Highveld Gerbil
Gerbilliscus brantsii

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Gerbilliscus brantsii – (Smith, 1836)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Gerbilliscus – brantsii
Common Names: Highveld Gerbil Brants’s Gerbil (English), Hoëveldse Nagmuis, Basoetolandse Nagmuis, Springhaasmuis (Afrikaans), Letsoètè (Sesotho)
Synonyms: Tatera brantsii (Smith, 1836), breyeri, draco, griquae, humpatensis, joane, maccalinus, maputa, miliaria, montanus, namaquensis, natalensis, perpallida, ruddi, tongensis (Dempster 2013)
Taxonomic Note: This species was previously described as Tatera brantsii, and a number of subspecies have been described (Skinner & Chimimba 2005). Following the reclassification of this species as Gerbilliscus brantsii, additional research is necessary to delineate subspecies status. In comparison to other Gerbilliscus species in its range (G. afra and G. leucogaster), G. brantsii can be recognised by the white terminal end of its tail and its tail length, which is only slightly longer than its body.
Red List Status: LC – Least Concern
Assessment Information
Assessors: Smith, C.1 & da Silva, J.2
Reviewer: Taylor, P.J.3
Contributor: Patel, T.1
Institutions:1Endangered Wildlife Trust,2South African National Biodiversity Institute, 3University of the Free State
Previous Assessors: Watson, J, du Plessis, J. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern in view of its wide distribution within the assessment region, abundant population, ability to live in a wide variety of habitats, including agricultural landscapes, and because there are no major threats that could cause population decline. This species is considered an agricultural pest during population surges and holistic management control methods, including the use of Barn Owls (Tyto alba), should be encouraged.
Regional population effects: This species has a contiguous habitat across Botswana and southeast Namibia, thus dispersal is highly probable between these regions.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended Citation: Smith C & da Silva JM. 2025. A conservation assessment of Gerbilliscus brantsii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
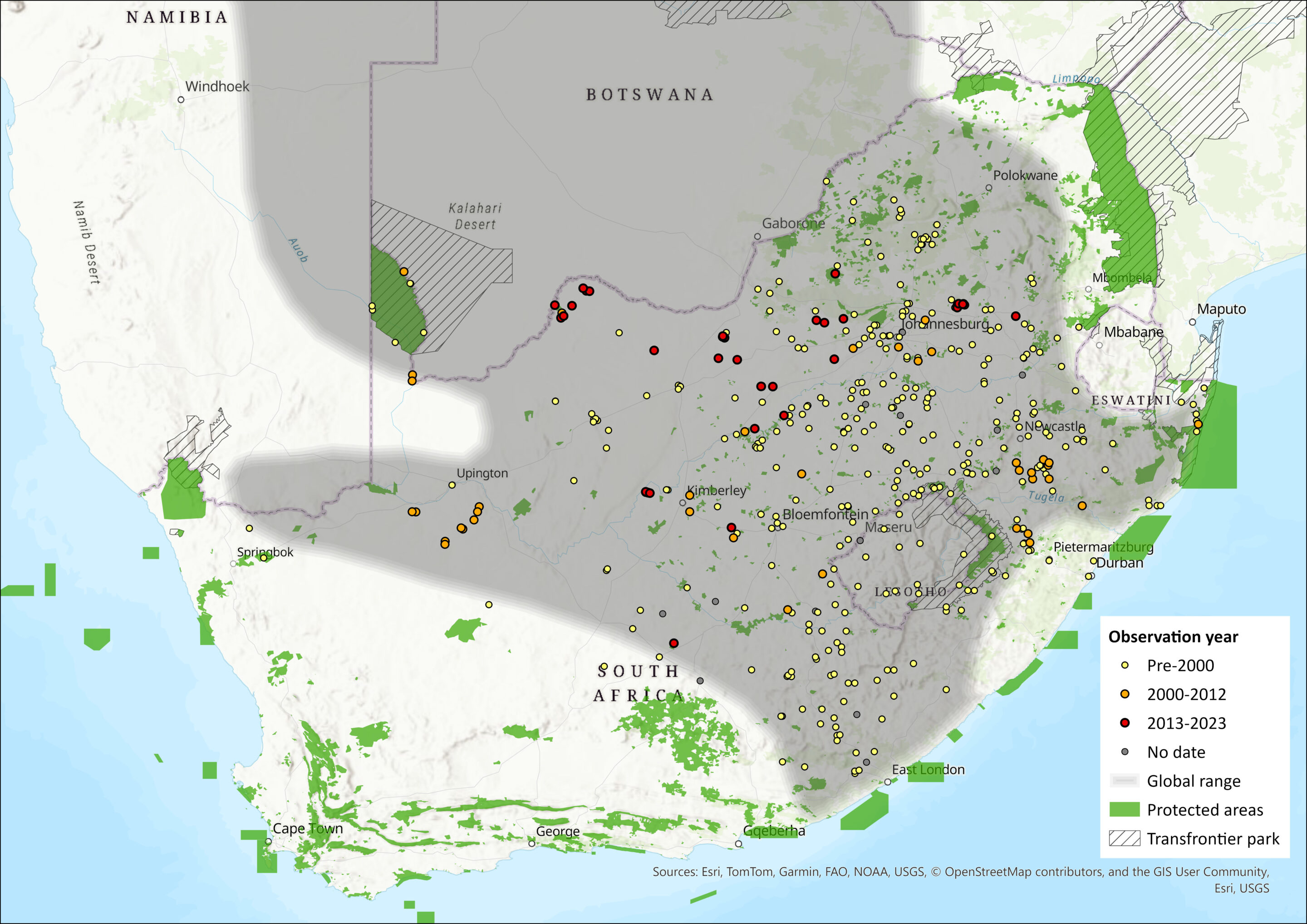
Highveld Gerbils are endemic to southern Africa (but absent from the mesic eastern savannahs and the arid western regions), where they mainly occur in open grasslands with sandy soils in the South-West Arid and Highveld biotic zones and marginally in the southern part of the Zambezian Woodland biotic zone (Dempster 2013; Monadjem et al. 2015). Their distribution includes the eastern half of Namibia, most of Botswana and marginal areas in southeastern Angola, a small western part of Zimbabwe and southwestern Zambia (Skinner & Chimimba 2005).
In the assessment region, they occur in South Africa throughout the North West, Gauteng and the Free State (Lynch 1983; Skinner & Chimimba 2005; Power 2014), in the western parts of Limpopo, the southern parts of Mpumalanga, the western and northeastern parts of KwaZulu-Natal, the northeastern parts of the Eastern Cape and the northern parts of the Northern Cape (Avery et al. 2002; Skinner & Chimimba 2005; Avery & Avery 2011). They are absent from the extremely arid western parts of South Africa (Dempster 2013). According to Lynch (1994) and Skinner and Chimimba (2005) they are rare throughout Lesotho, although later suggestions by Ambrose (2006) indicated they are more abundant in this country. They are absent from Esawtini (Monadjem 1997; Skinner & Chimimba 2005).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Highveld Gerbil (Gerbilliscus brantsii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future.
Population Information
It is a common species with expected cyclic variations in abundance (Avenant 2011). Korn (1987) estimated the density of G. brantsii in the Nylsvley Nature Reserve, Limpopo Province, as between 12 and 16 animals / ha during the dry period, while de Moor (1969) estimated their density to range from 14.8 to 27.1 animals / ha in the South African Highveld. Abundant food in the form of crops leads to higher population densities in this species, which in turn leads to crop damage, which leads to it being considered a pest species in agricultural settings (Skinner & Chimimba 2005; Power 2014). When the population density exceeds a critical limit, natural predators are not able to keep population numbers low enough to reduce crop damage to acceptable limits.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While this genus has been investigated in a phylogenetic context (Colangelo et al. 2007; Abiadh et al. 2010), no population genetic assessment has been undertaken on the species. It is abundant and seemingly connected across its range within the assessment region, and therefore is assumed to exist as a single metapopulation. There is no knowledge of other subpopulation extinctions, therefore the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 subpopulation remaining).
Because the species is locally abundant it is assumed to number in excess of 10 000 individuals throughout its range, hence exceeding the Ne 500 threshold (assuming a 0.1 conversion ratio of effective population size [Ne] to census size). While the 10 000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the proportion of populations with an Ne > 500 indicator, which is valued at 1.0 (1/1 subpopulation with Ne > 500).
Habitats and ecology
Highveld Gerbils are associated with open areas, or plains, in subtropical and wooded grasslands on sandy soils or sandy alluvium and may also be found in peaty soils around marshes or wetlands (Skinner & Chimimba 2005; Dempster 2013). They do not generally occur on heavy consolidated sands or very loose sandy soils and need cover of grass, scrub or open woodland (Skinner & Chimimba 2005; Dempster 2013). In Seekoeivlei Nature Reserve, Free State, the only specimen of G. brantsii was collected in a fallow maize field in sandy soil (Wandrag et al. 2002). In the Free State, they were found to be closely associated with cultivated lands (Lynch 1983), and may be a considerable pest in agricultural areas (Monadjem et al. 2015). For example, they are known to forage on germinating seeds and newly emergent seedlings in cropland (Verdoorn 2010; von Maltitz et al. 2014).
They tend to form small colonies with entrances to burrows five to ten paces apart. They sometimes use the tunnels of mole-rats (Cryptomys spp.) (Dempster 2013). Active burrows are recognised by fresh soil outside the burrow as these animals tend to clean the burrows every evening. They are nocturnal (Foster et al 2019). Despite the small number of individuals in a colony, colony warrens may cover areas as large as 70 ha (de Moor 1969). They are adapted to an omnivorous diet which consists mainly of plant material and a small proportion of insects. They mainly take the green parts, seeds and the roots of plants (Skinner & Chimimba 2005; Dempster 2013).
Ecosystem and cultural services: Highveld Gerbils maintain high plant diversity in savannah habitats (Korn & Korn 1989). This species is recognised as a major reservoir and vector of the bubonic plague, and resultantly plays a significant role in plague epidemiology in southern Africa (NICD 2005).
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
2.1. Savanna -> Savanna – Dry |
– |
Suitable |
– |
|
3.4. Shrubland -> Shrubland – Temperate |
– |
Suitable |
– |
|
3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry |
– |
Suitable |
– |
|
4.4. Grassland -> Grassland – Temperate |
– |
Suitable |
– |
|
8.2. Desert -> Desert – Temperate |
– |
Suitable |
– |
|
14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land |
– |
Suitable |
– |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats to this species. During population explosions it may become an agricultural pest, because they can feed on crops, in some areas which could result in persecution (Verdoorn 2010; von Maltitz et al. 2014, Foster et al 2019). Although this species is a major reservoir for the bubonic plague, it is likely not persecuted for this because it is known not to enter human establishments. Farmers often use poison/chemicals to control the population, which has knock-on effects for the ecosystem (Makundi & Massawe 2011).
Conservation
This species is present in many protected areas throughout the assessment region, including Kgalagadi Transfrontier Park, Sehlabathebe National Park (Lesotho), Golden Gate Highlands National Park, Pilanesberg National Park and Suikerbosrand Nature Reserve. There are no specific interventions necessary, and this species thrives in cultivated areas (predominantly on grain-growing lands). Selective chemical control (Verdoorn 2010; von Maltitz et al. 2014) or biocontrol of population explosions through the use of Barn Owls (Potter 2004) are viable control methods that are currently available and form part of holistic ecosystem management strategies. The use of Barn Owls for population control has been successfully implemented for the closely related G. afra, and is thus a potentially effective method for G. brantsii as well. In a study conducted on G. afra in the Western Cape this method was found to be twice as effective on the gerbil population, compared to the use of poison (Potter 2004). Barn Owls respond to prey explosions by increasing reproduction (Potter 2004; Makundi & Massawe 2011). This management intervention also saves farmers the cost of buying poison and the mortality of non-target species including domestic stock.
Recommendations for land managers and practitioners:
- Barn Owls are being used successfully to control the closely related species, G. afra. This should be encouraged as a holistic management intervention. In order to assist Barn Owls to manage the populations one should erect perches in the crop fields and then nesting boxes in appropriate sites close to the fields.
Research priorities:
- The practicalities and effectiveness of the use Barn Owls as a population control method for this species.
- Taxonomic resolution of the Gerbilliscus genus.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Install Barn Owl nest boxes and perches in crop fields to encourage biocontrol during population explosions.
Bibliography
Abiadh, A., Chetoui, M., Lamine-Cheniti, T., Capanna, E. & Colangelo, P. 2010. Molecular phylogenetics of the genus Gerbillus (Rodentia, Gerbillinae): Implications for systematics, taxonomy and chromosomal evolution,
Molecular Phylogenetics and Evolution, 56 (2), 513-518. https://doi.org/10.1016/j.ympev.2010.04.018.
Ambrose D. 2006. Lesotho annotated bibliography. Section 168: mammals including annotated species lists. House 9 Publications, National University of Lesotho, Roma.
Avenant N. 2011. The potential utility of rodents and other small mammals as indicators of ecosystem ’integrity’ of South African grasslands. Wildlife Research 38: 626–639.
Avery DM, Avery G. 2011. Micromammals in the Northern Cape Province of South Africa, past and present. African Natural History 7: 9–39.
Avery, D.M., Avery, G. and Roberts, A. 2002. A contribution from barn owl pellets to known micromammalian distributions in KwaZulu-Natal, South Africa. African Zoology 37: 131-140.
Colangelo, P., Granjon, L., Taylor, P.J. & Corti, M. 2007. Evolutionary systematics in African gerbilline rodents of the genus Gerbilliscus: Inference from mitochondrial genes, Molecular Phylogenetics and Evolution, 42(3), 797-806. https://doi.org/10.1016/j.ympev.2006.10.001
de Moor PP. 1969. Seasonal variation in local distribution, age classes and population density of the gerbil Tatera brantsi on the South African highveld. Journal of Zoology 157: 399–411.
Dempster ER. 2013. Gerbilliscus brantsii Highveld gerbil. Pages 273–274 in Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Foster, I.B., McIntyre, T. and Haussmann, N.S., 2022. Understanding the relationship between farmers and burrowing mammals on South African farms: are burrowers friends or foes?. In Rethinking Food System Transformation (pp. 69-81). Springer, Cham.
Korn H, Korn U. 1989. The effect of gerbils (Tatera brantsii) on primary production and plant species composition in a southern African savanna. Oecologia 79: 271–278.
Korn H. 1987. Densities and biomasses of non-fossorial southern African savanna rodents during the dry season. Oecologia 72: 410 –413.
Lynch CD. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum, Bloemfontein 10: 177–241.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Makundi RH, Massawe AW. 2011. Ecologically based rodent management in Africa: potential and challenges. Wildlife Research 38: 588–595.
Monadjem A. 1997. Habitat preferences and biomasses of small mammals in Swaziland. African Journal of Ecology 35: 64-72.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
NICD. 2005. Plague control guidelines for South Africa. National Institute for Communicable Diseases, Department of Health, South Africa.
Potter L. 2004. Raptors for rodent control: Is the Barn Owl a viable control agent for pest rodents on South African farmlands? University of Cape Town.
Power RJ. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Verdoorn GH. 2010. Environmentally compatible rodent management for agriculture. Griffon Poison Information Centre.
von Maltitz E, Kirsten F, Malebana P. 2014. Update on gerbil management in maize. SA Grain 16: 106–107.
Wandrag GF, Watson JP, Collins NB. 2002. Rodent and insectivore species diversity of Seekoeivlei Provincial Nature Reserve, Free State province, South Africa. South African Journal of Wildlife Research 32: 137–143.