388.
Krahn, M. M., Ford, M. J., Perrin, W. F., Wade, P. R., Angliss, R. P., Hanson, M. B., Taylor, B. L., Ylitalo, G. M., Dahlheim, M. E., Stein, J. E. and Waples, R. S. 2004. 2004 status review of southern resident killer whales (Orcinus orca) under the Endangered Species Act. NOAA Technical Memorandum NMFS-NWFSC 73: 133.
López, J. C. and López, D. 1985. Killer whales (Orcinus orca) of Patagonia, and their behavior of intentional stranding while hunting nearshore. Journal of Mammalogy 66: 181-183.
Matthews CJ, Luque SP, Petersen SD, Andrews RD, Ferguson SH. 2011. Satellite tracking of a killer whale (Orcinus orca) in the eastern Canadian Arctic documents ice avoidance and rapid, long-distance movement into the North Atlantic. Polar Biology 34: 1091-1096.
Morin, P.A., Archer, F.I., Foote, A.D., Vilstrup, J., Allen, E.E., Wade, P., Durvban, J., Parsons, K., Pitman, R., Li, L., Bouffard, P., Nielsen, S.C.A., Rasmussen, M., Willerslev, E., Gilbert, M.T.P., and Harkins, T. 2010. Complete mitochondrial genome phylogeographic analysis of killer whales (Orcinus orca) indicates multiple species. Genome Research 20: 908-916.
Mos L, Tabuchi M, Koop BF, Helbing CC, Ross PS. 2007. Toxicogenomics: a new tool for the assessment of health and toxicity in marine mammals. 17th Biennial Conference on the Biology of Marine Mammals.
Moura, A.E., Janse van Rensburg, C., Pilot, M., Tehrani, A., Best, P.B., Thornton, M., Plon, S., de Bruyn P.J.N., Worley, K.C., Gibbs, R.A., Dahlheim, M.E. & Hoelzel, A.R. 2014a. Killer Whale Nuclear Genome and mtDNA Reveal Widespread Population Bottleneck During the Last Glacial Maximum. Mol. Biol. Evol. 31, 1121-1131.
Moura, A.E., Kenny, J.G., Chaudhuri, R., Hughes, M.A., Welch, A., Reisinger, R.R., de Bruyn, P.J.N., Dahlheim, M.E., Hall, N., Hoelzel, A.R. 2014b. Population genomics of the killer whale indicates ecotype evolution in sympatry involving both selection and drift. Mol. Ecol. 23, 5179-5192
Moura AE, Kenny JG, Chaudhuri RR, Hughes MA, Reisinger RR, de Bruyn PJN, Dahlheim ME, Hall N, Hoelzel AR. 2015. Phylogenomics of the killer whale indicates ecotype divergence in sympatry. Heredity 114: 48-55.
Noël M, Barrett-Lennard L, Guinet C, Dangerfield N, Ross PS. 2009. Persistent organic pollutants (POPs) in killer whales (Orcinus orca) from the Crozet Archipelago, southern Indian Ocean. Marine Environmental Research 68: 196-202.
Olesiuk PF, Ellis GM, Ford JKB. 2005. Life history and population dynamics of northern resident killer whales (Orcinus orca) in British Columbia. Research document 2005/45. Canadian Science Advisory Secretariat, Fisheries and Oceans Canada, Ottawa, Canada.
Olesiuk, P., Bigg, M. A. and Ellis, G. M. 1990. Life history and population dynamics of resident killer whales (Orcinus orca) in the coastal waters of British Columbia and Washington State. Reports of the International Whaling Commission 12: 209-243.
Perrin, W.F. 2009. Species. In: W.F. Perrin, B. Wursig and J.G.M Thewissen (eds), Encyclopedia of Marine Mammals. Second edition, pp. 1084-1087. Academic Press, San Diego, California, USA.
Pitman R.L. and Durban J.W. 2010. Killer whale predation on penguins in Antarctica. Polar Biology 33: 1589-1594.
Pitman, R. L., and Durban, J. W. 2012. Cooperative hunting behavior, prey selectivity and prey handling by pack ice killer whales (Orcinus orca), type B, in Antarctic Peninsula waters. Marine Mammal Science 28: 16-36.
Pitman, R.L. and Ensor, P. 2003. Three forms of killer whales (Orcinus orca) in Antarctic waters. Journal of Cetacean Research and Management 5: 131-139.
Pitman, R.L., Durban, J.W., Greenfelder, M., Guinet, C., Jorgensen, M., Olson, P., Plana, J., Tixier, P., and Towers, J.R. 2011. Observations of a distinctive morphotype of killer whale (Orcinus orca), type D, from subantarctic waters. Polar Biology 34: 303-306.
Pitman, R.L., Perryman, W.L., Leroi, D. and Eilers, E. 2007. A dwarf form of killer whale in Antarctica. Journal of Mammalogy 88: 43-48.
Poncelet É, Barbraud C, Guinet C. 2010. Population dynamics of killer whales (Orcinus orca) in the Crozet Archipelago, southern Indian Ocean: a mark–recapture study from 1977 to 2002. Journal of Cetacean Research and Management 11: 41-48.
Rabearisoa N, Bach P, Tixier P, Guinet C. 2012. Pelagic longline fishing trials to shape a mitigation device of the depredation by toothed whales. Journal of Experimental Marine Biology and Ecology 432: 55-63.
Reisinger RR, Keith M, Andrews RD, de Bruyn PJN. 2015. Movement and diving of killer whales (Orcinus orca) at a Southern Ocean archipelago. Journal of Experimental Marine Biology and Ecology 473: 90-102.
Reisinger RR, de Bruyn PJN, Tosh CA, Oosthuizen WC, Mufanadzo NT, Bester MN. 2011a. Prey and seasonal abundance of killer whales at sub-Antarctic Marion Island. African Journal of Marine Science 33: 99-105.
Reisinger RR, de Bruyn PN, Bester MN. 2011b. Abundance estimates of killer whales at subantarctic Marion Island. Aquatic Biology 12: 177-185.
Reisinger RR, de Bruyn PN. 2014. Marion Island killer whales: 2006–2013. Mammal Research Institute, University of Pretoria 10.
Rendell L, Whitehead H. 2001. Cetacean culture: still afloat after the first naval engagement of the culture wars. Behavioral and Brain Sciences 24: 360-373.
Rice, D.W. 1998. Marine Mammals of the World: Systematics and Distribution. Society for Marine Mammalogy, Special Publication Number 4, Lawrence, Kansas.
Ross, P.S., Ellis, G.M., Ikonomou, M.G., Barrett-Lennard, L.G. and Addison, R.F. 2000. High PCB concentrations in free-ranging Pacific killer whales, Orcinus orca: effects of age, sex and dietary preference. Marine Pollution Bulletin 40: 504-515.
Shabangu FW, Daniels R, Jordaan RK, de Bruyn PJN, van den Berg MA, Lamont T. 2024 Killer whale acoustic patterns respond to prey abundance and environmental variability around the Prince Edward Islands, Southern Ocean. R. Soc. Open Sci. 10, 230903. (doi:10.1098/rsos.230903)
Shabangu FW, Yemane D, Best G, Estabrook BJ. 2022 Acoustic detectability of whales amidst underwater noise off the west coast of South Africa. Mar. Pollut. Bull. 184, 114122. (doi:10.1016/j.marpolbul.2022.114122)
Shabangu FW, Daniels R, Jordaan RK, de Bruyn PJN, van den Berg MA, Lamont T. 2024 Killer whale acoustic patterns respond to prey abundance and environmental variability around the Prince Edward Islands, Southern Ocean. R. Soc. Open Sci. 10, 230903. (doi:10.1098/rsos.230903)
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Tilney R, Purves MG. 1999. The status of integrated fisheries monitoring in South Africa. In: Nolan CP (ed.). Proceedings of the international conference on integrated fisheries monitoring, Sydney, Australia, 1–5 February 1999, FAO Rome.
Tixier P, Garcia JV, Gasco N, Duhamel G, Guinet C. 2014a. Mitigating killer whale depredation on demersal longline fisheries by changing fishing practices. ICES Journal of Marine Science: Journal du Conseil: 1-11.
Tixier P, Gasco N, Duhamel G, Guinet C. 2014b. Habituation to an acoustic harassment device (AHD) by killer whales depredating demersal longlines. ICES Journal of Marine Science: Journal du Conseil: 1-9.
Tixier P, Gasco N, Duhamel G, Guinet C. 2016. Depredation of Patagonian toothfish (Dissostichus eleginoides) by two sympatrically occurring killer whale (Orcinus orca) ecotypes: Insights on the behavior of the rarely observed type D killer whales. Marine Mammal Science 32: 983-1003.
Tixier P, Gasco N, Duhamel G, Viviant M, Authier M, Guinet C. 2010. Interactions of Patagonian toothfish fisheries with killer and sperm whales in the Crozet islands Exclusive Economic Zone: an assessment of depredation levels and insights on possible mitigation strategies. CCAMLR Science 17: 179-195.
Tixier P. 2012. Déprédation par les orques (Orcinus orca) et les cachalots (Physeter macrocephalus) sur les palangriers à la légine australe dans la ZEE de l’archipel de Crozet. Ph.D. Thesis. Université d’Aix Marseille II.
Tosh CA, de Bruyn PJN, Bester MN. 2008. Preliminary analysis of the social structure of killer whales, Orcinus orca, at subantarctic Marion Island. Marine Mammal Science 24: 929-940.
Visser, I.N., Smith, T.G., Bullock, I.D., Green, G.D., Carlsson, O.G. and Imberti, S. 2008. Antarctic peninsula killer whales (Orcinus orca) hunt seals and a penguin on floating ice. Marine Mammal Science 24: 225-234.
Wells RS, Rhinehart HL, Hansen LJ, Sweeney JC, Townsend FI, Stone R, Casper DR, Scott MD, Hohn AA, Rowles TK. 2004. Bottlenose dolphins as marine ecosystem sentinels: developing a health monitoring system. EcoHealth 1: 246-254.
Williams AJ, Petersen SL, Goren M, Watkins BP. 2009. Sightings of killer whales Orcinus orca from longline vessels in South African waters, and consideration of the regional conservation status. African Journal of Marine Science 31: 81-86.
de Bruyn, P.N., Tosh, C.A. and Terauds, A. 2013. Killer whale ecotypes: is there a global model? Biological Reviews 88: 62-80.
