Grey Climbing Mouse
Poemys melanotis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Poemys melanotis s.l.- (Smith, 1834)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Poemys – melanotis sensu lato
Common Names: Grey African Climbing Mouse, Grey Climbing Mouse, Grey Pygmy Tree Mouse (English), Grys Klimmuis, Grys Boommuis (Afrikaans)
Synonyms and previously recognised names: Dendromys melanotis (A. Smith, 1834), Dendromys subtilis (Sundevall, 1847), Dendromys pallidus (Fitzinger, 1867), Dendromys pallidus (von Heuglin, 1877); Dendromys nigrifrons (F. W. True, 1892); Dendromus spectabilis (E. Heller, 1911); Dendromus (Poemys) nigrifrons vulturnus (O. Thomas, 1916); Dendromus (Poemys) exoneratus (O. Thomas, 1918); Dendromus (Poemys) arenarius (A. Roberts, 1924); Dendromus melanotis (A. Roberts, 1924); Dendromus nigrifrons (A. Roberts, 1924); Dendromus nigrifrons nigrifrons (Granvik, 1924); Dendromus (Poëmys) concinnus (O. Thomas, 1926); Dendromus melanotis basuticus (A. Roberts, 1927); Dendromus (Poemys) melanotis chiversi (A. Roberts, 1929); Dendromus (Poemys) nigrifrons shortridgei (St. Leger, 1930); Dendromus melanotis capensis (A. Roberts, 1931); Dendromus melanotis pretoriae (A. Roberts, 1931); Dendromus melanotis thorntoni (A. Roberts, 1931); Pœmys concinnus Shortridge, 1934; Pœmys nigrifrons Shortridge, 1934; Poemys melanotis insignis Shortridge & T. D. Carter, 1938; Poemys melanotis capensis Shortridge, 1942; Poemys melanotis Shortridge, 1942; Poemys melanotis thorntoni Shortridge, 1942.
Taxonomic Note: The taxonomic status of this species is uncertain as evidenced by its karyotypic variability and many synonyms (Monadjem 2013). Analysis of chromosomal and molecular variation has revealed the presence of four putative cryptic species within the Poemys. melanotis (formerly Dendromus melanotis) clade in South Africa (Solano et al. 2014; Voelker et al. 2021). Meester et al. (1986) recognised five southern African subspecies, that have since been synonymised: D. m. arenarius from Botswana and the Northern Cape province of South Africa, D. m. concinnus from Namibia, D. m. vulturnus from the KwaZulu-Natal and Free State provinces of South Africa and Lesotho, and D. m. shortridgei from Botswana and Zimbabwe. Several of these previously listed synonyms, (arenarius, vulturnus, and basuticus), are likely to represent distinct species (Voelker et al. 2021). Furthermore, it is recommended that representatives of other synonymised taxa, including capensis, chiversi, pretoriae, and shortridgei, be incorporated into future integrative taxonomic studies to evaluate their status. Additional research is urgently needed to confirm the taxonomic validity of these putative species and to establish the appropriate nomenclature (Voelker et al. 2021; Mammal Diversity Database 2025). Due to taxonomic uncertainty and the absence of clear biogeographical boundaries for the genetic clades that may represent distinct species, the taxon is herein referred to as P. melanotis sensu lato.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Richards, L.R.1 & da Silva, J.M.2
Reviewers: Patel, T3.
Institutions:1Durban Natural Science Museum,2South African National Biodiversity Institute,3Endangered Wildlife Trust
Previous Assessors and Reviewers: MacFadyen, D. & Relton, C.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., Monadjem, A., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Listed as Least Concern as it has an expansive range within the assessment region, occurs commonly across a range of habitats, including many protected areas (such as Tswalu Kalahari Reserve, Telperion Nature Reserve, Rooipoort Nature Reserve, and Kruger National Park), and has been recorded from both intact and degraded areas. Currently there are no major threats expected to cause extensive population decline. However, continued habitat loss as a result of mining, urban expansion, intensive agriculture and forestry is suspected to be causing local declines or extinctions and should be monitored carefully. Population numbers also tend to fluctuate, with higher numbers recorded in autumn and winter on the Highveld. Finally, taxonomic resolution is required as four cryptic species may occur in South Africa, which may necessitate reassessment of this species.
Regional population effects: This species is expected to disperse from Mozambique, Zimbabwe, Botswana and Namibia into South Africa. As a generalist in its habitat preference, this species would allow for easy dispersal through modified habitats. Its range is largely contiguous except for isolated populations in northern Namibia and central Africa.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Richards LR & da Silva JM. 2025. A conservation assessment of Poemys melanotis s.l.. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
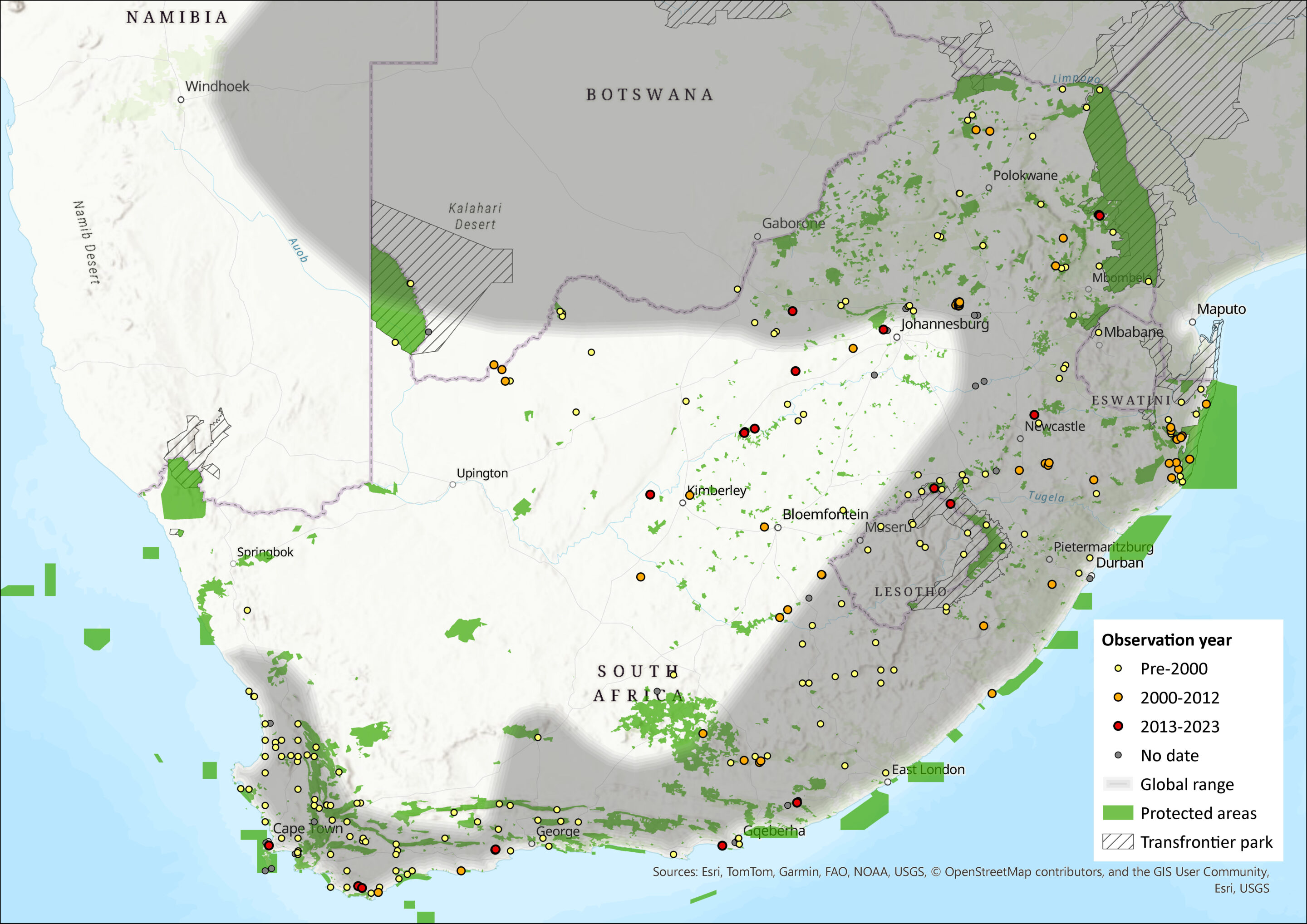
This species was historically widely distributed in southern and East Africa (Monadjem et al. 2015), including parts of Namibia, Botswana, and Zimbabwe, the central and southern parts of Mozambique, and all provinces in South Africa (Skinner and Chimimba 2005). It also occured in both Eswatini and Lesotho (Lynch 1994, Monadjem 1998). In Gauteng and Mpumalanga this species was recorded in high numbers in all habitats on Ezemvelo Nature Reserve and Telperion Nature Reserve (MacFadyen 2014). This species was also recorded in low numbers at Tswalu Kalahari Reserve and Rooipoort Nature Reserve in the Northern Cape (D. MacFadyen unpubl. data), and has been recorded in Tussen-die-Riviere Nature Reserve in the Free State (Ferreira and Avenant 2003). There have been no documented range expansions for the species (for example, Power 2014), and it is expected that its range has not decreased (MacFadyen 2014). However, the records from the interior of the country (Figure 1) require investigation as they have not yet been confirmed (see Monadjem et al. 2015). Recent phylogenetic evidence suggests that the true range of P. melanotis sensu stricto may be much more restricted, likely limited to South Africa’s Eastern Cape province. The broader distribution previously attributed to this species within the assessment region, appears to include at least three distinct putative species, which likely correspond to subspecies described earlier (Roberts 1954; Voelker et al. 2021).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 2,700 m asl (Rowe-Rowe and Meester 1982a)
Depth Lower Limit (in metres below sea level): N/A
Depth Upper Limit (in metres below sea level): N/A
Depth Zone: N/A
Map
Figure 1. Distribution records for Grey Climbing Mouse (Poemys melanotis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: N/A
FAO Area Occurrence
FAO Marine Areas: N/A
Climate change
To date, no formal studies have examined the potential impacts of climate change, along with associated bioclimatic and environmental shifts, on the biology, ecology, or physiology of this species. Although the species is found across a variety of habitats, it is commonly associated with grass-dominated landscapes (Skinner and Chimimba 2005). Midgley and Thuiller (2011) predicted a 30% replacement of grassland-dominated habitats by more arid-adapted or tolerant vegetation types (e.g. thorn-scrub savanna). This may result in the displacement or range contraction of grassland-associated species, that may include P. melanotis s.l. within the assessment region. Given its broad habitat tolerance (Skinner and Chimimba 2005; Monadjem 2013) and capacity to recolonise disturbed areas (Rowe-Rowe and Lowry 1981; Simelane et al. 2018; Mdluli et al. 2022), this species may experience less severe impacts from climate change compared to other Poemys and Dendromus species within the assessment region. However, this perspective could change once the taxonomic status of regional populations is fully resolved.
In general, rodent population declines tend to be more pronounced in habitats experiencing significant anthropogenic transformation or, alternatively, in high-altitude environments where the effects of climate change are likely to be intensified (Wan et al. 2022). Further research is required to assess the potential impacts of climate change on this species, and recently found cryptic species, within the assessment region.
Population
It is the most abundant of all climbing mouse species within the assessment region (Monadjem et al. 2013). For example, it was the third most common small mammal species in different high-altitude grassland habitats in KwaZulu-Natal (Rowe-Rowe and Meester 1982a), and the second most captured rodent across various habitats in the greater Telperion Nature Reserve (Gumbi et al. 2018; Mdluli et al. 2022). However, it is difficult to trap (Willan 1986), and thus population assessments are difficult. Population fluctuations occur seasonally and between different vegetation communities. MacFadyen (2014) recorded local movements within a subpopulation in the Highveld Grasslands of Gauteng and Mpumalanga in autumn and winter, revealing higher population numbers during these seasons. MacFadyen (2014) estimated density in Rocky Highveld Grassland at 5 animals / ha, with a higher density of 12 animals / ha recorded in degraded grassland. Gumbi et al. (2018) supported the notion that the species is locally abundant within disturbed habitats. This result is an indication that this species is able to inhabit a spectrum of habitats, from undisturbed to modified environments.
Population Information
Current population trend: Stable
Continuing decline in mature individuals? Unknown, but unlikely.
Extreme fluctuations in the number of subpopulations: This cannot be determined without phylogenetic and phylogeographic analyses that resolve the taxonomic status of local populations.
Continuing decline in number of subpopulations: This cannot be determined without phylogenetic and phylogeographic analyses that resolve the taxonomic status of local populations.
All individuals in one subpopulation: This cannot be determined without phylogenetic and phylogeographic analyses that resolve the taxonomic status of local populations.
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: This cannot be determined without phylogenetic and phylogeographic analyses that resolve the taxonomic status of local populations.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While a recent phylogenetic investigation of Climbing mice has been conducted, the authors were unable to include a ‘true’ P. melanotis specimen in their analyses, as none of the sampled regions encompassed those typically associated with the description “Port Natal” = greater Durban) (Voelker et al. 2021). As such, making strong inferences about this species in respect of its distributional limits and likely population characteristics is challenging. However, the authors report that the distribution of this previously assumed widespread species (i.e. P. melanotis) may be very narrowly distributed within the Eastern Cape Province of South Africa, under the assumption of additional cryptic taxa being described from the eastern regions of South Africa (Voelker et al. 2021). If this is true, the species may exist as a single metapopulation within the assessment region. However, this needs to be validated with additional molecular research, ideally incorporating comprehensive sampling across its presumed range. Such a study would also be invaluable in obtaining information on the genetic health/status/diversity of the species.
Habitats and ecology
The Grey African Climbing Mouse typically inhabits grassland and savannah regions, and is associated with a wide variety of habitats, including areas with a relatively low biomass, areas with stands of tall grass such as Hyparrhenia (Lynch 1994) and Merxmuellera spp., short montane grassland (Rowe-Rowe and Meester 1982a, Taylor 1998) and shrubs of the Savannah Biome (such as dry Kalahari scrub, river fringes and flood plains), as well as other areas where vegetation has encroached. It was recorded on Telperion Nature Reserve habitats ranging from densely grassed areas to rocky areas with sparse grass cover (MacFadyen 2014). In winter months, MacFadyen (2014) recorded this species in large numbers within Seriphum plumosum. Rautenbach et al. (2014) trapped this species at Phinda Private Game Reserve, KwaZulu-Natal, predominantly within Vachellia nilotica/Hyphaene coricea and Terminalia sericea woodlands, and one specimen was caught in the Drypetes arguta sandforest. Although considered to be a species preferring riverine conditions, it has been recorded in dry grassland, often some distance from water. This species is a habitat generalist, and has been recorded in both intact and degraded/disturbed areas, but is absent from overgrazed areas with very low biomass. It occurs from sea level right up to altitudes of approximately 2,700 m in the Drakensberg.
Like other species of climbing mice, this nocturnal and largely terrestrial species accesses seeds and invertebrates in low bushes and grass stalks, using its slender digits to grip and climb and its prehensile tail for balance. Thus, it is adapted to being semi-arboreal (Monadjem 2013). It weaves a grass nest with a single entrance usually attached to grass stems or shrubs up to 1 m above the ground (Skinner and Chimimba 2005; Monadjem 2013). The Grey Climbing Mouse has been observed to dig burrows approximately 0.5 m deep, featuring a nest chamber and an exit opposite the entrance tunnel (De Graaf 1981). These burrows are thought to protect the animals from veld fires, which may explain their ability to recolonise burned areas within a month (Monadjem, 2013). Several studies suggest that D. melanotis may represent a “pioneer species,” given its presence of the species in recently burnt grassland habitats (Rowe-Rowe and Lowry 1982; Simelane et al. 2018). However, this fairly rapid recolonization capability may be site specific as elsewhere the species was reported to suffer 100% post-burn mortality (Swanepoel 1981).
The species will feed on seeds, but also feeds on a wide range of invertebrates, including termites, crickets and moths. This species is solitary, or lives in pairs or small family groups; it breeds seasonally, with females usually producing litters of between two and four.
Ecosystem and cultural services: There are no recognised cultural uses for this species. Similar to other small mammals, this species plays a role in regulating invertebrate numbers, seed predation, nutrient cycling, and is an important prey species for predators (Monadjem 2013).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.2. Desert -> Desert – Temperate | – | Suitable | – |
Life History
Generation Length: Unknown
Age at Maturity: Female or unspecified: Unknown
Age at Maturity: Male: Unknown
Size at Maturity (in cms): Female: Mean total length = 15. 3 cm (Rautenbach 1982)
Size at Maturity (in cms): Male: Mean total length = 15. 2 cm (Rautenbach 1982)
Longevity: Unknown
Average Reproductive Age: Unknown
Maximum Size (in cms): Female maximum total length = 19.8 cm; Male maximum total length = 18.5 cm (Rautenbach 1982)
Size at Birth (in cms): Unknown
Gestation Time: Unknown
Reproductive Periodicity: Reproductively active within the summer and early autumn months, with pregnant females recorded between November to April within the assessment region (Jacobsen 1977; De Graaf 1981; Rowe-Rowe and Meester 1982b; Lynch 1994).
Average Annual Fecundity or Litter Size: Litter sizes are typically 2-4 pups (De Graaf 1981), however, litters containing up to 6-7 pups have been recorded (Hanney 1965; Jacobsen 1977).
Natural Mortality: As with Dendromus spp., predated upon by a variety of predators including snakes (De Graaf 1981), various raptors (Nel and Nolte 1965) and meso-carnivores such as Cape Fox (Vulpes chama; Klare et al. 2014) and Black-backed Jackal (Lupelella mesomelas; Rowe-Rowe 1983)
Does the species lay eggs? No
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Movement and dispersal patterns remain poorly understood. Jacobsen (1977) reported that the species can traverse considerable distances relative to its body size, as marked individuals were later found 60–100 meters away from their initial capture locations.
Congregatory: Bonded pairs may occupy the same burrow (De Graaf 1981). Family groups will continue to use the grass nest even after the young have been weaned (Skinner & Chimimba 2005).
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
Local Livelihood: None reported or known at present.
National Commercial Value: None reported or known at present.
International Commercial Value: None reported or known at present.
End Use: None reported or known at present.
Is there harvest from captive/cultivated sources of this species? None reported or known at present.
Harvest Trend Comments: N/A
Threats
There are no major threats to this fairly resilient species. However, habitat loss from mining, forestry, intensive agricultural expansion and urban expansion, combined with habitat degradation through overgrazing may lead to local declines or even extinctions. The impact of these threats on the population remains to be quantified. Following taxonomic resolution, the severity of regional threats facing any cryptic species should be established. Although they are able to survive in degraded areas, excessively overgrazed areas and areas transformed into habitats with little or no biomass would negatively impact this species.
Conservation
This species occurs in numerous protected areas across the assessment region, including Telperion Nature Reserve (Mpumalanga), Ezemvelo Nature Reserve and Rietvlei Nature Reserve (Gauteng), Tswalu Kalahari Reserve and Rooipoort Nature Reserve (Northern Cape), Venetia Limpopo Nature Reserve (Limpopo), many protected areas in KwaZulu-Natal including iSimangaliso Wetland Park, and the Kruger National Park. No specific conservation interventions are necessary, but the species would benefit from correct land management practices that restore degraded grassland areas, reduce overgrazing and implement a well-managed fire regimen, benefiting terrestrial small mammal communities as a whole.
Recommendations for land managers and practitioners:
- No specific management plan is necessary. However, this species would benefit from correctly managed agricultural lands, which restricts overgrazing as much as possible.
Research priorities:
- This is a species complex that requires taxonomic resolution.
- Population size and trend estimates, including population boundaries and overlaps within the species complex, as well as investigation into the reproductive rate and breeding success of this species.
- Quantification of the impacts of habitat loss across the species’ range.
- Understanding the local movements and dispersal mechanisms exhibited by this species.
- Research into the contribution made by this species to overall ecosystem functioning.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (e.g., iNaturalist and MammalMAP), especially outside protected areas.
- Deceased specimens should be submitted to the nearest accredited museum or biological repository for proper documentation and preservation.
- Report illegal mining practice and contribute to public pressure on post mining rehabilitation and restoration.
- Plant indigenous species in gardens and thus create suitable habitats.
- Create corridors to offset the impacts of urbanisation and ensure movement and gene flow between populations.
- Encourage organic agriculture and reduce the use of insecticides and herbicides as much as possible.
Bibliography
Avenant, N.L. 2000. Terrestrial small-mammal diversity in Korannaberg conservancy, Free State, South Africa: results. Navorsinge van die Nasionale Museum: Researches of the National Museum, 1;16(4):72-6.
Avenant, N.L, Cavallini P. 2007. Correlating rodent community structure with ecological integrity, Tussen‐die‐Riviere nature reserve, Free State province, South Africa. Integrative Zoology, 2(4):212-9.
Bowland, J.M. & Perrin, M.R. 1993. Wetlands as reservoirs of small-mammal populations in the Natal Drakensberg. South African Journal of Wildlife, 1;23(2):39-43.
De Graaff G. 1981. The Rodents of Southern Africa: Notes on Their Identification, Distribution, Ecology and Taxonomy. Butterworths, Pretoria.
Ferreira, S.M. and Avenant, N.L. 2003. Modelling the effects of trap-spacing on small mammal community descriptors in grasslands at Tussen-die-Riviere Nature Reserve, Free State Province, South Africa. Navorsinge van die Nasionale Museum, Bloemfontein 19: 21-30.
Gumbi, B.C., Shapiro, J., McCleery, R., Mahlaba, T., MacFadyen, D. & Monadjem, A. 2018. Assessing the impacts of domesticated versus wild ungulates on terrestrial small mammal assemblages at Telperion Nature Reserve, South Africa. African Zoology 53(1):23-9.
Hanney, P. 1965. The muridae of Malawi (Africa: Nyasaland). In: Proceedings of the Zoological Society of London 1965, Volume: 146(4), pp. 577-633. Blackwell Publishing Ltd, Oxford, U.K.
Jacobsen, N.H. 1977. An annotated checklist of the amphibians, reptiles and mammals of the Nylsvley Nature Reserve. Cooperative Scientific Programmes: CSIR.
Klare, U., Kamler, J.F. & Macdonald, D.W. 2014. Seasonal diet and numbers of prey consumed by Cape foxes Vulpes chama in South Africa. Wildlife Biology, 20(3):190-5.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum, Bloemfontein 10: 177–241.
MacFadyen, D.N. 2014. The dynamics of small mammal populations in Rocky Highveld Grassland, Telperion, South Africa. Ph.D. Thesis. University of Pretoria, Pretoria, South Africa.
Mammal Diversity Database. 2025. Poemys melanotis (A. Smith, 1834), Gray African Climbing Mouse. https://mammaldiversity.org/taxon/1003717
Mdluli, M.V., Bhembe, Z.D., Brown, L., MacFadyen, D.N., Mahlaba, T.A., Monadjem A. 2022. The loss of vegetation cover has distinct but short-term impact on multiple vertebrate taxa in a grassland ecosystem. Global Ecology and Conservation 38:e02198.
Meester, J.A.J., Rautenbach, I.L., Dippenaar,, N.J. & Baker, C.M. 1986. Classification of Southern African Mammals. Order Rodentia. Transvaal Museum Monographs, 5(1).
Midgley, G.F. and Thuiller, W. 2011. Potential responses of terrestrial biodiversity in Southern Africa to anthropogenic climate change. Regional Environmental Change, 11 (Suppl 1), pp.127-135.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45–59.
Monadjem, A. 2013. Dendromus melanotis Grey African Climbing Mouse. Pages 174–175 in Happold DCD, editor. Mammals of Africa. Volume III: Rodents, Hares and Rabbits. Bloomsbury Publishing, London, UK.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Nel, J.A.J. and Nolte, H. 1965. Notes on the prey of owls in the Kalahari Gemsbok National Park, with special reference to the small mammals. Koedoe, 8(1), pp.75-81.
Power, R.J. 2014. The Distribution and Status of Mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Rautenbach, I.L. 1982. Mammals of the Transvaal (No. 1). Ecoplan.
Rautenbach, A., Dickerson, T. & Schoeman, M.C. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Roberts, A. 1954. The Mammals of South Africa. Second Edition. Trustees of the Mammal of South Africa Book Fund, South Africa
Rowe-Rowe, D.T. & Lowry, P.B. 1982. Influence of fire small mammal populations in the Natal Drakensberg. South African Journal of Wildlife Research 12(4):130-9.
Rowe-Rowe, D.T., Meester, J. 1982a. Habitat preferences and abundance relations of small mammals in the Natal Drakensberg. South African Journal of Zoology 17: 202-209.
Rowe-Rowe, D.T. & Meester, J.A.J. 1982b. Population dynamics of small mammals in the Drakensberg, South Africa. Zeitschrift für Säugetierkunde 47: 347- 356.
Rowe-Rowe, D.T. 1983. Black-backed jackal diet in relation to food availability in the Natal Drakensberg. South African Journal of Wildlife Research, 13(1):17-23.
Simelane, F.N., Mahlaba, T.A., Shapiro, J.T., MacFadyen, D. & Monadjem, A. 2018. Habitat associations of small mammals in the foothills of the Drakensberg Mountains, South Africa. Mammalia 82(2):144-52.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Solano, E., Taylor, P.J., Rautenbach, A., Ropiquet, A. and Castiglia, R. 2014. Cryptic speciation and chromosomal repatterning in the South African climbing mice Dendromus (Rodentia, Nesomyidae). PloS one 9: e88799.
Swanepoel, C.M. 1981. The effect of fire on a small mammal community. African Zoology, 16(4):232-6.
Taylor, P.J. 1998. The Smaller Mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg, South Africa.
Vernon, C.J. 1972. An analysis of owl pellets collected in southern Africa. Ostrich, 43(2), pp.109-124.
Voelker, G., Huntley, J.B., Denys, C., Sumbera, R., Demos, T.C., Lavrenchenko, L., Nicolas, V., Gnoske, T.P., Kerbis Peterhans, J.C. 2021. Molecualr systematics and biogeographic history of the African climbing-mouse complex (Dendromus). Molecular Phylogenetics and Evolution 161: 107166.
Wan, X., Yan, C., Wang, Z. and Zhang, Z. 2022. Sustained population decline of rodents is linked to accelerated climate warming and human disturbance. BMC Ecology and Evolution, 22(1), p.102.
Willan K. 1986. Trap selection by some southern African small mammals. South African Journal of Wildlife Research 16(3):53-7.