Greater Kudu
Tragelaphus strepsiceros

2025 Red list status
Least Concern
Regional Population Trend
Increasing
Change compared to 2016
No Change
Overview
Tragelaphus strepsiceros – (Pallas, 1766)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Tragelaphus – strepsiceros
Common Names: Greater Kudu (English), Ibhalabhala (Ndebele), Thôlô (Sepedi, Sesotho, Setswana), Lishongololo (Swati), Nhongo (Tsonga), Tholo, Tholo-lurango (Venda), Iqudi (Xhosa), Igogo, Igoqo, iMbodwane, Umgankla (Zulu), Koedoe (Afrikaans), Grand Koudou (French), Grosskudu (German), Kudu or Cudo (Portuguese)
Synonyms: No Synonyms
Taxonomic Note:
Based on the number of pale transverse stripes on the body (Haltenorth 1963), four African subspecies of Tragelaphus strepsiceros have been recorded (Ansell 1972); although only T. s. strepsiceros is present in southern Africa (Skinner & Chimimba 2005).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Smith, C.1 & da Silva J.2
Reviewer: Anderson, J.3,4
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3IUCN SSC Antelope Specialist Group, 4International Conservation Services
Previous Assessor: Parrini, F.
Previous Reviewer: Child, M.F.
Previous Contributor: Selier, J.
Assessment Rationale
This species remains Least Concern as it is widespread and abundant within the assessment region, occurring in numerous protected areas across its range. There is an estimated mature population size of 63,708–67,383 animals (2013–2015 counts) across the country, with the majority of the population occurring on private land. The largest subpopulation is in Kruger National Park (KNP) with an estimated 8,239–13,490 animals (2012 count). Using a sample of 23 formally protected areas across its range with adequate long-term data, the population has increased by 72–81% over three generations (1990–2015). Similar increases are inferred on private lands. The high numbers of this species on private land reflect its value as one of Africa’s major trophy animals and it should continue to be utilised sustainably as part of the green economy. The wildlife industry is thus important for ensuring the continued existence of large numbers of Greater Kudu on private land. However, care should be taken to not establish further extra-limital subpopulations to prevent competition with local browsers. There are no major threats to this species and thus no immediate conservation interventions are necessary.
Regional population effects: There is presumably dispersal along the northern border of South Africa between Botswana, Zimbabwe and Mozambique through the Mapungubwe and Great Limpopo Transfrontier areas and northeastern KwaZulu-Natal.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Tragelaphus strepsiceros. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Historically, the Greater Kudu occurred over much of eastern and southern Africa, from Chad nearly to the Red Sea, south to the Eastern Cape, west to Namibia and north to mid-Angola (IUCN SSC Antelope Specialist Group 2016). While it has disappeared from substantial areas, mainly in the north of its range, it generally persists in a greater part of its former range than other large antelope species, as a result of its secretiveness and its ability to survive in settled areas with sufficient cover (IUCN SSC Antelope Specialist Group 2016).
Within the assessment region, it historically occurred in the Northern Cape, northeastern KwaZulu-Natal, North West, Limpopo, Mpumalanga and Gauteng provinces, with an isolated subpopulation in the Eastern Cape and scattered subpopulations in the Free State (Owen-Smith 2013). They currently occur in all provinces and Eswatini but are absent from Lesotho. They have remained fairly common throughout most of their range, even on ranchlands and close to settlements (Owen-Smith 2013). The expansion of wildlife ranching across the country is helping to increase the area of occupancy for the species, both inside and outside of the natural distribution. Natural range expansion, perhaps due to climate change and bush encroachment and the introduction of water points for livestock in the karoo, may also be occurring (Power 2014). While most kudu occur on private game farms and in protected areas, they also occur in free-roaming herds in the bushveld regions (Power 2014).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 2400
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
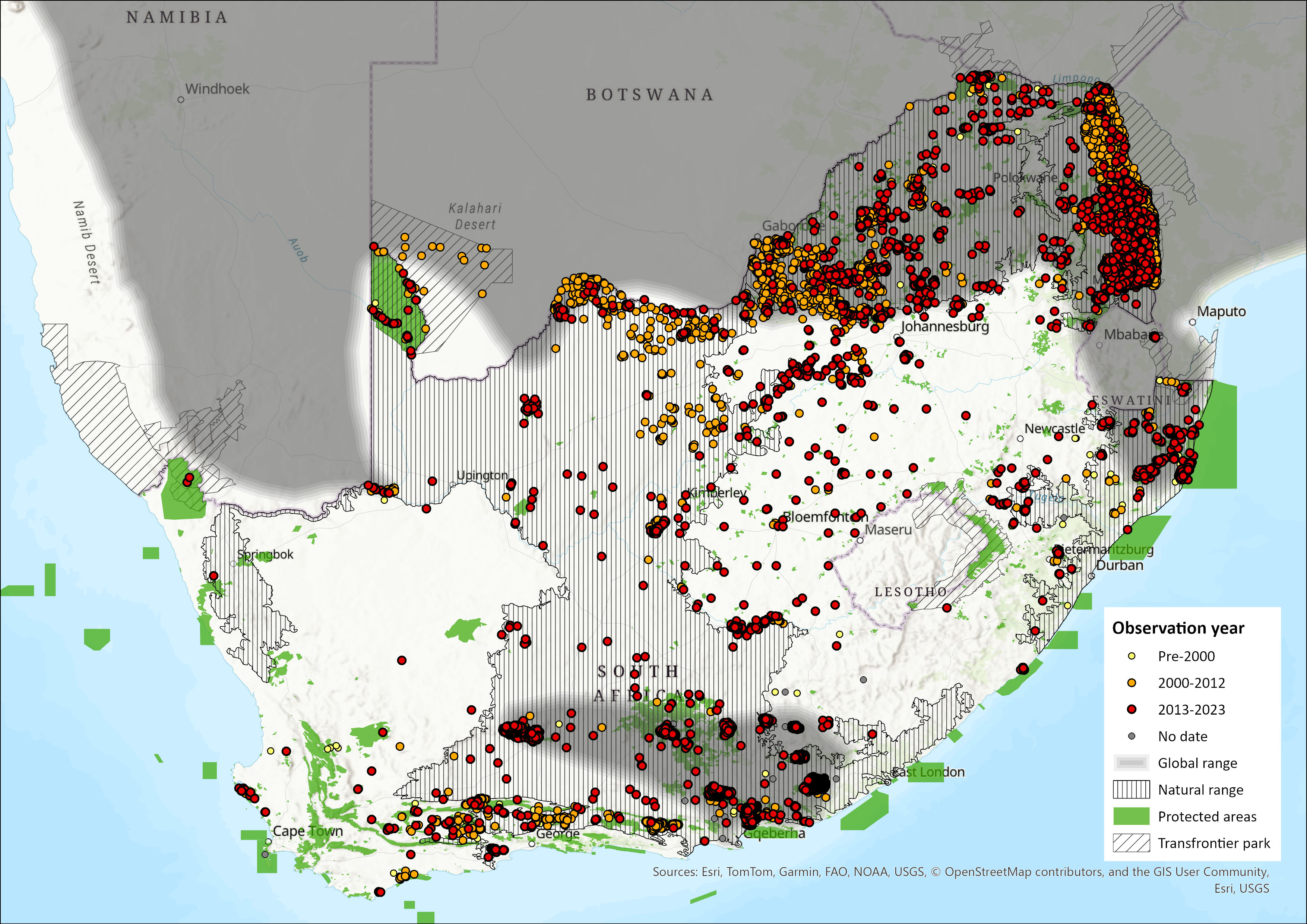
Map
Figure 1. Distribution records for Greater Kudu (Tragelaphus strepsiceros) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Djibouti | Possibly Extinct | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Somalia | Extinct Post-1500 | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Possibly Extinct | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future.
Population information
Throughout Africa, East (1999) estimated a total population of around 482,000 Greater Kudu with the largest populations found in Namibia and South Africa, where the species remains widely abundant on private farmland. Within the assessment region, there are estimated to be at least 25,794–31,045 animals on formally protected areas (68 areas; 2013–2015 counts) and an additional 65,217 animals on private game farms and ranches (740 properties; 2013–2014 counts) across the country; which yields an estimated current total of at least 91,011–96,262 animals in the assessment region. If we assume that 70% of these are mature individuals this yields a mature population size of 63,708–67,383 animals. KNP is the largest subpopulation, with an estimated 8,239–13,490 animals in 2012 (using distance sampling transects) (Ferreira et al. 2013).
Generation length is estimated to be 6.2–8.5 years (Pacifici et al. 2013; IUCN SSC Antelope Specialist Group 2016), which corresponds to an 18–25-year three generation period (1990/1996–2015). Limited subpopulation data are available over this period but using a sample of 23 formally protected areas across the range of the species, there has been an estimated 72–81% increase in the population over three generations (4,324–7,812 animals). We assume that similar increases have occurred on private lands. The number of free-roaming herds outside game farms or protected areas may have also increased since the 1990s (Power 2014). Throughout Africa, population trends are generally increasing in protected areas and on private land and decreasing elsewhere (IUCN SSC Antelope Specialist Group 2016).
Population densities estimated from aerial surveys are frequently less than 10 animals / 100 km², even in areas where this species is known to be at least reasonably common (IUCN SSC Antelope Specialist Group 2016). Higher densities of 20–40 animals / 100 km² have been estimated by aerial surveys in some other areas (IUCN SSC Antelope Specialist Group 2016). In general, aerial surveys underestimate Greater Kudu density as the species inhabits thicket areas. For example, ground counts in areas where the Greater Kudu is common have produced population density estimates from 30 animals / 100 km² to 410 animals / 100 km² (East 1999). Thus, estimating density and abundance of this species with distance sampling through line transects of pellet groups may be more accurate than aerial counts (Ellis & Bernard 2005), as estimates of abundance were two to three times greater than helicopter counts.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | – |
Current population trend: Increasing (based on 2016 assessment data)
Number of mature individuals in population: 63,708-67,383
Number of mature individuals in largest subpopulation: 5,767-9,443 (KNP)
Number of sites: c. 808, of which 68 occur on formally protected areas.
Extreme fluctuations in the number of subpopulations: None recorded
Continuing decline in number of subpopulations: (Not specified)
Severely fragmented: No. It is a browser found mainly in savannah woodlands and is difficult to contain within livestock fenced areas.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: Zero
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: Unlikely
Probability of extinction in the wild within 100 years: Not specified
Population genetics
Phylogeographic assessments on Tragelaphus strepsiceros revealed the existence of three evolutionary significant units (ESUs), with animals in the assessment region falling within the southern ESU, which corresponds to the subspecies T. s. strepsiceros (Nersting & Arctander 2001; Van Heerden et al. 2021). Additional structure within this ESU has also been detected, with two genetically distinct clades identified: an east South African clade (animals from North West, Free State, Limpopo, Mpumalanga, KwaZulu-Natal provinces, including animals from Botswana, Zimbabwe, Zambia) and a Cape clade (animals from Northern, Western and Eastern Cape) (Van Heerden et al. 2021; Jacobs et al. 2022). This suggests that despite the species being largely confined to several fenced (managed) properties, they exist as two metapopulations or subpopulations. Wildlife ranchers and hunters regard the Eastern Cape clade animals as being smaller than the northern clade and adult bulls in this clade have a back face (Flack 2012).
While population estimates are unknown for the two subpopulations separately, the estimate for the total population in the assessment region is between 63,708–67,380 (see Population section). Assuming the subpopulations are equally abundant, each would contain approximately 32,500 mature individuals. Assuming a Nc/Ne conversion ratio of 0.1-0.3, it is estimated that the Ne for each subpopulation is 3,250-9,750, exceeding the Ne 500 threshold. Quantification of Ne within each subpopulation is needed to validate these estimates.
Please scroll horisontal on mobile to view the table
Habitats and ecology
Preferred habitat includes plains, open woodland (Curlewis et al. 2018) mixed scrub woodland (O’Kane et al. 2013), Acacia, and Mopane bush on lowlands, hills, and mountains. Vegetation and environmental conditions can influence where kudus occupy (Curlewis et al. 2018). It is one of the few large mammals that can exist in settled areas, such as in the scrub woodland and bush that reclaims abandoned fields and degraded pastures (IUCN SSC Antelope Specialist Group 2016). They are browsers and can exist for long periods without drinking, obtaining sufficient moisture from their food, but become water dependent at times when the vegetation is very dry (Valeix et al. 2011; Owen-Smith 2013). Kudus browse a variety of trees, forbs and herbs during different seasons. Although seasonal changes have an influence on what the kudu selects (Makhado et al. 2020). In the Musina Nature Reserve, Limpopo, Kudu browsed several species, including Indigofera, during the dry season. However, during the wet season they preferred to browse species like Leucas and Monsonia (Makhado et al. 2020). In Limpopo, Mopane (Colophospermum mopane) leaves contributed most significantly (47% of intake) in the dry season (Makhado et al. 2016). Overlap in resource use with other browsers has been reported, and it might be higher in areas where kudu are synoptic with similar sized species such as Nyala (du Toit 1990; O’Kane et al. 2011). However, kudu have been observed forage mainly on branch ends while Nyala forage mainly on shoots and mature leaves. This species has been introduced into many extra-limital areas, including areas of Texas, USA where it may compete with native White-tailed Deer (Odocoileus virginianus) for browsing forage (Gray et al. 2007). The home range of a typical kudu herd ranges from 7.9–24 km2 (Owen-Smith 1979; du Toit 1990).
Ecosystem and cultural services: Key species for the sustainable, wildlife-based economy.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| Thicket – Succulent | – | Suitable – | |
| 8. Desert | – | Marginal | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: first conception and 2.3 years, first calf at 3 years
Age at Maturity: Male: Spermatogenesis at 18 months
Size at Maturity (in cms): Female: Sh Height 124 cm weight kg
Size at Maturity (in cms): Male: Sh height 134 cm, weight kg
Longevity: 15 years for females in the wild, up to 22 in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): Record horns 187.6 cms
Size at Birth (Kg): 16 Kg
Gestation Time: 259-271 days
Reproductive Periodicity: one calf annually
Average Annual Fecundity or Litter Size: One
Natural Mortality: 50% in the first year, primarily in the lying up phase. Prime age cows 8% per year, bulls higher (Owen-Smith 2013)
Does the species give birth to live young: Yes
Does the species exhibit parthenogenesis: No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Home ranges. 3-25 Km2 in Kruger as small as 1 Km2 in Succulent thicket (Owen-Smith 2013)
Congregatory: Live in a female group or “Clan” Owen-Smith 2013, males peripheral depending on breeding season)
Systems
System: Terrestrial
General Use and Trade Information
The Greater Kudu is much sought after by hunters, both for the magnificent horns of bulls and more generally for their high-quality meat (Owen-Smith 2013). They are one of the most commonly hunted species in southern Africa, and generate the highest proportion (13.2%) of hunting income in South Africa (Patterson & Khosa 2005). Greater Kudu are also a favoured game-ranching species, because as browsers they do not compete with domestic livestock (Owen-Smith 2013). The percentage of animals in offtake from ranching versus wild is not known. It also has subsistence value (both recreational and illegal bushmeat). Wildlife ranching and the private sector have thus generally had a positive effect on this species as it has been widely reintroduced onto private properties within its natural distribution. The demand for males with exceptional horns for breeding is high, with auction prices of up to R9.4 million (US $ 692,000).
| Subsistence: | Yes |
| Rationale | Bushmeat poaching, biltong hunting |
| Local Commercial: | Yes |
| Further detail including information on economic value if available: | Trophy hunting, live animal sales |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use |
1. Food – human 15. Sport hunting/specimen collecting |
| Subsistence |
1. true 15. – |
| National | True |
| International |
– True |
| Other (please specify) |
– – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Trophy hunting, live animal sales, bushmeat poaching
Threats
The Greater Kudu remains abundant and well managed in most parts of its range within the assessment region. There are no major identified threats. However, competition for resources from livestock ranching and bushmeat poaching could lead to localised declines. Farmers have also expressed concern about illegal poaching of this species from the roads (Power 2014), and numbers are unusually low in certain tribal areas where they were expected to be common (Buijs 2010). Additionally, kudu were one of the most poached species in Borakalalo National Park, North West Province (Nel 2015). However, this does not seem to be affecting the species’ overall long-term survival as they seem to be quite resilient to hunting pressure and remain abundant and well managed in other parts of its range (IUCN SSC Antelope Specialist Group 2016). In the North West Province, kudu have been regarded as a problem species by a small number of farmers owing to crop raiding (Power 2014). While hybridisation between the Greater Kudu and the Lesser Kudu (Tragelaphus imberbis) has not been recorded, hybridisation between Greater Kudu and Nyala can occur (Dalton et al. 2014). However, hybrid animals are considered to be sterile (Dalton et al. 2014); and thus, this is not a serious threat. The species is susceptible to rabies infection and there have been reports of losses of 30–70% of total populations, leading to important economic repercussions (Scott et al. n.d.). However, no rabies cycles have yet been reported for South Africa.
The decline in capacity in provincial conservation agencies in South Africa (Patel et al. 2023) is a valid cause for concern, as the invasion by squatters in some protected areas is not under control (J. Anderson pers. comm.).
Current habitat trend: Stable.
Conservation
Greater Kudu are well represented in protected areas. It also occurs widely outside protected areas, including large numbers on private farms and conservancies in southern Africa (Namibia, Zimbabwe and South Africa), where it is a mainstay of the trophy hunting industry. The species remains widely abundant on private farmland in South Africa and seem to be expanding their distribution outside protected areas. For example, within the North West Province in 2010, there were an estimated 1,771 individuals on provincial protected areas and 13,789 individuals on private lands in the province (Power 2014). No major conservation interventions are necessary for this species. However, ongoing harvest and trade management is necessary to ensure the sustainability of offtake on small ranchlands and protected areas. Internal fences should be removed to form conservancies and allow greater available space for the species. Terrain is a big influence on the kudu’s distribution in its home range and should be taken into account when considering managing inventions (Curlewis et al. 2018).
The animals in the isolated Eastern Cape clade are widely regarded as being smaller than the larger more northern clade (Flack 2012). This may induce some landowners to introduce animals from the northern clade in the hope of increasing trophy horn size.
Recommendations for land managers and practitioners:
- Landowners should continue to form conservancies to sustain wild and free-roaming herds.
- Maintain the genetic integrity of the Eastern Cape clade by not introducing animals from the northern clade and to have a dedicated section in the Rowland Ward and SCI Record books.
Research priorities:
- Effects of wildlife ranching on this species.
- Niche overlap and competition with other browsers.
- Population and harvest level trends, especially on private land.
- Research whether the Eastern Cape Kudu clade could be another subspecies.
Encouraged citizen actions:
- Report sightings of free-roaming individuals outside private lands or protected areas on virtual museum platforms (for example, iNaturalist and MammalMAP).
- Encourage communities to stock and manage Kudu in suitable habitats in communally owned land.
Bibliography
Ansell, W.F.H. 1972. Part 2, 15 Family Artiodactyla. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-84. Smithsonian Institution Press, Washington, DC, USA.
Buijs, D. 2010. Aerial survey of wildlife and domestic stock on tribal land in North West Province 2010. DACERD, Mmabatho.
Curlewis, B.J., Luus-Powell, W.J., Potgieter, M.J. and Makhado, R.A., 2018. Terrain encountered along the browsing path of Tragelaphus strepsiceros in the Musina Nature Reserve, Limpopo Province. Transactions of the Royal Society of South Africa, 73(3), pp.264-269.
Dalton DL, Tordiffe A, Luther I, Duran A, Wyk AM van, Brettschneider H, Oosthuizen A, Modiba C, Kotzé A. 2014. Interspecific hybridization between greater kudu and nyala. Genetica 142: 265-271.
du Toit JT. 1990. Home range—body mass relations: a field study on African browsing ruminants. Oecologia 85: 301-303.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Ellis AM, Bernard RTF. 2005. Estimating the density of kudu (Tragelaphus strepsiceros) in subtropical thicket using line transect surveys of dung and DISTANCE software. African Journal of Ecology 43: 362-368.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Summary Report: Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Flack. P. 2012. Eastern Cape Greater Kudu. 97-123 in Hunting the Spiral Horns: Kudu the Top African Antelope. Rowland Ward Publications . 278 pp.
Gess D, Swart S. 2014. The stag of the Eastern Cape: Power: status and kudu hunting in the Albany and Fort Beaufort districts, 1890 to 1905. African Historical Review 46: 48-76.
Gray SS, Simpson TR, Baccus JT, Manning RW, Wayne Schwertner T. 2007. Seasonal diet and foraging preference of Greater Kudu Tragelaphus strepsiceros in the Llano Uplift of Texas. Wildlife Biology 13: 75-83.
Haltenorth T. 1963. Klassifikation der Saügetiere: Artiodactyla I. Handbuch der Zoologie 8: 1-167.
IUCN SSC Antelope Specialist Group. 2016. Tragelaphus strepsiceros. The IUCN Red List of Threatened Species 2016: e.T22054A50196734.
Jacobs, R., Coetzer, W.G. and Grobler, J.P., 2022. A phylogeographic assessment of the greater kudu (Tragelaphus strepsiceros) across South Africa. Conservation Genetics, 23(5), pp.919-933.
Makhado R, Potgieter M, Luus-Powell W, Cooper S, Oppong C, Kopij G, Mutisi C, Makhabu S. 2016. Tragelaphus strepsiceros browse during the wet season in the mopani veld of Limpopo Province, South Africa. Transactions of the Royal Society of South Africa 71: 17-21.
Makhado, R.A., Curlewis, B.J., Luus-Powell, W.J. and Potgieter, M.J., 2020. Forbs and herbs diet of the Tragelaphus strepsiceros at Musina Nature Reserve, Limpopo Province, South Africa. Afr. J. Ecol, 58, pp.897-901.
Nel, P. 2015. Population estimates for large herbivores and predators in protected areas in the North West Parks Board November 2015. North West Parks Board.
Nersting, L.G. and Arctander, P., 2001. Phylogeography and conservation of impala and greater kudu. Molecular Ecology, 10(3), pp.711-719.
Owen-Smith N. 1979. Assessing the foraging efficiency of a large herbivore, the kudu. South African Journal of Wildlife Research 9: 102-110.
Owen-Smith N. 2013. Tragelaphus strepsiceros Greater Kudu. In: Kingdon JS, Hoffmann M (ed.), The Mammals of Africa. Volume VI: Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer, and Bovids, pp. 152-155. Bloomsbury Publishing, London, UK.
O’Kane CA, Duffy KJ, Page BR, Macdonald DW. 2011. Overlap and seasonal shifts in use of woody plant species amongst a guild of savanna browsers. Journal of Tropical Ecology 27: 249-258.
O’Kane CA, Duffy KJ, Page BR, Macdonald DW. 2013. Effects of resource limitation on habitat usage by the browser guild in Hluhluwe-iMfolozi Park, South Africa. Journal of Tropical Ecology 29: 39-47.
Patel. T., Cowan, O., Little, I., Friedmann, Y. & Blackmore, A. 2023. The State of Provincial Reserves in South Africa, Challenges and Recommendations. https://ewt.org.za/resources/cpsu-programme/
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Patterson C, Khosa P. 2005. Background Research Paper: A status quo on the professional and recreational hunting industry in South Africa. Report prepared for the panel of experts appointed by the Minister of Environmental Affairs and Tourism. TRAFFIC – The wildlife trade monitoring network.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Scott T, Hasses R, Nel L. (n.d.). Rabies in kudu (Tragelaphus strepsiceros). Berliner und Muenchener Tieraerzliche Wochenschrift 125: 141-236.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Valeix M, Fritz H, Sabatier R, Murindagomo F, Cumming D, Duncan P. 2011. Elephant-induced structural changes in the vegetation and habitat selection by large herbivores in an African savanna. Biological Conservation 144: 902-912.
Van Heerden, F.J., 2020. Phylogeography of Tragelaphus strepsiceros (Pallas 1766) in southern Africa (Doctoral dissertation, University of Pretoria).