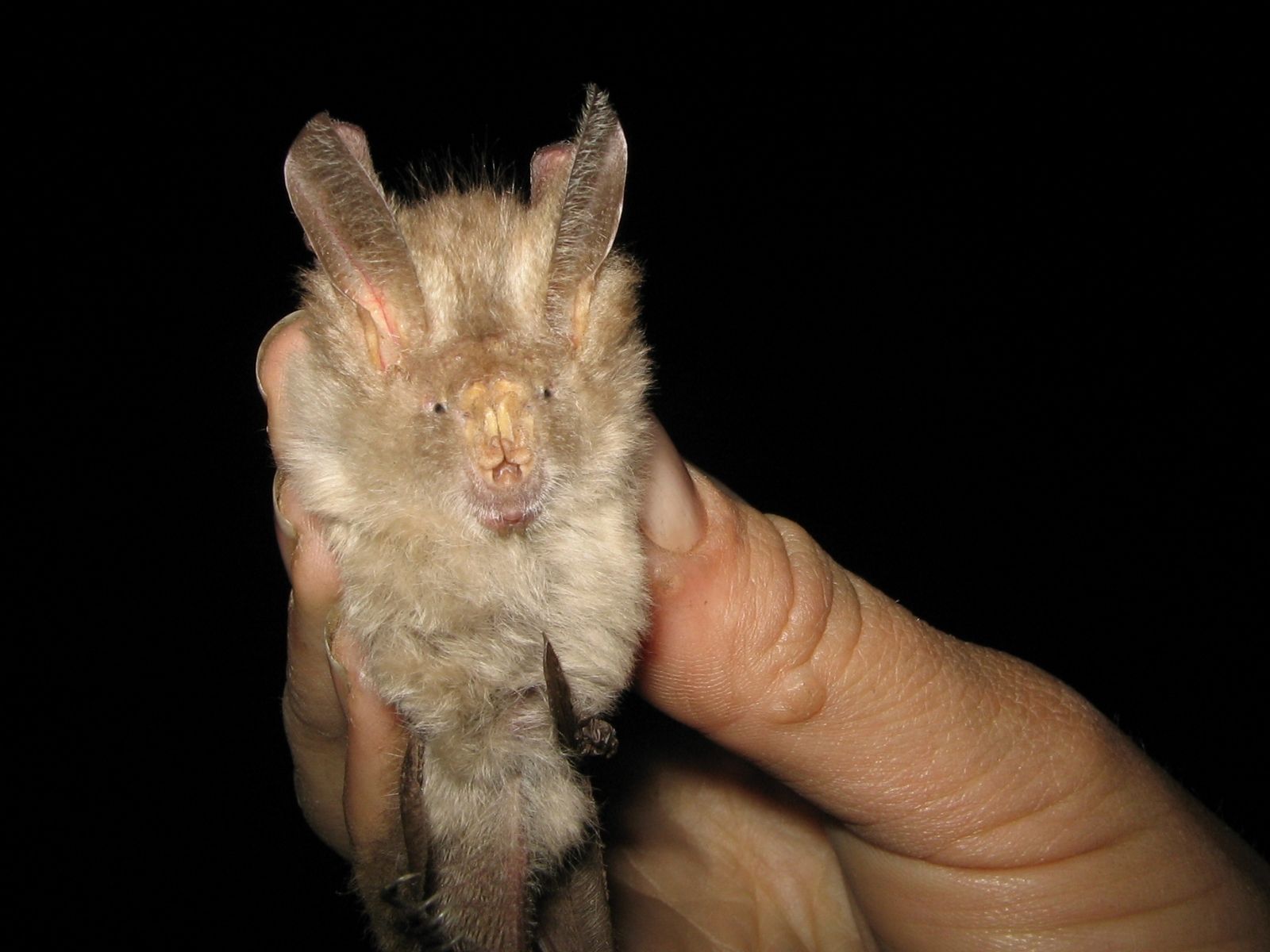
Hairy Slit-faced Bat
Nycteris hispida

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Nycteris hispida – (Schreber, 1775)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – NYCTERIDAE – Nycteris – hispida
Common Names: Hairy Slit-faced Bat, Hairy Long-eared Bat (English), Harige Spleetneusvlermuis (Afrikaans)
Synonyms: hispidus (von Schreber, 1774); barbata (P. L. S. Müller, 1776); daubentonii Ã. Geoffroy Saint-Hilaire, 1813 [nomen novum]; martini (L. Fraser, 1843); poensis J. E. Gray, 1843; villosa W. C. H. Peters, 1851; pilosa (J. E. Gray, 1866); pallida J. A. Allen in J. A. Allen, Lang, & Chapin, 1917; daubentoni Simmons, 2005 [incorrect subsequent spelling, not used as valid] (see Mammal Diversity Database 2025)
Taxonomic Note:
The species was originally described in Senegal (Skinner & Chimimba 2005). Meester et al. (1986) recognised two subspecies, with the nominate subspecies occurring throughout Africa and N. hispida villosa limited to south of the Zambezi River. However, these two subspecies do not appear to occupy geographically distinct regions and van Cakenberghe and de Vree (1993) concluded that N. hispida is uniform throughout its entire distribution area and thus cannot be divided into subspecies (although they were not able to examine specimens of the villosa form). Hence, in the absence of genetic data, no subspecies are recognised.
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Balona, J.1, Richards, L.R.2 & da Silva, J.M.3
Reviewer: Smith, C.4
Institutions: 1Gauteng and Northern Regions Bat Interest Group, 2Durban Natural Science Museum, 3South African National Biodiversity Institute, 4Endangered Wildlife Trust
Previous Assessors and Reviewers: Monadjem, A., Jacobs, D., MacEwan, K., Cohen, L., Richards, L.R., Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Raimondo, D., Nicholson, S.K. & Child, M.F.
Assessment Rationale
Although only recorded from a few localities in a restricted range within the assessment region (extent of occurrence estimated as 35,636 km2), records from the Eastern Cape coast indicate a wider distribution than previously known and the species is believed to be widespread (they are not often caught in mist nets but are not rare north of the assessment region). There remains limited information on the species, and therefore potentially warrants a Data Deficient classification. However, despite its relatively small EOO, it appears to be an adaptable species and is found in a few large reserves (e.g., iSimangaliso Wetland Park), and the Ingwavuma District. While deforestation from agricultural expansion and mining is a threat to this species, it is well protected in the north of its range and can occur in modified landscapes. Further field surveys are needed to more accurately delimit its southern distribution and to determine subpopulation trends. Currently, in the absence of any evidence for decline, we list as Least Concern. Given its rarity in southern Africa, this species should be reassessed once further population data are available.
Regional population effects: Although this species has low wing loading (Norberg & Rayner 1987), and thus dispersal is presumably limited, there is assumed to be an inflow from Mozambique as the habitat is mostly connected and thus rescue effects are probably possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommend citation: Balona J, Richards LR & da Silva JM. 2025. A conservation assessment of Nycteris hispida. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species has a wide range, encompassing much of sub-Saharan Africa in forest and savannah habitats (Skinner & Chimimba 2005), with the exception of the Horn of Africa and parts of southern Africa (ACR 2024). While sparsely distributed in South Africa, Mozambique and northern Zimbabwe, niche models indicate suitable habitat over much of central Mozambique (from where it has not been recorded) and along the eastern coastline of South Africa (Monadjem et al. 2020). Indeed, while only having been recorded from northern KwaZulu-Natal Province in the previous assessment (Friedmann & Daly 2004), records from the Eastern Cape Province extend the extent of occurrence for this species within South Africa (ACR 2015, 2024). The estimated extent of occurrence (EOO) is now 35,636 km2. However, this range expansion was not mentioned in Monadjem et al. (2020). It may occur more widely in the assessment region, but further field surveys are needed to confirm this. Just including the northern KwaZulu-Natal records yields an EOO of 8,304 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 3 m asl (Monadjem et al. 2024)
Elevation Upper Limit (in metres above sea level): ~3,900 m asl (Monadjem et al. 2024)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
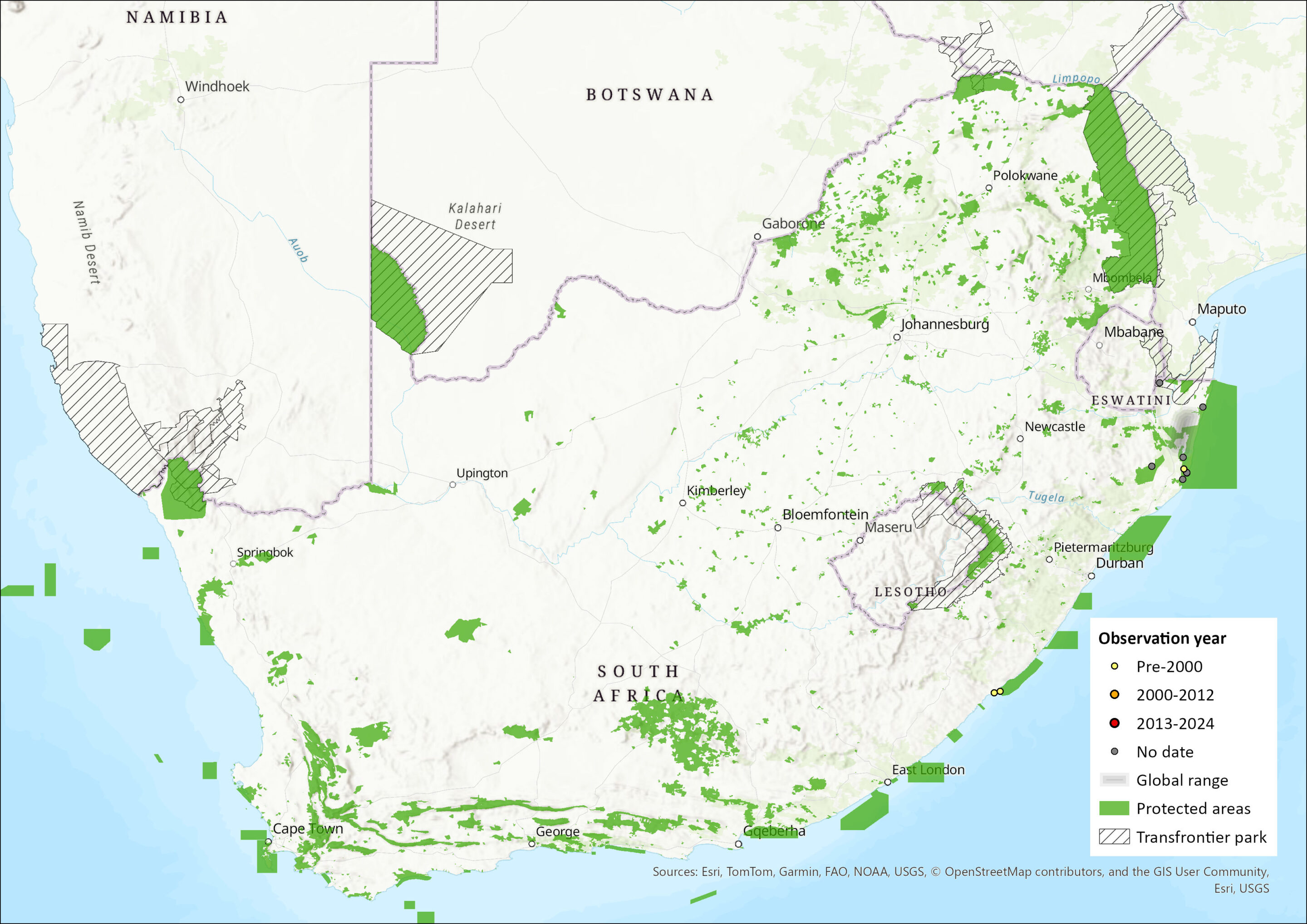 Figure 1. Distribution records for Hairy Slit-faced Bat (Nycteris hispida) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Hairy Slit-faced Bat (Nycteris hispida) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burkina Faso | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Equatorial Guinea -> Bioko | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Guinea-Bissau | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mali | Extant | Native | – | – |
| Mauritania | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Niger | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| Sierra Leone | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Sudan | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
There are no studies that have investigated the potential effects of climate change on the species distribution, ecology or physiology. Most studies on the effects of climate change on bats are based on predictive species distribution modelling, thus there is a lack of empirical studies measuring behavioural, physiological, phenological or genetic responses to extreme and seasonal climatic changes, especially in the Global South (Festa et al. 2023; Pio et al. 2014). Globally, there have been documented declines in bat populations, species richness and distributions in relation to water availability with increasing global aridity which may become a growing concern as heat waves and maximum temperatures are expected to increase over Sub-Saharan Africa (Adams & Hayes 2021).
Population Information
This is a very common bat in most parts of its range. However, it is not common in southern Africa (Skinner & Chimimba 2005), where only 28 specimens were examined in Monadjem et al. (2020). However, it may be under-sampled as it is rarely caught in mist nets. It is largely a solitary species but colonies range in size from individuals and pairs up to c. 20 bats (Skinner & Chimimba 2005; ACR 2024). The number of individuals present in colonies in the assessment region is unknown.
Current population trend: Stable
Continuing decline in mature individuals: No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Not suspected as it occurs largely in protected areas in the assessment region.
All individuals in one subpopulation: Presumed so
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Presumed to be one metapopulation
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unknown
Probability of extinction in the wild within 100 years: Unknown
Population Genetics
A phylogenetic study on slit‐faced bats (Chiroptera: Nycteridae) has uncovered deeply divergent African lineages within the family (Demos et al. 2019). However, regarding N. hispida, only a single sample from South Africa attributed as N. hispida/aurita was incorporated, clustering closely with individuals from Malawi (Demos et al. 2019). Any further information related to the species’ population genetic structure and diversity cannot be inferred from the available information. Consequently, it is recommended that more comprehensive surveys be undertaken to facilitate a population genetic study within the region.
Habitats and ecology
It occurs in a variety of habitats, from forest to dry savannah woodland to papyrus swamps, and has a broad tolerance of vegetation types (Rosevear 1965; Skinner & Chimimba 2005), including modified and disturbed habitats. It avoids open grasslands, montane areas semi-arid zones (Monadjem et al. 2020). In the assessment region, the species is recorded from the Lowveld and Indian Ocean Coastal Belt bioregions. While commonly roosting in dense bush, it can also use a diverse range of shelters; for example, Aardvark (Orycteropus afer) burrows, holes in termitaria, hollow trees, papyrus crowns and granite caves (Skinner & Chimimba 2005). In contrast to most other Nycteris species, N. hispida does not require a darkened cavity for a day roost (Monadjem et al. 2020). Individuals can weigh up to 10 grams with a variable pelage colour (Monadjem et al. 2020). It is an insectivorous clutter-edge forager, attracted by lights at night to hawk insects and may enters verandas and houses in search of insect prey drawn to the artificial light (Grubb et al. 1998; Skinner & Chimimba 2005).
Ecosystem and cultural services: As this species is insectivorous, it plays an important role in controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Often, bats prey on the insect species which destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats reduces the need to use pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 1.8. Forest -> Forest – Subtropical/Tropical Swamp | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unkown
Size at Maturity (in cms): Female: Mean forearm length = 4.16 ±0.22 cm; mean total length = 9.5 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Mean forearm length = 4.02 ±0.18 cm; mean total length = 9.08 ±0.67 cm (Monadjem et al. 2020)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Unknown
Size at Birth (in cms): 1.5 cm (Happold 2013)
Gestation Time: unknown
Reproductive Periodicity: very little information for the assessment region, however, the species is predicted to exhibit bimodal polyoestry based on breeding information collected elsewhere (Happold 2013). Births have been reported to take place in autumn (March/April) with females feeding young through to June (Verschuren, 1957). Births have also been reported in September, with lactating females captured in November (Verschuren, 1957; Pakenham, 1984).
Average Annual Fecundity or Litter Size: One pup born
Natural Mortality: Nocturnal avian predators and tree snakes (Happold 2013)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Unknown, but owing to low wing loading, unlikely capable of long-distance movement or migration.
Congregatory: Yes, it can be found singly or in groups of between 3-6 animals (Happold 2013). Colonies of 20 individuals have been reported (ACR 2024).
Systems
System: Terrestrial
General Use and Trade Information
This species has not been found to be traded or utilised.
Local Livelihood: Unknown
National Commercial Value: Provides essential ecosystem services in the form of insect population control.
International Commercial Value: May provides essential ecosystem services in the form of insect population control in relation to exported produce.
End Use: None reported
Is there harvest from captive/cultivated sources of this species? Not to our knowledge.
Harvest Trend Comments: N/A
Threats
Overall, no major threats have been identified. However, it is potentially threatened by loss of habitat in KwaZulu-Natal Province, due to deforestation from agricultural expansion, titanium mining, firewood and charcoal production (Friedmann & Daly 2004; ACR 2024). Pesticide use will presumably reduce its prey base in agricultural landscapes.
Conservation
In the assessment region, the species has been recorded in the protected Hluhluwe-iMfolozi Game Reserve and the iSimangaliso Wetland Park. No direct conservation measures are currently needed. However, further protected area expansion, especially transfrontier parks (sensu Smith et al. 2008), will presumably benefit this species.
Recommendations for land managers and practitioners:
- Reduce pesticide use in agricultural landscapes.
- Do not persecute bats that may make use of buildings and other man-made structures for roosting or hunting grounds.
Research priorities:
- Targeted surveys in areas where the species has been previously recorded to affirm current distribution and determine current population size.
- Survey areas
- Field surveys are needed to more accurately delimit its distribution in the southern parts of its range (for example, the Eastern Cape).
- To determine how climate change may affect the species, specifically their narrow distribution in South Africa.
Encouraged citizen actions:
- Citizens can assist the conservation of the species by reporting sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) and therefore contribute to an understanding of the species distribution.
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa
ACR. 2024. African Chiroptera Report 2025. AfricanBats NPC Pretoria, South Africa.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Demos, T.C., Webala, P.W., Kerbis Peterhans, J.C., Goodman, S.M., Bartonjo, M. and Patterson, B.D. 2019. Molecular phylogenetics of slit‐faced bats (Chiroptera: Nycteridae) reveal deeply divergent African lineages. Journal of Zoological Systematics and Evolutionary Research, 57(4), pp.1019-1038.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Friedmann, Y. and Daly, B. 2004. Red Data Book of the Mammals of South Africa: A Conservation Assessment. CBSG Southern Africa, IUCN SSC Conservation Breeding Specialist Group, Endangered Wildlife Trust, South Africa.
Grubb, P., Jones, T.S., Davies, A.G., Edberg, E., Starin, E.D. & Hill, J.E. 1998. Mammals of Ghana, Sierra Leone and The Gambia. The Trendrine Press, Zennor, St. Ives.
Happold, M. 2013. Nycteris hispida; pp 448-449 In: Mammals of Africa Volume IV – Hedgehogs, Shrews and Bats. Happold, M. & Happold, D. C. D. London UK · Bloomsbury Publishing.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: implications for biodiversity. South African Journal of Science 111: 1–9.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
Mammal Diversity Database. 2025. Mammal Diversity Database (Version 2.2). Zenodo. https://doi.org/10.5281/zenodo.15007505
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis, Second Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Norberg, U.M. and Rayner, J.M. 1987. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philosophical Transactions of the Royal Society B: Biological Sciences 316: 335–427.
Pakenham, R.H.W. 1984. The mammals of Zanzibar and Pemba Islands. Printed privately, Harpenden: 82 pp. URL: http://www.scribd.com/doc/14538645/The-Mammals-of-Zanzibar-and-Pemba.
Pio, D.V., Engler, R., Linder, H.P., Monadjem, A., Cotterill, F.P., Taylor, P.J., Schoeman, M.C., Price, B.W., Villet, M.H., Eick, G. and Salamin, N. 2014. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Global Change Biology, 20(5), pp.1538-1549.
Rosevear DR. 1965. The Bats of West Africa. British Museum (Natural History), London, UK.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Smith, R.J. et al. 2008. Designing a transfrontier conservation landscape for the Maputaland centre of endemism using biodiversity, economic and threat data. Biological Conservation 141: 2127–2138.
van Cakenberghe, V. and de Vree, F. 1993. Systematics of African Nycteris (Mammalia: Chiroptera). Part II. The Nycteris hispida group. Bonner Zoologische Beiträge 44: 299–332.
Verschuren, J. 1957. Ecologie, biologie et systématique des cheiroptères. Mission H. De Saeger et al., 1949 – 1952. Exploration du Parc national de la Garamba, 7: 1 – 473.
