
Heaviside’s Dolphin
Cephalorhynchus heavisidii

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Cephalorhynchus heavisidii – (Gray, 1828)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Cephalorhynchus – heavisidii
Common Names: Heaviside’s Dolphin, Benguela Dolphin, South African Dolphin (English), Heaviside se dolfyn (Afrikaans), Céphalorhynque du Cap, Dauphin de Heaviside (French), Delfín de Heaviside, Tunina de Heaviside (Spanish; Castilian), Heaviside-Delfin, Kapdelfin (German)
Synonyms: Grampus heavisidii Gray, 1828
Taxonomic Note:
No subspecies have been described for Cephalorhynchus heavisidii (Skinner and Chimimba 2005). Recent genetic research using mitochondrial DNA (mtDNA) control region and thirteen microsatellite loci to determine population genetic structure and gene flow revealed contrasting patterns of geographical variation among seven sampling sites (N = 395 specimens) across South Africa and Namibia (Gopal et al. 2019). Mitochondrial DNA suggested fine-scale division, with six subpopulations identified, whilst microsatellite markers indicated two widespread populations. These results are in contrast to earlier genetic work that found no evidence of population structure between South Africa and Namibia (van Vuuren et al. 2002), due to poor sampling. Gopal et al.’s (2019) results highlight the importance of evaluating multiple markers to have a comprehensive understanding of population structure in order to implement the correct conservation measures and for continual monitoring to take place to ensure the survivorship of this species.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessor: Probert, R.1
Reviewer: Purdon, J.2,3
Contributor: da Silva, J. M.4
Institutions: 1Department of Botany and Zoology, Stellenbosch University, South Africa, 2TUT Nature Conservation, 3The Whale Unit, University of Pretoria, 4South African National Biodiversity Institute
Previous Assessors: Gopal, K. & Plön, S.
Previous Reviewers: Elwen, S. & Child, M.F.
Previous Contributors: Atkins, S., Findlay, K., Meyer, M., Oosthuizen, H. & Relton, C.
Assessment Rationale
This is the only near endemic cetacean species within the assessment region, ranging northwards along the west coast of southern Africa from Cape Point to southern Angola. Recent research indicates a large population within the assessment region: estimated population size is 6,345 individuals (95% CI 3,573–11,267) along ~ 390 km of coastline from Table Bay to Lambert’s Bay. Thus, we infer that there are certainly > 1,000 mature individuals within the assessment region and possibly > 10,000 mature individuals along the entire west coast of the assessment region. Although we suspect that competition with hake (Merluccius spp.) fisheries could represent a threat to this species, this is indirect, as Heaviside’s Dolphins target smaller-sized prey and remain largely inshore of the fishery. A potential emerging threat is the experimental mid-water trawl fishery for Horse Mackerel (Trachurus capensis) operating of the West Coast. Heaviside’s Dolphin bycatch must be monitored, and the potential effects of the full-scale operation should be mitigated if it potentially increases mortality of the species in the future. As this species is limited by water temperature, climate change represents an additional emerging threat. A recent population viability analysis indicated that the population may decline if 63 animals per year are removed (assuming population size is 10,000 individuals), which emphasises the need to quantify mortality rates.
At present, there is no evidence for population decline and thus we list the species as Least Concern. Long-term monitoring of population size and trends is recommended to detect any significant effects of identified threats to this flagship cetacean species. This species should be re-assessed when mortality rates from bycatch and/or subpopulation trends have been quantified.
Regional population effects: The lack of definite population structure between South African and Namibian Heaviside’s Dolphin populations suggests dispersal between the two regions. Furthermore, subpopulation estimates from two high-use areas in Namibia (Walvis Bay: 508 individuals, 95% CI 461–833, and Lüderitz: 494 individuals, 95% CI 403–607) suggest a stable extra-regional population. Most recently, estimates within the Namibian Islands Marine Protected Area (NIMPA) from 2012–2014 using novel visual and acoustic surveys (Gridley et al. 2020) yielded an abundance of 1,594 individuals, 95% CI 776–3,275 (Martin et al. 2020).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Probert R. 2025. A conservation assessment of Cephalorhynchus heavisidii. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Heaviside’s Dolphins are near endemic to South African waters and are non-migratory. They are restricted to the cold continental shelf in waters between 13°c and 17°c (Purdon et al. 2020) of the Benguela ecosystem off southwestern Africa, where they are found in waters between the surf zone and 200 m depth, although most records occur in less than 100 m of water (Findlay et al. 1992; Best 2007). The northernmost record occurs at 16° 30’S (Baia dos Tigros, southern Angola) and the southernmost record at the southwestern tip of the continent (34° 20’S) at Cape Point (Best 2007). The cetacean fauna of Angola is poorly known, and it is uncertain how far north the species’ distribution extends, but it is likely that the distribution is closely linked to the cool waters of the Benguela ecosystem. Records available from both summer and winter surveys in the coastal waters of the Namibe Province, Angola (15° 33’S), do not include sightings of the species (Weir 2010). Although sightings have occurred in the warmer waters to the east of Cape Point, these are considered vagrants (Best 2007; Vinding et al. 2015). Within the assessment region specifically, the species’ range extends from the continental shelf waters off Cape Point to the Orange River. They appear to occur continuously along the coast within this geographic range, with areas of higher density associated with higher levels of prey availability in both South Africa (Elwen et al. 2010) and Namibia (Golaski 2015).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): 100-200 m
Depth Upper Limit (in metres below sea level): 0 m
Depth Zone: (Not specified)
Map
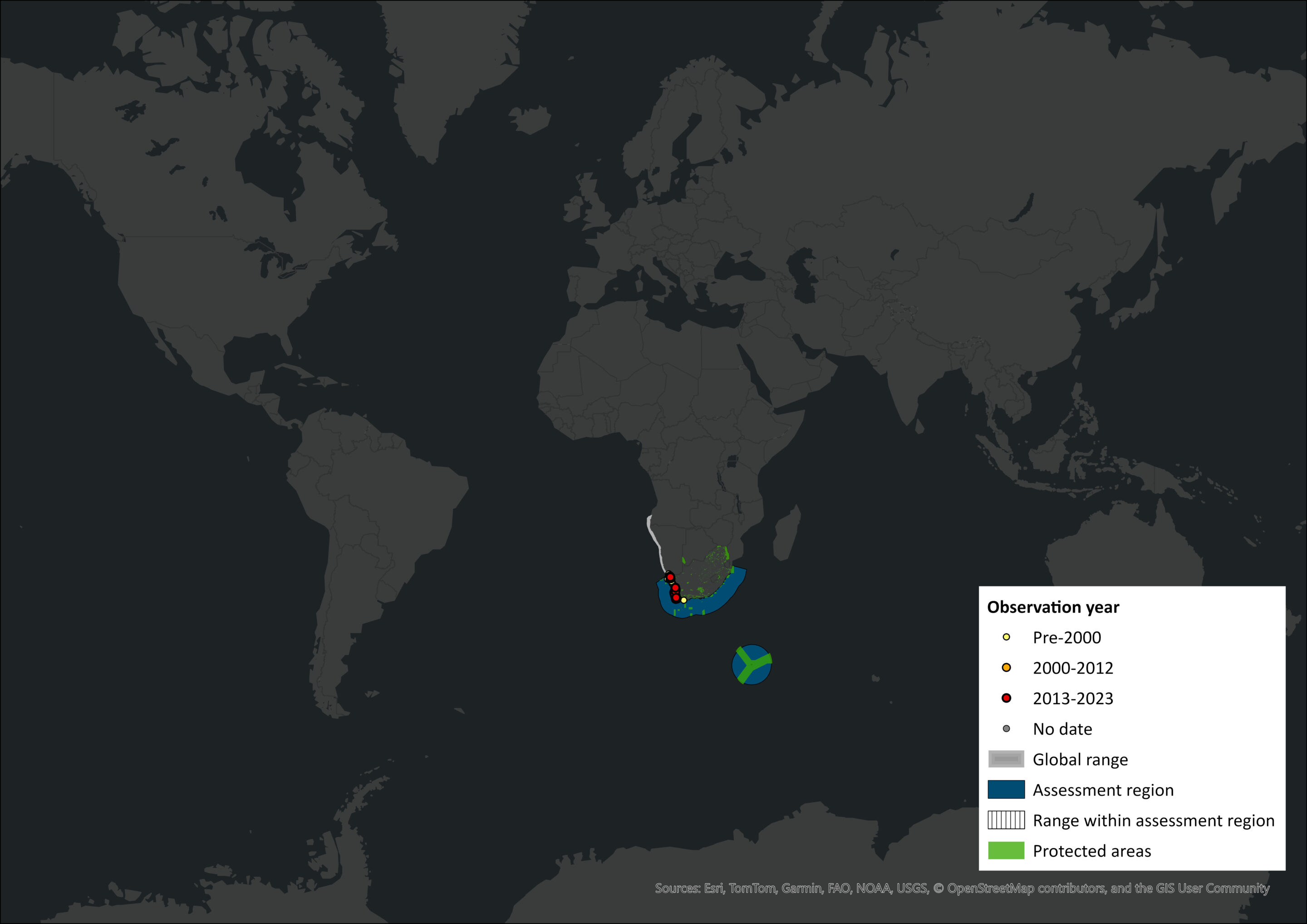 Figure 1. Distribution records for Heaviside’s Dolphin (Cephalorhynchus heavisidii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Heaviside’s Dolphin (Cephalorhynchus heavisidii) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | Resident |
| Namibia | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Northern Cape Province | Extant | Native | – | Resident |
| South Africa -> Western Cape | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: Benguela Current System LME.
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 47. Atlantic – southeast | Extant | Native | – | Resident |
Climate change
Significant shifts in the Benguela Current Large Marine Ecosystem (BCLME) have been documented over the years, including changes in wind patterns, upwelling regimes, and air and sea temperatures (Hutchings et al. 2009; Moloney et al. 2013; Jarre et al. 2015). These changes can have both direct and indirect effects on Heaviside’s Dolphin populations. Heaviside’s Dolphins are cooler and warmer water limited (CWWL) species, narrowing their range to intermediate temperatures that do not cross equatorial or polar boundaries. As this species is limited to the cold temperate shelf waters of the Benguela current, the ability to shift its range southward in the event of increasing sea surface temperatures (SST) is restricted. Even a small shift in SST can impact the species significantly due to the existing narrow habitat range. MacLeod (2009) estimated that the species will be at a 100% risk of extinction due to complete theoretical southward habitat shifts associated with a 5 °C increase in SST. Therefore, Heaviside’s Dolphins are extremely vulnerable to oceanic shifts associated with climate change, particularly increasing temperatures, and they are likely to be severely impacted by these oceanic changes.
Climate change can also exacerbate existing threats on Heaviside’s Dolphins such as prey availability and risks of bycatch. Using simulations of future climate change, increasing temperatures are shown to have the greatest impact on fish biomass in the Benguela current (Ortega-Cisneros et al. 2018a). Amongst the prey species favourable to Heaviside’s Dolphins, Cape hake (Mercillus spp.) is listed as one of the least sensitive to climate change, with other demersal prey fish, such as kingklip, moderately-highly sensitive (Ortega-Cisneros et al. 2018b). Fishing pressure is already increasing within the Benguela system, and with an, albeit low to moderate, potential decline in target fish species, small and large-scale fishing effort may increase. As a result, further pressure will be put on prey resources, and with more fleets and fishing effort, Heaviside’s Dolphins will be at an increased risk of entanglement and bycatch. Given that fishing pressures and climate change both play a role in fluctuating fish biomass, it is difficult to disentangle the role that each of these plays. Further investigation into climate change-associated effects on Heaviside’s Dolphins through prey availability/dietary plasticity and bycatch is needed in this highly complex ecosystem.
Given that rising temperatures are linked to both direct and indirect impacts on Heaviside’s Dolphins, climate change is of critical concern, particularly due to the limited range and distribution of the species. The Benguela current is warming at a “fast” rate (0.15-0.22 oC per decade), with cooling cells located along Cape Columbine, South Africa, and Lüderitz, Namibia (Sweijd and Smit 2020). It is necessary to further understand the distribution of warming rates in the Benguela system and the impacts on the distribution of Heaviside’s Dolphins within their range. But overall, climate change should be considered amongst the major threats to this species, both within the assessment range and extending into the northern Benguela boundary.
Population
No range-wide survey has been conducted for this species. However, population estimates are available at several spatial scales from photographic mark-recapture studies over three years (1999–2001) between Cape Town and Lambert’s Bay, South Africa: 527 animals (95% CI 272–1,020) using 20 km of coastline in western St Helena Bay estimated over 6 weeks of survey effort; using 150 km of coastline around St Helena Bay, estimated over three summer seasons: 3,429 animals (95% CI 1,721–6,828); and using ~ 390 km of coastline from Table Bay to Lambert’s Bay, estimated between two summer field seasons: 6,345 animals (95% CI 3,573–11,267) (Elwen et al. 2009b). Considering the fact that Table Bay to Lambert’s Bay represents roughly half the length of the west South African coastline, doubling this estimate would yield a total of 12,690 individuals within the assessment region. This estimate may be likely given the small home ranges, apparently continuous distribution within the overall range, and the size of estimates available for the widely spaced areas mentioned above. Corroborating this are population estimates from two high-use areas in Namibia (Walvis Bay: 508 animals, 95% CI 461–833, over eight weeks; and Lüderitz: 494 animals, 95% CI 403–607, 14 survey days over one year), and an overall estimate within the Namibian Islands Marine Protected Area (NIMPA: 1,594 individuals, 95% CI 776–3,275, between 2012 and 2014) using a novel method combining the use of photographs and acoustic surveys (Gridley et al. 2020; Martin et al. 2020). Similar densities exist in the northern Benguela (S. Elwen et al. unpubl. data). Although no data are currently available to estimate a population trend, we suspect the population is stable due to its large size and relatively minor threats. However, a recent population viability analysis indicated that the population may decline if 63 animals per year are removed (assuming the population size is 10,000 individuals), which emphasises the need to quantify mortality rates (Gopal 2014).
Continuing decline in mature individuals? No
Current population trend: Unknown, but likely to be stable.
Number of mature individuals in population: c. 12,690
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Six
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: (Not specified)
Number of Subpopulations: 6
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Within the South African habitat range, subpopulations have been identified using two genetic methods applied to seven geographic areas between 2009 and 2012 (Gopal et al. 2019). Analysis of mitochondrial DNA suggests four subpopulations within South Africa. In contrast, microsatellite data suggest two widespread subpopulations: one comprising Table Bay and St Helena Bay further south, and one comprising Lambert’s Bay, Hondeklipbaai and Port Nolloth further north, also including Lüderitz and Walvis Bay, Namibia. The discrepancies between the datasets could reflect differences in dispersal between males and females or historical patterns. Overall, the nuclear microsatellite dataset is thought to represent more contemporary neutral genetic structure.
To date there have been no temporal genetic monitoring studies of Heaviside’s Dolphins within the assessment region. There are no data available to suggest extinction of subpopulations, nor a decline in overall population size or subpopulation sizes. Although gene flow is limited between major bays described above (Gopal et al. 2019), there are no known ecological or behavioural boundaries between subpopulations, aside from apparent difference in diel movement patterns offshore in South Africa compared to Namibia. Furthermore, Heaviside’s Dolphins are thought to exist continuously throughout their range within the Benguela system.
Population estimates from photographic mark-recapture studies estimate a population of 6,345 individuals (95% CI 3,573-11,267) over around half of the South African range (390 km of coastline) between Table Bay and Lambert’s Bay (Elwen et al. 2009b). Doubling this would, therefore, yield 12,690 individuals within South African waters, which constitutes around one-third of the entire Benguela habitat range of the species. From these estimates it can be inferred that there are possibly > 10,000 mature individuals within the assessment area and extending into their northern Benguela range. Given that two subpopulations are found within the region, the number within each can be assumed to be half of the total number. Employing a Ne/Nc conversion ratio of 0.1-0.3, which has been deemed most appropriate for large mammals, the effective population size for each subpopulation is projected to be between 500-1,500 individuals, which is at or exceeding the Ne 500 threshold. However, the reported numbers were estimated over 20 years ago (1999-2001), which highlights the need for more recent population assessments, particularly in these named areas, as well as in the northern part of this assessment region.
Habitats and ecology
The Heaviside’s Dolphin is strongly associated with the cold, northward-flowing Benguela ecosystem off the west coast of southern Africa. This is a shelf-dwelling dolphin, seen mainly in waters less than 100 m deep, where more than 85% of sightings occur in waters with surface temperatures of 9–15 °C (Best & Abernethy 1994). Evidence from photographic mark-recapture (Elwen 2008) and satellite telemetry (Elwen et al. 2006; Davis et al. 2014) suggest individuals maintain small home ranges, extending approximately 50–80 km along the shore, resulting in total home range sizes of between 300 and 2,300 km2, depending on the measure used.
The diet of Heaviside’s Dolphins, as ascertained from caught, stranded and bycaught animals, consists mainly of juvenile hake Merluccius spp. (49% by modified volume) as well as juvenile Goby, Sufflogobius bibarbatus (13.6%), kingklip Ophiphidae spp. (8.5%) and a range of other predominantly demersal fish and cephalopods, including Cape Gurnard (Chelidonichthys capensis), octopus, and Chokka Squid (Best 2007). Most fish caught by Heaviside’s Dolphins are well below the modal length of commercially caught fish (Sekiguchi et al. 1992), and there is little spatial overlap in fishing effort and dolphin habitat (Fairweather et al. 2006). In South African waters, Heaviside’s Dolphins mainly feed nocturnally on prey associated with the deep scattering layer, which migrates closer to the surface at night (Sekiguchi et al. 1992; Elwen et al. 2006, 2009a). By day the majority of dolphins rest and socialise close to shore (Elwen et al. 2006, 2009a). This feeding habit results in a strong diurnal movement pattern, with animals being closest to shore between 06h00 and noon and farthest offshore between 15h00 and 05h00 (Elwen et al. 2006). Although the species similarly exhibits a strong diurnal movement pattern in Namibia, the pattern appears to be reversed with acoustic detections in coastal habitats higher during the night than in the day (Leeney et al. 2011). This is likely driven by differences in prey habitat use and behaviour.
They reach a maximum age of 26 and become sexually mature at 7.5 years. Calving is thought to occur seasonally in summer, and females are able to be simultaneously pregnant and lactating (Skinner & Chimimba 2005).
Ecosystem and cultural services: This is South Africa’s only near endemic cetacean species and a flagship species for the West Coast.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Suitable | Yes |
Life History
Generation Length: 14.4 years
Age at Maturity: Female or unspecified: 7.5 years
Age at Maturity: Male: 7.5 years
Size at Maturity (in cms): Female: 156 – 159 cms
Size at Maturity (in cms): Male: 156 – 159 cms
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): 174 cms
Size at Birth (in cms): 85 cms
Gestation Time: (Not specified)
Reproductive Periodicity: Seasonally in summer
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: Non-migratory
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
Some illegal hunting has been reported, but it is probably not at a significant level (Best & Abernethy 1994; Best 2007). There is presently no known trade in the region, although some localised, illegal hunting or opportunistic use of by-caught animals may still take place on the South African west coast.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Opportunistic use. |
Yes |
Non-consumptive, only for ecotourism. |
National Commercial Value: Yes
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Illegal hunting on small scale.
Threats
Heaviside’s Dolphins are suspected to face fewer threats than many other cetacean species due to limited human population density on the West Coast and the lack of direct competition with the hake industry. The following threats are described and should be quantitatively evaluated for their effect on the species:
Direct mortality from bycatch is probably the largest threat. They are susceptible to entanglement in inshore fishing gear, such as beach seines, purse seines, trawls and gillnets (Best & Abernethy 1994; Peddemors 1999; Elwen et al. 2010). Numbers killed are unknown, but are believed to be < 100 annually, because of short soak times of nets. However, this could be an underestimate, because fishermen may hide the carcasses (Best & Abernethy 1994). A potential emerging threat is the experimental mid-water trawl fishery for horse mackerel operating off the West Coast. Heaviside’s Dolphin bycatch must be monitored, and the potential effects of the full-scale operation should be considered if it might significantly increase mortality of the species in the future. A potential threat is competition with hake (Merluccius spp.) fisheries, which may reduce the prey base or kill animals as bycatch. There is no direct competition for hake as these dolphins target a smaller size class than the commercial fisheries (Sekiguchi et al. 1992). Furthermore, since Heaviside’s Dolphins occur mainly in waters less than 100 m deep, there is little overlap with the commercial hake bottom trawl, which occurs in waters between 100–1,000 m (Fairweather et al. 2006). Since the fishery is well managed and the stocks in South Africa and Namibia are considered sustainable (by the Marine Stewardship Council), although well below pristine levels (van der Westhuizen 2001; Rademeyer et al. 2008), there is no reason to expect a current or increasing threat to Heaviside’s Dolphins from the fishery.
Although there is no evidence for an overall population decline at present, small home range size may facilitate local declines and climate change may exacerbate existing threats. Significant changes in marine ecosystems have already been recorded in terms of air and sea temperatures, wind patterns, ocean current speed and upwelling regimes (Mead et al. 2013; Moloney et al. 2013; Jarre et al. 2015). Heaviside’s Dolphins are limited by water temperatures that are either too cold or too warm (so-called CWWL species) and are listed amongst the species most vulnerable to climate change (MacLeod 2009).
Although fully legally protected, some direct illegal harvesting has been reported in the past (Rice & Saayman 1984; Best & Abernethy 1994), but there are no known records of this recently.
Concern has been expressed about the potential effects of boat traffic and pollution (Culik 2005; MacLeod 2009). Although behavioural changes in response to boat traffic have been identified in some areas in Namibia (Elwen et al. in review; MacLeod 2009), low human population densities and the scarcity of large ports along most of the species’ range probably help reduce the possibility of adverse effects from boat traffic at a population level (P. Best pers. comm. 2013). However, there was one incident reported in 2010 where a single individual was observed with series of propellor strike injuries, which subsequently healed (Elwen & Leeney 2010). It should be noted that vessel collisions with small cetaceans are rare, but high site fidelity and higher densities of dolphins in certain areas may result in localized impacts in areas with high levels of boat traffic.
Organochlorine levels in a small sample of Heaviside’s Dolphins failed to indicate significant exposure to DDT in the coastal waters of South Africa’s west coast, where the scarcity of arable land and low rainfall may help minimise pesticide residue inputs to the marine environment (de Kock et al. 1994). A more recent analysis of heavy metals in the skin of Heaviside’s Dolphins from South Africa and Namibia revealed that, while levels were higher than expected for some metals, sampled individuals were deemed to be at relatively minimal risk of lethal poisoning (Serot 2013). Additionally, clear differences between sampling sites were detected, thereby corroborating the existence of population structure and small home range.
In recent years, noise pollution has become a great concern, particularly for coastal cetaceans. To date there have been no studies done on the direct effects of underwater noise on Heaviside’s Dolphins, however there is increasing evidence of noise impacts on the behaviour of other coastal dolphin species, including other Cephalorhynchus spp. (Martinez et al. 2012; Paiva et al. 2015; Marley et al. 2017; Carome et al. 2023). As there are areas of high levels of anthropogenic activity, such as tourism and commercial shipping, throughout the range of Heaviside’s Dolphins, the effects of anthropogenic noise on the species needs to be considered in future studies.
Population viability analysis (PVA) is a commonly used tool to forecast extinction risk as well as assessing a species’ threat category, whereas sensitivity analyses consider the effects that changes in demographic parameters (population size, age, birth and death rate and migration) or environmental variations can have on the resilience of wildlife populations, and the effect of different management approaches that can be tested. Sensitivity analysis was examined with various hypothetical scenarios whereby demographic parameter values were varied to examine potential population responses to threats. The modelled PVA exercise was inconclusive, because parameter values used were based on a threatened sister species, Cephalorhynchus hectori, and did not produce models that showed stable populations, suggesting these values do not apply to Heaviside’s Dolphins. However, when the model parameters were modified to produce a stable population, simulated rates of removal (for example, from bycatch and/or illegal harvesting) suggested that the population might decline under removal rates of 63 animals per year if population size is 10,000 individuals. Because of the seriousness of this modelling result, there is an urgent need for long-term life history data, inclusive of the direct and indirect threats faced by this species, to completely understand the biology and behaviour of the population (Gopal 2014).
Current habitat trend: Declining in quality. The West Coast has become cooler over the past 20–30 years (Mead et al. 2013), where simultaneously oxygen concentrations have decreased (Moloney et al. 2013). The specific effects of these patterns on Heaviside’s Dolphin distribution, either directly or indirectly through resource shifts, have not been documented and should be monitored.
Conservation
This species occurs in the West Coast National Park Marine Protected Area (MPA) and is listed on Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and the Marine Living Resources Act (No. 18 of 1998). Suggested interventions are further establishments of MPAs along the west coast, stricter regulation of the hake and horse mackerel fisheries, and zoning of boat traffic in high-density use areas, such as Granger Bay, Cape Town:
- MPAs would be best established at Table Bay, Yzerfontein or Lambert’s Bay where Heaviside’s Dolphin abundance is highest, due to the use of predictable breeding and foraging sites.
- Inshore net management of fisheries can be achieved by assessment of risk to dolphin populations and education of artisanal fishers.
- Zoning boat traffic could be achieved by relating traffic patterns to dolphin high-use areas and activity patterns, where boat traffic is restricted between 06h00 and noon when dolphins are most likely to be closest to the shore (Elwen et al. 2009a).
More research emphasis should be placed on possible detrimental interactions due to tour vessels and the overfishing of prey stocks, especially hake, and resilience of the species to adapt to alternate prey. Increased knowledge of how Heaviside’s Dolphins respond to boat traffic and the impact of this at a subpopulation level would be informative to potentially modify vessel behaviour/area use and reduce impacts. The direct and indirect effects of climate change on the distribution and mortality of Heaviside’s Dolphins needs attention as the species is extremely vulnerable, particularly to increasing sea surface temperatures. Habitat use, survival, and prey selection of the species should be assessed concurrently with the changes in the Benguela Current Large Marine Ecosystem associated with climate change to further understand the magnitude of the climatic impacts within this highly complex ecosystem.
Recommendations for managers and practitioners:
- The systematic monitoring of identified subpopulations to determine subpopulation size and trends is recommended. Both line-transect (Slooten et al. 2006) or mark-recapture (Gormley et al. 2005) methodologies are suitable for this species and have been used on the closely related Hector’s Dolphin (C. hectorii).
- Use of set nets in inshore waters of the Western Cape and Northern Cape requires careful monitoring and management to determine levels of bycatch and, if necessary, the initiation of mitigation measures.
Research priorities:
Most research on the species is, or has been, conducted or coordinated by members of the Mammal Research Institute of the University of Pretoria with research focussed on a number of topics, including distribution (Elwen et al. 2010), abundance and trends (Elwen et al. 2009b; Martin et al. 2020), foraging strategies (Elwen et al. 2009a, 2010; Leeney et al. 2011), ecological niche (stable isotope analysis), genetic population structure (Gopal et al. 2019) and heavy metal analysis (Serot 2013). Research has also been conducted on vocalisation characteristics (Morisaka et al. 2011; Martin et al. 2018, 2019) and response to tagging attempts (Sakai et al. 2011). Specific research priorities include:
- Density and abundance estimates along the entire range are needed to estimate total population size.
- For a comprehensive picture of the population genetic structure, additional samples (biopsy/stranded) should be obtained from areas not sampled previously.
- Estimates of dispersal rates and bycatch (including mapping geographical overlap with fisheries) are needed to evaluate their impacts on the population.
- Studies of ecological niche and adaptability to different foraging environments are necessary to understand likely impacts of environmental change due to climate change and other anthropogenic impacts.
- Investigate the impacts of anthropogenic noise, such as vessel noise, seismic explosions, and pile driving, on the behaviour, presence, and distribution of Heaviside’s dolphins throughout their range.
Encouraged citizen actions:
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, for example wwfsa.mobi, FishMS 0794998795.
- Save electricity and fuel to mitigate CO2 emissions and hence rate of climate change.
- Buy local products that have not been internationally shipped.
- Reduce boat speed in high-density Heaviside’s Dolphin areas.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution.
Bibliography
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Best, P.B. and Abernethy, R.B. 1994. Heaviside’s dolphin Cephalorhynchus heavisidii (Gray, 1828). In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Volume 5: The first book of dolphins, pp. 289-310. Academic Press.
Carome W, Rayment W, Slooten E, Bowman MH, Dawson SM. 2023. Vessel traffic influences distribution of Aotearoa New Zealand’s endemic dolphin (Cephalorhynchus hectori). Marine Mammal Science 39(2):626-47.
Culik, B. M. 2004. Review of small cetaceans: Distribution, behaviour, migration and threats. Marine Mammal Action Plan/Regional Seas Reports and Studies 177: 343 pp.
Davis, R.W., David, J.H.M., Meÿer, M.A., Sekiguchi, K., Best, P.B., Dassis, M., and Rodríguez, D.H. 2014. Home range and diving behaviour of Heaviside’s dolphins monitored by satellite off the west coast of South Africa. African Journal of Marine Science 36: 455-466.
De Kock, A.C., Best, P.B., Cockcroft, V.G. and Bosma, C. 1994. Persistent organochlorine residues in small cetaceans from the east and west coasts of southern Africa. Science of the Total Environment 154: 153-162.
Elwen SH, Golaski SB, Leeney RH, Shaanika T, Roux J-P, Gridley T. in review. Boat avoidance as a source of capture heterogeneity in Heaviside’s dolphins (Cephalorhynchus heavidii) in Namibia. PLoS One..
Elwen SH, Thornton M, Reeb D, Best PB. 2010. Near-shore distribution of Heaviside’s (Cephalorhynchus heavisidii) and dusky dolphins (Lagenorhynchus obscurus) at the southern limit of their range in South Africa. African Zoology 45: 78-91.
Elwen SH. 2008. The distribution, movements and abundance of Heaviside’s dolphins in the nearshore waters of the Western Cape, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Elwen, S.H., Best, P.B., Reeb, D., and Thornton, M. 2009a. Diurnal movements and behaviour of Heaviside’s dolphins, Cephalorhynchus heavisidii, with some comparative data for dusky dolphins, Lagenorhynchus obscurus. South African Journal of Wildlife Research 39: 143-154.
Elwen, S.H., Meÿer, M.A., Best, P.B., Kotze, P.G.H., Thornton, M,. and Swanson, S. 2006. Range and movements of female Heaviside’s dolphins (Cephalorhynchus heavisidii), as determined by satellite-linked telemetry. Journal of Mammalogy 87(5): 866-877.
Elwen, S.H., Reeb, D., Thornton, M., and Best, P.B. 2009b. A population estimate of Heaviside’s dolphins, Cephalorhynchus heavisidii, at the southern end of their range. Marine Mammal Science 25: 107-124.
Fairweather TP, Booth AJ, Sauer WHH, Leslie RW. 2006. Spatial description of hake-directed fishing activity off the west coast of South Africa. African Journal of Marine Science 28: 13-24.
Elwen SH, Leeney RH. 2010. Injury and subsequent healing of a propeller strike injury to a Heaviside’s dolphin (Cephalorhynchus heavisidii). Aquatic Mammals 36(4): 382-387.
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Golaski S. 2015. Spatial and temporal patterns of habitat use of Heaviside’s dolphins in Namibia. M.Sc. Thesis. University of Pretoria, Pretoria, South Africa.
Gopal, K. 2014. A genetic study of two inshore dolphin species (Cephalorhynchus heavisidii and Tursiops aduncus) found along the coast of South Africa. PhD thesis, University of Pretoria.
Gopal K, Karczmarski L, Tolley KA. 2019. Patterns of geographic variation between mitochondrial and nuclear markers in Heaviside’s (Benguela) dolphins (Cephalorhynchus heavisidii). Integrative Zoology 14(5):506-26.
Gormley AM, Dawson SM, Slooten E, Bräger S. 2005. Capture-recapture estimates of Hector’s dolphin abundance at Banks Peninsula, New Zealand. Marine Mammal Science 21: 204-216.
Gridley T, Martin MJ, Slater J, Roux JP, Swift RJ, Elwen SH. 2020. Towed passive acoustic monitoring complements visual survey methods for Heaviside’s dolphins Cephalorhynchus heavisidii in the Namibian Islands Marine Protected Area. African Journal of Marine Science 42(4):495-506.
Hutchings L, Van der Lingen CD, Shannon LJ, Crawford RJ, Verheye HM, Bartholomae CH, Van der Plas AK, Louw D, Kreiner A, Ostrowski M, Fidel Q. 2009. The Benguela Current: An ecosystem of four components. Progress in Oceanography 83(1-4):15-32.
Jarre A, Hutchings L, Kirkman SP, Kreiner A, Tchipalanga PC, Kainge P, Uanivi U, van der Plas AK, Blamey LK, Coetzee JC, Lamont T. 2015. Synthesis: climate effects on biodiversity, abundance and distribution of marine organisms in the Benguela. Fisheries Oceanography 24:122-49.
Leeney, R.H., Carslake, D., and Elwen, S.H. 2011. Using static acoustic monitoring to describe echolocation behaviour of Heaviside’s dolphins (Cephalorhynchus heavisidii) in Namibi. Aquatic Mammals 37: 151-160.
MacLeod, C.D. 2009. Global climate change, range changes and potential implications for the conservation of marine cetaceans: a review and synthesis. Endangered Species Research 7: 125-136.
Marley SA, Salgado Kent CP, Erbe C, Parnum IM. 2017. Effects of vessel traffic and underwater noise on the movement, behaviour and vocalisations of bottlenose dolphins in an urbanised estuary. Scientific Reports 7(1):13437.
Martin MJ, Gridley T, Elwen SH, Jensen FH. 2018. Heaviside’s dolphins (Cephalorhynchus heavisidii) relax acoustic crypsis to increase communication range. Proceedings of the Royal Society B: Biological Sciences 285(1883):20181178.
Martin MJ, Elwen SH, Kassanjee R, Gridley T. 2019. To buzz or burst-pulse? The functional role of Heaviside’s dolphin, Cephalorhynchus heavisidii, rapidly pulsed signals. Animal behaviour 150:273-84.
Martin MJ, Gridley T, Roux JP, Elwen SH. 2020. First abundance estimates of Heaviside’s (Cephalorhynchus heavisidii) and dusky (Lagenorhynchus obscurus) dolphins off Namibia using a novel visual and acoustic line transect survey. Frontiers in Marine Science 7:555659.
Martinez E, Orams MB, Pawley MD, Stockin KA. 2012. The use of auditory stimulants during swim encounters with Hector’s dolphins (Cephalorhynchus hectori hectori) in Akaroa Harbour, New Zealand. Marine Mammal Science 28(3):E295-315.
Mead A, Griffiths CL, Branch GM, McQuaid CD, Blamey LK, Bolton JJ, Anderson RJ, Dufois F, Rouault M, Froneman PW, Whitfield AK. 2013. Human-mediated drivers of change—impacts on coastal ecosystems and marine biota of South Africa. African Journal of Marine Science 35(3):403-25.
Moloney, C.L., Fennessey, S.T., Gibbons, M.J., Roychoudhury, A., Shillington, F.A. von der Heyden, B.P. and Watermeyer, K. 2013. Reviewing evidence of marine ecosystem change off South Africa. African Journal of Marine Science 35(3): 427-448.
Morisaka T, Karczmarski L, Akamatsu T, Sakai M, Dawson S, Thornton M. 2011. Echolocation signals of Heaviside’s dolphins (Cephalorhynchus heavisidii). The Journal of the Acoustical Society of America 129: 449-457.
Ortega‐Cisneros K, Cochrane KL, Fulton EA, Gorton R, Popova E. 2018a. Evaluating the effects of climate change in the southern Benguela upwelling system using the Atlantis modelling framework. Fisheries Oceanography 27(5):489-503.
Ortega-Cisneros K, Yokwana S, Sauer W, Cochrane K, Cockcroft A, James NC, Potts WM, Singh L, Smale M, Wood A, Pecl G. 2018b. Assessment of the likely sensitivity to climate change for the key marine species in the southern Benguela system. African Journal of Marine Science 40(3):279-92.
Paiva EG, Salgado Kent CP, Gagnon MM, McCauley R, Finn H. 2015. Reduced detection of Indo-Pacific bottlenose dolphins (Tursiops aduncus) in an inner harbour channel during pile driving activities. Aquatic Mammls 41(4):455-68.
Peddemors, V. M. 1999. Delphinids of southern Africa: a review of their distribution, status and life history. Journal of Cetacean Research and Management 1(2): 157-165.
Rademeyer RA, Butterworth DS, Plagányi É. 2008. Assessment of the South African hake resource taking its two-species nature into account. African Journal of Marine Science 30: 263-290.
Rice, F.H. and Saaymann, G.S. 1984. Movements and behaviour of Heaviside’s dolphins (Cephalorhynchus heavisidii) off the western coasts of southern Africa. Investigations in Cetacea 16: 49-63.
Sakai M, Karczmarski L, Morisaka T, Thornton M. 2011. Reactions of Heaviside’s dolphins to tagging attempts using remotely-deployed suction-cup tags. South African Journal of Wildlife Research 41: 134-138.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Serot, J.L. 2013. Heavy metal analysis in Heaviside’s dolphins (Cephalorhynchus heavisidii). Master of Science thesis, University of Hong Kong.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Slooten E, Rayment W, Dawson S. 2006. Offshore distribution of Hector’s dolphins at Banks Peninsula, New Zealand: is the Banks Peninsula Marine Mammal sanctuary large enough? New Zealand Journal of Marine and Freshwater Research 40: 333-343.
Sweijd NA, Smit AJ. 2020. Trends in sea surface temperature and chlorophyll-a in the seven African Large Marine Ecosystems. Environmental Development 36:100585.
Vinding K, Bester M, Kirkman SP, Chivell W, Elwen SH. 2015. The use of data from a platform of opportunity (whale watching) to study coastal cetaceans on the southwest coast of South Africa. Tourism in Marine Environments 11: 33-54.
Weir CR. 2010. Cetaceans observed in the coastal waters of Namibe Province, Angola, during summer and winter 2008. Marine Biodiversity Records 3: e27.
van Vuuren BJ, Best PB, Roux J-P, Robinson TJ. 2002. Phylogeographic population structure in the Heaviside’s dolphin (Cephalorhynchus heavisidii): conservation implications. Animal Conservation 5: 303-307.
van der Westhuizen A. 2001. A decade of exploitation and management of the Namibian hake stocks. South African Journal of Marine Science 23: 307-315.
