Hottentot’s Golden Mole
Amblysomus hottentotus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Amblysomus hottentotus – (A. Smith, 1829)
ANIMALIA – CHORDATA – MAMMALIA – AFROSORICIDA – CHRYSOCHLORIDAE – Amblysomus – hottentotus
Common Names: Hottentot’s Golden Mole, Zulu Golden Mole (English), Hottentot-gouemol, Hotnot-Kruipmol (Afrikaans), Hottentotten-Goldmull (German)
Synonyms: Chrysochloris hottentotus A. Smith, 1829; Amblysomus iris Thomas & Schwann, 1905
Taxonomic Note:
Traditionally taken to include populations that Bronner (1996, 2000) recognised as valid species, namely A. septentrionalis, A. robustus, A. marleyi and A. corriae (in part).
Previously included five subspecies: hottentotus, pondoliae, iris, longiceps and meesteri (Bronner 1995, 2013, Taylor et al. 2018). However, recent phylogenetic analyses revealed substantial population structure (Mynhardt et al. 2015, 2020), including nesting of A. robustus and A. septentrionalis within the larger clade of A. hottentotus. Pending further study, we recognise four of the five subspecies as part of A. hottentotus, elevating A. meesteri to species status (Gilbert et al. 2008, Mynhardt et al. 2015, 2020, Bronner et al. 2024).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Mynhardt, S.1 & da Silva, J.2
Reviewers: Asher, R.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3University of Cambridge
Previous Assessors: Bronner, G. & Mynhardt, S.
Previous Reviewer: Taylor, A.
Previous Contributors: Child, M.F. & Relton, C.
Assessment Rationale
A widespread and adaptable species that does not appear to be in decline. Listed as Least Concern in view of its relatively large distribution, presumed large population, ability to thrive in mildly-transformed habitats and its occurrence in a number of protected areas.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Mynhardt S & da Silva JM. 2025. A conservation assessment of Amblysomus hottentotus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
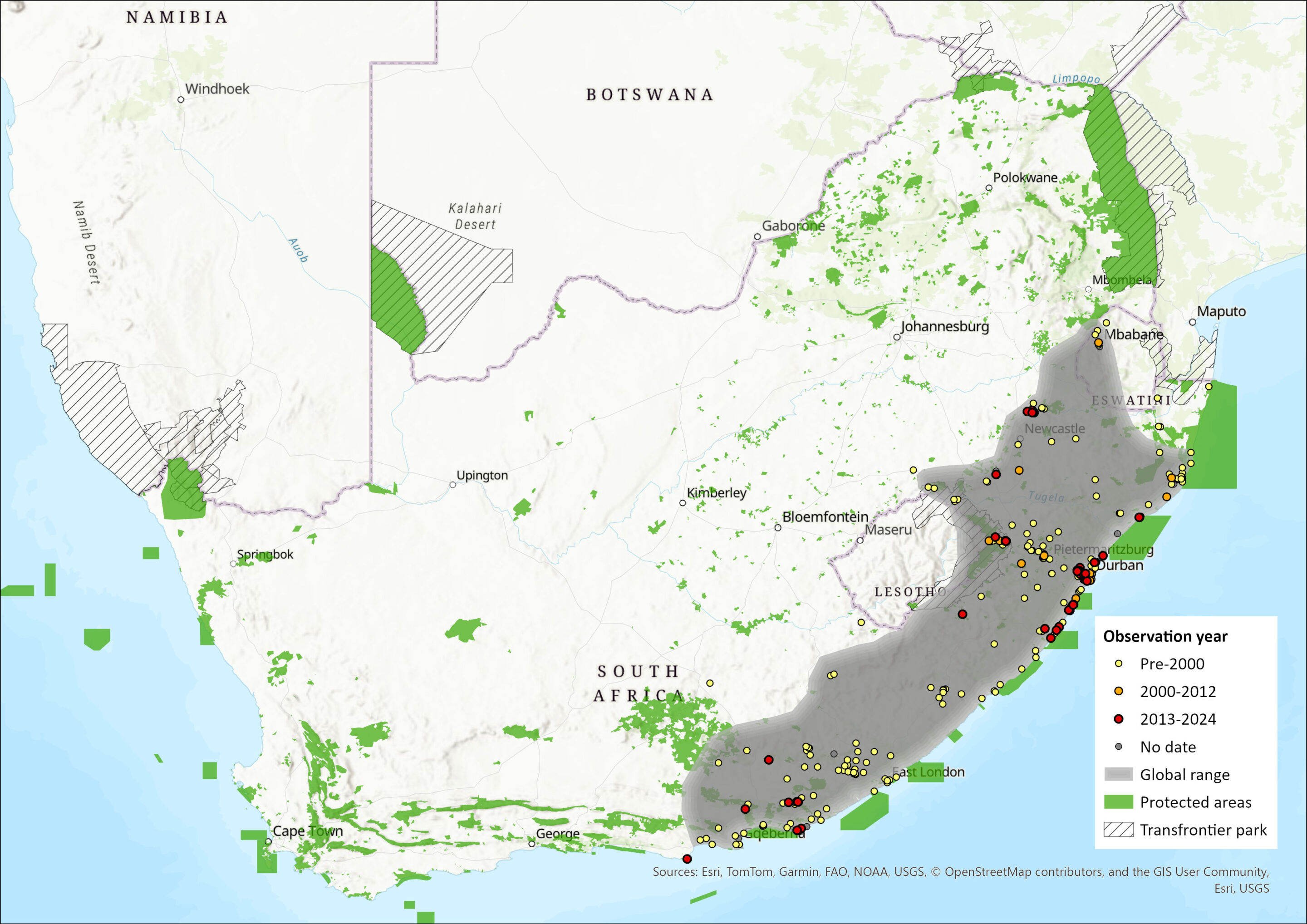
This species as presently known is found in South Africa and possibly also Swaziland (but whether the Swaziland specimens represent this species of Amblysomus septentrionalis, or both, awaits confirmation by genetic data). Occurs coastally from the Eastern Cape, in the vicinity of Van Staden’s River, northwards to St Lucia district in KwaZulu-Natal. Ranges inland to the foot of the Drakensberg escarpment, from Maclear/Ugie in the south to Van Reenen in the north, possibly with a marginal intrusion into northeastern Free State (Bronner 2013). Previously reported from Lesotho, based on a misidentified specimen (representing Chlorotalpa sclateri); a marginal occurrence in Lesotho in the northern Drakensberg (near Bethlehem) cannot, however, be discounted as species limits and distributions of this taxon and A. septentrionalis await clarification.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
MAP
Figure 1. Distribution records for Hottentot’s Golden Mole (Amblysomus hottentotus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Presence Uncertain | Native | – | – |
| Lesotho | Presence Uncertain | Origin Uncertain | – | – |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
| South Africa -> KwaZulu-Natal | Extant | Native | – | Resident |
| South Africa -> Mpumalanga | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Although the potential impacts of climate change on this species have not been directly assessed, climate change is expected to impact most golden mole species. Since these subterranean animals are restricted to soft soils for burrowing, and have very low vagility, their ability to migrate or shift their distribution ranges in response to climate change is very limited. Climate change is likely to impact golden moles through changes in vegetation type, soil moisture and prey availability, and coastal species additionally through reduced available habitat in developed/developing areas, as rising sea levels reduce the area of undeveloped coastal dune habitats between the coast and its associated developments.
Population Information
Common throughout range. Densities of up to 25 individuals/ha recorded in prime habitat (inferred from Kuyper 1985, Bronner 2013).
Current population trend: Unknown
Continuing decline in mature individuals? No
Severely fragmented: No
Extreme fluctuations in number of mature individuals: No
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: (Not specified)
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Recent genomic work using mtDNA and RADseq data has greatly informed the structure and composition of Amblysomus hottentotus across its distribution (Mynhardt et al. 2015; 2020). Phylogenetic analyses identified two highly divergent lineages – one associated with A. hottentotus and the other with the newly described A. meesteri (Mynhardt et al. 2020, Bronner et al. 2024).
Within A. hottentotus, additional structure was identified, with four, possibly five genetically distinct subpopulations identified within the species. These subpopulations include: 1) A. h. hottentotus (Eastern Cape, from Grahamstown to East London and inland to King Williams Town), 2) A. h. longiceps (Drakensberg), 3) A. h. pondoliae (KZN South coast), and 4) A. h. iris (KZN north coast, from Durban northwards to St. Lucia). A fourth genetically distinct subpopulation may be attributed to animals from Umtata, however, further investigation incorporating increased sampling is needed to verify the validity of this lineage, as results were inconsistent across analyses (Mynhardt et al. 2015; 2020). Accordingly, at this time, four genetically distinct subpopulations are recognised, all of which are currently extant within the assessment region.
While the effective population sizes (Ne) for each of the three subpopulations have not been quantified, given that they are considered common throughout their range, it is likely each subpopulation is genetically stable and healthy (e.g., Ne > 500). Verification of this can be made by analysing the existing RADseq data.
Habitats and ecology
This species occurs predominantly within the mesic eastern regions of South Africa, across a broad range of habitats from Afromontane and coastal dune forests to marshes, temperate grasslands and woodland savannahs (Skinner and Chimimba 2005). There is marginal intrusion of this species into the Fynbos and Nama-Karoo biomes in the southern part of its range. They seem to prefer moist soils near water sources, however they are able to survive far from water providing that soils remain soft enough for burrowing, and invertebrate prey is common. They adapt easily to modified landscapes, and are frequently associated with agricultural areas, golf courses and gardens but are less common in exotic plantations.
Similar to other golden moles, this species usually consumes earthworms and insects; however, it has also been documented feeding on snails and plant material, for example potatoes and bulbs (Kuyper 1985). Although largely solitary, A. hottentotus has been recorded in sympatry with African Mole-rats (Cryptomys hottentotus), even to the point of sharing burrow systems. Hottentot Golden Moles breed aseasonally, however they are significantly less active in winter than summer, and will burrow deeper during the colder winter months, often entering a state of torpor (Skinner and Chimimba 2005). Generally, this species produces two young per litter (Bernard et al. 1994, Schoeman et al. 2004). Studies have shown that when wet, a layer of air retained in their fur allows them to maintain a certain degree of buoyancy, thus enabling them to swim (Kuyper 1985, Hickman 1986). This becomes a valuable survival technique when burrows flood during heavy rainfall events.
Ecosystem and cultural services: This species is not known to provide any specific ecosystem services, but this may simply reflect the paucity of information available for this poorly-known species. They have, however, been identified as a source of food for predators, such as Barn Owls (Tyto alba). They become vulnerable to predation when they leave the safety of their burrows to feed, as well as following rainfall events, when activity increases substantially (Skinner & Chimimba 2005).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | Yes |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | Yes |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | Yes |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | Yes |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | No |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Marginal | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | Yes |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | No |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Marginal | – |
| 14.2. Artificial/Terrestrial -> Artificial/Terrestrial – Pastureland | – | Suitable | No |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Suitable | No |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
| 16. Introduced vegetation | – | Marginal | – |
Continuing decline in area, extent and/or quality of habitat: No. However, across the Eastern Cape, Mpumalanga and KwaZulu-Natal Provinces, urban development has increased by 6–11%, and rural development by 1–7%, between 2000 and 2013 (GeoTerraImage 2015). Similarly, there is an ongoing loss of natural habitat in KwaZulu-Natal at an average of 1.2% per year since 1994 (Jewitt et al. 2015).
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: 10-14
Size at Maturity (in cms): Male: 11-14
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): 4-5
Gestation Time: (Not specified)
Reproductive Periodicity: Aseasonal with enhanced fecundity in wet summer months
Average Annual Fecundity or Litter Size: Litter size 1-3 (usually 2)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
.
General Use and Trade Information
General notes regarding trade and use of this species: This species is not known to be utilised or traded in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Major threats include habitat transformation in coastal urban and resort areas, particularly on the “north coast”, just north of Durban. Inland subpopulations may be facing threats related to poor agricultural practices in some parts of their distribution range, as well as habitat transformation due to alien invasives. Inferred minor threats include persecution and poisoning by landowners and predation by domestic dogs and cats.
Conservation
The species is adequately conserved in many protected areas; see Bronner (1995) for a list of these. Currently, no specific interventions are required for this species. However, this may change should molecular research reveal more range-restricted endemic species.
Recommendations for land managers and practitioners: None
Research priorities:
- Studies on subpopulation sizes, trends and distributions.
- Studies assessing the severity of threats faced by this species.
- Molecular studies to disentangle the species complex.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Deposit any dead specimens found in a state or provincial museum, together with information on the date and site where found.
- Create indigenous vegetation gardens.
Bibliography
Afrotheria Specialist Group. 2014. Specialist Group website. Available at: http://www.afrotheria.net/index.php.
Bernard, R.T.F., Bronner, G.N., Taylor, P.J., Bojarski, C. and Tsita, J.N. 1994. Aseasonal reproduction in the Hottentot golden mole, Amblysomus hottentotus, from the summer rainfall region of South Africa. South African Journal of Science 90: 547-549.
Bronner, G.N. 1995. Systematic revision of the golden mole genera Amblysomus, Chlorotalpa and Calcochloris (Insectivora: Chrysochloromorpha; Chrysochloridae). Ph.D. Thesis, University of Natal.
Bronner, G.N. 1995. Systematic revision of the golden mole genera Amblysomus, Chlorotalpa and Calcochloris (Insectivora: Chrysochloromorpha; Chrysochloridae). Ph.D. thesis, University of Natal (Durban), South Africa.
Bronner, G.N. 1996. Geographic patterns of morphometric variation in the Hottentot golden mole, Amblysomus hottentotus (Insectivora: Chrysochloridae): a multivariate analysis. Mammalia 60: 729–751.
Bronner, G.N. 2000. New species and subspecies of golden mole (Chrysochloridae: Amblysomus) from Mpumalanga, South Africa. Mammalia 64: 41–54.
Bronner, G.N. 2013. Amblysomus hottentotus. In: J. Kingdon, D. Happold, T. Butynski, M. Hoffmann, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria, pp. 228-230. Bloomsbury Publishing, London.
Bronner, G.N. and Jenkins, P.D. 2005. Order Afrosoricida. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 70-81. The Johns Hopkins University Press, Baltimore, MD, USA.
Bronner, G.N., Mynhardt, S., Bennett, N.C., Cohen, L., Crumpton, N., Hofreiter, M., Arnold, P. and Asher, R.J., 2024. Phylogenetic history of golden moles and tenrecs (Mammalia: Afrotheria). Zoological Journal of the Linnean Society, 201(1), pp.184-213.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Gilbert, C., Maree S. and Robinson T.J. 2008. Chromosomal evolution and distribution of telomeric repeats in golden moles (Chrysochloridae, Mammalia). . Cytogenetic and Genome Research 121(2): 110-119.
IUCN. 2015. The IUCN Red List of Threatened Species. Version 2015.2. Available at: www.iucnredlist.org. (Accessed: 23 June 2015).
Hickman, G.C. 1986. Swimming of Amblysomus hottentotus (Insectivora: Chrysochloridae) with notes on Chrysospalax and Eremitalpa. Cimbebasia A 8: 55-61.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kuyper, M.A. 1985. The ecology of the golden mole Amblysomus hottentotus. Mammal Review 15(1): 3-11.
Mynhardt, S., Bennett, N.C. and Bloomer, P., 2020. New insights from RADseq data on differentiation in the Hottentot golden mole species complex from South Africa. Molecular phylogenetics and evolution, 143, p.106667.
Mynhardt, S., Maree, S., Pelser, I., Bennett, N.C., Bronner, G.N., Wilson, J.W. and Bloomer, P., 2015. Phylogeography of a morphologically cryptic golden mole assemblage from South-Eastern Africa. PLoS One, 10(12), p.e0144995.
Schoeman, S., Bennett, N.C., van der Merwe, M. and Schoeman, A.S. 2004. Aseasonal reproduction in the Hottentot golden mole, Amblysomus hottentotus (Afrosoricida: Chrysochloridae) from KwaZulu-Natal, South Africa. African Zoology 39: 41-46.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, United Kingdom, Cambridge.
Taylor WA, Mynhardt S, Maree S (2018). Family Chrysochloridae. In: Handbook of the Mammals of the World – Volume 8 Insectivores, Sloths and Colugos. DE Wilson & RA Mittermeier (eds). Lynx Edicions. ISBN-13: 978-84-16728-08-4.