Karoo Round-eared Sengi
Macroscelides proboscideus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Macroscelides proboscideus – (Shaw, 1800)
ANIMALIA – CHORDATA – MAMMALIA – MACROSCELIDEA – MACROSCELIDIDAE – Macroscelides – proboscideus
Common Names: Karoo Round-eared Sengi, Karoo Round-eared Elephant-shrew, Short-eared Elephant-shrew (English), Ronde-oorklaasneus (Afrikaans)
Synonyms: Macroscelides proboscideus ssp. proboscideus (Shaw, 1800); Sorex proboscideus Shaw, 1800
For general biological information on all sengi species, consult: Species accounts: Order Macroscelidea, in: Perrin, M. & Rathbun, G. B. 2013. Mammals of Africa, Volume I (Ed. by Kingdon, J., Happold, D., Hoffmann, M., Butynski, T., Happold, M. & Kalina, J.), pp. 261-278. London: Bloombury; Order Macroscelidea, in: Rathbun, G. B., (subeditor). 2005. The Mammals of the Southern African Subregion, 3rd edition (Ed. by Skinner, J. D. & Chimimba, C. T.), pp. 22-34. Cape Town, South Africa: Cambridge University Press; Species accounts, in: Rathbun, G. B. 2013. Mammals of Africa, Volume I (Ed. by Kingdon, J., Happold, D., Hoffmann, M., Butynski, T., Happold, M. & Kalina, J.), pp. 279-287. London: Bloomsbury. Order MACROSCELIDEA, in: Heritage, S. 2018. Handbook of the Mammals of the World, Volume 8. Insectivores, Sloths and Colugos (D.E. Wilson and R.A. Mittermeier, eds), Lynx Edicions, Barcelona, Spain. For current and more technical information, search the on-line bibliography at www.sengis.org.
Taxonomic Note:
Until the 1970s, the single family was included in the order Insectivora, but now the family is in the monophyletic order Macroscelidea and the superorder Afrotheria, which was created in 1998. Currently, there are 20 living species recognised in six genera. The soft-furred sengis or elephant-shrews include five genera: Petrodromus and Petrosaltator are monospecific, Macroscelides contains three species, Galegeeska has two species, while Elephantulus contains 8 species. The five species of giant sengis belong to the genus Rhynchocyon (Rathbun 2009). The common name “sengi” is being used in place of elephant shrew (with a hyphen) by many biologists to try and disassociate the Macroscelidea from the true shrews (family Soricidae) in the order Eulipotyphla (Rathbun and Kingdon 2006).
In their revision of the genus Macroscelides, Dumbacher et al. (2012) elevated the two generally accepted subspecies in the genus (Corbet and Hanks 1968) to full species: M. proboscideus and M. flavicaudatus. In 2014, a third species for the genus was described from northwestern Namibia (Dumbacker et al. 2014). Taxonomic information in Perrin and Rathbun (2013) is out-of-date because of the recent changes in taxonomy of this genus.
See www.sengis.org and also www.afrotheria.net for additional information and literature citations.
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessor: Schradin, C.1 & da Silva, J.2
Reviewer: Roxburgh, L.3
Institutions: 1Institut pluridisciplinaire Hubert Curien, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors: Rathbun, G.B. & Smit-Robinson, H.
Previous Reviewer: Taylor, A.
Previous Contributors: Child, M.F. & Nicholson, S.
Assessment Rationale
Although this species is not locally abundant, it is widespread in suitable habitats over a wide extent of occurrence in South Africa. Because it occupies habitats that are semi-arid to arid that will not support most development without the availability of water, there are few known threats to the vast majority of the habitats occupied by the Karoo Round-eared Sengi. Areas close to rivers or reliable sources of water may have been developed, or may be developed in the future, as agricultural and urban areas. For example, a narrow area adjacent to and along the Orange River between Namibia and South Africa has been developed, but this is a relatively small area compared to the overall distribution of the Karoo Round-eared Sengi. Relatively small areas may also be impacted by mineral extraction activities, such as around the town of Springbok in South Africa, along the N14 highway up to Kuruman, and north towards the border with Namibia. Again, this disturbance is confined to a relatively small area compared to the overall distribution of the species. Past, current, and future development in this region of Africa is not expected to have a significant impact on this sengi or its habitats. However, a number of wind and solar farms are proposed throughout the distribution of the species in South Africa and the potential displacement impacts of these renewable energy facilities should be monitored as emerging threats. On the other hand, bush encroachment and desertification, especially related to widespread intensive goat and sheep grazing outside of protected areas, might adversely alter habitats that these sengis occupy, and these processes should be monitored for possible negative impacts on sengi populations, especially as the species lives in flats that are more likely to be overgrazed than mountain ranges (as used by many other sengi species). The species remains listed as Least Concern.
Regional population effects: More or less continuous distribution with rest of African range (Namibia and Botswana).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Schradin C & da Silva JM. 2025. A conservation assessment of Macroscelides proboscideus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
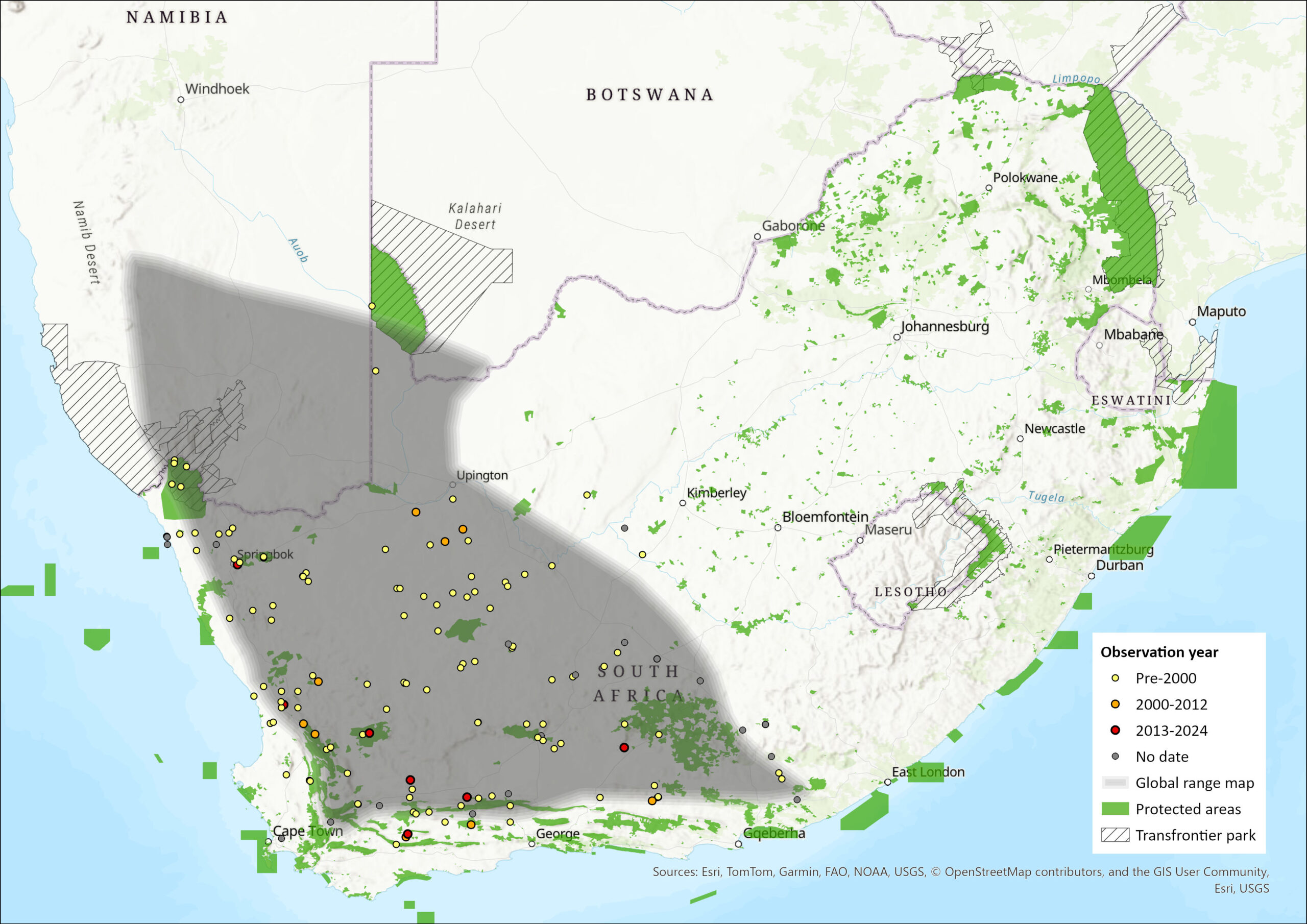
This sengi species is fairly widespread (Rathbun 2009; Dumbacher et al. 2012). Its distribution includes the southern and eastern regions of Namibia, extreme southwestern Botswana and South Africa (Rathbun 2009; Dumbacher et al. 2012). Within South Africa the species occurs in the Northern, Western and Eastern Cape provinces (Rathbun 2009; Dumbacher et al. 2012). This species is confined to a comparatively narrow part of the south-western subregion in the Nama-Karoo and Succulent Karoo biomes (Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 1400
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Karoo Round-eared Sengi (Macroscelides proboscideus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
|
Country |
Presence |
Origin |
Formerly Bred |
Seasonality |
|
Botswana |
Extant |
Native |
– |
Resident |
|
Namibia |
Extant |
Native |
– |
Resident |
|
South Africa |
Extant |
Native |
– |
Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified
Climate change
The species is adapted to semi-arid and arid environments, making it unlikely that future aridification due to climate change endangers it, though it might lead to local reduction of population density or local extinction due to reduced food availability. However, as the species does not use burrows but typically rests above ground inside shrubs, it might be vulnerable to heat waves. Under various climate change scenarios from 1.5 to 4°C warming, however, the range of this species will experience a) a 20 to 40% decline in rainfall, b) a 2 to >6°C increase in temperature and c) increases in drought, and heat wave and fire risk days (Engelbrecht et al. 2024). These are significant changes and would increase the heat burden on the animals significantly.
Population information
Population numbers across much of the species’ range are assumed to be relatively low (Corbet & Hanks 1968; Rathbun 2005; Smit et al. 2010; Schubert 2011; Dumbacher et al. 2012; Perrin & Rathbun 2013). Almost no data on population dynamics of this species is available (Rathbun & Smit-Robinson 2015), but it is expected that subpopulation sizes will vary greatly in the arid habitats where it occurs (Rathbun & Smit-Robinson 2015). This is possibly correlated to climate regimes and climatic variability (Rathbun & Smit-Robinson 2015). Population density in the Goegap Nature Reserve ranges from 0.125 to 3.0 individuals per hectare with a mean of 1.3 individuals (Schradin, unpubl annual data 2008-2024).
Population Information
| Continuing decline in mature individuals | Unknown |
| Qualifier | – |
| Justification | – |
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A population genetic study using two mitochondrial genes (cytB, control region) detected an overall pattern of isolation by distance in this species suggesting a continuous distribution with gene flow taking place among closest M. proboscideus groups (Smit et al. 2010). However, a spatial analysis of molecular variance was still able to identify two distinct genetic populations, with the two most eastern localities, Aberdeen and Steytlerville, forming a distinct group and the remaining distribution forming another as a metapopulation. Aberdeen and Steytlerville are situated on the limit of the eastern distribution of the species, in a region where the vegetative Fynbos biome changes to Albany thicket (Mucina &Rutherford, 2006; Smit et al. 2010). This transitional zone between biomes has previously been identified as a region where distinct genetic lineages characterise species (see for example the study by Tolley et al. 2008), thereby providing added support of the designation of at least two genetic subpopulations within this species. Given that no other subpopulations are thought to have gone extinct, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (2/2 subpopulations remaining).
While population size estimates within each subpopulation are unknown, a rough approximation may be provided based on density and AOO. Based on density estimates from Goegap Nature Reserve (see Population section) and extrapolating this across the species range within the assessment region based on their Area of Occupancy (AOO), which is estimated at 1,856 km2 (using the IUCN’s method of a 2×2 km grid), the entire population is estimated to be 241 280 individuals. Even if the eastern subpopulation represents only 5% of the entire range, which seems likely given that the AOO for occurrence records within or close to the Albany Thicket biome make up around 5% of the AOO, the smallest genetic subpopulation would still equate to approximately 12,064 individuals. Applying a 0.1-0.3 Ne/Nc conversion ratio, the smallest subpopulation is likely to have an effective population size between 1,200 and 3,600 breeding individuals. This exceeds the Ne 500 threshold that represents genetically stable populations into the long-term. From this we can calculate the GBF’s headline indicator – proportion of species with an effective population size greater than 500 – which is valued at 1.0 (2/2 subpopulations with Ne >500).
Given the genetic study used to define genetic subpopulations was based on broad mitochondrial DNA markers (Smit et al. 2010) and the population estimates were based on extrapolated densities and AOO, it is highly recommended that a more fine-scale study be undertaken to get a better idea of the recent genetic structure and more accurately quantify effective population size from all identified genetic subpopulations.
Habitats and ecology
The species occurs within open country with a preference for shrub bush and areas with sparse grass cover (Skinner & Chimimba 2005). The Karoo Round-eared Sengi is a habitat specialist, which occupies sand and gravel plains associated with alluvial plains and relatively flat areas between higher elevation areas such as outcrops, scarps, hills, and mountains (Corbet & Hanks 1968; Rathbun 2005, 2009; Rathbun & Smit-Robinson 2015). Such areas are sparsely vegetated with bunch grasses and widely spaced small bushes (Kerley 1992; Dumbacher et al. 2012; Perrin & Rathbun 2013). In addition, these areas can vary from hard gravel to looser sandy soils (Rathbun & Smit-Robinson 2015). Key vegetation types for the species includes the Succulent Karoo and the Nama Karoo Vegetation biomes.
Densities range between 0.4 and 3.0 individual / ha, and home range areas tend to overlap by a male and female, with male home ranges tending to be larger than those of females (Schubert 2011; Schubert et al. 2009a, 2009b). Smit et al. (2010) indicate that the genetic structure within the species is mostly a pattern of isolation-by-distance, typical of a species with a continuous rather than clustered distribution.
Ecosystem and cultural services: Sengi species are thought to be included in San art and are therefore subject to local folklore.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | Resident | Suitable | Yes |
| 8.1. Desert -> Desert – Hot | Resident | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: 4 months (Olbricht et al. 2006)
Age at Maturity: Male: 3 months (Olbricht et al. 2006)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: In the field in Namaqualand (Goegap) the oldest female was 2.2 years, the oldest male 2.5 years old. In captivity, the oldest age is 8.9 years (Weigl 2005, AnAge database)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 13.5 cm
Size at Birth (in cms): (Not specified)
Gestation Time: 60-61 days (Olbricht et al. 2006)
Reproductive Periodicity: Bi-Annual (In captivity throughout the year (Olbricht et al. 2006). In Namaqualand during the moist warm spring with 2 litters per female).
Average Annual Fecundity or Litter Size: 70% twins, 29% singletons (Olbricht et al. 2006)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Not a Migrant
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is no indication that this species has ever been used by people for any purposes. It has been infrequently exported to various zoological gardens over the last few decades, where husbandry techniques have been developed, breeding has been achieved, and research results have been published (Olbricht 2009). It is also kept as an exotic pet by private people in Europe, especially Germany.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no known major threats to the species. However, local declines due to habitat modification in relatively small areas may occur near rivers and human population centres due to small-holder and industrial agriculture, mineral extraction, and urban development (Rathbun & Smit-Robinson 2015). Changes in habitats due to desertification and bush encroachment and proposed wind and solar energy facilities (especially in the Northern Cape) may adversely alter habitats for sengis and displace them from such areas, but at present these changes appear neither widespread nor serious, especially since the species is associated with arid habitats.
Current habitat trend: This species occurs in rocky habitats unlikely to be transformed extensively. However, livestock and wildlife ranching may have detrimental effects on habitat quality through overgrazing.
Conservation
As this species is widespread, it is not in conflict with most human activities; and it likely occurs in many protected areas (for example, Goegap Nature Reserve, Gamkaberg Nature Reserve, Tankwa Karoo National Park, Mokala National Park, Richtersveld National Park, Augrabies Falls National Park, Namaqua National Park, and Karoo National Park), thus there are no conservation actions recommended at present or in the foreseeable future. Protected area expansion would, however, benefit this species by connecting subpopulations and mitigating fragmentation from land transformation and development including renewable energy generation.
Recommendations for land managers and practitioners:
- Continue to accumulate information on occurrence points (see www.sengis.org).
- Land managers should stock cattle or game at ecological levels.
Research priorities:
- Determining the impacts of habitat shifts, including livestock grazing, on local populations.
- Determining the possible impacts of climate change, including habitat shifts, physiological and behavioural impacts, availability of food and water and changes in population demographics.
Encouraged citizen actions:
- Citizens are encouraged to report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) with photographic confirmation.
Bibliography
Corbet, G.B. and Hanks, J. 1968. A revision of the elephant-shrews, family Macroscelididae. Bulletin of the British Museum of Natural History (Zoology) 16: 1-111.
Dumbacher, J.P., Rathbun, G.B., Osborne, T.O., Griffin, M. and Eiseb, S. 2014. A new species of round-eared sengi (genus Macroscelides) from Namibia. Journal of Mammalogy 95(3): 443-454.
Dumbacher, J.P., Rathbun, G.B., Smit, H.A. and Eiseb, S.J. 2012. Phylogeny and taxonomy of the round-eared sengis or elephant-shrews, genus Macroscelides (Mammalia, Afrotheria, Macroscelidea). PLOS One 7(3): e32410. doi:10.1371/journal.pone.0032410 (12 pp + 4 suppl).
Engelbrecht, F.A., Steinkopf, J., Padavatan, J., Midgley, G.F. 2024. Projections of Future Climate Change in Southern Africa and the Potential for Regional Tipping Points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7.
Everson, T.M. and Morris, C.D. 2006. Conservation of biodiversity in the Maloti-Drakensberg Range. In: E.M. Spehn, Land Use Change and Mountain Biodiversity, pp. 285-291. CRC Press, Boca Raton, USA.
Heritage, S. (2018). Order MACROSCELIDEA. Pp. in Handbook Mammals of the World, 8. Insectivors, Sloths and Colugos (D.E. Wilson, D.E. and R.A. Mittermeier, eds), Lynx Edicions, Barcelona, Spain.
Kerley, G.I.H. 1992. Ecological correlates of small mammal community structure in the semi-arid Karoo, South Africa. Journal of Zoology 227: 17-27.
Olbricht, G. 2009. Reproduction and growth of elephant shrews or sengis (Macroscelidea). Sudwestdeutscher Verlag fur Hochschulschriften, Saarbrucken, Germany.
Olbricht, V.G., Kern, C. and Vakhrusheva, G. 2006. Einige Aspekte der Fortpflanzungsbiologie von Kurzohr- Rüsselspringern (Macroscelides proboscideus A Smith, 1829) in Zoologischen Gärten unter besonderer Berücksichtigung von Drillingswürfen. Der Zoologische Garten, 75, 304- 316.
Olivier, C.-A., A. V. Jaeggi, L. D. Hayes and C. Schradin (2022) Revisiting the components of Macroscelidea social systems: Evidence for variable social organization, including pair-living, but not for a monogamous mating system. Ethology 128: 383-394.
Perrin, M. and Rathbun, G.B. 2013. Species accounts: Order Macroscelidea, Family Macroscelididae, Genus Elephantulus, E. edwardii, E. intufi, E. myurus, E. rozeti, E. rufescens; Genus Macroscelides; species account M. proboscideus. In: J.S Kingdon, D.C.D. Happold, M. Hoffmann, T.M. Butynski, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria, pp. 261-278. Bloomsbury Publihing, London.
Rathbun G.B. and Smit-Robinson H. 2015. Macroscelides proboscideus. The IUCN Red List of Threatened Species 2015.
Rathbun, G.B. (subeditor). 2005. Macroscelidea. In: J.D. Skinner and C.T. Chimimba (eds), The Mammals of the Southern African Subregion, 3rd edition, pp. 22-34. Cambridge University Press, Cambridge, UK.
Rathbun, G.B. 2009. Why is there discordant diversity in sengi (Mammalia; Afrotheria; Macroscelidea) taxonomy and ecology? African Journal of Ecology 7(3): 1-13.
Rathbun, G.B. and Kingdon, J. 2006. The etymology of “sengi”. Afrotherian Conservation, Newsletter of the IUCN-SSC Afrotheria Specialist Group 4: 14-15.
Schubert M. 2011. A summary of the social system of the round-eared sengi. Afrotherian Conservation 8: 12-13.
Schubert, M., N. Pillay, D. O. Ribble and C. Schradin (2009) The round-eared sengi and the evolution of social monogamy: factors that constrain males to live with a single female. Ethology 115: 972-985.
Schubert, M., C. Schradin, H. Rödel, N. Pillay and D. Ribble (2009) Male mate guarding in a socially monogamous mammal, the round-eared sengi: on costs and trade-offs. Behavioral Ecology and Sociobiology 64: 257-264.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smit, H.A., Watson, J. and Jansen van Vuuren, B. 2010. Relative importance of habitat connectivity in shaping the genetic profiles of two southern African elephant-shrews. Journal of Biogeography 37: 857-864. https://doi.org/10.1111/j.1365-2699.2009.02249.x
Weigl, R. 2005. Longevity of Mammals in Captivity; from Living Collections of the World. Kleine Senckenberg-Reihe 48: Stuttgart