Honey Badger
Mellivora capensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Mellivora capensis – (Schreber, 1776)
ANIMALIA – CHORDATA – MAMMALIA – CARNIVORA – MUSTELIDAE – Mellivora – capensis
Common Names: Honey Badger (English), Ratel (Afrikaans), Insele (isiZulu, Swati), Matshwane (Setswana), Ulinda (Ndebele), Matshwane, Magôgô, Magôgwê, Magogwe, Magwagwê (Setswana), Sere, Tsere (Shona), Xidzidzi (Tsonga), Tshiselele (Venda)
Synonyms: Viverra capensis Schreber, 1776
Taxonomic Note: Coetzee (1977) described 10 subspecies for Africa based primarily on size and pelage (mantle) variation, with only M. c. capensis present in the assessment region. However, no DNA investigation of subspecies has been completed so far and, therefore, subspecies denoted only by morphometrics, or pelage colour and pattern, are of dubious validity. There may even be a large variation in pelage pattern (length and size of white stripe) within populations and in size between localities within the same geographical areas (for example between Kalahari, a semi-arid environment, and the Zambezi Valley, a mesic environment) (C.M. Begg & K.S. Begg pers. obs. 1994–1999). This perceived variation may also be biased by lumping sexes, despite the fact that Honey Badgers are significantly sexually size-dimorphic, with males at least one-third larger than females (Begg 2001a).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Power, R.J.1, Nicholson, S.K.2& Hlako, J.2
Reviewer: Do Linh San, E.3
Contributor: da Silva, J.M.4 & Patel, T.2
Institutions:1North West Provincial Government, 2Endangered Wildlife Trust, 3Sol Plaatjie University, 4South African National Biodiversity Institute
Previous Assessors and Reviewers: Begg, C., Begg, K., Power, J., van der Merwe, D., Camacho, G., Cowell, C. & Do Linh San, E.
Previous Contributors: Abramov, A.V., Child, M.F., Isham, J., Duckworth, J.W. & Nicholson, S.
Assessment Rationale
This species has a wide habitat tolerance, a catholic diet and a large area of occupancy (AOO) in the assessment region. Although persecution – both direct for beehive damage and poultry losses, and incidental as bycatch in damage-causing animal controls – is ongoing and suspected to be resulting in localised declines, such threats can and are being mitigated by active and successful conservation projects and education programmes. Hence, there is no evidence for, nor any reason to suspect an overall population decline. The estimated population size ranges from a minimum of 741 (which is improbable due to their wide occurrence on protected areas and game farms) to a likely 13,200 mature individuals. We retain the Least Concern listing, but caution that the species may warrant re-assessment and listing in a threatened category if evidence of a decline or of increasing threat level is produced.
Regional population effects: There is a broad front on South Africa’s northern borders of Namibia, Botswana, Zimbabwe and Mozambique, from whence there is likely to be contiguous populations with all these countries. The species is contiguous with the Botswana population and sightings are frequent along the Nossob riverbed in Kgalagadi Transfrontier Park (KTP). It is suspected that there is immigration from neighbouring countries into the assessment region, especially as the dispersal ability is good and Honey Badgers cover large daily distances in search of food (Begg et al. 2005b). This is moderately strong enough evidence to warrant a strong rescue effect, but it is of moderate significance.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Power RJ, Nicholson SK & Hlako J. 2025. A conservation assessment of Mellivora capensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Honey Badger has an extensive historical range which extends through most of sub-Saharan Africa from the Western Cape, South Africa, to southern Morocco and southwestern Algeria, and outside of Africa through Arabia, Iran and western Asia to Middle Asia and the Indian peninsula (Proulx et al. 2016).
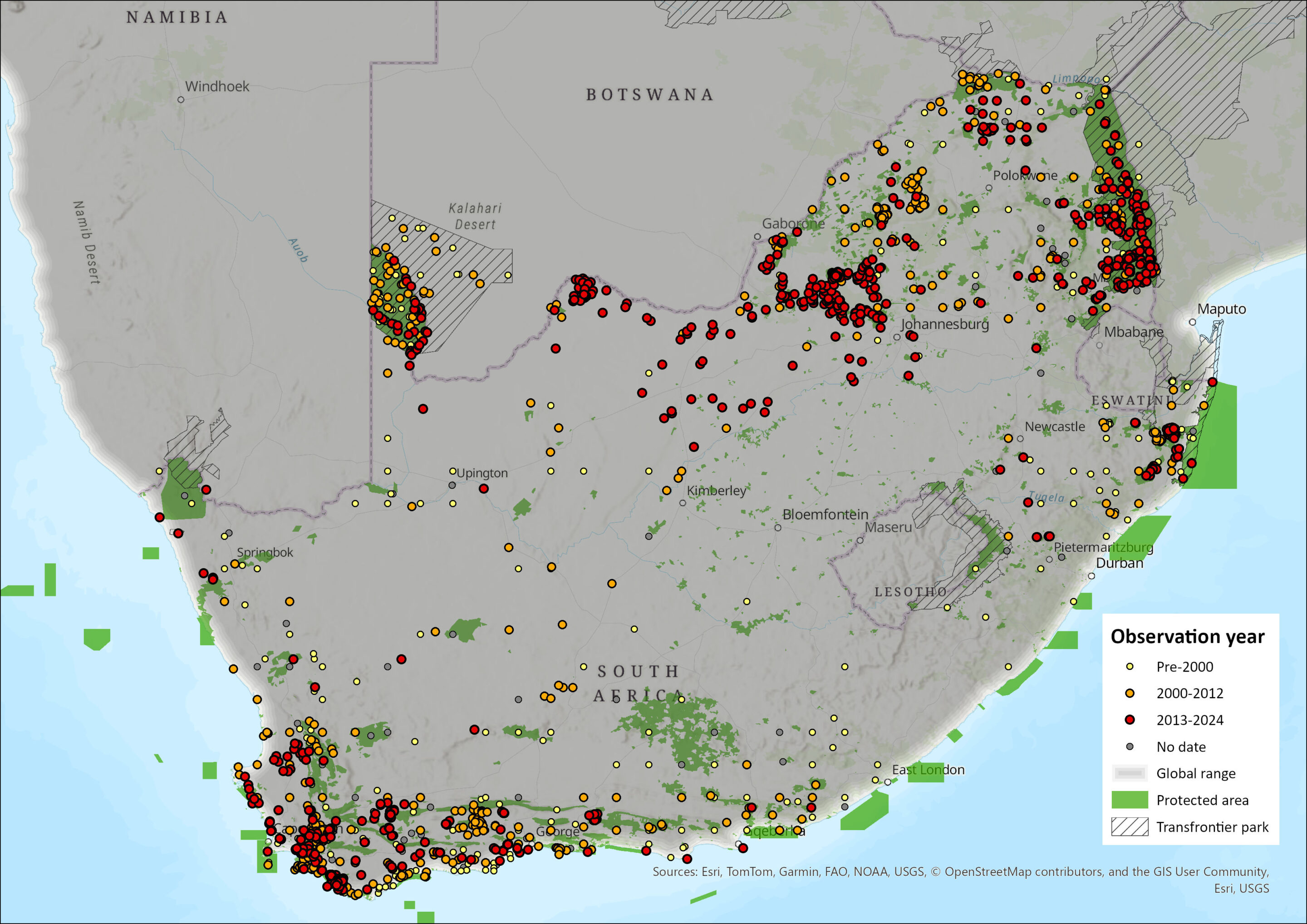
Within the assessment region, the species occurs in South Africa and the Lowveld regions of Eswatini (Monadjem 1998) but is absent from Lesotho (Lynch 1994; Proulx et al. 2016). In South Africa, Honey Badgers historically occurred in all provinces except the Free State (Lynch 1983). The reason for the absence of badgers in this area is likely because of the suboptimal, open-steppe nature of this region (Begg 2001b; Begg et al. 2016c). A single record was received from the eastern border of the Free State during a critical assessment of the Badger Friendly Labelling (BFL) Project (Irlich & Davies-Mostert 2009), but it remains unclear whether this represents range expansion, lack of surveying in the area, or spill-over from suitable habitats in the KwaZulu-Natal Province across the Drakensberg range. However more recently, there appears to be no records in that province (see Figure 1), though sporadic occurrences over time will always be possible.
Range expansion onto the Highveld has already been documented in the previous assessment (Power 2014; Begg et al. 2016), and they occur as far east into Mpumalanga too (Loock et al. 2019). The species was absent from the southern Highveld grasslands during the 1970s (Rautenbach 1982), and even early 2000s (Friedmann & Daly 2004; Skinner & Chimimba 2005). Honey Badgers were record in 2010 in the Cradle of Humankind area of Gauteng (Kuhn 2014), and still seem to be in the same areas, as in July 2021, a Honey Badger was reported to have bitten a dog from the Kromdraai area (Mohale et al. 2022). Elsewhere on the Highveld of the province, two Honey Badgers were captured on a camera trap on the Highveld east of Pretoria in 2018 (Keyes et al. 2021). Elsewhere on the Highveld, in the North West, the species still occurs in this ecoregion (Power et al. 2019; Figure 1). In August 2020, on the Highveld near Schweizer-Reneke, video footage was obtained of a mother and pup (Mr Johan Scribante, reported to Directorate of Biodiversity Management). More recently, also on the Highveld, a sighting report was made of two was made at Rondepan, near Barberspan in December 2023 (Miss Annemarie Schutte, reported to Mr Sampie Van der Merwe, Park Manager of Barberspan reserve). Given the extended period of maternal dependence (Begg et al. 2005a), the likelihood is always higher of any two seen being likely a mother and offspring, even if similarly sized, which alludes to reproduction being evident in these areas, which suggest the species is present and breeding on the Highveld of the North West and Gauteng at least. They are still present on Madikwe (Smith et al. 2023), and seems to be all over the North West (Figure 1). Because Honey Badgers are known to be able to swim (Kingdon 1997), it remains to be seen whether they ever have forded the Vaal River to enter the Free State.
The species occurs westwards across North West (Power et al. 2019), into the Northern Cape (Figure 1). Honey Badgers occur along the Orange River in the Northern Cape (Tshabalala et al. 2021), and the Namaqualand area.
In the Eastern Cape, the old Transkei (eastern parts of the Eastern Cape) always had a low prevalence of this species (see Stuart 1981; Lloyd & Millar 1983). This is still reflected currently, and more recent occurrences seem more coastal (Figure 1). In protected areas of the old Transkei, a mammalian survey carried out in 2003 showed no evidence for this species’ occurrence (Hayward et al. 2005). There was only one record from Lynch (1989) in the northeastern Eastern Cape from the Jamestown District, and this seems still to be the case (Figure 1). Similarly, since the last assessment (Rowe-Rowe 1992; Friedmann & Daly 2004), there seems to be a decline in occurrence of the species in southern KwaZulu-Natal, which may be genuine or an artefact of no recent records being available. If the former, this could be cause for concern. The species is prevalent in Zululand of north-eastern KwaZulu-Natal (Kheswa et al. 2018; Ramesh et al. 2017; Pretorius 2019) and is mostly present in the northern half of the province (Figure 1).
The species occurs across the northern South African provinces of Limpopo and Mpumalanga and in north-eastern Limpopo, they avoided settlements and croplands and occurred in rangelands (Williams et al. 2018). They are present in several reserves in Limpopo (Comley et al. 2020; Smith et al. 2023).
Honey Badgers are prominent in the Western Cape, occurring across most bioregions (Tshabalala et al. 2021), and prevalent in the Cape Winelands Biosphere Reserve (Nieman et al. 2021) as well as the Overberg area (Ross et al. 2022). Interestingly, in 2019, the Honey Badger was first detected on wildlife-friendly farms of the Nuweveld mountains of the Western Cape Karoo, possibly due to more favourable conditions in this arid environment (Schurch et al. 2021).
The same high population concentrations as reported for Mpumalanga and Limpopo, the Kalahari in the Northern Cape as well as the Western Cape’s coastal lowlands (Begg 2001b; Irlich and Davies-Mostert (2009), are still applicable.
The current distribution is similar to the 2016 baseline (Begg et al. 2016c), though with a possible range loss from the hinterland of the Eastern Cape, and deeper penetrations of the range into the Highveld of North West, Gauteng and Mpumalanga provinces (see Figure 1).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 4000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Honey Badger (Mellivora capensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Indomalayan, Palearctic
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Afghanistan | Extant | Native | – | – |
| Algeria | Extant | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| India | Extant | Native | – | – |
| Iran, Islamic Republic of | Extant | Native | – | – |
| Iraq | Extant | Native | – | – |
| Israel | Extant | Native | – | – |
| Jordan | Extant | Native | – | – |
| Kazakhstan | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Kuwait | Extant | Native | – | – |
| Lebanon | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Morocco | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nepal | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Oman | Extant | Native | – | – |
| Pakistan | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Saudi Arabia | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Syrian Arab Republic | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Turkmenistan | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| United Arab Emirates | Extant | Native | – | – |
| Uzbekistan | Extant | Native | – | – |
| Western Sahara | Extant | Native | – | – |
| Yemen | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
It has been hypothesised that increased woody cover, due to climate change-induced bush encroachment onto the Grassland Biome, has facilitated the greater occurrence of this species (Power 2014).
It is important to consider a species’ ecological and biological traits to determine the likely mechanisms of climate change impact and quantify these using expert knowledge (Foden & Young 2016), and given the species is dependent on vegetative cover as a variable (Ramesh et al. 2017; Kheswa et al. 2018; Pretorius 2019), it may benefit from bush encroached conditions which may prevail under conditions of warming in the future (Foden & Young 2016). The species is also a generalist in both habitat (Kheswa et al. 2018), and diet (Begg 2003a), so this will also enable it to adapt, whether dry or wetter even.
Population
Honey Badgers are considered to be rare or to exist at low densities across most of their range (Begg et al. 2013). Densities based on night counts have been estimated at 0.1 individual / km² in the Serengeti National Park, Tanzania (Waser 1980) and 0.03 adult / km² in the Kgalagadi Transfrontier Park (KTP) (Begg 2001a). There is unfortunately no density data from the mesic savannahs, such as Kruger National Park (KNP), and it is unknown at this stage as to which areas have higher densities, i.e. KNP vs KTP. Given these density estimates (0.10–0.03 individual / km²), and an estimated 200,000 km² total AOO across the assessment region, the overall population is between 6,000 and 20,000 individuals, which is likely to be comprised of 3,960–13,200 mature (assuming that 66% of the population is mature, sensu Friedmann & Daly 2004). At the very minimum, the AOO is 37,416 km² based on confirmed presence in national parks across South Africa, which yields a population size of 1,122–3,742 individuals (741–2,470 mature). Thus, the estimated population size ranges from a minimum of 741 to a more likely 13,200 mature individuals. This range encompasses the previous assessment estimate of 4,000 individuals (2,600 mature) using the same area estimate and assuming a 50 km² home range for breeding pairs (Friedmann & Daly 2004). Further density estimates, both inside and outside protected areas, are required to more systematically estimate population size.
The species is often detected as by-catch on studies focussed on other species (Pretorius 2019; Ross et al. 2022). If one indirectly gleans information from one particular study, the highest photographic captures of the species are western Limpopo (Ross et al. 2022), in what appears to be Atherstone Nature Reserve, as well as the Maputaland coastline (Ross et al. 2022), in isiMangaliso Wetland Park, where it seems the species is well known from camera trap studies (Ramesh et al. 2017; Kheswa et al. 2018; Pretorius 2019). Though this does not give information on numbers, it renders relative abundance only (see also Nieman et al. 2021).
From 2016 to 2024 in the North West, there were on average 2±2,2 incidents (range 0 – 7) per year involving the species in the province. The number of incidents peaked in 2019 (NW Directorate of Biodiversity Management records, 2016-2023). This period contrasts with a previous 8-year period (2008-2015), where a lot fewer incidents were reported, where this was an average of 0.37±0,5 (range 0-1) (NW Directorate of Biodiversity Management records, 2008-2015). Crudely, this information may indicate a general increase, given the same officer has been in the employ of the Department over the period and there has been operational consistency over the period. Although it is poor quality data to make such an inference.
We suspect that the population is stable or increasing given the stable or increasing AOO of the species since the last assessment (Begg et al. 2016c). Additionally, retaliatory killings from beekeepers have not necessarily declined since the last assessment (Begg et al. 2016c), but may be stable, but at lower levels than previously. Although Honey Badgers may be experiencing local declines outside of protected areas due to accidental persecution, in some cases the reverse is true, so population abundances may be counterbalanced.
Population Information
Current population trend: Probably stable due to wide habitat tolerance, large area of occupancy and active and successful conservation projects and education programmes.
Continuing decline in mature individuals? Probably not
Number of mature individuals in population: 741-13,200
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: Unknown
Severely fragmented: No. They have a broad habitat tolerance and can exist in at least some agricultural and rural landscapes.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While the species has been investigated in a phylogenetic context, no population genetic study has been undertaken. However, an analysis by Rhodes (2006), using both nuclear and mitochondrial data from samples across Africa, showed that there was a pattern of isolation by distance, yet phylogeographic structuring between eastern and southern Africa was present (Rhodes 2006). This supported the identification of distinct management units (eastern and southern African populations). Consequently, within the assessment region, there is likely a single metapopulation with animals capable of dispersal to neighbouring countries/regions.
Based on density estimates and AOO (see Population section), it is estimated that approximately 13,200 mature individuals exist within the assessment region (although lower limits predict only 740). Based on these estimates and a Ne/Nc conversion ratio between 0.1-0.3, the effective population size (Ne) for this population is approximated at 1,320- 3,960. This is based in the more likely (higher) estimates, and hence these values should be treated with caution. It is recommended that more detailed surveys or a population genetic study be undertaken to verify these values.
Dominant male Honey Badgers are the most likely father of 50% of the cubs in the Kalahari, while subordinate males are also known to father cubs, and brief mating opportunities may be afforded to subordinate males which leads to fertilisation (Verwey et al. 2004), which might be beneficial from a genetic point of view.
The white phenotype (likely albino) Honey Badgers is a rare phenomenon. Records of this white phenotype appear localized to a relatively small area within the Eastern and Western Cape (Ross et al. 2022). Inbreeding is often invoked when albinism is detected, though suspected to be less important given the reserve where it was discovered in is not insular to the surrounding areas (Ross et al. 2022). Given albinism can be linked to inbreeding depression (Laikre et al. 1996), and high historical persecution (Hey 1964; Stuart 1981; Kruger et al. 2021) it is not impossible that this subpopulation at the southern tip of Africa went through a bottleneck.
However, despite fitness costs associated with being albino, Honey Badgers still seem to survive better in the wild than albino individuals from other species, i.e. nocturnality inter alia (Ross et al. 2022), and perhaps it can become fixed in a sense.
Habitats and ecology
Honey Badgers live in a wide variety of habitat types within the assessment region. However, they are generally absent from the more open and central parts of the Grassland and Nama Karoo biomes, which suggests cover to be important, which has since been confirmed (Kheswa et al. 2018; Pretorius 2019). In northern KwaZulu-Natal, Honey Badgers require closed, wooded habitats (Ramesh et al. 2017; Kheswa et al. 2018; Pretorius 2019). Locally, the species also prefers to be near water in some ecosystems (Kheswa et al. 2018). In the St Lucia area of KwaZulu-Natal, Honey Badger were commonly found in Eucalyptus plantations, even more so than in natural habitat types (Kheswa et al. 2018). Human-modified landscapes may not be detrimental to Honey Badgers (Kheswa et al. 2018 but see Williams et al. 2018). However, in Limpopo, they avoided human settlements and croplands (Williams et al. 2018). Honey Badgers are habitat-generalists that are able to exploit new habitat opportunities (Kheswa et al. 2018).
They are opportunistic, generalist carnivores (Begg et al. 2003a), and feed on a range of prey items varying in size from small insect larvae to animals similarly sized to themselves (Kruuk & Miller 1983), and sometimes the young of ungulates, i.e. sheep Ovis sp. (Stuart 1981; Kruger et al. 2021) and domestic poultry (Stuart 1981) or take food from camp kitchens and bins (Bird & Mateke 2013). Honey Badgers were recorded to scavenge on carcasses in an agricultural landscape (Stuart 1981; Keyes et al. 2021). In the Kalahari at least, small mammals and reptiles (<100g) are the most common prey of badgers contributing >75%of the prey items (Begg et al. 2003a). They dig up prey from the soil surface, and these may include scorpions, gerbils, snakes and small reptiles (Kruuk & Mills 1983; Begg et al. 2003a).
All mammalian carnivores smaller than Honey Badgers are considered prey items, as are the young of medium-sized carnivores (Begg et al. 2022. In the Bushmanland Karoo of the Northern Cape, Honey Badgers are known to specialise on tortoises and are able to break into them (Lloyd & Stadler 1998).
Although they are primarily hunters of their own food, they may pirate food from other carnivores and will also scavenge from the kills of larger animals (Begg et al. 2013). In the Kalahari, intraguild predation was common and Honey Badgers preyed or attempted to prey on all mammalian carnivores smaller than themselves, which included the young of medium-sized carnivores (Begg et al. 2022). Honey Badgers themselves have been killed by Lions Panthera leo and Leopards Panthera pardus, while cubs were killed by Black-backed Jackals (Begg et al. 2022).
Honey Badgers are essentially nocturnal, but they may be active during the day in areas where there is little human disturbance, and during seasons when day temperatures are cooler (Begg et al. 2016a). Honey Badgers are primarily solitary, with a non-territorial polygynous or promiscuous mating system (Begg et al. 2005a). Males may range over areas as large as 551 km2 (Begg et al. 2005a), and scent-marking plays an important role in communication (Begg et al. 2003b). Female home-ranges were significantly smaller than that of the males ˜126km2) (Begg et al. 2005b). Female Honey Badger home-range size were five times larger than that predicted from body mass. The extensive home ranges of females were likely to be a function of relatively low prey availability in the semi-arid Kalahari and the long period of cub dependence (12–16 months) (Begg et al. 2005b). Female Honey Badger did not appear to actively defend a territory and scent marking appears to mediate spatio-temporal separation between them (Begg et al. 2003b), such that a loosely territorial spacing pattern exists (Begg et al. 2005b). In contrast, males did not support the typical mustelid pattern of intra-sexual territoriality but instead encompassed the overlapping home ranges of up to 13 females (Begg et al. 2005b). Young male Honey Badgers tended to have smaller home ranges (151±45km2) than adult males and showed a spacing pattern more similar to adult females than adult males (Begg et al. 2005b).
In the Kalahari, Begg et al. (2022) recorded foraging associations between Honey Badgers and seven other species (two mammals, five birds). Commensalistic interactions between badgers and Pale Chanting-goshawks (Melierax canorus) and Black-backed Jackals were the most common. Goshawks and jackals experienced increased hunting opportunities and intake rate and therefore benefited from the association through facilitation. Honey Badgers, in contrast, did not show any significant differences in capture success, intake rate or predator vigilance when foraging in association compared to foraging alone.
Honey Badgers seem to have some immunity to bee stings, but they are cautious and can be killed by bees as seen when badgers are caught in gin traps around commercial beehives and when badgers break into wild hives (C.M. Begg and K.S. Begg pers. obs. 2000). They also have developed some immunity to snake venom which is thought to be the result of numerous minor envenomation events from bees, scorpions and smaller snakes (see also Drabeck et al. 2015). When interacting with larger carnivores, they have a formidable display of a rattling roar, rushing at predators and the release of scent that dissuades many opponents at close contact; and when fighting is inevitable, their coarse, loosely-fitted skin and thick sub-cutaneous fat deposits have an important protection function (Begg et al. 2022).
Ecosystem and cultural services: Since they feed extensively on rodents and arthropods (Smithers 1971; Begg et al. 2003a), which are agricultural pests, Honey Badgers can serve a useful role in the agriculture industry (Begg et al. 2016c). Since Honey Badgers are a generalist carnivore, they can prey on several agricultural pest species, such as Springhare Pedetes capensis (Kruuk & Mills 1983; Begg et al. 2003a), small rodents, such as gerbils (Begg et al. 2003a), and even the young of similarly sized carnivores such as the African Wild Cat (Begg et al. 2003a), and Black-backed Jackal (Begg et al. 2022). This may impart some importance to an agro-ecosystem, though it is not always appreciated (Williams et al. 2018). Williams et al. (2018) reckon a carnivore needs to have an excess of 20% of rodents in its diet to be able to provide a meaningful ecosystem service, and the Honey Badger may just make the cut.
Mesopredators, such as Honey Badgers, can be ecological indicators as they tend to correlate with areas with high mammalian species richness (Tshabalala et al. 2021), and can thus be good biodiversity surrogates.
As alluded to in the diet section, Goshawks and jackals benefited from increased hunting opportunities and intake rate. In addition, goshawks showed increased strike success and an expanded prey base when hunting with Honey Badgers compared to hunting alone in similar habitat elsewhere. Overall, Honey Badgers did not show any significant differences in foraging success, when foraging in association compared to foraging alone (Begg et al. 2022). They thus can be an important agent in an ecosystem in terms of forage facilitation.
Honey Badgers are believed to have a mutualistic association with the Greater Honeyguide (Indicator indicator); the latter would lead a badger to the beehives, where the badger would break open the hive and feed on the bee larvae (not the honey) and leave scraps for the bird (Friedmann 1955). There is significant anecdotal evidence of this across Africa (Kingdon 1997), and although not scientifically proven, there is a real possibility that badgers do actually engage in such behaviour. However, one should also consider that this association might have been misinterpreted due to the real guiding behaviour that honeyguides provide for people. Honey Badgers can easily find hives themselves, and C.M. Begg and K.S. Begg (pers. obs. 2004) have seen honeyguides arrive at a hive once a badger was already breaking in. As for other bird species, observations to date seem to suggest that the honeyguide may rather opportunistically follow the Honey Badger (Begg et al. 2016c). The most likely scenario is that the interaction between Honey Badgers and honeyguides does occur but is highly localised or extremely difficult to observe (Van der Wal et al. 2023).
Honey Badger (Mellivora capensis) extracting and eating bee larvae from a honeycomb (Keith S. Begg and Colleen M. Begg)
The species is well known for its ferocity and antics of unprovoked attacks on other larger species, including man (Smithers 1971; Mills 1997; Skinner & Chimimba 2005), and notwithstanding its small size (Skinner & Chimimba 2005), the character of bravery, irascibility and courage emerge, which are naturally immortalised in western culture. There have been numerous recent documentaries and media clips showcasing the aggressiveness and tenacity of Honey Badgers (Somerville 2022). A particular animal name “Stoffel” has captured the public’s imagination with its incessant antics of escaping its enclosure (a subtle reminder that should not be there).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.9. Forest -> Forest – Subtropical/Tropical Moist Montane | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 3.7. Shrubland -> Shrubland – Subtropical/Tropical High Altitude | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
Life History
Generation Length: 6 years
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Body parts use:
Honey Badger body parts (particularly paws, skin and organs) are commonly used locally in traditional medicine because of the species’ reputation for fearlessness and tenacity, which may be a particular problem in KwaZulu-Natal (Ngwenya 2001). Honey Badgers may be increasingly used in the bushmeat trade due to the decline in other more favoured bushmeat species (Colyn et al. 2004; Begg et al. 2013).
In some areas of South Africa, there is an overall clientele consisting of 60 – 80% of South African citizens who seek cures from wild animals (Nieman 2018). In the Western Cape, there has been an increase in medicinal use trade (Nieman 2018). In traditional healer communities in the Western Cape, body parts of the Honey Badger appeared in 47.1% of communities and they appeared important there (Nieman 2018). The skin pieces of the Honey Badger have been used to ostensibly protect against evil spirits and bad muti (Nieman et al. 2018), and could be sold for R500 – R1700 (Nieman 2018). The paws and bones are believed to protect against bad muti (Nieman et al. 2018) and could sell for between R1000 – R1700 (Nieman 2018). The subcutaneous fats and oils are believed to protects infants from evil spirits (Nieman et al. 2018), and can sell for R700 (Nieman 2018). In Mpumalanga and Limpopo, the Honey Badger is believed to be able to predict the future (Mashele et al. 2021).
Trophy hunting:
There has been an increase in the use of the species for trophy hunting though it is likely not detrimental yet. They were hunted as trophy animals (average of 16 ± 6 per year exported from South Africa between 2002 and 2012; CITES trade database, Begg et al. 2016c).
Interest in trophy hunting this species has come to the fore in the Limpopo Province where a number of permits have been requested for this purpose. There has been a steady increase in hunting of Honey Badgers (SANBI Professional Hunters register, 2010-2019), with over 94% of hunts of the species taking place in Limpopo, suggesting the biggest off-takes there too. In the North West province, since 2020, there have been 16 applications to hunt the species, and three have been rejected, related to a regional level quota which has been instituted as well as spatial clumping (North West Directorate of Biodiversity Management Directorate, 2020-2023). The interest in hunting them has increased since 2016 in the province (NWPG 2017).
Tourism
Honey Badgers are intriguing and rarely seen, and there is a desire by naturalists to view them. They are often seen in the rest camps of Kruger where they can become habituated, and they are seen frequently in the Kgalagadi Transfrontier Park. There are a number of captive facilities, including rehabilitation centres where people visit and learn about them.
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Used locally and opportunistically as bushmeat. | Yes | Local commercial use in traditional medicine trade. |
National Commercial Value: Yes
International Commercial Value: No
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | – | – | – | – |
| 3. Medicine – human & veterinary | true | true | – | – |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: Possibly increasing with rural settlement expansion.
Threats
Like any species, habitat loss and degradation is a possible threat (Ray et al. 2005), though this species seems quite adaptable (Kheswa et al. 2018).
As their scientific name suggests (melis means honey and voro means devour), conflict has occurred between Honey Badgers and beekeepers as they share a common interest. Beehive damage by Honey Badgers is a significant threat to beekeeping productivity, particularly around protected areas. Honey Badgers have been persecuted by farmers since the early 1800s as they were classified as “vermin” or problem animals, and even well into the 20th century (Hey 1964). At the time, Begg (2001b) found that Honey Badgers were directly causing in excess of R500,000 worth of damage per annum in the Western Cape and Mpumalanga provinces alone. Thus, the main threat to Honey Badgers was direct persecution through the use of, for example, steel-jawed traps and poisons, by apiculturists throughout their range. In contemporary South Africa, Honey Badgers are still known to cause conflict in the honey industry by raiding beehives and are persecuted as a result (Begg 2001b; Nieman et al. 2020; Hendricks & Masehela 2024).
However, in the Boland Mountain Complex, interestingly, despite the damage suffered by 8% of landowners, who had apiaries, landowners were relatively tolerant of the species (Nieman et al. 2020). In the North West, Honey Badger are a minor problem in the honey industry (Power 2014), but the species is more problematic with poultry, even if just slightly. Of the incidents since 2016, 25% involved poultry depredations, and 18,7 % (n=16) involved beehive conflict (NW Directorate of Biodiversity Management records, 2016-2023). From 2016 to 2024, there was an average of 2±2,2 incidents (range 0 – 7) per year involving the species in the province. The number of incidents peaked in 2019 (NW Directorate of Biodiversity Management records, 2016-2023). These incident reports involve advising a complainant on a problem involving the species, or needing to catch and remove a species, whether it be an injured animal or it is conflict causing (NW Directorate of Biodiversity Management records, 2016-2023).
Honey Badgers are also indirectly killed by non-selective control programmes targeting other species, such as Black-backed Jackal and Caracal (Caracal caracal) (Begg et al. 2013) and are intentionally targeted too (Begg et al. 2001b). Considering that Honey Badgers are scavengers as well, they are likely to become victims of poisoning. There is evidence to suggest that they have gone locally extinct in many areas due to poisoning (Begg et al. 2016c). Honey Badgers are known to kill poultry (Hey 1964; Stuart 198a), as well as sheep, in particular the lambs (Kruger et al. 2021). In response, Honey Badger have sometimes been killed using hounds and by trapping (Hey 1964; Stuart 1981; Kruger et al. 2021). In 1956, 40 Honey Badger were killed for bounties in the then Cape Province, which was a lot fewer than other species (Hey 1964), which may have reflected rarity then, or difficulty to trap them (see Kruger et al. 2021).
Earlier on, Honey Badgers were responsible for large numbers (18.7%) of small livestock losses between 1978 and 1991 in the Mossel Bay District in the late 1980s, second to the Caracal (Kruger et al. 2021). From 1978 to 1988, up to 10 Honey Badger were killed annually in the Mossel Bay District owing to depredations (Kruger et al. 2021). According to comments recorded by hunters in the hunt reports, Honey badger control was more challenging than controlling Caracals (Kruger et al. 2021).
From 2016 to 2023, of 16 cases dealt with, four have resulted in mortalities (NW Directorate of Biodiversity Management records, 2016-2023). In 2017, one badger succumbed to starvation in a reservoir where it could not climb out of (NW Directorate of Biodiversity Management records, 2016-2023). This was thus accidental, but points to the anthropogenic environment. Two mortalities were reported which were related to conflict, where in 2023, community members killed one they trapped, and one died in 2019 from excessive bee stings, owing to a 3 day period in a trap cage, set adjacent a beehive (NW Directorate of Biodiversity Management records, 2016-2023). These cases point to this being an unabated threat to the species. Since 2016, five Honey badgers have been captured and released elsewhere (NW Directorate of Biodiversity Management records, 2016-2023), as part of conflict mitigation.
In the Waterberg of the Limpopo in 2011, 0.5% of game, was attributed to Honey Badger (Thorn et al. 2013), though one cannot be certain of the provenance. Preying upon larger mammals is very rare (Begg et al. 2003b), though not impossible (Kruuk & Mills 1983), given sizes of juvenile ungulates. Honey Badgers have been persecuted, and the off-take of Honey Badger from Waterberg ranches in the Limpopo in 2011 was estimated at 0,04 Badgers/ 100 km2 (Th0rn et al. 2013), which was likely to be low, and not a great threat.
Additional threats:
Wire snares
In 2019, one Honey badger was retrieved from a snare, treated and re-released again (NW Directorate of Biodiversity Management records, 2016-2023). This points to snaring as a possible threat, though it is likely still be low, as snaring is reported frequently across a spectrum of other species (Leopard Panthera pardus). It is possible that their heads are too low to be caught in most snares, which are aimed at the Common Duiker Sylvicapra grimmia (see Nieman 2018), or they strong enough to break out of them.
Roads
Roadkill appears to be a minor threat to Honey Badgers (Ray et al. 2005). There have however been a few records from Limpopo (Collinson et al. 2015), Western Cape (Serieys et al. 209), and North West provinces (Power 2014).
In northern Limpopo, Collinson et al. (2015) drove a total of 14,400 km over 120 days spanning 10 months across 2011 and 2012, on a 120 km circuit around the Venetia Limpopo Nature Reserve, and a stretch that abutted the Mapungubwe National Park. On this circuit, only one Honey Badger was reported to have been killed on these roads (Collinson et al. 2015). Given the 10-month study period, this would amount to 1.2 Badgers killed by roads per annum.
In the North West, since 2010, only two Honey Badger roadkills have been recorded, and this is despite extensive province wide driving, as well as a commissioned project from 202o on this (NW Directorate of Biodiversity Management records, 2010-2023), which suggests this threat is likely low, unless such species are quickly removed and not detected.
Body parts uses trade
In the Western Cape, Honey Badgers appeared in 47,1% of communities which traded with animal parts (Nieman 2018). Animal body parts, such as the Honey Badger body parts are increasingly more important in South Africa (Nieman et al. 2018). The Honey Badger has a Cultural Significance Index (CSI) of 4.5, which is moderate amongst several others that are a lot more important such as the leopard (Panthera pardus) for instance (Nieman 2018). In Mpumalanga, a concern had been expressed by Traditional Healers was that acquiring vultures through poisoning was unsustainable, because birds were killed en masse, and this may reduce the sustainability of vulture populations for traditional use by future generations. They thus suggested the Honey Badger as one of the alternatives for vultures in traditional medicine (Mashele et al. 2021). This could be of concern.
Disease
In July 2021, a Honey Badger from the Kromdraai area, Gauteng Province, bit a dog on a small farm (Mohale et al. 2022). The following day the same Honey Badger attacked three adults in the area, with one of the victims requiring hospitalisation for management of her injuries. The badger was subsequently shot and the carcass examined and a positive rabies diagnosis was confirmed and phylogenetic analysis of the amplified glycoprotein gene of the rabies virus demonstrated the virus to be of dog origin (Mohale et al. 2022). Disease may not be nationally important for the species but could have local level impacts on society where certain diseases are detected, i.e. rabies.
Pesticides
Trace residues of Anticoagulant Rodenticides have been discovered in Honey Badger from the Cape Peninsula (Serieys et al. 2019). Small residues concentrations of difenacoum were found in one Honey Badger specimen examined, so there has been exposure to these anticoagulant rodenticides, probably via ingestion of prey species, or even scavenging (Serieys et al. 2019).
Trophy hunting
This removal of Badgers has been on the increase (Begg et al. 2016c), though it is uncertain whether it is yet valid for an emerging threat.
It seems the same anthropogenic threats are still around, but perhaps not as high as before, as there seems to be more of an inclination to get the authorities involved, though mortalities have still occurred, it may be less than before, or the same. The threat of use for medicinal purposes may however be seen as an increasing threat. The types of anthropogenic mortality may not necessarily be counteracted by natural recolonisation as they have a slow recolonisation rate and currently only a small percentage of South African nature reserves are large enough to sustain viable subpopulations of these animals, leaving the larger part of South Africa’s Honey Badger population unprotected (Begg 2001b). Compounding this, Honey Badgers also have low natural reproduction rates. There is generally only one cub per litter which reaches independence at the age of 12–16 months, and cub mortality is 47% (Begg 2001a; Begg et al. 2005a), This, together with large home range sizes (e.g. Begg et al. 2005b), suggest that these mustelids live at low densities and are therefore vulnerable to even modest levels of persecution. Not unlike other larger mammals, the Honey Badger is a K-selected species, when compared to other species within the Mustelidae. The long time to independence of cubs (12–16 months) results in birth intervals being longer than 12 months. It furthermore has an unusually small litter size of one cub (Begg et al. 2005b), and they may be vulnerable to extirpation as a result (Carter et al. 2017). These life history parameters can suggest that its population can be decrease by excess mortalities in whatever form.
Threat categories have been assessed for various carnivore species in Africa, and each species is differentially impacted by a set of ten external threats commonly encountered by African carnivores (Ray et al. 2005). Each species was subjected to a scoring process for each threat that assessed the relative severity (its effect on the species), urgency (timescale over which it is most likely to occur), probability (likelihood of occurrence), and geographical extent of the threat through the species’ range in Africa. The threat score for the African Honey Badger was 26, and of 20 carnivores in Africa it ranked 18th (Ray et al. 2005). This pan-African assessment suggests the species to be relatively at low risk especially when considering the other species, and it may be related to its generalist habits and wide distribution (Ray et al. 2005).
While continentally the species may be minimally threatened (Ray et al. 2005), nationally some of the same threats are still present, though not necessarily increasing, but specific threats may supersede previously mentioned threats.
Conservation
Honey Badgers are found in many protected areas throughout the assessment region.
However, they are persecuted by apiculturists for the damage caused to commercial honey production. Begg (2001b) and Begg and Begg (2002) showed that hive damage could be reduced from 24% to 1% with the help of hive-protection methods; for example, by securing beehives 1 m or more above the ground on a stand or trestle, thereby minimising conflicts between Honey Badgers and apiculturists. Begg (2001b) also highlighted that it was economically more viable for beekeepers to be “Badger Friendly”.
The Badger–Beekeeper Extension Programme (BBEP) was established in 2002 to educate beekeepers on effective beehive protection measures and Honey Badger conservation, as well as public awareness on the topic (Isham et al. 2005). As part of the project, a “Badger Friendly Label” (BFL) was developed. The purpose of this project was to assist South African beekeepers in preventing damage by Honey Badgers to beehives by the use of initiatives that prevent Honey Badgers from damaging hives – a non-lethal control method. This prevents the use of other lethal control methods such as poison and gin traps that have a negative impact on Honey Badger and other carnivore populations. This was a long-term solution that secured valuable habitat for Honey Badger populations on farmland in South Africa. Hence, the label was a voluntary incentive to be used only by beekeepers that effectively protected their beehives and thus removed the need to harm Honey Badgers. Furthermore, this label was used to indicate to the public which honey products were produced by Badger Friendly practices and thus provided leverage for consumers to put pressure on the beekeeping industry to change their ways. The labels were sold at a small cost to beekeepers that had signed a declaration copy and which after an inspection of their apiary sites were accredited with Badger Friendly Status” (Isham et al. 2005).
Isham et al. (2005) found that of the protection methods implemented, approximately 90% of the beekeepers protecting their hives made use of methods that raised the hives off the ground, while only about 10% used on‐ground protection methods. It was confirmed that a hive in good condition, securely strapped to a stand of at least 1 m reduced the chance of badger access significantly.
During a critical assessment of the BFL project (Irlich & Davies-Mostert 2009), a total of 46 records of hive damage caused by badgers were collected. This was significantly fewer when compared to a total of 179 records of hive damage recorded by Begg (2001b). According to the beekeepers, badgers were not the most severe threat to honey production anymore. Instead, vandalism and theft are the most severe threats, as they indirectly decrease the number of available sites where beekeepers can keep their beehives. This appears to still be the case (see Hendricks & Masehela 2024).
The threat of direct persecution for beehive depredation was thus inferred to have lessened over the past decade (Begg et al. 2016c). Reports of beehive damage have declined between 2001 and 2009, by an estimated 66% through the work of conservationists in promoting hive protection methods (Begg 2001b; Irlich & Davies-Mostert 2009). Of the 62 beekeepers audited by Irlich and Davies-Mostert (2009), only 16 sustained hive damage from Honey Badgers, while 46 did not sustain any damage since protecting their hives despite high badger activity in the regions. Similarly, while 64% of beekeepers listed badgers as the top threat to productivity in Begg’s (2001b) report, only 12% did so in 2009. Of those who listed badgers as a threat, 85% said that the impact of these mustelids was negligible compared to only 33% in 2001 (Begg 2001b; Irlich & Davies-Mostert 2009). During the course of this assessment (2008–2009) only a single report was received of a badger being killed by beekeepers in Mpumalanga (Irlich & Davies-Mostert 2009). Thus, the overall consensus then was that the problem of Honey Badgers raiding beehives was decreasing in intensity or, at worst, staying constant, with no beekeepers mentioning that the conflict was increasing in intensity or frequency of hive damage. This status quo may thus be relevant in the present.
A 2016 Badger Friendly Audit was conducted by the Endangered Wildlife Trust during March and April 2016 at a number of beekeepers’ properties in Gauteng, Northern Cape, Eastern Cape, Western Cape and Limpopo provinces. A total of 27 beekeepers were audited and 26 of them complied with the criteria in order to obtain the BFL (Begg et al. 2016c). Overall, the 2016 audit confirmed that the BFL project had decreased the number of Honey Badgers being killed by beekeepers as a result of effective hive protection methods, and can thus be considered a genuine conservation success (Begg et al. 2016c; Carter et al. 2017).
However, more work needs to be done to increase consumer awareness of Badger Friendly products and thus to incentivise beekeepers to continue with the project. In 2009, only 2.8% of consumers said that their first choice was whether the honey displayed a Badger Friendly label and the number of stickers sold to retailers declined from 2005 to 2008 (Irlich & Davies-Mostert 2009). Clearly, more work needs to be done to raise the public profile of the label and increase consumer buy-in. The retail of Badger Friendly honey is an intervention similar to “green labelling” that holds sway at the level of the consumer. To increase the number of consumers buying Badger Friendly honey, additional intensive consumer awareness should be undertaken. It would be beneficial to involve members of the public nationally in using consumer power to support the initiative, as well as contributing their data on Honey Badger sightings and become engaged with badger conservation in general. This could be carried out in conjunction with more public awareness and social marketing on badgers and the BFL project.
The Badger-Friendly eco-label marked the start of a conservation movement unique to South Africa (also see Begg 2001b). It also brought awareness to the consumer and is a well-intentioned measure for protecting the Honey Badger and its conservation status. Its effectiveness is, however, inconclusive, not least because the eco-label application procedure does not account for a chain of custody audit to certify honey provenance, i.e. ‘Badger-Friendly’ or not (Hendricks & Masehela 2024).
It has become routine for many South African citizens to purchase such labelled products (Begg et al. 2016c). However, it seems that since, the project may be stagnated (Hendricks 2021), despite the applauded efforts of the EWT and others (Van Wilgen 2007; Carter et al. 2017). Of a survey of 39 respondents in the honey industry, only 56% of them cited correctly what ‘Badger friendly’ honey meant, i.e. raising hives above reach of Honey Badgers, and no poisons or trapped used inter alia (Hendricks 2021). Some landowners mentioned that they once used the Badger-Friendly label, however they no longer do so, as they feel it is an untrue claim and it is impossible to audit all beekeepers who had this eco-label (Hendricks 2021). According to Hendricks (2021), there are contradictions in the attainment and the implementation of the eco-label, especially considering the ‘Badger-Friendly’ label requirements. Beekeepers were even found to be printing their own stickers (labels), and that there were no known cases of rejections or refusals to issue the ‘Badger-Friendly’ eco-label. Respondents in a survey, also reported some label holders had not undergone third-party audits in a decade, when they should be biennial (Hendricks & Masehela 2024).
Additionally, aspects of the feasibility of the auditing process were seen as impractical. This was validated as not all the hives at the apiary sites could reasonably be accounted for, and the method of the inspection would therefore be a sample audit or ‘merely a paper exercise’, as stated by one of the respondents. This was explained in the context of an apiary landscape (commercial scale), where there would be multiple apiary sites (hosting a number of hives) and a separate manufacturing or bottling facility, serving as a common collection point for incoming honey from the producer. Respondents validated this argument by stating that the honey is only being audited at the supplier’s manufacturing facilities, not at all of the producer-beekeeper’s apiary sites, due to its impracticality. Others argued that beekeepers work for themselves, and that it is difficult to monitor their beekeepers’ social conditions as all their honey is externally sourced (Hendricks & Masehela 2024). One respondent— a bottler and holder of the Badger-Friendly eco-label—made it known that keeping track of the conditions from where their honey is originating is a difficult task. This could mean that locational masking also creates limitations regarding the effectiveness of the label as a conservation measure, as it does not account for imported honey, and beekeeping conservation methods against predators. This could mean that any honey is labelled as ‘Badger-Friendly’ (Hendricks & Masehela 2024).
In terms of protecting bee hives, this has been important where beehive fences are used to deter African Elephant Loxodonta africana (Johnson 2019). Prior to the start of an intervention of placing deterrents at beehives, 77.1% percent of hives were destroyed within a week following a Honey Badger attack. After the addition of deterrents (motion activated light deterrent, a preventative cone structure and a caged hive), only 11.1% percent of the hives were attacked, suggesting the deterrents effectively reduced the amount of successful honey badger attacks (Johnson 2019), but did not stop them. All deterrent methods were effectively preventing Honey Badgers from raiding hives with variance in the success rates and economic feasibility. This project complemented the Elephants and Bee Project’s ongoing research by providing much-needed insight into the role Badger deterrents could play in preventing damage to the elephant deterring beehive fences. This research suggests management recommendations through these deterrents to not only reduce Badger hive raiding but also to improve human-Honey Badger coexistence (Johnson 2019).
Translocations and rehabilitation
In the North West, since 2016, five Honey badgers have been captured and released elsewhere (NW Directorate of Biodiversity Management records, 2016-2023), as part of conflict mitigation. Within geographic areas, because of a genetic pattern of isolation by distance, it is recommended that individual Honey Badgers for translocations come from geographically proximate populations (Rhodes 2006).
Conservation efforts and solutions have been successfully implemented and the Honey Badger is now a success story in terms of conservation, but the long-lasting effect on the sustainability of this on the species needs to be evaluated in the future (Carter et al. 2017), and aspects of this, have in fact been done (Nieman et al. 2020; Hendricks & Mashela 2024).
Recommendations for land managers and practitioners:
- Monitoring should be established to measure local subpopulation trends. This should be done in the different provinces and simple relative abundance indices may even suffice.
- Continue to encourage beehive protection methods. Beekeepers must be informed via the media and popular magazines (for example, Farmers Weekly) of the most reliable and cost-effective measures for protecting beehives.
- All apiaries situated within biosphere reserves or along the borders of protected areas must be adequately protected, as conflict with Honey Badgers is inevitable. This should be mandatory.
- Given the species propensity to easily dig under fences (Kesch et al. 2014), and livestock or poultry holding enclosures should be secure to prevent access by the species.
- Reinvigorate ‘Badger friendly honey initiative.
Research priorities:
- Intensive research into the current distribution of Honey Badgers, together with possible estimates of their abundance (both inside and outside of protected areas) is required to successfully address distributional changes in the future. To carry out such an investigation will prove to be time-consuming, and thus citizen science schemes (for example, through social media) should be established to aid with data collection.
- Quantifying long-term population trends in different biomes and land-use areas, in particular to focus on the isiMangaliso Wetland Reserve where there has at least been a history of several studies done over time (see Ramesh et al. 2017; Kheswa et al. 2018; Pretorius 2019), and this could be seen as a sentinel site of sorts.
- The work done on relative occurrence, farmers tolerance and use of the species in the Boland area of the Western Cape (see Nieman 2018; et al. 2020; 2021), would be worth continuing in some manner, as this area was also an apiculturist area too.
- Although sometimes mesocarnivores are less detected than larger carnivores, especially in a biased design study (see Pretorius 2019), but secondary use of the information is always useful (Pretorius 2019; Ross et al. 2022), i.e. the National Leopard Monitoring Project etc.
- Trends in the numbers of animals killed by farmers.
- Trends in the numbers of animals accidentally killed in traps intended for other target species.
- Assess the impact of trophy hunting on the species
- Assess the future of the medicinal trade use in the species
- Home range size for different vegetation types (particularly mesic/semi arid Savannas, Fynbos, Karoo and Renosterveld types) so as to be able to evaluate space requirements for viable populations at the local scale.
A wealth of information has been obtained from one particular ecosystem, the arid Kalahari savannahs (see Mills 1997; Begg 2001a; Begg et al. 2003a,b, 2005a,b, 2016a,b, 2016a; 2016b, 2022), in the western part of South Africa. The following biogeographical areas need to be focussed on, should there be such a further need:
- Spatial and population ecology in mesic savannah ecosystems.
- Spatial and population ecology in Fynbos ecosystems.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP). Priority areas include southern KwaZulu-Natal and the far Eastern Cape Province. Camera-trappers should strategically deploy their camera traps in pursuit of this species and upload their records to bona fide data repositories.
- Only purchase Badger Friendly honey. Contact the Endangered Wildlife Trust (ewt@ewt.org) for further information.
Bibliography
Allsopp MH, De Lange WJ, Veldtman R. 2008. Valuing insect pollination services with cost of replacement. PLoS One 3.
Begg, C.M. 2001a. Feeding ecology and social organization of honey badgers Mellivora capensis, in the southern Kalahari. Ph.D. Thesis, University of Pretoria, Pretoria.
Begg KS. 2001b. Report on the conflict between beekeepers and honey badgers Mellivora capensis, with reference to their conservation status and distribution in South Africa. Endangered Wildlife Trust, Johannesburg, South Africa.
Begg, K.S. and Begg, C.M. 2002. The conflict between beekeepers and honey badgers in South Africa: a western Cape perspective. The Open Country 4: 25-37.
Begg CM, Begg KS, Do Linh San E, du Toit JT, Mills MGL. 2016a. Sexual and seasonal variation in the activity patterns and time budget of honey badgers in an arid environment. In: Proulx G, Do Linh San E. (ed.), Badgers: Systematics, Biology, Conservation and Research Techniques, pp. 161–192. Alpha Wildlife Publications, Alberta, Canada.
Begg CM, Begg KS, Do Linh San E, du Toit JT, Mills MGL. 2016b. An evaluation of techniques used for the capture, immobilization, marking, and habituation of honey badgers (Mellivora capensis). In: Proulx G, Do Linh San E. (ed.), Badgers: Systematics, Biology, Conservation and Research Techniques, pp. 339–362. Alpha Wildlife Publications, Alberta, Canada.
Begg CM, Begg KS, Do Linh San E, du Toit JT, Mills MGL. 2022. Interactions between honey badgers and other predators in the southern Kalahari: intraguild predation and facilitation. In: Do Linh San E, Sato JJ, Belant JL, Somers MJ. (ed.), Small Carnivores: Evolution, Ecology, Behaviour & Conservation, Wiley-Blackwell, Oxford, UK. P 323-346.
Begg, C.M., Begg, K.S., du Toit, J.T. and Mills, M.G.L. 2003a. Sexual and seasonal variation in the diet and foraging behaviour of a sexually dimorphic carnivore, the honey badger (Mellivora capensis). Journal of Zoology 260: 301-316.
Begg, C.M., Begg, K.S., du Toit, J.T. and Mills, M.G.L. 2003b. Scent-marking behaviour of the honey badger, Mellivora capensis (Mustelidae) in the southern Kalahari. Animal Behaviour 66: 917-929.
Begg, C.M., Begg, K.S., du Toit, J.T. and Mills, M.G.L. 2005a. Life history variables of an atypical mustelid, the honey badger Mellivora capensis). Journal of Zoology 265: 17-22.
Begg, C.M., Begg, K.S., du Toit, J.T. and Mills, M.G.L. 2005b. Spatial organization of the honey badger (Mellivora capensis). Journal of Zoology 266: 23-35.
Begg, C., Begg, K. and Kingdon, J. 2013. Mellivora capensis Ratel (Honey Badger). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. V. Carnivores, Pangolins, Equids and Rhinoceroses, pp. 119-125. Bloomsbury, London, UK.
Begg CM, Begg KS, Power RJ, van der Merwe D, Camacho G, Cowell C, Do Linh San E. 2016c. A conservation assessment of Mellivora capensis. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Bird, T.L.F. and Mateke, C.W. 2013. A preliminary survey of the presence and distribution of small carnivores in the Lower Zambezi Protected Area Complex, Zambia. Small Carnivore Conservation 48: 47-59.
Carter S, Du Plessis T, Chwalibog A, Sawosz E. 2017. The honey badger in South Africa: biology and conservation. Int J Avian Wildl Biol. 2(2), p.00091.
Coetzee, C.G. 1977. Order Carnivora. Part 8. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-42. Smithsonian Institution Press, Washington, DC, USA.
Collinson WJ, Reilly BK, Parker DM, Bernard RT, Davies-Mostert HT. 2015. An inventory of vertebrate roadkill in the greater Mapungubwe Transfrontier conservation area, South Africa. African Journal of Wildlife Research 45(3): 301-311.
Colyn, M., Dufour, S., Condé, P.C. and Van Rompaey, H. 2004. The importance of small carnivores in forest bushmeat hunting in the Classified Forest of Diecké, Guinea. Small Carnivore Conservation 31: 15-18.
Comley J, Joubert CJ, Mgqatsa N, Parker DM. 2020. Lions do not change rivers: complex African savannas preclude top-down forcing by large carnivores. Journal for Nature Conservation, 56:125844.
Drabeck DH, Dean AM, Jansa SA. 2015. Why the honey badger don’t care: convergent evolution of venom-targeted nicotinic acetylcholine receptors in mammals that survive venomous snake bites. Toxicon 1(99):68-72
Foden WB, Young BE. 2016. IUCN SSC Guidelines for Assessing Species’ Vulnerability to Climate Change. Version 1.0. Occasional Paper of the IUCN Species Survival Commission No. 59. Cambridge, UK and Gland, Switzerland: IUCN Species Survival Commission. x+114pp.
Friedmann, H. 1955. The Honey-Guides. Bulletin of the United States National Museum, 208.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. Conservation Breeding Specialist Group (SSC/IUCN) and Endangered Wildlife Trust, Parkview, South Africa.
Hayward MW, White RM, Mabandla KM, Bukeye P. 2005. Mammalian fauna of indigenous forest in the Transkei region of South Africa: an overdue survey. South African Journal of Wildlife Research 35: 117-124.
Hendricks LL. 2021. An analysis of eco-labelled honey practices employed in the South African beekeeping industry: implications for agricultural sustainability. MA degree thesis, University of Stellenbosch, Stellenbosch.
Hendricks L, Masehela TA. 2024. Synopsis of Crimes in the South African Beekeeping Industry: Contextualising the Industry Harms, Malpractices, and Risks. International Journal for Crime, Justice and Social Democracy. 13(1):29-40.
Hey D. 1964. The control of vertebrate problem animals in the province of the Cape of Good Hope, Republic of South Africa. In: Proceedings of the Vertebrate Pest Conference. 2(2): 57-70.
Irlich U, Davies‐Mostert HT. 2009. Critical Assessment of the Honey Badger Friendly Label Programme. Endangered Wildlife Trust, Johannesburg, South Africa.
Isham J, Begg KS, Begg CM. 2005. Honey badger and beekeeper extension programme. Final Report. Carnivore Conservation Group, Endangered Wildlife Trust.
Johnson AS. 2019. The effects of tactile and visual deterrents on honey badger predation of beehives. MA degree thesis, University of New York, New York.
Kesch KM, Bauer DT, Loveridge AJ. 2014. Undermining game fences: who is digging holes in K alahari sands?. African Journal of Ecology. 52(2):144-150.
Keyes CA, Myburgh J, Brits D. 2021. Scavenger activity in a peri-urban agricultural setting in the Highveld of South Africa. International Journal of Legal Medicine 135:979-91.
Kheswa EZ, Ramesh T, Kalle R, Downs CT. 2018. Habitat use by honey badgers and the influence of predators in iSimangaliso Wetland Park, South Africa. Mammalian Biology 90: 22-29.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals (first edition}. Academic Press, San Diego, California, USA.
Kruger Q, Avenant NL, De Waal HO. 2021. The contribution of historical hunt club records to inform the management of damage-causing animals in South Africa. Indago 37: 17-30.
Kruuk H, Mills ML. 1983. Notes on food and foraging of the honey badger Mellivora capensis in the Kalahari Gemsbok National Park. Koedoe. 6(1):153-157.
Kuhn, B.F. 2014. A preliminary assessment of the carnivore community outside Johannesburg, South Africa. South African Journal of Wildlife Research 44(1): 95-98.
Laikre L, Andrén R, Larsson HO, Ryman N. 1996. Inbreeding depression in brown bear Ursus arctos. Biological conservation. 76(1):69-72.
Lindsey PA, Romanach SS, Davies-Mostert HT. 2009b. The importance of conservancies for enhancing the value of game ranch land for large mammal conservation in southern Africa. Journal of Zoology 277: 99-105.
Lloyd PH, Millar JCG. 1983. A questionnaire survey (1969-1974) of some of the larger mammals of the Cape Province. Bontebok 3: 1-49.
Lloyd, P, Stadler DA. 1998. Predation on the tent tortoise Psammobates tentorius: a whodunit with the honey badger Mellivora capensis as prime suspect. African Zoology 33(4): 200-202.
Loock D, SwanepoeL L. 2019. Assessing local patterns in carnivore occupancy and richness by camera trapping: a monitoring of diversity at Verloren Vallei Nature Reserve, Dullstroom, Mpumalanga. Wildlife & Resource Association (WRA), Bloemfontein.
Lynch C.D. 1989. The mammals of the north-eastern Cape Province. Navorsinge van die Nasionale Museum Bloemfontein 25: 1-116.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Mashele NM, Thompson LJ, Downs CT. 2021. Uses of vultures in traditional medicines in the Kruger to Canyons Biosphere Region, South Africa. Journal of Raptor Research. 55(3):328-339.
Mills G. 1997. Honey badger Mellivora capensis. In: Mills G, Hes L. (ed.), The Complete Book of Southern African Mammals, pp. 202. Struik, Cape Town, South Africa.
Mohale DK, Ngoepe E, Mparamoto M, Blumberg L, Sabeta CT. 2023. A case report on a human bite contact with a rabid honey badger Mellivora capensis (Kromdraai Area, Cradle of Humankind, South Africa). Tropical medicine and infectious disease 8(4), p186. https://doi.org/10.3390/tropicalmed8040186
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
NAMC. 2008. The South African Beekeeping Industry: A Section 7 Committee Investigation. . National Agricultural Marketing Council, Pretoria, South Africa.
Ngwenya MP. 2001. Implications of the medicinal animal trade for nature conservation in KwaZulu-Natal, Report No. NA/124/04. Ezemvelo KZN Wildlife.
Nieman WA. 2018. Culture, Conflict, and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human Wildlife Interface. Msc thesis, University of Stellenbosch, Stellenbosch.
Nieman, W.A., Leslie, A.J. and Wilkinson, A., 2018. The Use of Animals and Animal-Derived Constituents in African Traditional Medicine and Other Cultural Applications: Townships in the Western Cape Province. Culture, Conflict and Cuisine: A Quantitative Assessment of Terrestrial Vertebrate Off-Take at the Human-Wildlife Interface, p.215.
Nieman WA, Wilkinson A, Leslie AJ. 2020. Farmer Attitudes and Regional Risk Modelling of Human–Wildlife Conflict on Farmlands Bordering the Boland Mountain Complex, South Africa. African Journal of Wildlife Research 50: 36. https://doi. org/10.3957/056.050.0036.
Nieman WA, Schultz BC, Wilkinson A, Leslie AJ. 2021. Stakeholders’ perceptions of mammal occurrence and abundance on agricultural properties bordering the Boland Mountain Complex, South Africa. African Zoology 56(2): 104-116.
NWPG (North West Provincial Government) 2017. The status of free ranging carnivores in Bokone Bophirima and impacts of hunting them. Department of Rural, Environment and Agricultural Development, North West Provincial Government, Mahikeng.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Power RJ, Van Straaten A, Schaller R, Mooke M, Boshoff T, Nel HP. 2019. An inventory of mammals of the North West Province, South Africa. Annals of the Ditsong Museum of Natural History 8: 6-29.
Proulx G, Abramov AV, Adams I, Jennings A, Khorozyan I, Rosalino LM, Santos-Reis M, Veron G, Do Linh San E. 2016. World distribution and status of badgers – A review. In: Proulx G, Do Linh San E. (ed.), Badgers: Systematics, Biology, Conservation and Research Techniques., pp. 31–116. Alpha Wildlife Publications, Alberta, Canada.
Ramesh T, Kalle R, Downs CT. 2017. Staying safe from top predators: patterns of co-occurrence and inter-predator interactions. Behavioral Ecology and Sociobiology. 71:1-4.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Ray JC, Hunter L, Zigouris J. 2005. Setting Conservation and Research Priorities for Larger African Carnivores. WCS Working Paper No. 24. Wildlife Conservation Society, New York.
Rhodes JI. 2006. Phylogeographic structure of the honey badger (Mellivora capensis). Msc thesis, University of Stellenbosch, Stellenbosch.
Ross MD, Hargey AS, Smyth LK, Mann GK, O’Riain M. 2022. New records of white phenotype honey badgers (Mellivora capensis) from South Africa. African Journal of Wildlife Research 52(1): 12-16.
Rowe-Rowe, D.T. 1992. The Carnivores of Natal. Natal Parks Board, Pietermaritzburg, South Africa.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Schurch MP, McManus J, Goets S, Pardo LE, Gaynor D, Samuels I, Cupido C, Couldridge V, Smuts B. 2021. Wildlife-friendly livestock management promotes mammalian biodiversity recovery on a semi-arid Karoo farm in South Africa. Frontiers in Conservation Science 2:6. https://doi.org/10.3389/fcosc.2021.652415
Smith K, Venter JA, Peel M, Keith M, Somers MJ. 2023. Temporal partitioning and the potential for avoidance behaviour within South African carnivore communities. Ecology and Evolution. 13(8):e10380.
Somerville K. 2022. Jackals, Golden Wolves, and Honey Badgers: Cunning, Courage, and Conflict with Humans. Routledge, London.
Stuart, C.T. 1981. Notes on the mammalian carnivores of the Cape Province, South Africa. Bontebok 1: 1-58.
Thorn M, Green M, Scott D, Marnewick K. 2013. Characteristics and determinants of human-carnivore conflict in South African farmland. Biodiversity and conservation 22: 1715-1730.
Tshabalala T, McManus J, Treves A, Masocha V, Faulconbridge S, Schurch M, Goets S, Smuts B. 2021. Leopards and mesopredators as indicators of mammalian species richness across diverse landscapes of South Africa. Ecological Indicators. 121:107201.
Van der Wal JE, Afan AI, Anyawire M, Begg CM, Begg KS, Dabo GA, Gedi II, Harris JA, Isack HA, Ibrahim JI, Jamie GA. 2023. Do honey badgers and greater honeyguide birds cooperate to access bees’ nests? Ecological evidence and honey‐hunter accounts. Journal of Zoology 321(1):22-32.
Van Wilgen B. 2007. Gums, badgers, & economics?: your questions answered. Quest 3(4), p.41.
Verwey R, Begg C, Begg K, Matthee CA. 2004. A microsatellite perspective on the reproductive success of subordinate male honey badgers, Mellivora capensis. African Zoology. 39(2):305-308.
Waser, P.M. 1980. Small nocturnal carnivores: ecological studies in the Serengeti. African Journal of Ecology 18: 167-185.
Williams ST, Maree N, Taylor P, Belmain SR, Keith M, Swanepoel LH. 2018. Predation by small mammalian carnivores in rural agro-ecosystems: an undervalued ecosystem service?. Ecosystem Services 30: 362-371.
Winter S, Fennessy J, Janke A, Nilsson MA. 2023. Northern olingo (Bassaricyon gabbi), zorilla (Ictonyx striatus), and honey badger (Mellivora capensis) mitochondrial genomes and a phylogeny of Musteloidea. Frontiers in Ecology and Evolution. 10:1089641.