Hairy-footed Gerbil
Gerbillurus paeba

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Gerbillurus paeba – (A. Smith, 1836)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – MURIDAE – Gerbillurus – paeba
Common Names: Hairy-footed Gerbil, Pygmy Gerbil, Pygmy Hairy-footed Gerbil (English), Haarpootnagmuis (Afrikaans)
Synonyms: Gerbillurus paeba (A. Smith 1936)
Taxonomic Note: This species was previously described under the genus Gerbillurus, which included three other species, of which only Gerbilliscus vallinus (previously Gerbillurus vallinus) occurs within the assessment region. Until recently Gerbillurus and Gerbilliscus were treated as separate genera (Musser & Carleton 2005). However, chromosomal, mitochondrial and nuclear molecular data suggest that they should be combined into one genus (see Monadjem et al. 2015). Gerbilliscus was also previously included in Tatera, but evidence suggests that the genus Tatera is restricted to a single Asian species and that all African species formerly grouped in Tatera form a distinct clade that corresponds with Gerbilliscus (see Monadjem et al. 2015).
This is one of four species from the “Gerbillurus” group that can be distinguished from other Gerbilliscus species by the hairy soles of their hindfeet, their relatively long tails, and their small body size (Monadjem et al. 2015). When compared to G. vallinus, G. paeba can be recognised by its lighter pelage colouration, which is generally light reddish-brown/-grey, shorter hindfeet (< 30 mm), and the lack of red/black hairs on the end of its tail. Taxonomic revision of the peripherally distributed subspecies from the assessment region, including G. p. mulleri (Western Cape Province), G. p. exilis (Eastern Cape Province), G. p. coombsi (Limpopo Province) and the nominate G. p. paeba is urgently required (Qumsiyeh et al. 1991).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Smith, C.1 & da Silva, J.M2
Reviewer: Taylor, P.J.3
Contributors: Roxburgh, L.1 & Patel, T.1
Institutions:1Endangered Wildlife Trust,2South African National Biodiversity Institute,3University of the Free State
Previous Assessors and Reviewers: Schlitter, D. & Kerley, G.
Previous Contributors: Avenant, N., Avery, M., Baxter, R., Child, M.F., MacFadyen, D., Monadjem, A., Palmer, G., Relton, C., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern in view of its wide distribution within the assessment region, presumed large population, and because there are no major threats that could cause population decline. It occurs in a number of protected areas as well as in agricultural landscapes. However, each of the peripheral subspecies, G. p. mulleri in extreme southwestern South Africa, G. p. exilis along the Indian Ocean coast and G. p. coombsi in extreme northeastern South Africa, is morphologically distinct from the centrally occurring nominate subspecies as well as other subspecies in adjacent parts of the species’ geographic range. An isolated population of G. p. exilis, which occurs in the dunefields of the northern shores of Algoa Bay, between the Swartkops River and Woody Cape, may especially warrant separate assessment. Although most of this subpopulation’s habitat is included in the Addo Elephant National Park, it may be threatened by property development in other parts of its range. Although not differing in terms of chromosome structure, this subpopulation may represent a new species pending molecular research. Once the taxonomy is resolved, this form may warrant separate assessment.
Regional population effects: There is possible dispersal between Namibia, South Africa and Botswana. However, each of the three peripheral subspecies occurring in South Africa are isolated in unique habitats with no evidence of dispersal from the nominate subspecies in South Africa or those in neighbouring Zimbabwe, Botswana or Namibia. Populations of G. p. coombsi in northeastern South Africa seem to be isolated from each other and those in neighbouring Zimbabwe and Mozambique.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Gerbillurus paeba. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The Hairy-footed Gerbil is located across western and central southern Africa, from western and northern South Africa northwards throughout Namibia, marginally into southwestern Angola, through most of Botswana (absent from the extreme north), partially into western and southeastern Zimbabwe and into the southwestern parts of Mozambique (Skinner and Chimimba 2005). Substrate and vegetation cover are important factors determining distribution (Perrin and Dempster 2013).
Although widespread in South Africa, three allopatric subspecies are distributed peripherally to the nominate subspecies. In the southwestern coastal region, Gerbilliscus paeba mulleri occurs from the vicinity of Eendekuil and Langebaan to Mitchell’s Plain on the Cape flats; G. p. exilis occurs along the south Indian Ocean coast from east of Witsand to the vicinity of Port Shepstone in suitable solidified coastal white sand dunes; while G. p. coombsi is found in sandveld habitat in the extreme northeast of South Africa. Additionally, a disjunct population, recognised as G. p. exilis, occurs in the coastal dunefields on the northern shores of Algoa Bay in the Eastern Cape. This population is isolated from those in the hinterland by the thicket vegetation of the lower Sundays River valley.
The broad hiatus between the distributions of the nominate subspecies, G. p. paeba, and the northeastern subspecies, G. p. coombsi, is valid. The region has been extensively sampled over the years and no records have been taken. The relationship between the distributions of the other two coastal subspecies and the nominate subspecies is unclear. Both of the former subspecies seem to be more of a habitat specific while the nominate subspecies is more habitat generalist. Interestingly, samples from nearly sympatric localities show no integration of morphological characters (D. Schlitter unpubl. data).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
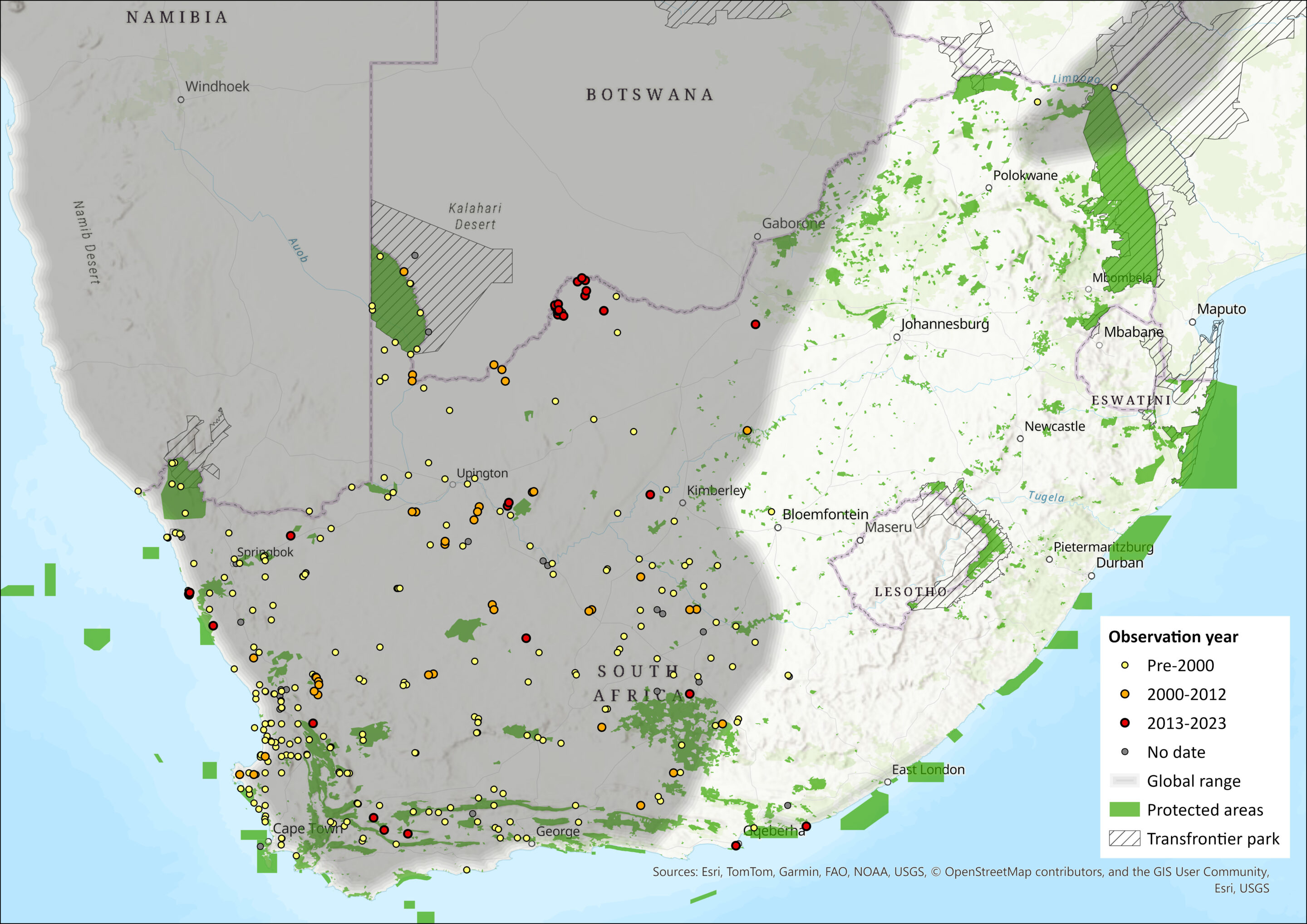
Map
Figure 1. Distribution records for Hairy-footed Gerbil (Gerbillurus paeba) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
This species occurs across western and central southern Africa. It might be affected by the 1.5-4°C increase in temperature predicted under various climate change scenarios (Engelbrecht et al. 2024). Rainfall is predicted to decrease in its range. These decreases will result in a more arid and drought-prone habitat which could affect food availability in its distribution range. The population in one locality collapsed during an El Niño-related drought in the Karoo (Kerley and Erasmus 1992), suggesting a vulnerability to predicted climate extremes under climate change. However, the species is widespread, and rescue effects could occur provided there is connectivity between localities.
Population Information
A common and abundant species exhibiting population fluctuations, often dominating small mammal communities numerically. For example, they are the most common rodent in the Kalahari Desert, constituting 90% of the rodent community in sand dune habitats and 54% in riverbed habitats (see Perrin and Dempster 2013). However, only the nominate subspecies is common in suitable habitats, especially during years of population explosions. The three peripheral subspecies seem to occur in low numbers in South Africa, Zimbabwe and Mozambique.
Population densities vary considerably: 5–32 individuals / ha were recorded at one site in the semiarid Karoo (Kerley 1992a), 1–9 individuals / ha for six sites across the southern Karoo (Kerley and Erasmus 1992), and 0.7–3.3 individuals / ha in the Alexandria Dunefield (Ascaray et al. 1991). One locality collapsed during an El Niño-related drought in the Karoo (Kerley and Erasmus 1992), suggesting a vulnerability to predicted climate extremes under global change.
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No, population is contiguous in the arid and semi-arid western southern Africa, with the exception of the Algoa dunefield population.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While this genus has been investigated in a phylogenetic context (Qumsiyeh et al. 1991), no population genetic assessment has been undertaken on the species, nor have the subspecies been investigate using molecular techniques. They have been differentiated solely based on morphological evidence and are located in different biogeographic zones.
Assuming their validity, all four occur in the assessment region, with the nominate subspecies G. p. paeba being relatively more abundant. It is assumed dispersal within a subspecies group is possible; however further substructure would need to be investigated using fine scale molecular techniques. While population fluctuations are observed within the species, no local extinctions have been recorded to date. Based on the available information, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (4/4 subpopulations remaining).
Only the nominate subspecies is considered common; therefore, if should be safe to assume that its numbers are in excess of 10 000 individuals, translating to an effective population size (Ne) of approximately 1000 (assuming a 0.1 conversion ratio of Ne to census size). While the 10 000 value is not an actual census count, the quantity can still be used as a proxy. Given the size of the other subspecies are unknown, Ne estimates are not possible for these subpopulations. This uncertainty makes it difficult to quantify the proportion of populations with an Ne > 500 indicator, but at least one subpopulation should exceed the Ne 500 threshold.
Further certainty and validation of these proxy metrics should be investigated using actual DNA-based genetic diversity metrics to ensure the genetic health of the species is sufficiently accounted for.
Habitats and ecology
The Hairy-footed Gerbil is a nocturnal species that occurs extensively throughout the Nama Karoo and Succulent Karoo biomes, generally within sandy soils, or sandy alluvium associated with grass, scrub or thin woodland cover (Nowak 1999, Perrin and Dempster 2013). Their occurrence extends into savannah, desert and dunefield habitats. For example, they are common in the Kalahari Desert in lightly vegetated dune slopes, calcrete riverbanks and pans (Perrin and Dempster 2013). The population in the dunefields of Algoa Bay favours the dune swales habitat, avoiding dense vegetation and dune crests. In Rolfontein Nature Reserve, Northern Cape Province, a single specimen was caught in the Rhus ciliata community (Jooste and Palmer 1982). Within the North West Province, the species is common on Kalahari sands and found in all the western Kalahari vegetation types and may occur in the Mafikeng Bushveld, but so far, no evidence has been found (Power 2014). The populations of G. p. exilis in the coastal white sand dunes seem to be found most commonly on the interface between the grass vegetation and the open sand dunes but not on the bare sand dunes themselves. Such grass cover may be critical to their survival and, if so, must be a part of any conservation effort to protect tracts of white dunes along the coastal region.
Abundance is negatively correlated with plant cover (Kerley and Erasmus 1992), and it is thus an open habitat specialist (Kerley et al. 1990). This is a solitary species, which dwells in simple burrows with a single entrance hidden by vegetation, and a tunnel extending to about 220 mm underground (Downs and Perrin 1989, De Graaf and Nel 1992, Skinner and Chimimba 2005). As omnivorous rodents (Kerley and Erasmus 1992), G. paeba consume arthropods, seeds and plant material (Downs and Perrin 1989, Griffin 1990, Kerley et al. 1990, Kerley 1992b, Perrin et al. 1992). Plant material was found to constitute the majority of their diet in the Karoo, while seeds and insects made up a lesser proportion (Skinner and Chimimba 2005). Contrastingly, in the Kalahari, seeds made up a much larger proportion of their diet (Nel et al. 1984). Although insect consumption did not vary seasonally in the Karoo, consumption of plant material was highest in late summer (February/March), while seeds were eaten far more commonly in winter (June/July) (Kerley 1992b).
It appears able to breed opportunistically throughout the year, although, in the Karoo, pregnancies were only recorded from October to May (White et al. 1997), and in the Kgalagadi Transfrontier Park, reproductive potential increases in summer. The gestation period is 21 days (Ascaray 1986), and litter size has been recorded as 3.7 in Botswana (Smithers 1971), 3.0 in the Karoo (White et al. 1997), 4.1 in the Kgalagadi Transfrontier Park (Nel et al. 1984), and 4.6 in captivity (Dempster and Perrin 1989).
Ecosystem and cultural services: Rodents are both predators and dispersers of plant seeds in the environment. G. paeba is a disperser of seeds in the sandy regions of the Cape Floristic Region (CFR) (Weighill et al. 2016). Since these gerbils are hoarders, they may serve in a limited capacity as seed dispersers. Seeds taken into the burrows are likely to be consumed, but some of the seeds scatter-hoarded in caches or buried are often forgotten or abandoned, and if these escape other seed predators, they may germinate and establish seedlings. This species is both a consumer and cacher of Willdenowia incurvata (Weighill et al. 2016). Additionally, this species is a valuable food source for small carnivores (Perrin and Dempster 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.4. Shrubland -> Shrubland – Temperate | – | Suitable | – |
| 8.1. Desert -> Desert – Hot | – | Suitable | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This species is not traded or utilised in any form and has not entered the international pet trade, nor is it utilised in traditional medicine.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
No major threats have been identified for this species. However, an observed local collapse of the population under an El Niño-related drought (Kerley and Erasmus 1992) suggests a vulnerability to predicted climate extremes under global change.
The nominate subspecies seems to have no threats over its wide geographic range. However, the three peripheral subspecies are under a number of local threats: G. p. coombsi is threatened by habitat change mostly due to agricultural development and the need for both housing and small agricultural plots for the expanding local populations; G. p. exilis is confined to a narrow band of suitable coastal white sand dune habitat that is undergoing increasing housing and industrial developments all along the Indian Ocean coast; and G. p. mulleri is losing suitable habitat to expanding housing developments, especially informal housing in the Cape Town region, and agricultural and industrial development in the northern parts of its distribution, including irrigated monocultures.
Current habitat trend: Stable
Conservation
No direct interventions are necessary as this species can exist in agricultural landscapes and urban areas. Additionally, this species is present in many protected areas within the assessment region, including the Karoo, Tankwa Karoo, Namaqua, Richtersveld, Camdeboo, Augrabies Falls and Addo Elephant National Parks. The isolated population in the coastal dunefields of the Eastern Cape is well protected in the Addo Elephant National Park Woody Cape section, which comprises over 60% of the known range of this form and is thus critical for the conservation of the G. p. exilis subspecies. Gerbilliscus p. coombsi is present within both Kruger and Mapungubwe National Parks, and based on its distribution, it is possible that G. p. mulleri occurs within the West Coast National Park. However, continued taxonomic investigations are required to confirm the conservation status of these peripheral subspecies. These two subspecies are under threat from habitat loss due primarily to expanding agriculture and informal housing, especially in the Cape Town region. Protected area expansion and stewardship may be necessary for the subspecies once taxonomic and distributional limits are more clearly established.
Recommendations for land managers and practitioners: No specific management plan is currently necessary.
Research priorities:
- An extensive study of the distributions and local conditions of each of the three subspecies, so that the exact ranges and preferred habitats of each can be delineated.
- A study of the population ecology, numbers and reproduction of each subspecies so that a better understanding of the status of each subspecies is known, and a better management strategy can be done after the amount of available and suitable habitat is known.
- Continued taxonomic research is necessary to discern whether the Eastern Cape subpopulation may represent a new species/subspecies.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas.
.
Bibliography
Ascaray, C.M. 1986. An ecological study of the hairy-footed gerbil, Gerbillurus paeba in an eastern Cape duneveld. M.Sc.Thesis, University of Port Elizabeth.
Ascaray, C.M., Perrin, M.R., McLachlan, A. and Els, S.F. 1991. Population ecology of the hairy-footed gerbil, Gerbillurus paeba, in a coastal dunefield of South Africa. Zeitschrift für Säugetierkunde 56: 296–305.
Crawford-Cabral, J. 1998. The Angolan rodents of the superfamily Muroidea. An account on their distribution. Estudos, Ensaios e Documentos 161: 1-222.
De Graaf G, Nel JAJ. 1992. Notes on a single burrow system of the fat mouse Steatomys pratensis in the Kruger National Park. Koedoe 35:123–124.
Dempster, E.R. and Perrin, M.R. 1989. Maternal behaviour and neonatal development in three species of Namib Desert rodents. Journal of Zoology 218: 407–419.
Downs, C.T. and Perrin, M.R. 1989. An investigation of the macro-and micro-environments of four Gerbillurus species. Cimbebasia 11: 41–54.
Engelbrecht, FA., Steinkopf, J., Padavatan, J. & Midgley, GF. 2024. Projections of future climate change in southern Africa and the potential for regional tipping points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7
Griffin, M. 1990. A review of taxonomy and ecology of gerbilline rodents of the central Namib Desert, with keys to the species (Rodentia: Muridae). In: Seely, M.K. (ed.), Namib Ecology: 25 Years of Namib Research, pp. 83–98. Transvaal Museum Monograph No. 7, Transvaal Museum, Pretoria.
Jooste JF, Palmer NG. 1982. The distribution and habitat preference of some small mammals in the Rolfontein Nature Reserve. South African Journal of Wildlife Research 12: 26-35.
Kerley, G.I. 1992. Small mammal seed consumption in the Karoo, South Africa: further evidence for divergence in desert biotic processes. Oecologia 89: 471–475.
Kerley, G.I.H, Knight, M.H. and Erasmus, T. 1990. Small mammal microhabitat use and diet in the southern Kalahari, South Africa. S. Afr. J. Wildl. Res. 20: 123-126.
Kerley, G.I.H. 1992. Trophic status of small mammals in the semi-arid Karoo, South Africa. Journal of Zoology 226: 563-572.
Kerley, G.I.H. and Erasmus, T. 1992. Small mammal communities in the semi-arid Karoo, South Africa: biomass and energy requirements. Journal of Arid Environments 22: 251–260.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa – a biogeographic and taxonomic synthesis. De Gruyter, Berlin/Munich/Boston.
Musser, G.G. and Carleton, M.D. 2005. Superfamily Muroidea. In: D.E. Wilson and D.A. Reeder (eds), Mammal Species of the World: a geographic and taxonomic reference, pp. 894-1531. The John Hopkins University Press, Baltimore, USA.
Nel, J.A., Rautenbach, I.L., Els, D.A. and De Graaff, G. 1984. The rodents and other small mammals of the Kalahari Gemsbok National Park. Koedoe 27: 195–220.
Nowak, R.M. 1999. Walker’s Mammals of the World. The Johns Hopkins University Press, Baltimore, USA and London, UK.
Perrin, M. and Dempster, E.R. 2013. Gerbillurus paeba Pygmy Hairyfooted Gerbil (Paeba Hairy-footed Gerbil). In: Happold, D.C.D. (ed.), Mammals of Africa. Volume III: Rodents, Hares and Rabbits, pp. 288–290. Bloomsbury Publishing, London.
Perrin, M.R., Boyer, H. and Boyer, D.C. 1992. Diets of the hairy-footed gerbils Gerbillurus paeba and G. tytonis from the dunes of the Namib Desert. Israel Journal of Zoology 38: 373–383.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Qumsiyeh, M.B., Hamilton, M.J., Dempster, E.R. and Baker, R.J. 1991. Cytogenetics and systematics of the rodent genus Gerbillurus. Journal of Mammalogy 72: 89–96.
Seely, M.K. 1977. Sand solidified by gemsbok urine as selected burrow sites by gerbils. Zoologica Africana 12: 247–249.
Skinner J.D. and Chimimba C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Weighill, B., Huysamer, A. and Anderson, B., 2017. The nightshift: seed dispersal and consumption differences by rodents before and after dark. South African Journal of Botany, 108, pp.267-271.
White, R.M. Kerley, G.I.H. and Bernard, R.T.F. 1997. Pattern and controls of reproduction of the southern African rodent Gerbillurus paeba in the semi-arid Karoo, South Africa. Journal of Arid Environments 37: 529–549.