
Hourglass Dolphin
Sagmatias cruciger

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Sagmatias cruciger – (Quoy & Gaimard, 1824)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Sagmatias – cruciger
Common Names: Hourglass Dolphin (English), Dauphin Crucigère, Lagénorhynque Crucigère (French), Delfín Cruzado (Spanish; Castilian)
Synonyms: Delphinus albigena (Quoy & Gaimard 1824); Delphinus bivittatus (Lesson & Garnot 1826); Electra clancula (Gray 1868); Electra cruciger (Gray 1870); Lagenorhynchius clanculus (Gray 1846); Lagenorhynchus latifrons (True 1889); Lagenorhynchus supercillosus (Trouessart 1904); Lagenorhynchus wilsoni (Lillie 1915); Phocaena crucigera (Philippi 1893); Phocaena d’orbignyi (Philippi 1893)
Taxonomic Note:
A recent taxonomic revision of the genus Laegenorhynchus that incorporated multiple lines of evidence (morphology, genetics, historical biogeography, acoustics and phylogenetics) consistently reported that the six species currently assigned to Lagenorhynchus do not form a monophyletic group (Vollmer et al. 2019). While Lagenorhynchus albirostris (White-Beaked Dolphin) has been retained, the remaining five species have been reassigned to existing generic names, namely Leucopleurus acutus (Atlantic White-Sided Dolphin), Sagmatias australis (Peale’s Dolphin), Sagmatias cruciger (Hourglass Dolphin), Sagmatias obliquidens (Pacific White-Sided Dolphin), and Sagmatias obscurus (Dusky Dolphin).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Probert, R.1 & da Silva, J. M.2
Reviewer: Smith, C.3
Institutions: 1Department of Botany and Zoology, Stellenbosch University, 2South African National Biodiversity Institute, 3Endangered Wildlife Trust
Previous Assessors: Plön, S. & Cockcroft, V.
Previous Reviewer: Child, M.F.
Contributors: Relton, C., Atkins, S., Findlay, K., Meyer, M., Oosthuizen, H. & Elwen, S.
Assessment Rationale
Although this species is frequently sighted in Antarctic waters, and has a circumpolar and widespread distribution, it is one of the least-known of all dolphin species. This species is considered fairly abundant within its range, and no major threats have been recognised. Thus, in line with the global assessment, the Hourglass Dolphin is listed as Least Concern within the assessment region.
Regional population effects: The Hourglass Dolphin is wide-ranging throughout the pelagic waters of the southern oceans, and no obvious barriers to dispersal have been identified, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Probert R & da Silva JM. 2025. A conservation assessment of Sagmatias cruciger. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Hourglass Dolphin has a circumpolar distribution within the higher latitudes of the Southern Ocean (Goodall 1997; Goodall et al. 1997; Brownell & Donahue 1998). It is one of three species of delphinid which is considered truly Antarctic (Boyd 2002), and the only delphinid regularly occurring in the Antarctic Polar Front (Brownell & Donahue 1998). They predominantly inhabit sub-Antarctic and Antarctic waters between 45° and 67°S, and although primarily occur offshore, they are occasionally sighted along banks or islands (Goodall 1997, 2002). A recent study into the occurrence of Hourglass Dolphins in the south-west Atlantic and Southern Oceans (Dellabianca et al. 2012) found the presence of two group separated by environmental conditions. The first of these groups occurs in shallow, coastal waters, while the other is primarily located in deeper, colder regions offshore (Dellabianca et al. 2012). Other localised concentrations have been identified around the southern tip of South America, South Georgia, the Falkland Islands (Goodall 1997; Goodall et al. 1997), and north of the South Shetland Islands in the southern Drake Passage (Santora 2012). Although the northern extent of its range is uncertain (there are occasional records from around 33°S), the southern parameters of its distribution extend to the ice-edges in the south. Within the assessment region, this species occurs around the Prince Edward Islands. The Hourglass Dolphin is the only small delphinid that regularly frequents areas south of the Antarctic Convergence and along the Agulhas Current.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
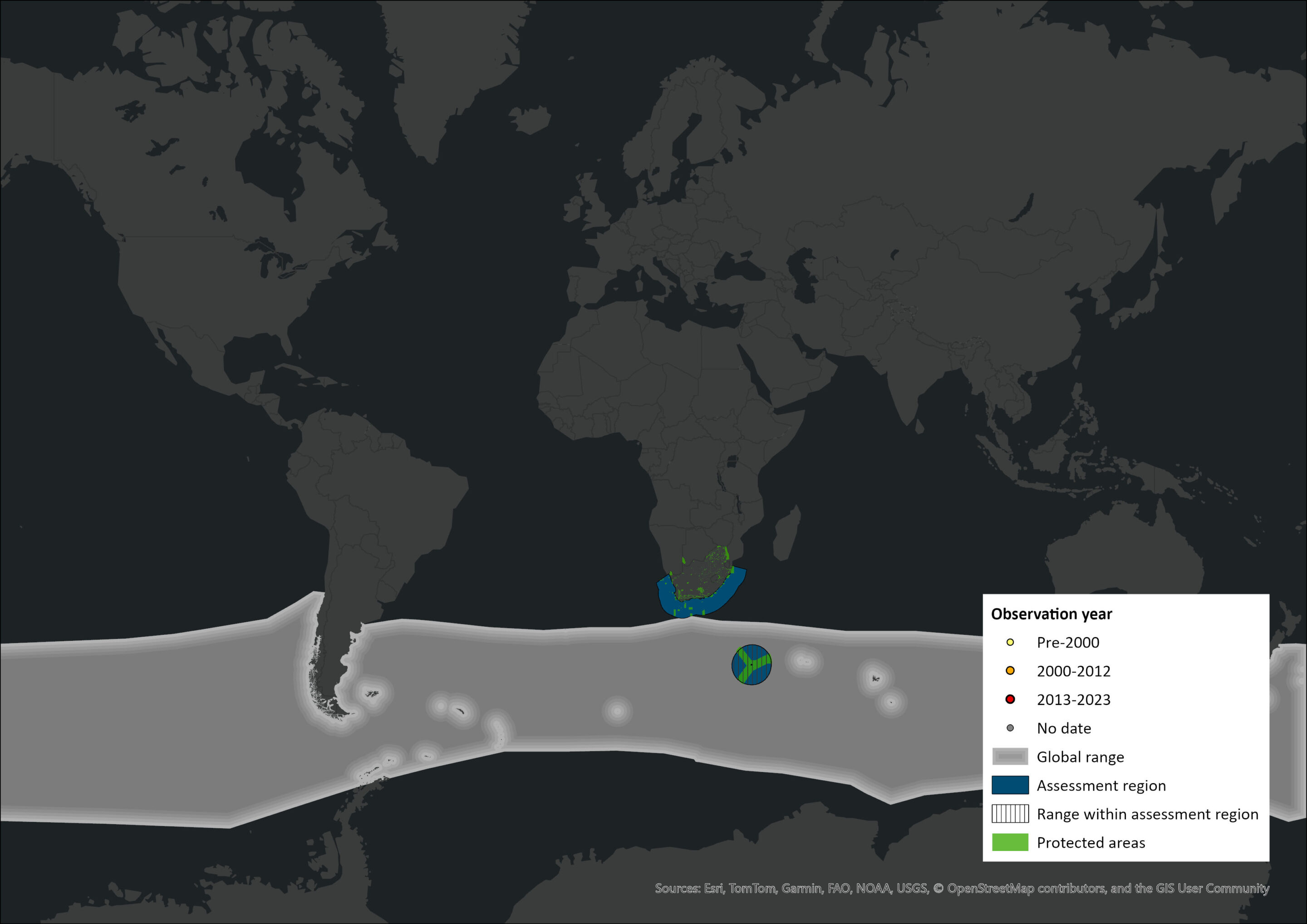 Figure 1. Distribution records for Hourglass Dolphin (Sagmatias cruciger) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Hourglass Dolphin (Sagmatias cruciger) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Antarctic, Australasian, Neotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Antarctica | Extant | Native | – | – |
| Argentina | Extant | Native | – | – |
| Australia | Extant | Native | – | – |
| Bouvet Island | Presence Uncertain | Native | – | – |
| Chile | Extant | Native | – | – |
| Falkland Islands (Malvinas) | Extant | Native | – | – |
| French Southern Territories | Extant | Native | – | – |
| French Southern Territories -> Crozet Is. | Extant | Native | – | – |
| French Southern Territories -> Kerguelen | Extant | Native | – | – |
| New Zealand | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Africa -> Marion-Prince Edward Is. | Extant | Native | – | – |
| South Georgia and the South Sandwich Islands | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 41. Atlantic – southwest | Extant | Native | – | – |
| 47. Atlantic – southeast | Extant | Native | – | – |
| 48. Atlantic – Antarctic | Extant | Native | – | – |
| 57. Indian Ocean – eastern | Extant | Native | – | – |
| 58. Indian Ocean – Antarctic | Extant | Native | – | – |
| 81. Pacific – southwest | Extant | Native | – | – |
| 87. Pacific – southeast | Extant | Native | – | – |
| 88. Pacific – Antarctic | Extant | Native | – | – |
Climate change
Hourglass Dolphins are cooler and warm water-limited (CWWL), with a generally narrow temperature range, ranging from -0.3 to 13.4 °C with a mean of 4.8°C (Goodall 1997, 2002; MacLeod 2009). The Prince Edward Islands are influenced by the surrounding cooler Antarctic Circumpolar Current, and inputs from the warmer Agulhas Current. Over the years, significant climatic shifts have been documented in the Agulhas Current System and around the Prince Edward Islands. The average sea surface temperature (SST) in the Agulhas Bank increased by 1.5°C between the 1980s and the early 2000s (Mead et al. 2013) and is estimated to further increase by 2 °C by 2100 (Asdar et al. 2022). Mean SSTs within the Prince Edward Islands have increased by 1.4 °C over the past 50 years (up to 2000, Melice et al. 2003). There are no recent SST estimates and future predictions for this region. As a CWWL species, an increase in SST may alter the distribution and movement patterns of Hourglass Dolphins, decreasing their northward distribution boundary resulting in unfavourable conservation implications (MacLeod 2009; Bhagarathi et al. 2024).
The Prince Edward Islands, much like other sub-Antarctic islands, form very sensitive ecosystems, where changes in ocean dynamics associated with the Antarctic Circumpolar Current can impact the foraging behaviour of top predators (Ansorge et al. 2009). Allan et al. (2013) demonstrated long-term declines in phytoplankton productivity along the shelf of the Prince Edward Islands, which have influenced the productivity of the surrounding ecosystem and, thus, impacted populations of top predators (various species of sea birds and seals). Stomach contents from two Hourglass Dolphins from Patagonian waters revealed a diet of various cephalopods (primarily squid), fish (lanternfish and hake), and crustaceans. Although the negative impacts of climate change on cetacean species through shifts in prey are unknown, the climate-driven changes in productivity are also likely to affect the prey of Hourglass Dolphins, as well as other delphinid species feeding in this region, as many cephalopod (including squid) and fish (including lanternfish) species in the Southern Ocean are likely to be impacted by climate change (Caccavo et al. 2021). Furthermore, shifts in prey availability may result in increased competition (Bhagarathi et al. 2024), which may negatively impact predator populations.
Population
There are no recent global estimates of abundance for this species, although Kasamatsu and Joyce (1995) integrated abundance and distribution data collected during sighting surveys from 1976/77 to 1987/88, generating a population estimate for the region south of the Antarctic Convergence of 144,300 (CV = 17%). Additionally, during the Southern Hemisphere Minke Whale Assessment Cruises (IWC/ IDCR), which took place between 1978/79 and 1987/88, 233 schools of Hourglass Dolphins were recorded, including a total of 1,634 individuals (Kasamatsu et al. 1990). No records of abundance are available for the assessment region, where they occur around the Prince Edward Islands.
| Continuing decline in mature individuals? | Qualifier | Justification |
| Unknown | – | – |
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
No population genetic study has been undertaken on this species; however, because it is wide-ranging throughout the pelagic waters of the southern oceans, and there are no obvious barriers to dispersal identified, it is expected to exist as a single population.
No population sizes have been recorded within the assessment region, and global estimates are very outdated (see Kasamatsu & Joyce 1995), making it difficult to estimate the effective population size.
Habitats and ecology
This species is primarily associated with waters exhibiting sea surface temperatures ranging from 0.3–13.4 °C (at an average of 4.8 °C; Goodall 1997), and within deep, open ocean waters (Dellabianca et al. 2012), but has also occasionally been sighted in the region of the Antarctic Peninsula where waters become fairly shallow, and at temperatures as low as -0.3 °C (Goodall 2002). Around southern South America and the Antarctic Peninsula, this species was recorded in groups ranging from 1–14 individuals, during 27 sightings, mostly around islands or banks, in waters less than 200 m deep (Goodall & Polkinghorn 1979). Due to its pelagic distribution and the shortage of stranding records, very little information is available regarding the habitat and diet preferences of this species, as well as its interactions with other marine mammals (Ashford et al. 1996; Goodall et al. 1997; Fernández et al. 2003; Goodall 2009).
An assessment of the stomach contents of two individuals from Patagonia was conducted by Fernández et al. (2003), revealing a diet of squid (primarily Semirossia tenera and Loligo gahi), Argentine Hake (Merluccius hubbsi), pelagic lanternfish Protomyctophum spp., several crustaceans, and other cephalopods, such as the benthic Eledone massyae, and the demersal-pelagic Illex argentinus. Although no specific predators have been identified, considering the distribution of Hourglass Dolphins south of the Antarctic Convergence, the Killer Whale (Orcinus orca) may very likely prey upon this species (Brownell & Donahue 1998).
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales, and therefore they are prime sentinels of marine ecosystem change; polar cetaceans are useful for assessing the effects of rapid changes in sea ice conditions on food webs in these strongly seasonal ecosystems (Moore 2008).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Marginal | – |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
Although one scientific sample was collected during commercial whaling operations, and a number of other individuals have been caught during research cruises (Brownell & Donahue 1998), there is considered to be no contemporary trade or use of this species.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There is very little anthropogenic influence within their distribution and hence there are no known major threats to this species. However, there may be some risk of entanglement in deep-sea fisheries set in the southern oceans, even though records of this are extremely rare. A Japanese experimental drift net fishery, which was set for squid in the southern Indian Ocean, incidentally, caught at least one individual (Brownell & Donahue 1998).
MacLeod (2009) suggests that some cetacean species are more vulnerable to the effects of climate change than others, due to certain ecological characteristics, such as a preference for non-tropical regions and shelf waters, which are exhibited by most porpoises and two of the four Sagmatias species (previously identified as Lagenorhynchus species). In fact, a framework for assessing predicted range changes in response to climate change revealed that all four Sagmatias species have either a high or unfavourable risk of extinction of at least one population (MacLeod 2009). Additionally, a range contraction is predicted for Hourglass Dolphins, as they are expected to track a range of water temperatures, to which they are adapted. Furthermore, indirect effects of climate change include changes in prey availability, which in turn, may affect dolphin abundance, distribution, movement patterns, social structure, reproductive rate, as well as their susceptibility to disease and the build-up of toxins (Learmonth et al. 2006).
Conservation
The Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) listed the Hourglass Dolphin on Appendix II, and this species is also protected by national legislation under the Marine Living Resources Act (No. 18 of 1998). Although no species-specific conservation initiatives have been identified for the Hourglass Dolphin, it is likely that this species would benefit from the ongoing collection of sightings records during ship-based surveys directed at other species, in order to monitor the abundance and distribution of this unfamiliar species. Additionally, comprehensive data collection of bycatch in deep-sea fisheries is necessary in order to determine the severity of this threat to Hourglass Dolphins within the assessment region.
Recommendations for managers and practitioners:
- Accurately assess bycatch in the Southern Ocean deep-sea fisheries.
- Enforce regulations associated with deep water fisheries, including bycatch mitigation efforts.
- Record sightings data during systematic monitoring of other marine species.
Research priorities:
- Basic life history parameters, population size, structure and trends within the waters around the Prince Edward Islands.
- Bycatch assessments in deep-sea Southern Ocean fisheries.
- Current threats, including possible range shifts or indirect effects on prey resources, associated with global climate change.
Encouraged citizen actions:
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, e.g. wwfsa.mobi, FishMS 0794998795.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution.
Bibliography
Allan, L. E., Froneman, W. P., Durgadoo, J. V., McQuaid, C. D., Ansorge, I. J., & Richoux, N. B. 2013. Critical indirect effects of climate change on sub‐A ntarctic ecosystem functioning. Ecology and Evolution 3(9): 2994-3004.
Ansorge, I. J., Durgadoo, J. V., & Pakhomov, E. A. 2009. Dynamics of physical and biological systems of the Prince Edward Islands in a changing climate. In Papers and Proceedings of the Royal Society of Tasmania (Vol. 143, No. 1, pp. 15-18).
Asdar S, Jacobs ZL, Popova E, Noyon M, Sauer WH, Roberts MJ. 2022. Projected climate change impacts on the ecosystems of the Agulhas Bank, South Africa. Deep Sea Research Part II: Topical Studies in Oceanography 200: 105092.
Ashford JR, Rubilar PS, Martin AR. 1996. Interactions between cetaceans and longline fishery operations around South Georgia. Marine Mammal Science 12: 452-457.
Bhagarathi, L. K., DaSilva, P. N., Maharaj, G., Balkarran, R., Baksh, A., Kalika-Singh, S., … & Cossiah, C. 2024. The impact of climate change on the ecology, reproduction and distribution of marine mammals and the possible legislation, conservation and management approaches to protect these marine mammal species: A systematic review. Magna Scientia Advanced Biology and Pharmacy 13: 045-084.
Boyd IL. 2002. Antarctic marine mammals. In: Perrin WF, Würsig B, Thewissen JGM (ed.), Encyclopedia of Marine Mammals., pp. 30-36. Academic Press, San Diego, California, USA.
Brownell Jr., R. L. and Donahue, M. A. 1999. Hourglass dolphin Lagenorhynchus cruciger (Quoy and Gaimard, 1824). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Vol. 6: The second book of dolphins and the porpoises, pp. 121-135. Academic Press.
Caccavo, J. A., Christiansen, H., Constable, A. J., Ghigliotti, L., Trebilco, R., Brooks, C. M., … & Xavier, J. C. 2021. Productivity and change in fish and squid in the Southern Ocean. Frontiers in Ecology and Evolution 9: 624918.
Dellabianca N, Scioscia G, Schiavini A, Rey AR. 2012. Occurrence of hourglass dolphin (Lagenorhynchus cruciger) and habitat characteristics along the Patagonian Shelf and the Atlantic Ocean sector of the Southern Ocean. Polar Biology 12: 1921-1927.
Fernández M, Beron-Vera B, Raga JA, Crespo EA. 2003. Food and parasites from two hourglass dolphins, Lagenorhynchus cruciger (Quoy and Gaimard, 1824), from Patagonian waters. Marine Mammal Science 19: 832-836.
Goodall RNP, Polkinghorn JT. 1979. Preliminary report on sightings of small cetaceans off southern South America and the Antarctic Peninsula. Paper SC/31/SM2 presented to the International Whaling Commission Scientific Committee.
Goodall, R. N. P. 1997. Review of sightings of the hourglass dolphin, Lagenorhynchus cruciger, in the South American sector of the Antarctic and the sub-Antarctic. Reports of the International Whaling Commission 47: 1001-1014.
Goodall, R. N. P. 2002. Hourglass dolphin Lagenorhynchus cruciger. In: W. F. Perrin, B. Wursig and J. G. M. Thewissen (eds), Encyclopedia of Marine Mammals, pp. 583-585. Academic Press, San Diego, California, USA.
Goodall, R. N. P. 2009. Hourglass dolphin Lagenorhychus cruciger. In: Perrin, W. F., Würsig, B., and Thewissen, J. G. M. (eds), Encyclopedia of Marine Mammals, pp. 573-576. Academic Press, Amsterdam.
Goodall, R. N. P., Baker, A. N., Best, P. B., Meyer, M. and Miyazaki, N. 1997. On the biology of the hourglass dolphin, Lagenorhynchus cruciger (Quoy and Gaimard, 1824). Reports of the International Whaling Commission 47: 985-999.
Hammond, P. S., Bearzi, G., Bjørge, A., Forney, K., Karczmarski, L., Kasuya, T., Perrin, W. F., Scott, M. D., Wang, J. Y., Wells, R. S., and Wilson, B. 2008. Lagenorhynchus cruciger. In ‘The IUCN Red List of Threatened Species’. Version 2012.2. Available at: www.iucnredlist.org. (Accessed: 13 April 2013).
IUCN (International Union for Conservation of Nature). 2012. Lagenorhynchus cruciger. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org.. Downloaded on 21 February 2016
Kasamatsu F, Joyce G, Ensor P, Mermoz J. 19990. Current occurrence of cetacea in the Southern Hemisphere minke whale assessment cruises, 1978/79–1987/88. Paper SC/42/015 presented to the International Whaling Commission Scientific Committee.
Kasamatsu, F. and Joyce, G.G. 1995. Current status of odontocetes in the Antarctic. Antarctic Science 7(4): 365-379.
LeDuc, R.G., Perrin, W.F. and Dizon, A.E. 1999. Phylogenetic relationships among the delphinid cetaceans based on full cytochrome b sequences. Marine Mammal Science 15: 619-648.
Learmonth, J.A., Macleod, C.D., Santos, M.B., Pierce, G.J., Crick, H.Q.P. and Robinson, R.A. 2006. Potential effects of climate change on marine mammals. Oceanography and Marine Biology: An Annual Review 44: 431-464.
MacLeod, C.D. 2009. Global climate change, range changes and potential implications for the conservation of marine cetaceans: a review and synthesis. Endangered Species Research 7: 125-136.
Mead JG, Brownell RL. 1993. Order Cetacea. In: Wilson DE, Reeder DM (ed.), Mammal Species of the World: A taxonomic and geographic reference, pp. 349-364. Smithsonian Institution Press, Washington, DC, USA.
Mead A, Griffiths CL, Branch GM, McQuaid CD, Blamey LK, Bolton JJ, Anderson RJ, Dufois F, Rouault M, Froneman PW, Whitfield AK. 2013. Human-mediated drivers of change—impacts on coastal ecosystems and marine biota of South Africa. African Journal of Marine Science 35(3):403-25.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Santora, J.A. 2012. Habitat use of hourglass dolphins near the South Shetland Islands, Antarctica. Polar biology 35: 801-806.
Vollmer, N. L., Ashe, E., Brownell Jr, R. L., Cipriano, F., Mead, J. G., Reeves, R. R., … & Williams, R. 2019. Taxonomic revision of the dolphin genus Lagenorhynchus. Marine Mammal Science 35(3): 957-1057.
