Hippopotamus
Hippopotamus amphibius

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Hippopotamus amphibius – Linnaeus, 1758
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – HIPPOPOTAMIDAE – Hippopotamus – amphibius
Common Names: Hippopotamus, Common Hippopotamus, Large Hippo (English), Seekoei (Afrikaans), Imvubu (Ndebele, Xhosa, Zulu, Swati), Kubu (Sepedi, Sesotho, Setswana), Mvuvhu (Venda), Mpfuvu (Tsonga), Hipopótamo Anfibio (Spanish; Castilian), Hippopotame (French)
Synonyms: No Synonyms
Taxonomic Note:
Grubb (1993) lists five subspecies: H. a. amphibius from eastern Gambia to Sudan, Ethiopia, the northern Democratic Republic of the Congo (DRC), Tanzania and Mozambique, H. a. tschadensis from Nigeria and Chad, H. a. kiboko from Somalia and Kenya, H. a. constrictus from Angola, the southern DRC and Namibia, and finally, H. a. capensis from Zambia southwards to South Africa. It is important to note, however, that this classification has not been widely accepted (Eltringham, 1999; Beckwitt et al. 2016), and recent taxonomic research on this species is lacking. Furthermore, these five subspecies are visually indistinguishable in the wild, and the geographic extent of each range remains vague (Eltringham, 1993).
| Red List Status |
| LC – Least Concern, (IUCN version 3.1) |
Assessment Information
Assessors: Selier, SAJ.1, Fritsch, C.2, Egan, V.T.3 & da Silva, J.1
Reviewers: Ferreira, S.M.4 & Downs, C.T.2
Contributors: Eksteen, J.5 & Goodman, P.6
Institutions: 1South African National Biodiversity Institute, 2University of KwaZulu-Natal, 3Limpopo Department of Economic Development, Environment & Tourism, 4South African National Parks, 5Mpumalanga Tourism & Parks Agency, 6Wildlife Conservation Solutions
Acknowledgements: I. Pienaar, D., Relton, C., Child, M.F., & Page, S.
Assessment Rationale
Range-wide populations of the Common Hippopotamus (hereafter hippo) have declined over the past several decades because of habitat loss and degradation, ongoing droughts, and poaching. Within the assessment region, however, the Least Concern listing remains as the minimum population is currently estimated as at least 9,060 individuals, which equates to a minimum of c. 5,436 –6,342 mature individuals (assuming a 60–70% mature population structure; Lacy et al. 2024).
An updated hippo population estimate of at least 5,700 individuals was reported in 2024 (Lacy et al. 2024) for the Great Limpopo Transfrontier Conservation Area (incl. Kruger National Park (KNP, South Africa), Limpopo National Park (Mozambique) and Gonarezhou National Park (Zimbabwe)) insinuating a likely decrease from the previous 2015 estimates. This is largely because of drought events that caused major declines in many large mammal species (Malherbe et al. 2020). However, a recent update indicates that the KNP hippo population is estimated at 5,889 individuals (end of 2023) (Greaver et al. 2024). Hippo numbers respond dynamically to management and drought. Pre- and post-drought abundances suggest that hippos have a high resilience and recovery potential in protected areas and highlight the ‘boom bust’ nature of the species (Marshall and Sayer, 1976; Chansa et al. 2011; Chomba, 2013; Chomba et al. 2014).
Similar trends have been observed in the St Lucia Game Reserve component of the iSimangaliso Wetland Park KwaZulu-Natal (KZN), where the hippo population has increased from 416 individuals (1978) to approximately 1,000 individuals (2023) (Pers. comms. Ezemvelo KZN Wildlife). This population fluctuates as environmental conditions change. Hippo subpopulations in Mpumalanga increased by 78% between 2003 and 2013 and by 20–30% in Limpopo over the same period (Eksteen et al. 2016). The hippo subpopulations in Mpumalanga and Limpopo provinces are currently considered to be stable. The trend in KZN between 2004 and 2011 was an increase of 4% per year but the population declined between 2011 and 2013 (Eksteen et al. 2016). Additionally, reintroductions into private protected areas will also continue to bolster population size. Thus, overall, the South African population is considered to be stable to increasing at present.
Increasing frequencies of drought spells because of climate change may become a threat to hippos as reductions in grazing areas cause both direct mortality and force hippos into surrounding agricultural landscapes, which may lead to conflict – both increased persecution and poaching. The latter is already manifesting in some regions. For example, in KZN, poaching rates of hippos increased by an average of 21%/year between 2004 and 2013, which represents an emerging threat to this species (Eksteen et al. 2016). Additionally, continued land transformation, altered hydrological patterns, and deteriorating water conditions because of human development will decrease the available area of occupancy. Although these threats are not suspected of causing a net decline in the hippo population over three generations, they should be carefully monitored, and further research should quantify more accurate population size and trends for the assessment region. Once such data are available, this species should be reassessed as the cumulative impacts of intensifying threats may cause a population decline in future.
Despite being an iconic African species, relatively little is published on aspects of hippo ecology and behaviour. Management planning should include identifying areas where hippos can be reintroduced to enhance ecosystem functioning, identifying areas where sustainable use can be implemented, and identifying interventions to mitigate local- or regional-scale threats.
Regional population effects: The hippo distribution range is connected to Mozambique through the waterways of the Great Limpopo Transfrontier Park, Limpopo Province, and Ndumo Game Reserve, KZN. Since rivers traverse borders, rescue effects are possible. However, the many small hippo subpopulations on game farms and small game reserves are not connected. There is a genetic isolation of these small and genetically non-viable subpopulations.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Selier SAJ, Fritsch C, Egan VT & da Silva JM. 2025. A conservation assessment of Hippopotamus amphibius. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
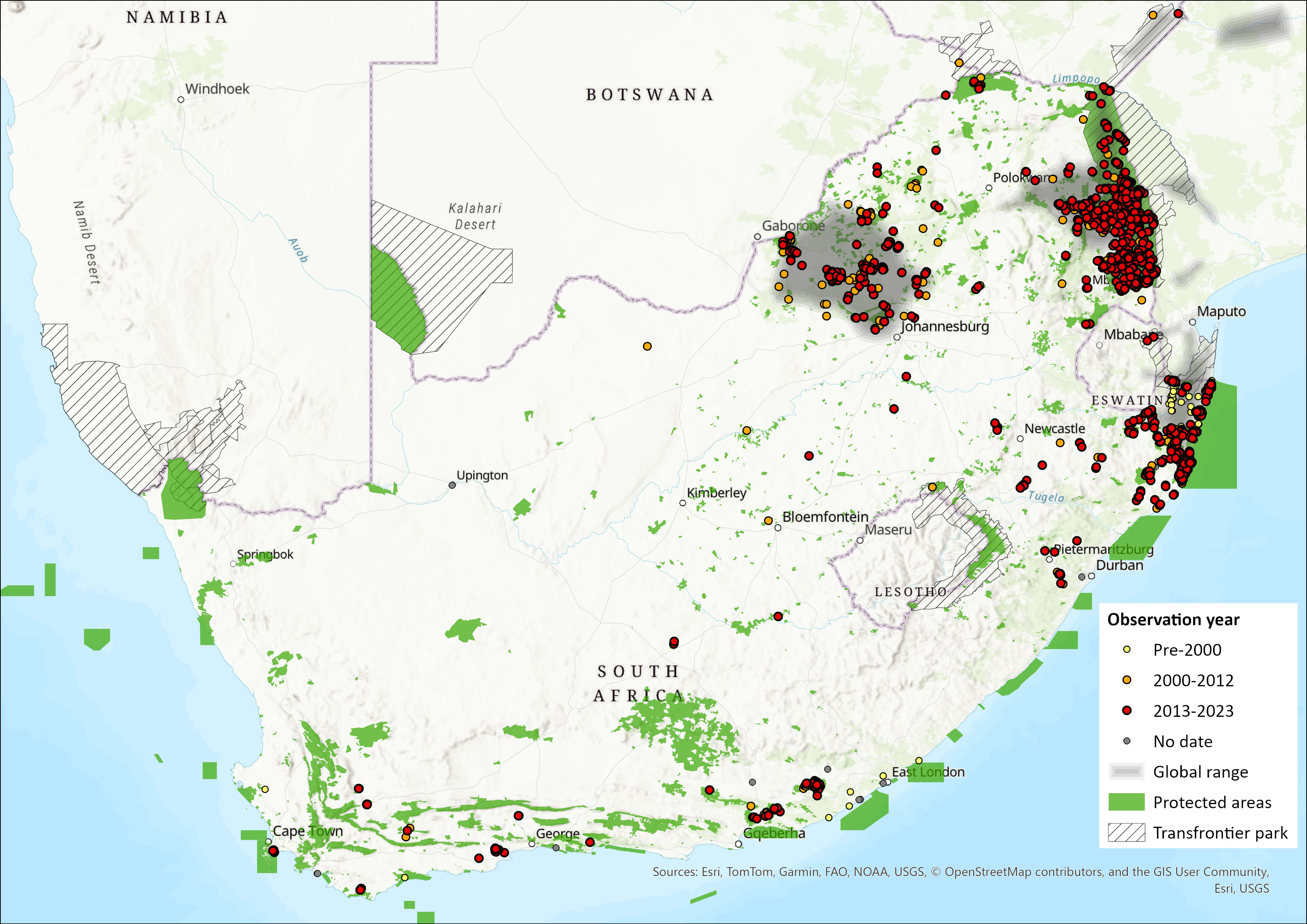
Historically, hippos were widespread throughout sub-Saharan Africa, occurring in most suitable habitats (rivers throughout savannah biomes), but avoiding deserts. Their range distribution extended along the coastline below the escarpment from the Western Cape to KZN to Mozambique, through the Lowveld/bushveld of the former Transvaal and into Zimbabwe, with extensive use of the Orange River system. Bernard and Parker (2006) describe the presence of this species in engravings, near Carnarvon in the Northern Cape Province dating back between 3,200–2,500 years. It is suggested that the change in hippo distribution since the engravings were made may be attributable to overexploitation by humans (Bernard and Parker, 2006). The presence of hippos were reported in a number of areas within the Cape provinces (Skead et al. 2011). These areas include Church Square (Skinner and Smithers, 1990), the Orange River in northern Namaqualand (Boshoff et al. 2013), Table Bay, Malmesbury district, Rondebosch, and the region between the West Coast and Port Elizabeth (Skead et al. 2011) (Fig. 1). In addition, the presence of hippos were recorded in the Northern Cape (Skead et al. 2011) as well as in the Eastern Cape, along the Orange River along the northern border of the Eastern Cape Midlands and the former Transkei (Skead et al. 2007) (Fig. 1). Historical records also note the presence of hippos in the Free State along rivers with suitable grazing resources close to water such as the Klip and Vaal rivers (Boshoff and Kerley, 2013).
Within the assessment region, hippos still occupied much of their former distribution range in 1959, although they had thereafter disappeared from most of South Africa except for the Kruger National Park (KNP) (Sidney, 1965) and northeastern KZN, where subpopulations survived in the coastal lakes and estuaries of St Lucia and Kosi, Lake Sibaya and the Pongola River downstream of Jozini Dam. Historical decline of hippos was probably because of over-hunting (Bernard and Parker, 2006) and dams that changed natural processes in rivers. For example, in Mpumalanga, several rivers often stopped flowing in dry seasons.
Currently, hippo populations are considered fragmented, but widespread across the country. They occur in low-lying areas of northeastern KZN, isolated localities in the Limpopo Province and North West Province, and have been reintroduced into the Fish and Sundays Rivers of the Eastern Cape and the northern bushveld parks in North West Province (Power, 2014). While the majority of the hippos within the Eastern Cape occur within enclosed State protected areas (Fish River Nature Reserve and Addo Elephant National Park) and game farms, some hippos from the State protected areas are likely to move along the Sundays and Fish rivers and their tributaries. Likewise, in Gauteng hippos are mostly restricted to protected areas and game farms, occurring naturally in the Dinokeng area and having been reintroduced into the Cradle of Humankind World Heritage Site, while in the Limpopo, North West and Mpumalanga provinces, hippos now also occur outside of protected areas and game farms along all major rivers. They occur throughout KNP, mainly in the major rivers, but also in isolated pools and man-made dams throughout the park.
In KZN, hippos occur at ten localities within protected areas and in 22 localities on communal and private land (Pers. comms. Ezemvelo KZN Wildlife). The species is mainly confined to the large rivers, coastal lakes and estuaries of north-eastern Zululand and the Maputaland regions of KZN. The hippo population has contracted on the Phongola floodplain because of human population growth and the increasing intensity of agriculture on the region’s floodplains. The remnants of this population survive in Ndumo Game Reserve, where changes in daily, monthly, and annual minimum population numbers have been observed (Fritsch and Downs, 2020). Natural expansions have taken place up the Mkuze River into irrigation and other small dams on game ranches. Additionally, a small population is confirmed to reside in southern Swaziland on a small extension of the Phongola Poort (Jozini) Dam, which protrudes into the country. Other subpopulations are expected to occur in Eswatini but we would have to consult with conservation authorities there for confirmation.
The species has been widely reintroduced into the northern bushveld parks in North West Province (Power 2014). They once occurred in the Orange River (Skead, 1980), as well as the Vaal River, while there exists even older (11,000–12,000 years ago) archaeological evidence from engravings in the Northern Cape to suggest the species did occur widely during sufficiently wet years (Plug and Badenhorst, 2001).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Click on map to view/enlarge
Figure 1. Distribution records for Hippopotamus (Hippopotamus amphibius) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extinct Post-1500 | Native | – | – |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Egypt | Extinct Post-1500 | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extinct Post-1500 | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change poses a significant threat to hippo populations, primarily through alterations in water availability and habitat quality. Increased frequency and severity of droughts in combination with anthropogenic landscape change and water abstraction in Africa have led to the seasonal and permanent distribution of wading habitat in rivers, lakes, and wetland areas (Stommel et al. 2016; Stears et al. 2019; Fritsch et al. 2022). During drought conditions, hippo populations exhibit a decreased birth rate and an increase mortality, because of heat stress, enhanced susceptibility to disease and a decline in forage resources (Smuts and Whyte, 1981; Malherbe et al. 2020; Fritsch et al. 2021). Subpopulations can decline rapidly in these conditions. For example, during the 1991/92 drought, the KNP subpopulation declined by 12.6% in two years (Viljoen, 1995). A similar die-off was experienced in KNP in 2015 (Malherbe et al. 2020). Recovery however was fast since the drought with similar numbers than pre-drought by 2023 (Greaver et al. 2024).
Increasing frequencies of drought spells because of climate change reduce grazing areas, thus causing direct mortality and forcing hippos to move into surrounding agricultural landscapes, which may lead to increased persecution and poaching (Eksteen et al. 2016). Smaller, isolated hippo populations are particularly vulnerable to these pressures, as their limited numbers make recovery from population declines more challenging.
Conversely, climate change can also lead to unseasonal and intense flooding events (Fritsch et al. 2023a; Dube et al. 2023). Rapid-onset floods can inundate grazing areas, reducing the availability of forage and displacing hippo populations (pers. obs. Fritsch). Similarly, fencing may isolate hippo populations from grazing where these are placed close to riverbanks (pers. obs. Egan). Swift currents during such events can erode riverbanks and degrade wading habitats, further limiting suitable environments for hippos. These altered hydrological patterns also disrupt breeding and feeding behaviours, adversely affecting population dynamics (Fritsch et al. 2023a, b).
The congregation of hippos in shrinking water bodies because of prolonged droughts exacerbates water quality issues (Subalusky et al. 2014; Dawson et al. 2016; Stears et al. 2018a, b). High densities of hippos lead to increased defecation in confined areas, elevating nutrient loads and causing eutrophication. This process depletes dissolved oxygen levels, resulting in fish kills and altered aquatic ecosystems (Stears et al. 2018a). Such environmental degradation not only impacts hippos but also the broader freshwater systems.
Climate change-induced drying of rivers can expose Bacillus anthracis (anthrax) spores in sediment – coupled with hippo wallowing and distribution, this can increase the risk of anthrax outbreaks (Stears et al., 2021). As hippos are forced to move in search of wading habitat during periods of low water, they disturb and then transport these spores across the landscape, facilitating the potential spread of disease to other wildlife, livestock, and humans. Finally, human-hippo conflict is increasing and climate-change in combination with anthropogenic expansion and landscape change, will increase instances of conflict as hippos will travel into human areas in search of food and wading resources (Mmbaga, 2022; Tefera et al. 2024).
In summary, climate change-induced alterations in temperature and hydrological cycles, including both droughts and floods, pose substantial risks to hippo populations. These changes affect habitat availability, water quality, and disease transmission, and increase human-hippo conflicts, necessitating proactive conservation strategies to mitigate impacts.
Population information
The overall hippo population in Africa is estimated at between 115,000 and 130,000 (Lewison and Pluháček, 2017), and, across the southern African region, populations are generally considered stable but are declining in other parts of Africa (Lewison and Pluháček, 2017). Hippo populations are naturally regulated by rainfall, because of the fact that they spend much of the day in or near water (Field, 1970). Within the assessment region, there are two major subpopulations, occurring in the Lowveld of northeastern South Africa and that of northern KZN, and several minor subpopulations. Most groups are fragmented by fences or other barriers to movement across the water-savanna ecotone. Water quality and quantity have declined, which has possibly led to further fragmentation. Overall, the minimum current (2018-2023 counts) population size is observed (based on game censuses using aerial surveys) to be 10,000 individuals, which equates to c. 6,000-7,000 mature individuals (assuming a 60–70% mature population structure). Most subpopulations around the country have been stable or increasing over the last decade (SANBI, 2022). However, some localized declines were noted. The total hippo count in KNP in 1986 was estimated at 2,510 individuals across the five major rivers (Joubert 2007). In 2008, an aerial census put the population size at 3,100 individuals (Ferreira et al. 2013). As a result of the 2015 – 2016 drought, hippo numbers in KNP declined by 45.3% from approximately 7,270 individuals in 2015 (prior to the drought) to approximately 3,986 individuals at the end of 2017. The population has since recovered and increased to 5,889 individuals (end of 2023) (Greaver et al. 2024). Since the cessation of population control of hippos within KNP in the late 1980s, the hippo population increased from 2,510 in 1986 to 7,270 in 2015. Hippo culling was reinstated in KNP in 2016, because of a lack of forage resources in the park, though less than 100 individuals have been culled annually within the park since then. The hippo population with KNP appears to be stable.
Hippo numbers respond dynamically to management and drought. Pre- and post-drought abundances suggest that hippos have a high resilience and recovery potential in protected areas and highlight the ‘boom bust’ nature of the species (Marshall and Sayer, 1976; Chansa et al. 2011; Chomba, 2013; Chomba et al. 2014). Similar trends have been observed in the St Lucia Game Reserve component of the iSimangaliso Wetland Park KwaZulu-Natal, where the population has increased from 416 individuals (1978) to approximately 1,000 individuals (2023). This population fluctuates as environmental conditions change. In areas, such as the Associated Private Nature Reserves, bordering KNP, with high densities of artificial waterholes, the hippo numbers have increased from 147 individuals 2004 to 556 individuals at the end of 2022. Hippos, thus, have a high resilience of and recovery ability in response to disturbances in protected areas within South Africa. This is most likely a feature of the species’ biology.
In Mpumalanga, the current hippo subpopulation is estimated at ~ 1,000 animals, where 782 animals occur in provincial and private nature reserves, whilst an estimated 260 to 300 hippos occur in the provincial river systems. In Mpumalanga, the numbers of hippos increased from 562 animals in 2003 to 1,003 animals in 2013, an increase of 78%. These totals are based on game censuses on provincial nature reserves, private nature reserves on the western boundary of KNP, and regular hippo counts along the Lowveld rivers. Most of the hippo subpopulations in the protected areas and rivers close to the KNP show strong positive growth trends, in spite of significant hippo removals through damage-causing animal (DCA) complaints and pro-active hippo capture operations.
In Limpopo, outside the KNP, the hippo numbers are currently estimated at ~ 751 animals. All permanent rivers within the province were surveyed in February 2019. During this survey, 197 hippos were counted along the Olifants River, while 384 were counted within the Letaba River and 25 and 145 in the Levubu and Limpopo rivers respectively (Pers comms. Limpopo Department of Economic Development, Environment and Tourism). A small population of hippos was also observed in the upper Matlabas River. It should be noted that the hippo population in the lower Olifants River is mostly restricted to the Lowveld, with a small, historical population in the upper Olifants having largely retracted into Mpumalanga – it is likely that the intervening area is unoccupied because of high human densities, industrialisation and agriculture, with associated sedimentation of suitable deep water habitats (Ashton et al. 2001). Hippo persistence in the Limpopo River is enabled by the presence of suitable aquatic habitat near weirs (Jacobsen and Kleynhans, 1993). Hippos are also present in low numbers in Mapungubwe and Marakele National Parks, as well as in three provincial nature reserves. In Mpumalanga and Limpopo many individuals on private land are not counted. Since 2017 the hippo population in the province increased from 462 in 2017 to 751 individuals in February 2019. Private landowners are also establishing hippos in dams. The population in Limpopo Province is currently considered stable to increasing but are likely to be greatly influenced by population trends in the adjacent KNP.
According to the North West Provincial Government, there is approximately 200 hippos occur on private land and in protected areas within the North West Province. In the Eastern Cape Province, the hippo population is estimated to be c. 100 (about 26 occurring on state land and 38 occurring on private land in the Cacadu Region) (Pers. comms. Eastern Cape Department of Economic Development, Environmental Affairs and Tourism). While the majority of the hippos within the Eastern Cape occur within enclosed state protected areas (Fish River Nature Reserve and Addo Elephant National Park) and game farms, some hippos from the State protected areas are likely to move along the Sunday and Fish rivers and their tributaries. The hippo population within the Sunday River in Addo Elephant National Park is estimated at between 10 and 20 (2020) and the population within the Great Fish River Nature Reserve is estimated at nine (2018) (Pers. comms. Eastern Cape Parks Board). Upstream of the reserve, there is a hippo conservancy and farmers within this area are accepting of hippos in this stretch of the Great Fish River.
Small groups of hippos occur within Gauteng (approx. 35 individuals) (pers. comms. Gauteng Department of Agriculture and Rural Development), the Western Cape (44 individuals) (Pers. comms. CapeNature) and the Free State (approx. 25 individuals) (Pers. comms. Department of Economic, Small Business Development, Tourism and Environmental Affairs, Free State).
In Eswatini the population is currently estimated at about 120 animals (Eksteen et al. 2016). The hippo distribution expanded between 2004 and 2014, and some DCA hippo control is done to limit conflict between hippos and humans.
The generation length for this species is calculated as 10 years (Pacifici et al. 2013), which yields a 30-year three-generation period. Collation of accurate long-term data is needed to estimate population trends on a national scale. No systematic monitoring data over this time period are currently available.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | Locally (for example, poaching in KwaZulu-Natal) |
Current population trend: Stable with localised declines.
Extreme fluctuations in the number of subpopulations: No
Continuing decline in number of subpopulations: No
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Approx. 3533 – 4122 individuals
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
A study examining the phylogeographic history and structure of H. amphibius across Africa failed to find any clear genetic structure throughout Africa (based on both mitochondrial and nuclear haplotype networks) (Stoffel et al. 2015). Despite this, most sampling areas were found to possess high proportions of private haplotypes associated with river basins, suggesting some level of contemporary genetic isolation. These did not, however, correspond to subspecific delineations (Stoffel et al. 2015), Given that this study only incorporated a single sample from the assessment region (namely South Africa), it cannot be used to infer subpopulations within the region. However, it is likely that hippos once existed as a single metapopulation, but is currently fragmented because of anthropogenic influences, resulting in increased genetic structure because of high levels of inbreeding from small founder groups (e.g., unique haplotypes within certain groups; Beckwitt et al. 2016).A comprehensive population genomic study across the assessment area would help better inform the population genetic structure and diversity within this species.
Assuming H. amphibius exists as a single population, and based on census estimates (Nc) of between 6000-8000 mature individuals and a Nc/Ne conversion ratio between 0.1-0.3, the effective population size (Ne) for this species is estimated at 600-2,400 individuals – above the Ne 500 threshold for a health and stable population. However, if H. amphibius exists as separate subpopulations (and hence the number of mature individuals and Ne are dividing between them, the genetic health of this species within the assessment region may be of concern.
Habitats and ecology
The hippo is an amphibious creature, spending the majority of its day in water, and emerging at night to feed on dry land (Eltringham, 1999). Subtropical floodplain forests, grasslands and coastal grasslands are especially important habitat types for this species. Thus, the ecological requirements for hippos include a supply of permanent water, large enough for the territorial males to spread out at a depth of about 1.4 m (Taylor, 2013), and adequate grazing on open grassland within a few kilometres of the daytime resting sites. Freshwater for drinking is essential when they live in a saline environment – such as in St Lucia (Taylor, 2013). Although they are restricted to regions in the proximity of water, they are able to disperse efficiently from one water source to another. Open water is not always essential as hippos can survive in muddy wallows but must have access to permanent water to which they can return in the dry season. The essential factor is that the skin must remain moist as it will crack if exposed to the air for long periods. A curious feature is the red secretion from modified sweat glands, which is thought to have an antibiotic function.
Wright (1964) argued very convincingly that the greatest benefit of an amphibious lifestyle is thermoregulation as a large body produces a considerable amount of metabolic heat. The water environment acts as a heat sink. Hippos leave their wallows soon after sunset and graze nocturnally on short grass swards up to several kilometres from water. These swards, which are kept short by the grazing activities of the hippo, are known as hippo lawns. Although the hippo grazes every night, except for mothers with very young calves, there are usually individuals present in the water all night, as some return after a few hours and others leave later. Hippos consume approximately 40 kg of grass each night (Klingel, 1983), and may walk up to 35 km during these nocturnal foraging activities. Their movements are not hindered by general fences. Natal buffalo grass (Panicum maximum), bushveld signal grass (Urochloa mosambicensis) and couch rass (Cynodon dactylon) were commonly selected for in northern KZN (Scotcher et al. 1978), whereas Ischaemum fasciculatum is known to be a preferred species in the St Lucia region. During droughts, hippos need to range further, as food resources near to water become depleted. In severe droughts, when food resources are too far for a daily return to their pools, some hippos may not return to water every night, and will lie up in the shade of a tree during daylight hours. Hippos feed by plucking grass with their wide, muscular lips and passing it to the back of the mouth to be ground up by the molars. The front teeth (incisors and canines) play little part in feeding (Skinner & Chimimba 2005). The amount of food ingested is small relative to the size of the animal but its resting habits by day reduce its energetic demands. The stomach is a complex four-chambered structure in which fermentation digestion occurs, i.e. this species does not ruminate (Langer, 1976).
The hippo social system is based on mating territoriality (Klingel, 1991). Hippos are gregarious, social and polygamous animals. Territorial males monopolise a length of the shoreline of the river or lake but tolerate bachelors in their territory provided they behave submissively. Non-breeding males may also settle outside of territorial areas. Fights for the possession of a territory can be fierce and the animals may inflict considerable damage on each other with their huge canines. Minor conflicts are usually settled by threat displays, of which the “yawn” is the most conspicuous. There is little association between hippos when they are feeding at night, except between females and their dependent young. During these periods, males do not behave in a territorial fashion. Both males and females spread their dung by wagging their tails vigorously while defecating, both in the water and on land, where it is thought to have a signalling rather than a territorial function. These dung piles may serve for orientation. Vocalisations take the form of complex bellows and grunts, which presumably also have a signalling function. Sounds may be made either on land or in the water and may be transmitted simultaneously through air and water (Barklow, 2004).
A study in the KNP (Smuts and Whyte, 1981) examined carcasses of 225 hippos randomly culled between 1974 and 1975, and 238 between 1976 and 1977 during population reduction programs. This study showed that hippos are born at a sex ratio of 1♂:1♀. Male mortality rates appear to be much higher than females as this ratio changed to 1♂:2.97♀♀ in the adult age classes. Calves weigh 50 kg at birth and may be born at any time of the year, but the majority are born during the mid-summer (rainy) months. The gestation period is just 8 months – an extraordinarily short time for such a large animal. Growth of the hippo foetus in the womb is, therefore, very rapid. Black rhinos (Diceros bicornis), which are very similar in size to hippos, produce a calf weighing about 40 kg, but their gestation time is almost twice as long as hippos. Growth for the first ten years of life is also very rapid, the average increase in weight is in the order of 100 kg per year, so that at 10 years old they can weigh in excess of 1,200 kg (Whyte, unpubl. data). Males and females are very similar in size, males being only slightly larger than the females. The average weight for 86 adult males culled in KNP was 1,546 kg while 192 females averaged 1,385 kg (Whyte unpubl. data). Females may conceive as early as eight years old, but the average age of sexual maturity is 9–10 years. The mean calving interval is 21.8 months (Smuts and Whyte, 1981), and lactation lasts for 10–12 months (Laws and Clough, 1966). The comparison of data collected in the droughts of 1964 (Pienaar et al. 1966) and data of Smuts and Whyte (1981), which were acquired during a period of above average rainfall (1974/75), showed that Hippo reproduction is very sensitive to deteriorating environmental conditions. Both conception and the percentage of females lactating were significantly higher in the pluvial 1970s.
Ecosystem and cultural services: Hippos form a vital component and are a flagship species of natural water ecosystems. They are important ecosystem engineers, acting as carbon and nutrient vectors between savanna grassland and aquatic habitats (Subalusky et al. 2015). They also physically alter the environment they live in, creating feeding lawns, paths, and channels in swampland. Hippos may be responsible for increasing the fertility of coastal waters, for example, Taylor (2013) estimated the quantities of nutrients hippos bring into the St Lucia estuarine system.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.7. Forest -> Forest – Subtropical/Tropical Mangrove Vegetation Above High Tide Level | – | Marginal | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Marginal | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
| 3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist | – | Marginal | – |
| 4.6. Grassland -> Grassland – Subtropical/Tropical Seasonally Wet/Flooded | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.2. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent/Irregular Rivers/Streams/Creeks | – | Suitable | – |
| 5.3. Wetlands (inland) -> Wetlands (inland) – Shrub Dominated Wetlands | – | Marginal | – |
| 5.5. Wetlands (inland) -> Wetlands (inland) – Permanent Freshwater Lakes (over 8ha) | – | Suitable | – |
| 5.6. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Freshwater Lakes (over 8ha) | – | Suitable | – |
| 5.7. Wetlands (inland) -> Wetlands (inland) – Permanent Freshwater Marshes/Pools (under 8ha) | – | Suitable | – |
| 5.8. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Freshwater Marshes/Pools (under 8ha) | – | Suitable | – |
| 5.13. Wetlands (inland) -> Wetlands (inland) – Permanent Inland Deltas | – | Suitable | – |
| 5.14. Wetlands (inland) -> Wetlands (inland) – Permanent Saline, Brackish or Alkaline Lakes | – | Suitable | – |
| 5.15. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Saline, Brackish or Alkaline Lakes and Flats | – | Marginal | – |
| 5.16. Wetlands (inland) -> Wetlands (inland) – Permanent Saline, Brackish or Alkaline Marshes/Pools | – | Marginal | – |
| 5.17. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent Saline, Brackish or Alkaline Marshes/Pools | – | Marginal | – |
| 9.10. Marine Neritic -> Marine Neritic – Estuaries | – | Marginal | – |
| 13.4. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Brackish/Saline Lagoons/Marine Lakes | – | Marginal | – |
| 13.5. Marine Coastal/Supratidal -> Marine Coastal/Supratidal – Coastal Freshwater Lakes | – | Suitable | – |
Life History
Generation Length: 10 years
Age at maturity: female or unspecified: 9-10 years (Smuts & Whyte 1981)
Age at Maturity: Male: 6 years (Smuts & Whyte 1981)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: Approximately 35 years in the wild
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: Between 225 and 257 days (Apps, 2012; Estes, 1991; Smith, 1988)
Reproductive Periodicity: 18 months (optimal conditions) to 2 years
Average Annual Fecundity or Litter Size: Single calf
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns:(Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial, Freshwater (=Inland waters), Marine
General Use and Trade Information
The hippo is traded both locally and internationally for meat, trophies and in the live animal trade on a subsistence and commercial level. The trade in hippo ivory is regulated by the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES), and currently, this trade is not expected to have any severe effects on the population. In fact, some expect that trade in hippo resources has warranted the introduction of hippos into additional suitable habitats.
An experimental cull of hippos in the KNP was conducted in 1966, and subsequently, culls were executed annually between 1974 and 1982, during which a total of 1,105 individuals were culled (Joubert, 2007). Population declines in the 1980s resulted in the end of culling, and in the late 1980s, culling was no longer considered a necessary strategy for population management, especially considering that hippo abundance was naturally regulated by environmental conditions, e.g. drought (Whyte, 1987). Culling at St Lucia has been documented by Taylor (2009). In the period from 1981 to 1983, a total of 184 hippos were removed. Since 2005, a small number (up to 22 per year) of hippos have been captured each year (Eksteen et al. 2016). The purpose is to dampen the estimated 3% per annum population increase and also to provide hippos for conservation needs elsewhere. At the same time, an objective has been to gain the necessary expertise in the capture of hippos.
More recently (2016), hippo culling has been reinstated in the KNP, where a population of over 7,000 hippos was recently recorded (S. Ferreira unpubl. data). Culling is managed adaptively given the current drought conditions (M. Hofmeyr pers. comm. 2016). These culls are attributable to a lack of forage resources in the park as a result of the severe drought across South Africa during late 2015/early 2016.
Harvesting of hippos, including trophy hunting in South Africa, is well-regulated, and off-takes are managed through an adaptive framework at a local level. While the species is noton the list of Threatened or Protected Species (TOPS) published in terms of the National Environmental Management: Biodiversity Act, 2004 (Act No. 10 of 2004) (NEMBA), sufficient protection exists on a provincial level. The species is listed as protected in all nine provinces, which means that a permit is required for the keeping, translocation and harvesting of the species. The robust system in place in South Africa thus negates the risks associated with trophy hunting that have been observed in some parts of Africa such as the extirpation of hunted species where off-takes are excessive (Lindsey et al. 2007) or the negative evolutionary consequences associated with selective harvesting (Balme et al. 2010; Crosmary et al. 2013; Selier et al. 2014). Trophy hunting in South Africa and across southern Africa is not a threat to the persistence of the species in those countries. Trophy hunting is an important management tool and not only provides revenue that contributes to the conservation and management of the species, but also creates incentives for the keeping of the species and tolerating negative impacts experienced by landowners and communities living with these dangerous animals.
Management responses to damage-causing animals (DCAs) may not necessarily only be paying for damages, but softer conservation outcomes such as addressing local needs, engaging on issues of mutual interest and re-establishing and maintaining amicable relationships with neighbouring communities, thereby providing benefits to human wellbeing as well as conservation (Swemmer et al. 2017). In cases where hippos are controlled through DCA permits, meat distribution to local residents provides benefits and builds positive relationships between protected areas and their neighbours. The removal of DCA hippos is done in cases where hippos pose a risk to people and/or their property. This is primarily done in the context of environmental justice, where people have a constitutional right to a safe and secure environment that does not damage their health or well-being (See section 24 of the South African constitution).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Meat, trophies (including ivory) and live animal trade. | Yes | Meat, trophies (including ivory) and live animal trade. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | – | – |
| 12. Handicrafts, jewellery, etc. | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Meat, trophies (including ivory), harvest for biological control, killing of damage causing animals and live animal trade.
Threats
Habitat loss and land transformation have threatened and continue to threaten hippo populations across their African range, particularly with regard to the drainage of wetland regions and the expansion of agricultural development onto floodplains (Smuts and Whyte, 1981; Jacobsen and Kleynhans, 1993). With the increased droughts associated with climate change and the increased human demands for water, hippo subpopulations are further pressurised. Additionally, lack of grass foraging areas associated with drought, fencing and bush encroachment are a concern. The redirection of water from natural rivers and lakes towards agricultural areas, results in additional habitat loss and deterioration (Cole, 1992; Jacobsen and Kleynhans, 1993; Viljoen and Biggs, 1998). Additionally, poaching for meat and ivory (from their large canine and incisor teeth) has been recognised as a long-lasting and ongoing threat to this species (Vega, 1995), and is enhanced by the increase in human settlement along the boundaries of protected regions (Wittemyer et al. 2008). Estimates of the amount of hippo ivory illegally exported have also increased. A 1994 assessment by TRAFFIC, the monitoring agency of international trade for the IUCN, reported that illegal trade in hippo ivory increased sharply following the international elephant ivory ban in 1989. Between 1991 and 1992, approximately 27,000 kg of hippo canine teeth were exported, an increase of 15,000 kg from the 1989 and 1990 estimates (Weiler et al. 1994). In 1997, more than 1,700 hippo teeth en route from Uganda to Hong Kong were seized by customs officials in France (TRAFFIC, 1997). Five thousand kilograms of hippo teeth (from an estimated 2,000 hippos) of unknown origins were exported from Uganda in 2002 (New Vision, 2002). The Democratic Republic of the Congo once supported Africa’s largest hippo populations, but recent field assessments have revealed population declines of more than 95% because of unregulated hunting pressure (Hillman Smith et al. 2003). Within the assessment region, the projected number of hippo poached in KZN in 2010 was an underestimate, since poaching incident reports along the Pongolo River in Ndumo, KZN, ceased midway through the year. Between 2004 and 2013, hippo poaching incidents have shown an increasing trend, with average poaching in KZN increasing by 21% between 2004 and 2013 (average of six poached per year) (Goodman and Craigie 2014). Generally, within the assessment region, poaching is not considered a major threat to this species. Poaching of hippo is not considered a major treat to the species at present, and is currently localised to two areas. The population in Ndumo Game Reserve, KZN, has declined in recent years from 300 individuals to approximately 203 individuals because of ongoing poaching (Pers comms., Ezemvelo KZN Wildlife).
Mismanagement of water systems and siltation may lead to a decline in habitat and the availability of drinking water, particularly during low rainfall years. Hippo pools may dry out, leading to habitat loss, heat stress and increased competition. The hippo’s reliance on freshwater habitats appears to put them at odds with human populations and adds to their vulnerability, given the growing pressure on freshwater resources across Africa. Additionally, inappropriate fire management may threaten hippo forage availability. Partial loss of habitat in major river systems such as the Orange River has altered habitat, but hippos were long since removed from these areas and appear to be slowly re-colonising areas with suitable habitat. Fritsch et al. 2023a) found that hippos are dependent/controlled by timeous seasonal changes in flow, where wet season high flows permit movement of hippos to remote feeding areas, and allow mothers to give birth in seclusion, while dry season low flows purge weak animals and play a role in controlling numbers. As activities that negatively impact river discharge timing and quality increase, the impact on hippo populations is likely to increase.
During low rainfall years, hippos may move into high-risk areas, coming into conflict with other species or human settlements. Globally, reports of human mortalities from hippo interactions have also increased in recent years. Ten countries reported growing numbers of hippo-human conflicts, in several cases exacerbated by drought conditions (Eksteen et al. 2016). Similarly, Mpumalanga Province has recorded an increase in the number of DCA incidents over the past ten years as increasing human (especially adjacent to protected areas), and hippo populations escalate human-wildlife conflict (J. Eksteen, unpubl. data). Hippos are thought to be responsible for more human deaths than any other mammal, and have been known to attack and kill humans when provoked (Kingdon, 1979). Human and hippo fatalities may also occur because of vehicle collisions (pers. obs. Egan).
Lewison (2007) evaluated the relative impacts of the known threats to hippo persistence – habitat loss (from agricultural or larger-scale development) and hunting pressure – on a model population. While accounting for rainfall variability and demographic stochasticity, the model results suggest that combinations of habitat loss and even moderate levels of adult mortality from hunting (1% of adults) can lead to relatively high probabilities of population declines over the next 30–40 years over its global range. However, reviewing the model, there are several concerns highlighted, 1) there are no baseline model grounded in observational data, 2) the model was not validated with independent data, and 3) the parameters of disturbance are guesses and not grounded in observations, which means that all forecasts have little validity. Lastly, the data were based on one source from 1974 which was from an overabundant population on one river that was harvested. Generalised projections from the Lewison (2007) study may carry significant risks. For instance, populations grow at an intrinsic rate of between 8% and 11% (Marshall and Sayer, 1976). Population growth rates decrease with increase in density especially as the population approaches carrying capacity (K) (Chomba et al. 2012; 2014). The above suggests that hippo populations can withstand a maximum sustainable offtake higher than the 1% reported by Lewison (2007) as well as Lewison and Pluháček, (2017). An analysis suggested sustainable offtakes of up to 4% (Martin and Thomas, 1991) especially if sex or age categories are not targeted (IUCN /TRAFFIC, 2022). This is realised in the example where hippos were harvested (KNP, see above) indicating high resilience to responsible offtakes contrasting the 1% offtake threshold advocated by HSI, based on Lewison (2007).
Conservation
Most populations of hippos are located within protected areas and on game ranches, mostly in dams across South Africa. Only small proportions of the total hippo population in South Africa occur on non-protected private, communal and municipal lands, and may be regarded as a damage-causing animals in these areas, which requires training in holistic management techniques.
A primary conservation intervention, which applies to all aquatic species, is to manage agricultural and industrial development more stringently to ensure that water resources are not drained, diluted or polluted. The government’s Working for Water programme is expected to benefit this species.
The species is not recommended for reintroduction in many parts of the North West Province, for the following reasons (Power, 2014): a) winter frost, b) high risk of escape (and subsequent destruction), and c) their bulk grazing behaviour could have adverse effects on the grass sward (see Skinner and Chimimba, 2005). Reintroductions should follow the IUCN reintroduction and translocation guidelines and should be done for conservation rather than commercial purposes.
Recommendations for land managers and practitioners:
- Develop a Biodiversity Management Plan and identify areas to create conservation corridors.
- Continue and intensify the national population monitoring of all free-ranging hippo populations, and establish monitoring parameters, especially in the context of climate change.
- Ensure that illegal mortalities are reported to the relevant authorities.
- Employ correct fire regimes to ensure habitats remains suitable.
- The sustainable use of this species as part of wildlife-based rural economies should be carefully managed but is encouraged (M. Hofmeyr pers. comm. 2016).
Research priorities:
- The impact of illegal hunting or poaching on population stability and trends.
- The rates of land-use change near hippo subpopulations.
- The effect of climate change on population numbers, distribution and loss of Area of Occupancy.
- The effects of siltation of hippo pools and declining water quality on the dynamics and distribution of this species.
- Taylor (2014) presents the following recommendations which are specific for St Lucia, but generally apply to all the larger populations in South Africa:
-
- To gain an understanding of the hippo population dynamics and to develop a population dynamics model.
-
- To understand the social structure and interactions between hippos.
-
- To describe the impacts of Hippos on the environment in their role as ‘bio-engineers’.
-
- To determine the carrying capacity for the population – and what the consequences will be if this is exceeded.
-
- Understanding hippo-human interactions – both from the aspect of crop-damage and for tourism.
-
- The identification of key aquatic systems that could improve with the reintroduction of hippos and strategies to implement the reintroductions.
Encouraged citizen actions:
- Report hippo sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Manage water resources on private properties by clearing alien invasive vegetation and removing barriers to dispersal to different river systems in times of drought.
Bibliography
Apuuli B, Wright J, Elias C, Burton I. 2000. Reconciling national and global priorities in adaptation to climate change: with an illustration from Uganda. Environmental Monitoring and Assessment 61: 145-159.
Ashton, P.J., Love, D., Mahachi, H. and Dirks, P.H.G.M., 2001. An overview of the impact of mining and mineral processing operations on water resources and water quality in the Zambezi, Limpopo and Olifants Catchments in Southern Africa. Contract Report to the Mining, Minerals and Sustainable Development (Southern Africa) Project, by CSIR-Environmentek, Pretoria and Geology Department, University of Zimbabwe-Harare. Report No. ENV-PC, 42, pp.1-362.
Barklow, W. E. (2004). Amphibious communication with sound in hippos, Hippopotamus amphibius. Animal Behaviour, 68(5), 1125–1132. https://doi.org/10.1016/j.anbehav.2003.10.034
Beckwitt R, Barbagallo J, Breen N, Hettinger J, Liquori A, Sanchez C, Vieira N, Barklow W. 2016. Mitochondrial DNA sequence variation in Hippopotamus amphibius from Kruger National Park, Republic of South Africa. African Zoology 51: 77-82.
Bernard RT, Parker DM. 2006. The use of archaeological and ethnographical information to supplement the historical record of the distribution of large mammalian herbivores in South Africa. South African Journal of Science 102: 117.
Boko M, Niang I, Nyong A, Vogel C, Githeko A, Medany M, Osman-Elasha B, Tabo R, Yanda P. 2007. Africa. Climate Change 2007: Impacts, Adaptation and Vulnerability. In: M. L. Parry, O. F. Canziani, J. P. Palutikof, P. J. van der Linden, and C. E. Hanson (eds), Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change, pp. 433–467. Cambridge University Press, Cambridge, UK.
Cole, M. 1992. Zimbabwe’s hippos threatened by drought. New Scientist 134(1817): 9.
Dawson, J., Pillay, D., Roberts, P.J., & Perissinotto, R. 2016. Declines in benthic macroinvertebrate community metrics and microphytobenthic biomass in an estuarine lake following enrichment by hippo dung. Scientific Reports 6: 37359. https://doi.org/10.1038/srep37359.
Dube, K., Nhamo, G., Chikodzi, D., & Chapungu, L. 2023. Mapping and evaluating the impact of flood hazards on tourism in South African national parks. Journal of Outdoor Recreation and Tourism 43(5): 100661. https://doi.org/10.1016/j.jort.2023.100661.
Eksteen, J., Goodman, P., Whyte, I., Downs, C., & Taylor, R., 2016. A conservation assessment of Hippopotamus amphibius. In Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT, editors. The Red List of Mammals of South Africa, Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Eltringham SK. 1993. The common hippopotamus (Hippopotamus amphibius). In: W. L. R. Oliver (ed.), Pigs, Peccaries and Hippos. Status Survey and Conservation Action Plan, IUCN/SSC Pigs and Peccaries Specialist Group and IUCN/SSC Hippo Specialist Group.
Eltringham, S.K. 1999. The Hippos: Natural history and conservation. Academic Press, London & San Diego.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Field CR. 1970. A study of the feeding habits of the hippopotamus (Hippopotamus amphibius Linn.) in the Queen Elizabeth National Park, Uganda, with some management implications. Zoologica Africana 5: 71-86.
Fritsch, C.J.A., Hanekom, C., and Downs, C.T. 2021. Hippopotamus population trends in Ndumo Game Reserve, South Africa, from 1951 to 2021. Global Ecology and Conservation 32: e01910.
Fritsch, C.J.A., Plebani, M., and Downs, C.T. 2023a. Inundation area drives hippo group aggregation and dispersal in a seasonal floodplain system. Mammalian Biology 102: 1811–1821.
Fritsch, C., Streicher, J., Downs, C.T. 2023b. Activity and nocturnal home range size of the common hippopotamus in perennial river systems in the Kruger National Park, South Africa. African Journal of Ecology 61, 999-1005.
Goodman PS, Craigie J. 2014. KZN Biodiversity Status Assessment Report – 2013. Biodiversity Asset: Hippo (Hippopotamus amphibius).
Grubb P. 1993. The afrotropical hippopotamuses Hippopotamus and Hexaprotodon. In: W. L. R. Oliver (ed.), Pigs, Peccaries and Hippos. Status Survey and Conservation Action Plan, pp. 41-43. Gland, Switzerland.
Hillman Smith, A.K., Merode, E., Smith, F., Ndey, A., Mushenzi, N. and Mboma, G. 2003. Virunga National Park – North Aerial Census of March 2003. Unpublished report ICCN, ZSL, FZL, IRF, USFWS.
Jacobsen, N.H.G. and Kleynhaus, C.J. 1993. The importance of weirs as refugia for hippopotamus and crocodiles in the Limpopo River, South Africa. Water South Africa 19: 301-306.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Joubert SCJ. 2007. The Kruger National Park – a History. High Branching, Johannesburg.
Kingdon JS. 1979. East African Mammals. Academic Press, London.
Klingel H. 1983. Life with gentle giants. Swara 6: 24-27.
Klingel H. 1991. The social organisation and behaviour of Hippopotamus amphibius. In: F. I. Kayanja and E. L. Edroma, (eds), East African Wildlife Research and Management, International Council for Scientific Unions, Paris.
Lacy, H., Beger, M., and Traill, L.W. 2024. Present distribution of common hippopotamus populations in southern Africa, and the need for a centralised database. Biological Conservation 301(5): 110878. https://doi.org/10.1016/j.biocon.2024.110878.
Langer, P. 1976. Functional anatomy of the stomach of Hippopotamus amphibius L. 1758. S.A.J.Sci. 72: 12-16.
Laws RD, Clough G. 1966. Observations on reproduction in the hippopotamus (Hippopotamus amphibious Linn.). In: I. W. Rowlands (ed.), Comparative Biology of Reproduction in Mammals, Academic Press, London.
Lewison R, Oliver W. 2008. IUCN SSC Hippo Specialist Subgroup. Hippopotamus amphibius.The IUCN Red List of Threatened Species
Lewison R. 2007. Population responses to natural and human-mediated disturbances: assessing the vulnerability of the common hippopotamus (Hippopotamus amphibius). African Journal of Ecology 45: 407-415.
Malherbe, J., Smit, I., Wessels, K. J., & Beukes, P. J. (2020). Recent droughts in the Kruger National Park as reflected in the extreme climate index. African Journal of Range and Forage Science, 37(1), 1–17. https://doi.org/10.2989/10220119.2020.1718755
McCauley, D.J., Dawson, T.E., Power, M.E., Finlay, J.C., Ogada, M., Gower, D.B., Caylor, K., Nyingi, W.D., Githaiga, J.M., Nyunja, J., et al. 2015. Carbon stable isotopes suggest that hippopotamus-vectored nutrients subsidize aquatic consumers in an East African river. Ecosphere 6(4): 1-11. https://doi.org/10.1890/ES14-00514.1.
Mmbaga, N. 2022. Human-hippo (Hippopotamus amphibius) interactions in villages adjacent to Lake Babati, Tanzania. Global Ecology and Conservation 40(10): e02316. https://doi.org/10.1016/j.gecco.2022.e02316.
New Vision. 2002. UWA Allows Export of Questionable Teeth.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pienaar U deV, van Wyk P, Fairall N. 1966. An experimental cropping scheme of hippopotami in the Letaba River of the Kruger National Park. Koedoe 9: 1-33.
Plug I, Badenhorst S. 2001. The Distribution of Macromammals in Southern Africa Over the Past 30.000 Years. Transvaal museum.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
SANBI. 2011. Non-detriment finding for Hippopotamus amphibius (Hippopotamus). Scientific Authority, South African National Biodiversity Institute, Pretoria.
Scotcher JSB, Stewart DRM, Breen CM. 1978. The diet of the hippopotamus in Ndumu game reserve, Natal, as determined by faecal analysis. South African Journal of Wildlife Research 8: 1-11.
Skead CJ. 1980. Historical mammal incidence in the Cape Province. Department of Nature and Environmental Conservation, Cape Provincial Administration, Cape Town Volume 1.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smuts GL, Whyte IJ. 1981. Relationships between reproduction and environment in the hippopotamus Hippopotamus amphibius in the Kruger National Park. Koedoe 24: 169-185.
Stears, K., & McCauley, D. 2018a. Hippopotamus dung inputs accelerate fish predation by terrestrial consumers. African Journal of Ecology 56(4): 872-875. https://doi.org/10.1111/aje.12543.
Stears, K., McCauley, D., Finlay, J., Mpemba, J., Warrington, I., Mutayoba, B., Power, M., Dawson, T., & Brashares, J. 2018b. Effects of the hippopotamus on the chemistry and ecology of a changing watershed. Proceedings of the National Academy of Sciences 115: E5028–E5037. https://doi.org/10.1073/pnas.1800407115.
Stears, K., Nuñez, T., Muse, E., Mutayoba, B., & McCauley, D. 2019. Spatial ecology of male hippopotamus in a changing watershed. Scientific Reports 9: 17696. https://doi.org/10.1038/s41598-019-51845-y.
Stears, K., Schmitt, M., Turner, W., McCauley, D., Muse, E., Kiwango, H., Mathayo, D., & Mutayoba, B. 2021. Hippopotamus movements structure the spatiotemporal dynamics of an active anthrax outbreak. Ecosphere 12: e03540. https://doi.org/10.1002/ecs2.3540.
Stoffel, C., Dufresnes, C., Okello, J.B.A., Noirard, C., Joly, P., Nyakaana, S., Muwanika, V.B., Alcala, N., Vuilleumier, S., Siegismund, H.R. and Fumagalli, L. (2015), Genetic consequences of population expansions and contractions in the common hippopotamus (Hippopotamus amphibius) since the Late Pleistocene. Mol Ecol, 24: 2507-2520. https://doi.org/10.1111/mec.13179
Stommel, C., Hofer, H., and East, M. 2016. The effect of reduced water availability in the Great Ruaha River on the vulnerable common hippopotamus in the Ruaha National Park, Tanzania. PLOS ONE 11(6): e0157145. https://doi.org/10.1371/journal.pone.0157145.
Subalusky AL, Dutton CL, Rosi-Marshall EJ, Post DM. 2015. The hippopotamus conveyor belt: vectors of carbon and nutrients from terrestrial grasslands to aquatic systems in sub-Saharan Africa. Freshwater Biology 60: 512-525.
Sydney, J. 1965. The past and present distribution of some African ungulates. Transactions of the Zoological Society of London 3: 1–397.
Taylor RH. 2009. Biodiversity management strategy for hippos in the iSimangaliso Wetland Park. Unpublished document
Taylor RH. 2013. Hippopotamuses. In: R. Perissinotto, D. D. Stretch, and R. H. Taylor (eds), Ecology and Conservation of Estuarine Ecosystems: Lake St Lucia as a Global Model, pp. Cambridge. Cambridge University Press.
Taylor RH. 2014. Hippopotamuses. In: A. K. Whitfield (ed.), Proceedings of the St Lucia natural sciences workshop: change, connectivity and conservation in a major wetland system. Water Research Commission Research Report No.TT582/13, Pretoria.
Tefera, G.G., Tessema, T.H., Bekere, T.A., & Gutema, T.M. 2024. Human-common hippo (Hippopotamus amphibius) conflict in the Dhidhessa Wildlife Sanctuary and its surrounding, Southwestern Ethiopia. PLOS ONE 19(5): e0303647. https://doi.org/10.1371/journal.pone.0303647.
Vega, I. 1995. The hippo, threatened due to ivory trade. Quercus, III, Mayo.
Viljoen, P.C. 1995. Changes in number and distribution of hippopotamus (Hippopotamus amphibius) in the Sabie River, Kruger National Park, during the 1992 drought. Koedoe 38: 115-121.
Viljoen, P.C. and Biggs, H.C. 1998. Population trends of hippopotami in the rivers of Kruger National Park, South Africa. In: N. Dunstone and M. L. Gorman (eds), Behavior and Ecology of Riparian Mammals. Symposia of the Zoological Society of London, pp. 251–279. Cambridge Press, London, UK.
Weiler, P., De-Meulenaer, T. and Vanden-Block, A. 1994. Recent trends in the international trade of hippopotamus ivory. TRAFFIC Bulletin 15: 47-49.
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.
World Water Council – WWC. 2004. Analysis of the Third World Water Forum. Secretariat of the 3rd World Water Forum.
Wright PG. 1964. Wild animals in the tropics. Symposia of the Zoological Society of London 13: 17-28.