Karoo Bush Rat
Otomys unisulcatus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Otomys unisulcatus – F. Cuvier, 1829
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Otomys – unisulcatus
Common Names: Karoo Bush Rat, Bush Karoo Rat, Bush Vlei Rat (English), Karoose Bosrot, Boskkaroorot (Afrikaans)
Synonyms: Myotomys unisulcatus (F. Cuvier, 1829)
Taxonomic Note:
Some authors include Otomys unisulcatus and O. sloggetti in the genus Myotomys (Pocock 1976; Musser & Carleton 2005) because O. unisulcatus is closer related to the two Parotomys than to other Otomys species (Mizerovska et al. 2023).
Red List Status: Least Concern
Assessment Information
Assessors: Schradin, C.1,2, Makuya, L.1 & da Silva, J.M.3
Reviewers: Oosthuizen, M.4
Contributor: Do Linh San, E.5
Institutions:1IPHC-DEPE, CNRS,2University of the Witwatersrand,3South African National Biodiversity Institute, 4University of Pretoria, 5Sol Plaatjie University
Previous Assessors and Reviewers: Do Linh San, E., Babu, N., Xalu, M., Le Gars, S., Perquin, J., Baxter, R.M., Hoepfl, J., Stuart, C. and Stuart, M.
Previous Contributors: Child, M.F., Avenant, N.L., Avery, M., MacFayden, D., Monadjem, A., Palmer, G., Taylor, P. and Wilson, B.
Assessment Rationale
The Karoo Bush Rat is listed as Least Concern because it has a wide distribution within the assessment region, can be abundant in suitable habitat, is present in several protected areas and because there are no identified threats that could cause widespread population decline. However, there are potentially synergistic effects of climate change drying up wetlands and overgrazing/ browsing removing at least part of the plant food and cover that this species relies upon. Such effects on subpopulation trends and population distribution should be monitored.
Regional population effects: This species is endemic to the assessment region. Its dispersal abilities are not well known. Subpopulations seem to be patchily distributed at the landscape level, according to the presence of favourable habitats. While it is likely that movements and possibly rescue effects exist between subpopulations, others might be physically and genetically isolated.
Red List Index
Red List Index: No change
Recommended Citation: Schradin C, Makuya L & da Silva JM. 2025. A conservation assessment of Otomys unisulcatus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
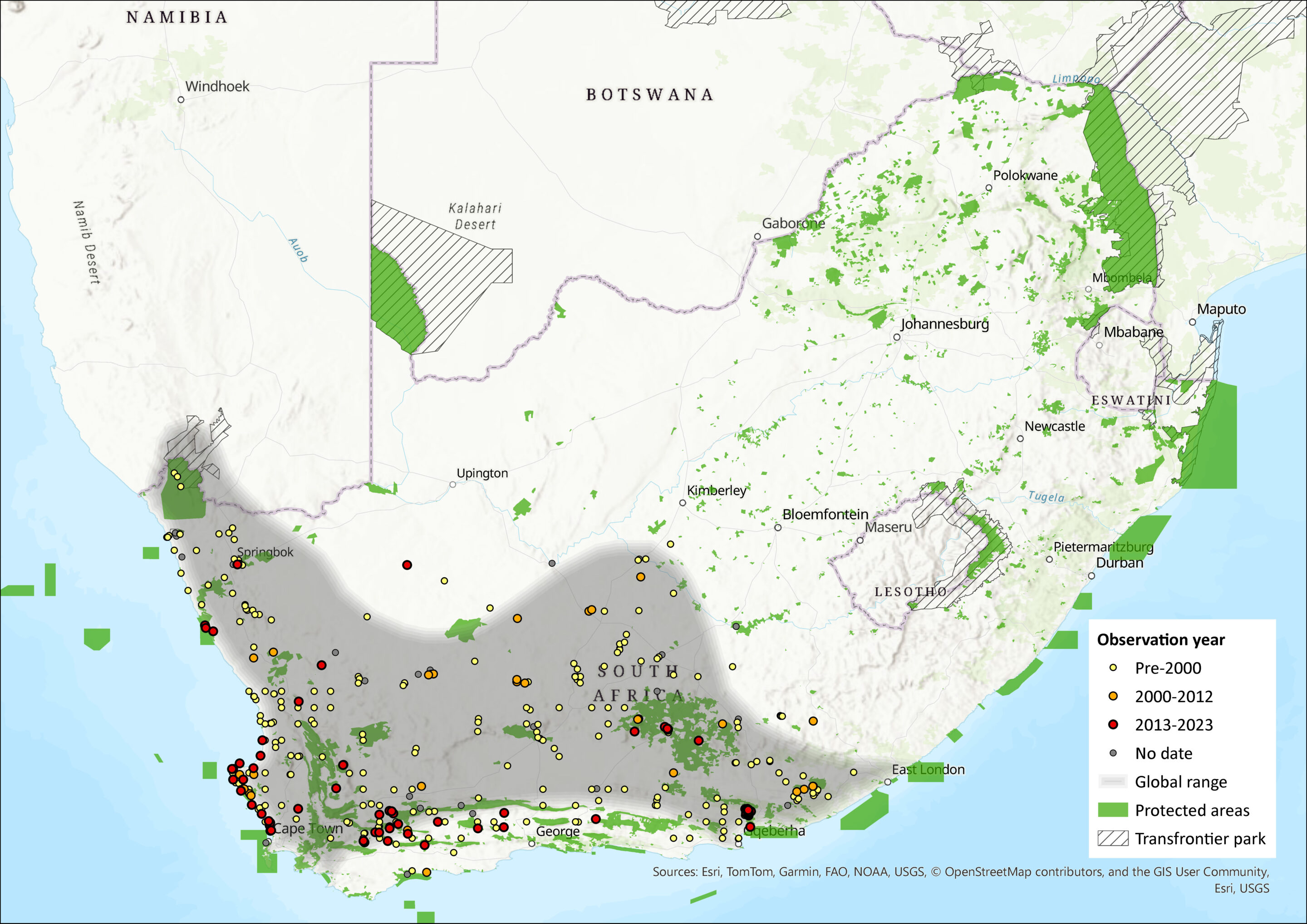
This species occurs throughout the semi-arid Succulent Karoo and Nama-Karoo of South Africa (Monadjem et al. 2015), specifically in the Eastern, Northern and Western Cape provinces, with some limited occurrence in the Fynbos Biome (Vermeulen & Nel 1988). It may marginally occur in southern Namibia, but further surveys are required to confirm this. Regardless, the bulk of the population occurs in South Africa. Kerley and Erasmus (1992) argued that the lodges built by this species are vulnerable to destruction by fire. As a result, they hypothesised that this shelter-building strategy is only viable in the absence of frequent burning, and therefore it is likely that the incidence of fire (or lack thereof) played an important role in shaping the range limits of this species. In addition, detailed studies have shown that niche separation between the Karoo Bush Rat and Brants Whistling Rat (Parotomys brantsii) occurs because the former selects areas with plant cover and dense foliage, while the latter selects areas with deep soil (du Plessis & Kerley 1991; du Plessis et al. 1992; Malan 2001; Jackson et al. 2002).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Karoo Bush Rat (Otomys unisulcatus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Namibia | Extant (surveys required to confirm this) | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Northern Cape Province | Extant | Native | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It is not known whether the species can persist in disturbed or modified habitats.
Population
Karoo Bush Rats are generally abundant in suitable habitat (see Habitats and Ecology). Little data are available on population densities per se. In the Great Fish River Nature Reserve (GFRNR), Eastern Cape, Le Gars (2005) trapped and marked 105 unique individuals in a 1.8 ha study area, corresponding to a density of 58 individuals / ha. For other studies, population density data can be calculated based on lodge density and average number of individuals per lodge. For example, in Postberg Nature Reserve, Western Cape, Vermeulen and Nel (1988) reported densities varying between 64 and 380 lodges / ha in four different study plots and found that most lodges were inhabited by one or two rats. This corresponds to population densities of between 64 and 760 individuals / ha over small, favourable habitat patches. Lower densities of 46 lodges / ha (Brown & Willan 1991) and 22–27 lodges / ha (Xalu 2009) were reported in the Eastern Cape. In terms of biomass, Malan (2001) estimated 89 kg / km2 for a Karroid Broken Veld habitat near Calitzdorp, Western Cape, conservatively assuming that one adult rat inhabits a nest. Both population densities and biomass data would have to be adjusted accordingly for habitats or during periods (years or seasons) where lodges are occupied by more or less individuals, on average. However, because Karoo Bush Rats often display a patchy distribution at the local scale, several lodges may be disused, and individual rats may exploit several neighbouring lodges (see Habitats and Ecology), more meaningful estimates at the landscape level can only be obtained through counting (marking) of individuals and including “barren” areas in the calculations. The current population size in the assessment region is unknown, but it is estimated to be well over 10,000 mature individuals considering the wide distribution range and the locally high densities reached by this species.
Population Information
Current population trend: Unknown but probably stable based on wide extent of occurrence and lack of threats.
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown, but probably > 10,000.
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No, not at the regional scale.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A study examining the mitochondrial DNA [mtDNA]) and morphological variation within O. unisulcatus detected two genetic assemblages (coastal and central) within the species (Edwards, 2009). The morphological evidence however suggests more fine-scale sub-structuring associated with biome boundaries (except for the Fynbos and Thicket Bushveld biomes which clustered together). Given that the mtDNA patterns are likely reflecting more distant relationships (gene flow), a more fine-scale nuclear (microsatellite) or genomic study would be beneficial and may detect the same structure uncovered by the morphometric dataset, which is associated with more recent adaptations.
Based on the available information, there is sufficient evidence for three geographically distinct populations. There is no evidence of population extinction. Therefore, the Convention on Biological Diversity’s populations maintained (PM) indicator receives a value of 1.0 (3/3).
No population estimates are available; hence the effective population size (Ne) and Ne 500 indicator cannot be quantified; however, it is expected that each of the three populations are genetically stable exceeding the 500 threshold. This should be verified using molecular techniques.
Habitats and ecology
The Karoo Bush Rat is found in Succulent Karoo habitat, Nama-Karoo and fynbos scrub. Contrarily to other vlei rats, it is mostly present in semi-arid and arid environments. Although this species is often found along the courses of ephemeral streams and rivers or near seasonal dams and pans, it is mostly the availability of fresh food and plant cover/dense foliage for shelter building (see below) that is determinant, as, similarly to all members of the subfamily Otomyinae, Karoo Bush Rats do not specifically drink water (Pillay et al. 1994). Surprisingly studies on thermoregulation and kidney morphology suggest that they are not physiologically well adapted to arid environments, having a higher than predicted basal metabolic rate and a thermoneutral zone at relatively low ambient temperature (du Plessis et al. 1989; Pillay et al. 1994). As a result, they can only cope with xeric conditions through using behavioural adaptations.
One unique feature of the Karoo Bush Rat is that it builds dome-shaped shelters or “lodges” made of sticks and
twigs, often using bushes and shrubs as supporting structures (see details in Brown 1987; Vermeulen & Nel 1988; du Plessis 1989; Le Gars 2005; Hoepfl 2013). Plant species used in this context often possess thorny branches and/or are densely tangled, such as Lycium spp., Acacia karoo, Grewia robusta or Exomys microphylla, and some of them are also important food sources. In the Little Karoo, Malan (2001) showed that the density of 76–150 cm high shrubs accounted for 60% of the variation in the densities of lodges, while at Tsolwana Game Reserve, Eastern Cape, the floristic composition and structure of the woody vegetation were critical determinants of habitat choice. Sticks and twigs are generally shorter than 20 cm in length (but some can be up to 50 cm long) and are collected in the surroundings and brought back to the lodge. Other materials locally and punctually used to build the lodges are as diverse as tree bark, pine needles, mussel shells, animal droppings, sheep wool, stones and bones, and even plastic and string. Lodge size varies tremendously within one site, possibly in relation to the time since construction, current use and number of rats occupying the lodge at any one time. The average diameter varies from 1.2–1.7 m (maximum 2.74 m) and the height from 0.38–0.52 m (maximum 0.75 m) depending on the study areas (Vermeulen & Nel 1988; Le Gars 2005; Hoepfl 2013). The volume occupied by the lodges may reach 4 m³ and represent nearly 40 kg of material. The lodges contain two to four internal chambers which are often lined up with grass (nests), as well as one to five latrines. Several entrances (sometimes over ten) associated to well-marked paths or “runways” surround the lodge. Lodges are interconnected by a
network of such runways which also leads to shrubs providing sticks and food. Shallow burrows are sometimes
excavated below the lodges, or nearby, and are also connected through runways (Le Gars 2005); in some
areas nest chambers are exclusively located underground (Brown & Willan 1991).
The stick lodges offer protection against avian and some terrestrial predators, as well as partially against harsh
environmental conditions such as extreme temperatures, wind and rain, because there is generally low plant cover in the Karoo regions (du Plessis & Kerley 1991; Kerley & Erasmus 1992; Schradin 2005). The temperature variation inside these shelters is lower than the ambient temperature variation, with temperatures during cold winter nights being about 4°C higher than outside and during hot summer days being 14°C lower than outside
(du Plessis et al. 1992). Several other rodent species may locally and/or seasonally use the lodges, and Karoo Bush Rats may compete with Rhabdomys spp. for favoured nesting sites, such as the shrub Lycium cinereum
(Schradin 2005). Lodges can also host amphibians and reptiles, including snake species such as Puffadders (Bitis arietans), Cape Cobras (Naja nivea) and Mole Snakes (Pseudapis cana), all potential predators of Karoo Bush
Rats.
The Karoo Bush Rat is a generalist herbivore, feeding on the leaves, stems, flowers and fruits of succulent karroid vegetation (Brown & Willan 1991; du Plessis et al. 1991; Hoepfl 2013). Locally, up to over 60 plants species have been recorded in the diet throughout the year (Kerley 1989; Kerley et al. 1991; Hoepfl 2013). This generalist dietary adaptation is to be expected in environments with low predictability of annual precipitation and plant growth, with perennials forming a stable dietary base. Karoo Bush Rats are both terrestrial and arboreal. They gather large amounts of plant material at the basis of, or on shrubs and bushes, which is then transported back to the nest for consumption, thereby minimising exposure to the macroclimate. Sometimes though, they feed on site, especially when active inside dense bushes (Photo 3). Like other Otomys species, Karoo Bush Rats practice coprophagy, hence eating their own faeces and those of other individuals. This is a particularly important habit in the weaning young, at it supplies them with the proper bacteria for the digestion of plant material. In addition, in the absence of free water in this species’ habitats, food –especially succulent plants – functions as the primary water source. The poor nutritive quality of the diet, requiring large volumes to be fermented in the caecum, is to some extent overcome by the rats collecting large quantities of food while foraging and consuming this later at the nest (Kerley 1989).
Activity patterns have only been studied in detail in the GFRNR. There radio-tracked Karoo Bush Rats showed an average activity budget of about 20% throughout the 24-hour cycle, with no difference between males and females. Rats exhibited a bimodal activity cycle, with a first peak in the morning (from 06:00–09:00) and a second one late afternoon (16:00–18:00), thereby avoiding being active during the hottest times of the day (> 40°C at midday in summer). The socio-spatial organization of this species seems to be variable, largely depending on cycles of rainfall and drought, and resulting variations in food availability and rat carrying capacity (Le Gars 2005; Xalu 2009). It also varies spatially within the same study site (N. Babu & E. Do Linh San unpubl. data). The basic social unit seems to be that of one male and one female sharing a common territory, with the current offspring. However, when conditions are favourable, groups using between one and four different nests may contain a second female and several subadults – likely offspring from previous litters that did not disperse due to high food availability and/or habitat saturation. Average number of individuals per nest varies between 0.36 and 3.78 (Vermeulen & Nel 1988; Le Gars 2005; Xalu 2009; Hoepfl 2013). In one extreme case, up to 11 different individuals had been observed on, or in close proximity to a lodge in a study site in the central Karoo plateau (Hoepfl 2013). In GFRNR, a maximum of 8 individuals – including a maximum of 6 adults, 6 juveniles, 5 males or 7 females – have been caught at a single nest at one given time (Le Gars 2005). Maximum numbers are likely higher for social units. Average maximal home range size of radio-tracked rats was about 590 m2 for males and 330 m2for females. Males were highly territorial. Females from different social units were also territorial, but females belonging to the same group had highly overlapping home ranges (N. Babu & E. Do Linh San unpubl. data).
In the central Karoo and in the Albany Thicket Biome, Karoo Bush Rats reproduce during the whole year, with
possibly an overall higher reproductive output during winter (Xalu 2009; Hoepfl 2013). The mean number of
young per litter in captivity averages 2.09 with a range of one to three (Pillay 2001). All males attain sexual maturity by 56 days (8 weeks, 70 g) and all females by 63 days (9 weeks, 77 g). The estimated gestation period is 37 days. Pillay (2001) suggested that the reproductive biology and postnatal development of O. unisulcatus appears to reflect a compromise between exploiting sporadically abundant resources and the occupation of stick lodges, which provide a favourable microclimate to buffer the harsh external climate.
Ecosystem and cultural services: Together with Brants’ Whistling Rat, the Karoo Bush Rat is a key prey species for Pale Chanting-Goshawk (Melierax canorus) in the Karoo, where the reproductive fitness of this diurnal bird of prey is associated with the Karoo Broken Veld vegetation type that contains the heuweltjies (raised soil mounds) and tall shrubs necessary to sustain high densities of both otomyine species (Malan 2001). It is likely that the Karoo Bush Rat is occasionally predated by other raptors, as well as herons and snakes (Hoepfl 2013). Karoo Bush Rat remains have also been found in the scats of a wide range of small carnivores, both diurnal (Cape Grey Mongoose Herpestes pulverulentus, Large Grey Mongoose Herpestes ichneumon, Yellow Mongoose Cynictis penicillata) and nocturnal (Water Mongoose Atilax paludinosus) (Stuart 1983; Cavallini & Nel 1990; Nqinana 2009; Hoepfl 2013; Bizani 2014).
Currently the hypothesis is tested that the building of lodges by O. unisulcatus might enrich soils with carbon and nutrients.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation |
– |
Suitable |
– |
Life History
|
Generation Length |
Justification |
Data Quality |
|
1-2 years |
see Pacifici et al. 2013 |
medium |
Age at maturity: female or unspecified: 6 weeks
Age at Maturity: Male: 6 weeks
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: 2 years
Average Reproductive Age: 5 weeks (Wolhuter et al. 2021)
Maximum Size (in cms): 15 cm (Pillay 2001)
Size at Birth (in cms): 6 cm (Pillay 2001)
Gestation Time: 37 days (Pillay 2001)
Reproductive Periodicity: seasonal breeding during the moist season
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: predation; starvation
Does the species lay eggs? no
Does the species give birth to live young: yes
Does the species exhibit parthenogenesis: no
Does the species have a free-living larval stage? no
Does the species require water for breeding? no
Movement Patterns
Movement Patterns: none
Congregatory: no
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
General notes regarding trade and use of this species: (Not specified)
Local Livelihood: no
National Commercial Value: none
International Commercial Value: none
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? no
Harvest Trend Comments: (Not specified)
Threats
There are no major identified threats to the species. However, the following two threats may reduce habitat quality:
- Overgrazing/browsing the vegetation around wetlands reduces habitat suitability and may cause local declines. The expansion of wildlife ranching will have to be monitored in this regard, as antelope overstocking may also affect wetland condition.
- Climate change may reduce area of occupancy significantly by drying up wetlands. For example, they are vulnerable to local extinctions following severe drought (Schradin 2005). As their lodges are comparatively poorer buffers from ambient temperature fluctuations than the burrows of Brants’ Whistling Rats (Jackson et al. 2002), this may make subpopulations increasingly threatened by climate change.
Overgrazing and climate change may synergise to cause non-linear and accelerating population decline. More research is needed to validate these hypotheses.
Habitat trend: Stable. This species inhabits arid areas that are unlikely to be transformed significantly. However, overgrazing may reduce habitat quality, and this should be monitored. For example, Masubelele et al. (2014) found a general increase in grass cover in eight sites in a 500 km ecotone between the Grassland and Nama-Karoo biomes between 1962 and 2009, and which is attributed to a general decrease in stocking rates in the region. More research is needed to assess the net loss or gain in habitat.
Conservation
The range of the species includes several protected areas, for example Postberg Nature Reserve (Vermeulen & Nel 1988), Tsolwana Game Reserve (Brown 1987), Addo Elephant National Park (E. Do Linh San pers. obs. 2014) and the GFRNR (Le Gars 2005; Xalu 2009). However, further protected areas should be documented. It is not known whether the species can persist in disturbed or modified habitats.
Although no specific interventions are necessary at present, the following interventions are likely to benefit the species:
- Wetland conservation and restoration: land managers should maintain a vegetation buffer to reduce impacts of land-use practices (Driver et al. 2012). Restoration will also create corridors between suitable habitat patches to allow for dispersal in responses to climate change.
- Holistic management of ranchlands: interventions including de-stocking, rotational grazing and buffering wetland vegetation are encouraged.
Recommendations for land managers and practitioners:
- Land managers should decrease stocking rates to conserve vegetation around wetlands.
- Long-term, systematic monitoring is needed to establish subpopulation trends and threat levels.
Research priorities:
- Effects of overgrazing and climate change affecting habitat suitability and population status.
- Fine scale studies on habitat loss and inferred impact on the species.
- Genetic research.
Encouraged citizen actions:
- Report Karoo Bush Rat records on virtual museum platforms (for example, iNaturalist and MammalMAP), both inside and outside protected areas. The presence of this species in an area is revealed by that of the conspicuous lodges it builds. Moist brownish pellets can be found next to some of the entrances or on the runways of lodges that are effectively inhabited. In addition, tell-tale piles of chopped sections of stem and other unpalatable parts are dropped under nearby bushes that are used as feeding areas.
Bibliography
Bizani M. 2014. Diet of the yellow mongoose (Cynictis penicillata) in the Albany Thicket Biome of South Africa. M.Sc. Thesis. University of Fort Hare, Alice, South Africa.
Brown ED. 1987. Comparative socio-ecology of Otomys irroratus and Otomys unisulcatus. M.Sc. Thesis. University of Fort Hare, Alice, South Africa.
Brown E, Willan K. 1991. Microhabitat selection and use by the bush Karoo rat Otomys unisulcatus in the Eastern Cape Province. South African Journal of Wildlife Research 21:69–74. Edwards S. 2009. Phylogeographic variation of the Karoo Bush Rat, Otomys unisulcatus: a molecular and morphological perspective. MSc thesis. Stellenbosch university. https://scholar.sun.ac.za/server/api/core/bitstreams/1bbdc457-ff42-4b2a-bc16-6bb5273647e4/content
Cavallini P, Nel JAJ. 1990. The feeding ecology of the Cape grey mongoose, Galerella pulverulenta (Wagner 1839) in a coastal area. African Journal of Ecology 28:123–130.
Driver A, Sink KJ, Nel JN, Holness S, Van Niekerk L, Daniels F, Jonas Z, Majiedt PA, Harris L, Maze K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
du Plessis A. 1989. Ecophysiology of the bush Karoo rat (Otomys unisulcatus) and the whistling Rat (Parotomys brantsii). M.Sc. Thesis. University of Port Elizabeth, South Africa.
du Plessis A, Erasmus T, Kerley GIH. 1989. Thermoregulatory patterns of two sympatric rodents: Otomys unisulcatus and Parotomys brantsii. Comparative Biochemistry and Physiology, Series A 94: 215–220.
du Plessis A, Kerley GIH. 1991. Refuge strategies and habitat segregation in two sympatric rodents Otomys unisulcatus and Parotomys brantsii. Journal of Zoology 224:1–10.
du Plessis A, Kerley GIH, Winter PED. 1991. Dietary patterns of two herbivorous rodents: Otomys unisulcatus and Parotomys brantsii in the Karoo. South African Journal of Zoology 26:51–54.
du Plessis A, Kerley GIH, Winter PED. 1992. Refuge microclimates of rodents: a surface nesting Otomys unisulcatus and a burrowing Parotomys brantsii. Acta Theriologica 37:351358.
Le Gars S. 2005. Social organization of Otomys unisulcatus, and cartography of their habitats and movements. Short-term research report. Université du Littoral, Dunkerque, France.
Jackson TP, Roper TJ, Conradt L, Jackson MJ, Bennett NC. 2002. Alternative refuge strategies and their relation to thermophysiology in two sympatric rodents, Parotomys brantsii and Otomys unisulcatus. Journal of Arid Environments 51:21–34.
Kerley GIH. 1989. Diet of small mammals from the Karoo, South Africa. South African Journal of Wildlife Research 19:67–72.
Kerley GIH, Erasmus T. 1992. Fire and the range limits of the bush Karoo rat Otomys unisulcatus. Global Ecology and Biogeography Letters 2:11–15.
Happold, M. and Happold, D.C.D. 2013. Chiroptera. In: M. Happold and D.C.D. Happold (eds), The Mammals of Africa, Volume IV Hedgehogs, Shrews, and Bats, Bloomsbury Publishing, London.
Hoepfl J. 2013. Aspects of the biology and behaviour of the Bush Karoo Rat (Otomys unisulcatus) in the Central Karoo, South Africa. Research report. African–Arabian Wildlife Research Centre, Loxton, South Africa.
IUCN. 2016. The IUCN Red List of Threatened Species. Version 2016-3. Available at: www.iucnredlist.org. (Accessed: 07 December 2016).
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-1. Available at: www.iucnredlist.org. (Accessed: 27 April 2017).
Makuya, L., Pillay, N. & Schradin, C. 2024. Kin based spatial structure in a solitary small mammal as indicated by GPS dataloggers. Animal Behaviour.215:45-54.
Malan G. 2001. Otomyine rodent refuge habitat: does it represent habitat quality for pale chanting-goshawk families? South African Journal of Wildlife Research 31:49–58.
Masubelele ML, Hoffman MT, Bond WJ, Gambiza J. 2014. A 50 year study shows grass cover has increased in shrublands of semi-arid South Africa. Journal of Arid Environments 104:43–51.
Mizerovská, D., Martynov, A. A., Mikula, O., Bryjová, A., Meheretu, Y., Lavrenchenko, L. A. & Bryja, J. 2023. Genomic diversity, evolutionary history, and species limits of the endemic Ethiopian laminate-toothed rats (genus Otomys, Rodentia: Muridae). Zoological Journal of the Linnean Society.
Monadjem A, Taylor PJ, Denys C, Cotterill FPD. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Nqinana A. 2009. Diet of the marsh mongoose Atilax paludinosus in the Andries Vosloo Kudu Nature Reserve (Eastern Cape Province, South Africa). B.Sc. Honours Thesis. University of Fort Hare, Alice, South Africa.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pillay N. 2001. Reproduction and postnatal development in the bush Karoo rat Otomys unisulcatus (Muridae, Otomyinae). J Zool 254:515–520. https:// doi. org/ 10. 1017/ S0952 83690 10010 17.
Pillay N, Willan K, Taylor P. 1994. Comparative renal morphology of some southern African otomyine rodent species. Acta Theriologica 39:37–48.
Pocock TN. 1976. Pliocene mammalian microfauna from Laangebaanweg: a few fossil genus linking the Otomyinae with the Murinae. South African Journal of Science 72:58–60.
Rathbun, G.B. (subeditor). 2005. Macroscelidea. In: J.D. Skinner and C.T. Chimimba (eds), The Mammals of the Southern African Subregion, 3rd edition, pp. 22-34. Cambridge University Press, Cambridge, UK.
Schradin, C. 2005. Nest site competition in two diurnal rodents from the succulent karoo of South Africa. J Mammal, 86, 757-765.
Stuart C. 1983. Food of the large grey mongoose Herpestes ichneumon in the south-west Cape Province. South African Journal of Zoology 18:401–403.
Vermeulen HC, Nel JAJ. 1988. The bush Karoo rat Otomys unisulcatus on the Cape West coast. South African Journal of Zoology 23:103–111.
Wolhuter, L., Thomson, J., Schradin, C. & Pillay, N. 2021. Life history traits of free-living bush Karoo rats (Otomys unisulcatus) in the semi-arid Succulent Karoo. Mammal Research, 67, 73-81.
Xalu M. 2009. Aspects of the population ecology of the bush Karoo rat, Otomys unisulcatus, in the Sam Knott Nature Reserve. BSc Honours Thesis. University of Fort Hare, Alice, South Africa.