Greater Cane Rat
Thryonomys swinderianus

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Thryonomys swinderianus – (Temminck, 1827)
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – THRYONOMYIDAE – Thryonomys – swinderianus
Common Names: Greater Cane Rat (English), Grootrietrot, Rietrot (Afrikaans), Tswidi (Sesotho), Sepoopoo, Letswidi, Lebodi, Tlhêkgê (Setswana), Livondo (Swati), Nhleti (Tsonga), Tshedzi (Venda), Ivondwe (Zulu)
Synonyms: No Synonyms
Taxonomic Note: No subspecies are currently recognised (Monadjem et al. 2015).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Patel, T.1 & da Silva, J.M.2
Reviewer: Smith, C.1
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute
Previous Assessors & Reviewers: Avenant, N., Power, J., MacFadyen, D. & Child, M.F.
Previous Contributors: Avery, M., Baxter, R., Mondajem, A., Palmer, G., Taylor, P., Wilson, B. & Relton, C.
Assessment Rationale
Listed as Least Concern because of its wide distribution within the assessment region, its documented range expansion and continuous expansion into large parts of the Free State and North West provinces, its tolerance of habitat modification and ability to thrive in agricultural landscapes, its occurrence in numerous protected areas, and because there are no major threats that would cause population decline. It has also become more common in urban areas and is regularly encountered in and on the edge of large cities and towns such as Pretoria, Johannesburg, Welkom and Kroonstad. No conservation interventions are necessary at present. Conversely, its spread along the Vaal and Orange River systems must be monitored as control operations may be necessary if research proves that they exacerbate ecosystem degradation. The consumption of this species as bushmeat or from captive-bred sources implies that it could potentially be utilised sustainably as a source of low-carbon protein for rural communities. Its occurrence in the central Free State (e.g. around the Welkom mine dams) has been attributed to direct human import as a protein source.
Regional population effects: Limited dispersal possibly occurs between Zimbabwe, Mozambique and South Africa, especially through Great Limpopo Transfrontier Park. While there are some local movements, the availability of suitable habitat may restrict movement. Within their natural/historical distribution area they are common but specialized in their habitat requirement; in the Free State they are less common, but their infiltration since the early 1980s has followed along a number of rivers. Populations are connected via reed bed-fringed rivers. In the Kruger National Park their mapped distribution is along all the rivers. This most likely continues along the east-flowing rivers into Mozambique. There are also suitable habitats that would connect populations in Mozambique to those in the KwaZulu-Natal Province. There is a low likelihood that Zimbabwe can serve as a source population, as the Limpopo River does not offer suitable habitat for this species.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Patel T & da Silva JM. 2025. A conservation assessment of Thryonomys swinderianus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
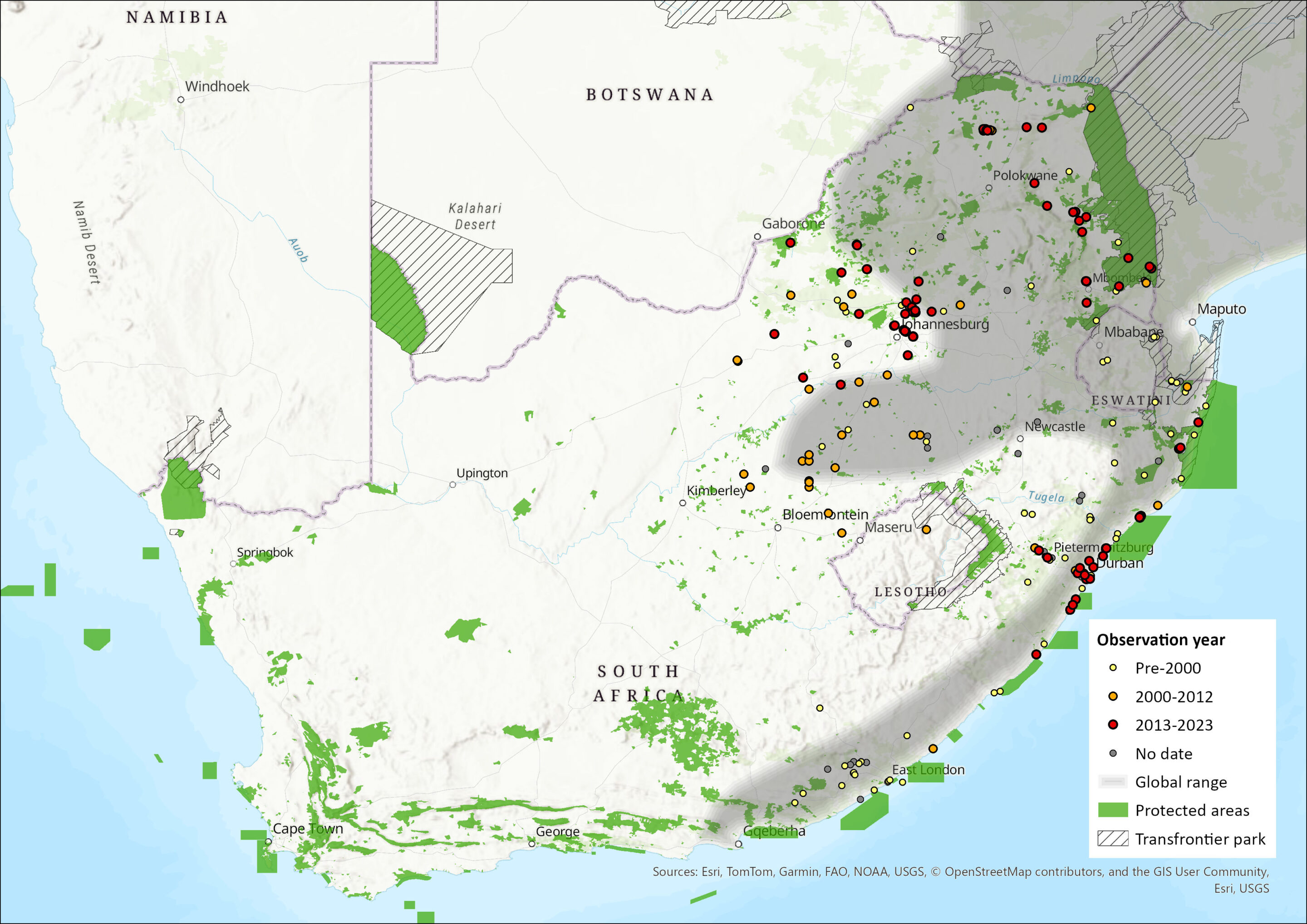
This species is widely distributed throughout the moister parts of sub-Saharan Africa, avoiding only deserts, dry savannahs and the interior of rainforests (Monadjem et al. 2015). However, its distribution is patchy and discontinuous, and they only occur where there is suitable habitat (Smithers 1983; van der Merwe & Avenant 2004; Skinner & Chimimba 2005). Within the assessment region, it occurs from the Grahamstown District in the Eastern Cape Province through KwaZulu-Natal, into Mpumalanga, and then its distribution arcs westwards across the central parts of the Limpopo and North West provinces (Rautenbach 1978; Skinner & Chimimba 2005; Power 2014) and, since the 1980s, also in Gauteng and the Free State (Figure 1; van der Merwe & Avenant 2004). The species occurs in the Albany Thicket, Savannah, Indian Ocean Coastal Belt and Grassland biomes (Mucina & Rutherford 2006), where it is confined to reed beds along drainage lines and rivers.
The western parts of its range are expanding. The most westerly records are from the Barberspan area in North West Province (Power 2014) and the pans to the west of Bultfontein in the Free State Province (N.L. Avenant unpubl. data), which is congruent with a general westward range expansion in the central interior. In the Free State, for instance, they have spread rapidly from east to west (first record in 1983) over a distance of 400 km within 20 years, presumably following rivers and a series of pans that are, during the wet season, connected by dense grass and Phragmites spp. reed bed vegetation (van der Merwe & Avenant 2004). While they were once reported not to occur in the Vaal and the Orange River systems (Rautenbach 1982; Lynch 1983; Smithers 1983), they now are regularly seen there and along its tributaries, as a breeding species (van der Merwe & Avenant 2004; Power 2014). They have been observed in the Leeu and Caledon Rivers that flow into the Orange River (N.L. Avenant unpubl. data). Greater Cane Rats have also been observed in the Katse Dam area of Lesotho (du Plessis et al. 2014). Further range expansions are expected in the assessment region as they do adapt well to cleared areas where food and industrial crops are grown, as well as to secondary savannahs formed as a result of forest destruction (Jori et al. 1995). As they are associated with Phragmites spp. reed beds, they will continue to expand along such river courses; even along ephemeral rivers in the drier interior where dam walls and weirs today support this vegetation throughout the wet and dry seasons.
While the presence of this species in Limpopo, Mpumalanga, KwaZulu-Natal and North West provinces are natural occurrences, its introduction in the interior (Free State Province) was most probably through either accidental import from KwaZulu-Natal in sugar cane trains, or by people using them as a source of food (van der Merwe & Avenant 2004). This species is regularly observed at Rietvlei Dam and Faerie Glen Nature Reserve in Pretoria East and has been observed as roadkill in other parts of Pretoria and Centurion, Gauteng Province, and has also been recorded in the Phragmites spp. reed beds at Telperion Nature Reserve in Mpumalanga Province (D. MacFadyen unpubl. data). It has also been seen at Punda Maria camp and in the vlei at the N’washitshumbe enclosure site in northern Kruger National Park (D. MacFadyen pers. obs.). As it is also kept as a pet (N.L. Avenant unpubl. data), this can also result in its introduction and spread. Supporting this, Greater Cane Rats have been observed being moved around in small cages and kept as a food source in pens like chickens, such as around mines in the Welkom area (N.L. Avenant pers. obs.). Similarly, Greater Cane Rats may have been brought to the Katse area in Lesotho as a sought after and easy-to-breed protein source (du Plessis et al. 2014). Once introduced to the wild, they are thought to benefit from the reed beds close to man-made weirs and dam walls as well as the decreased natural predator density and diversity.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Greater Cane Rat (Thryonomys swinderianus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Benin | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Congo | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Côte d’Ivoire | Extant | Native | – | – |
| Equatorial Guinea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Gabon | Extant | Native | – | – |
| Gambia | Extant | Native | – | – |
| Ghana | Extant | Native | – | – |
| Guinea | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Introduced | – | – |
| Liberia | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| Nigeria | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Senegal | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Free State | Extant | Introduced | – | – |
| South Africa -> Gauteng | Extant | Introduced | – | – |
| South Africa -> KwaZulu-Natal | Extant | Native | – | – |
| South Africa -> Limpopo Province | Extant | Native | – | – |
| South Africa -> Mpumalanga | Extant | Native | – | – |
| South Africa -> North-West Province | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Togo | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
From a study conducted on scientific publications of the Greater Cane Rat from 1969 to 2019, there is no research on the effect of climate change on the species (Mustapha et al. 2020).
Population Information
This is an increasingly common species within the assessment region. It is often abundant in agricultural lands. For example, in KwaZulu-Natal and Mpumalanga provinces, it can be common in sugar cane fields. It has been observed in relatively high numbers in Punda Maria, and the Nwashitshumbe enclosure site on the northern plains of Kruger National Park (D. MacFadyen pers. obs.). In the central interior it is found in small groups where it does occur and is considered adapted to the harsh cold and dry winters. In the Free State both adult and juvenile specimens have been recorded throughout the year; and it was also still present in all river catchments where it was located previously, which indicates that this species survives and breeds under local conditions (van der Merwe & Avenant 2004; N.L. Avenant unpubl. data). Furthermore, its abundance and range expansion should increase as the damming of rivers has created suitable habitat with water, reed and riverine bush throughout the year, while the predator communities in these areas are depressed compared to historical times. Additionally, there is a possibility that individuals are continuously being brought into the mine areas as a food source.
Population Information
|
Continuing decline in mature individuals? |
Qualifier |
Justification |
|
No |
Inferred |
No, although local losses may be incurred from bushmeat hunting. |
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Until recently, the genetic studies on T. swinderianus were scarce and focused on animals outside the assessment region (e.g., Adenyo et al. 2013; Adenyo et al. 2017a; Adenyo et al. 2017b, Coker et al. 2017, Kilwanila et al., 2023) Coetzer (2023) conducted the first phylogenetic study on the species across the assessment region and found animals from South Africa to form a genetically distinct clade from animals in West Africa. Additionally, South African animals were found to share haplotypes, indicating they represent a single population or metapopulation. Fine scale sampling and analyses across its distribution are needed however to obtain more accurate assessments on possible subpopulation structure. Assuming no genetically distinct subpopulations have gone extinct, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (1/1 population remaining).
While there are no population size or density estimates for this species (see Population section), it is considered common with an expanding range due to its ability to tolerate transformed habitats. Based on this it is assumed that well over 5000 individuals are likely to exist in the assessment region, translating to an effective population size (Ne) of over 500 (assuming a 0.1 conversion ratio of Ne to census size). While the 5 000 value is not a census count, the quantity can still be used as a proxy for quantifying the proportion of populations with an Ne > 500 indicator, which is valued at 1.0 (1/1 population with Ne > 500).
Habitats and ecology
This species is typically found within reed beds or in areas of dense, tall grass with thick reed or cane-like stems, typical of riverine and other similar habitats. For example, in the Free State Province, it is associated with reed beds and dense grass around rivers, streams, vleis and pans, as well as irrigation dams, some water canals and mine dams (van der Merwe & Avenant 2004). It also uses thatching grass, which is of a similar reed-like structure. It is seldom found far from water and tends to inhabit the warmer areas of South Africa, although it has adapted to the winters of the Highveld (van der Merwe & Avenant 2004; N.L. Avenant unpubl. data). Smithers (1983) and Skinner and Chimimba (2005) note that agricultural crops (such as maize, wheat and sugar cane) have greatly improved the habitat for this species, such that it has become an agricultural pest in some regions. In the Kruger National Park, its distribution is confined to both the perennial and seasonal rivers (Pienaar et al. 1987). It has also become more common in urban areas and is regularly encountered in and on the edge of large cities.
At more than 4.5 kg, Greater Cane Rats are the second largest rodents in Africa. They feed on roots, reeds and the stems of grasses along rivers and in other moist areas. Grasses eaten include Couch Grass (Cynodon dactylon), Swamp Grass (Hemarthria altissima), Elephant Grass (Pennisetum purpureum) and Guinea Grass (Panicum maximum). While grasses are their principal food, they raid crops (such as maize, millet, sorghum, wheat, and sugar cane) and cause damage by cutting the stems at their base with their broad sharp incisors (Skinner & Chimimba 2005).
Greater Cane Rats are predominantly nocturnal or crepuscular. They are polygamous, with a single male observed with a number of females and their offspring. Although they are generally reported to be solitary, family groups of up to ten may live in restricted areas (Skinner & Chimimba 2005). Two litters of as many as 12 young are born annually (Skinner & Chimimba 2005), with two to four pups per litter the norm in all southern African countries for which data are available (van der Merwe 1999). In Greater Cane Rat farms in Benin, however, they breed throughout the year and litters of > 14 are frequently observed (Asibey 1974a; A. Durojave pers. comm.). No reproductive information is available for Greater Cane Rats in the Free State Province, but this is expected to be similar to the rest of the southern African sub-region.
Ecosystem and cultural services: This species is an important forage species for Crowned Eagles (Stephanoaetus coronatus), as well as small and medium sized carnivores, such as Servals (Leptailurus serval), African Rock Pythons (Python sebae) and Leopards (Panthera pardus). It may also be an indicator and/or agent of ecosystem degradation.
In the Free State Province, for instance, the river systems in which Greater Cane Rats occur are all considered in poor to fair condition (River Health Programme 2003). Through competition with indigenous rodents, Greater Cane Rats may well exacerbate the poor state of these ecosystems, thereby degrading habitat integrity even further. Furthermore, although not much is known about their diet in the Free State, it is possible that they can become pests in lucerne, wheat and maize fields as their numbers increase. Studies elsewhere have indicated that a family of Greater Cane Rats can cause severe damage to such crops, stressing the importance and urgency of further studies on this issue in the South African interior for economic reasons. It is uncertain how they will affect the natural ecosystems and, therefore, their importance as an invader species in the Free State Province also needs further research.
Interestingly, this species is often portrayed as aggressive, and there are myths of them attacking sugar cane farmers, but in truth this is a very docile species. When caught, there is no form of aggression, and they have been found as pets in houses (N.L. Avenant pers. obs.). The skin of the cane rat breaks easily, possibly a drastic predator avoidance mechanism.
Finally, if managed correctly, Greater Cane Rats could become a source of cheap, low-carbon protein within the assessment region, through commercial farming on a sustainable basis (see Use and Trade).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 5.1. Wetlands (inland) -> Wetlands (inland) – Permanent Rivers/Streams/Creeks (includes waterfalls) | – | Suitable | – |
| 5.2. Wetlands (inland) -> Wetlands (inland) – Seasonal/Intermittent/Irregular Rivers/Streams/Creeks | – | Suitable | – |
| 5.3. Wetlands (inland) -> Wetlands (inland) – Shrub Dominated Wetlands | – | Suitable | – |
| 14.1. Artificial/Terrestrial -> Artificial/Terrestrial – Arable Land | – | Suitable | – |
| 14.3. Artificial/Terrestrial -> Artificial/Terrestrial – Plantations | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial, Freshwater (=Inland waters)
General Use and Trade Information
This species is very common in bushmeat markets and there have been numerous studies investigating the viability of farming this species to supply demands for protein in West and Central Africa (for example, Ajayi 1971; Asibey 1974b; Baptist & Mensah 1986; Kyle 1987; Jori et al. 1995). Already in 1919, Fitzsimons stated that cane rats could be domesticated and bred as a food source, their flesh being “equal to that of partridge and pheasant, and much superior to that of the hare and rabbit”. Today, Greater Cane Rat meat is a protein source highly in demand by all levels of society in many African countries, and often sells for more per kilogram than chicken, beef, pork, or lamb (Martin 1985; Baptist & Mensah 1986; National Research Council 1991).
The number of Greater Cane Rats hunted annually in West Africa was estimated to be 80 million, yielding the equivalent of 300,000 metric tons of meat (Jori et al. 1995). They are in such a high demand that, in an effort to capitalise on the markets for this delicacy, agricultural extension services of Cameroon, Ghana, Ivory Coast, Nigeria, Togo, and particularly Benin, are encouraging farmers to rear them as micro-livestock (National Research Council 1991). In West Africa, they are thus commercially farmed and are delicacies on most restaurant menus, where an estimated 16,000 rats are managed in 500 farms in Benin alone (Granjon & Duplantier 2009). In the assessment region, however, it is undervalued as a protein source and is yet to be farmed commercially. There is much potential for this practice to establish itself within the assessment region and enhance food security: they are relatively cheap to keep and feed (prefer grass above other plants), and are capable of growing when the fibre content of the grass is much higher than that accepted by many other domestic stock (Alexander 1991).
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
Used as food by farm and mine labourers. |
– |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: Possibly increasing
Threats
There are no major threats to this species. It is a major source of bushmeat in West Africa, but the threat is not as pronounced in South Africa. Rather, the bushmeat trade may be contributing to range expansion within the assessment region: there are indications that it is carried as a food source (for example, by mine workers transporting Greater Cane Rats from their natural range) into areas where it does not occur naturally (for example, the Free State gold fields); here it has adapted and spread further along rivers and drainage lines.
Greater Cane Rats are often considered a major agricultural pest, as they raid croplands and gardens (Roberts 1951; Skinner & Chimimba 2005), which has led to control measures being applied to keep them out of crops (Hoffmann 2008). It is almost certain that they will also raid lucerne fields within the assessment region (van der Merwe & Avenant 2004), which may become an issue with the expansion of game feeding operations as part of the wildlife ranching industry, and in irrigation fields along rivers in the drier interior. They are very fond of kikuyu grass and have been observed to raid lawns in urban areas where, as with some other grasses, they scratch the soil apart to reach the underground stems and roots (M. van der Merwe pers. obs.). So far, they have not yet been reported as a problem animal in either the Free State or North West provinces (van der Merwe & Avenant 2004; Power 2014).
Greater Cane Rats are also prone to be hit by vehicles and are regularly recorded dead on roads, especially in areas with thick vegetation on the road verges. Urban and rural sprawl will also increase predation by domestic pets. Thus, urbanisation and the increase of vehicle traffic is a minor threat to this species when invading cities and towns.
Conservation
This species occurs in many protected areas within the assessment region, including Kruger National Park (Limpopo/Mpumalanga), Faerie Glen Nature Reserve and Ezemvelo Nature Reserve (Gauteng), and Telperion Nature Reserve (Mpumalanga). No specific interventions are required at present, but farmers are encouraged to use holistic control techniques and perhaps be incentivised to harvest the species sustainably as a low-carbon source of protein. Protected area expansion (for example, through provincial stewardship programmes) and enforcement of legislation that protect riparian zone habitats would benefit this species. Within its historical distribution, corridors of suitable vegetation in urban areas should be connected to allow for dispersal. However, artificial range expansion and introduction of this species into regions outside of its historic distribution should not be encouraged.
Recommendations for land managers and practitioners:
- Farming of the species for a source of protein should be advocated for rural communities.
- In the central interior (Free State and North West provinces) the invasion of this species should be monitored. It can become a pest in agricultural fields and has now spread to both the Vaal and Orange River systems, where continuous reed beds occur – this means that it may (and probably will) invade further along these rivers and into their catchments.
Research priorities:
- Impacts of human settlement and agricultural expansion on the abundance and distribution of the species.
- Role of natural predators, and thus holistic management, in the control of damage-causing individuals.
- The ecological and ecosystem consequences of invasion into new areas.
- The economic consequences of invasion into new areas.
- Extensive and valuable research into the breeding rate of this species has been conducted by Prof. Mac van der Merwe at the University of Pretoria. These types of studies should be continued and expanded upon.
Encouraged citizen actions:
- Report sightings of this species, especially outside protected areas, on virtual museum platforms (for example, iNaturalist and MammalMAP) and/or the closest natural history museum, to help monitor invasion rates, directions and habitat preferences. Piles of droppings and the characteristic cut stems of grasses indicate their presence.
- Within their natural distribution area, landowners and city planners can leave corridors of rank vegetation and grass to encourage dispersal and connectivity for this species.
- Ensure thatching grass and reeds are cut on road verges to reduce road collisions.
- Take part in museum distribution projects (see, for example, van der Merwe & Avenant 2004).
Bibliography
Adenyo, C., Hayano, A., Kayang, B.B., Owusu, E.H. and Inoue-Murayama, M., 2013. Mitochondrial D-loop diversity of grasscutter (Thryonomys swinderianus Rodentia: Hystricomorpha) in Ghana.
Adenyo, C., Kayang, B.B., Owusu, E.H., Inoue, E. and Inoue-Murayama, M., 2017. Genetic diversity of grasscutter (Thryonomys swinderianus, Rodentia, Hystricomorpha) in Ghana based on microsatellite markers. West African Journal of Applied Ecology, 25(2), pp.1-15.
Adenyo, C., Ogden, R., Kayang, B., Onuma, M., Nakajima, N. and Inoue‐Murayama, M., 2017. Genome‐wide DNA markers to support genetic management for domestication and commercial production in a large rodent, the Ghanaian grasscutter (Thryonomys swinderianus). Animal Genetics, 48(1), pp.113-115.
Ajayi SS. 1971. Wildlife as a source of protein in Nigeria: some principles for Development. Nigerian Field 36: 115–127.
Alexander, A. 1991. Why is the cane rat not farmed in South Africa? Farmer’s Weekly March 29: 60–61.
Asibey EOA. 1974. Wildlife as a source of protein in Africa south of the Sahara. Biological Conservation 6: 32–39.
Asibey EOA. 1974a. Reproduction in the grasscutter in Ghana. Symposia of the Zoological Society of London 34: 251–263.
Avenant NL, du Plessis J, Senoge J. 2014. Mammals of the Mohale Dam catchment. Specialist report for Contract 1273: Biological Resources Monitoring within Phase 1 of the LHWP Catchments 2013-14. Report no. AEC/14/12 submitted by Anchor Environmental Consultants to the Lesotho Highlands Development Authority.
Baptist R, Mensah GA. 1986. The cane rat: farm animal of the future. World Animal Review 60: 2–6.
Coker, O.M., Omonona, A.O., Fagbohun, O.A., Pylant, C. and Austin, J.D., 2017. Genetic structure of wild and domesticated grasscutters (Thryonomys swinderianus) from south-western Nigeria. African Zoology, 52(3), pp.155-162.
Coetzer, W.G., 2023. A phylogeographic assessment of South African greater cane rats (Thryonomys swinderianus): Preliminary insights. Vertebrate Zoology, 73, pp.277-288.
Granjon, L. and Duplantier, J.M. 2009. Les Rongeurs de l’Afrique sahélo-soudanienne. IRD/MNHN (Collection Faune et Flore tropicale N°43). Marseille.
Hoffmann M. 2008. Thryonomys swinderianus.
Jori, F., Mensah, G.A. and Adjanohoun, E. 1995. Grasscutter production: an example of rational exploitation of wildlife. Biodiversity and Conservation 4: 257-265.
Kilwanila, S.I., Lyimo, C.M. and Rija, A.A., 2022. Mitochondrial genetic diversity of the Greater Cane rat (Thryonomys swinderianus) populations from the Eastern Arc Mountains ecosystem, Tanzania. Molecular Biology Reports, 49(11), pp.10431-10442.
Kyle R. 1987. A Feast in the Wild. Kudu Publishing, Oxford, UK.
Lynch, C.D. 1983. The mammals of the Orange Free State, South Africa. Navorsinge van die Nasionale Museum Bloemfontein 18: 1-218.
Martin GHG. 1985. West Africa: carcass composition and palatability of some wild animals commonly used as food. World Animal Review.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. De Gruyter, Berlin, Germany.
Mucina, L. and Rutherford, M.C. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria, South Africa.
Mustapha OA, Teriba EE, Ezekiel OS,Olude AM, Akinloye AK, Olopade JO. A study of scientific publications on the greater cane rat (Thryonomys swinderianus, Temminck 1827). Animal Model Exp Med. 2020; 3:40–46. https://doi.org/10.1002/ame2.12103
National Research Council. 1991. Micro livestock: little-known small animals with a promising economic future. National Academic Press, Washington, DC, USA.
Pienaar, U. de V., Joubert, S. C. J., Hall-Martin, A., de Graff, G. and Rautenbach, I. L. 1987. Field guide to the mammals of the Kruger National Park. Struik and the National Parks Board of Trustees, Cape Town and Pretoria.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Rautenbach, I.L. 1978. Ecological distribution of the mammals of the Transvaal (Vertebrata: Mammalia). Annals of the Transvaal Museum 31: 131-156.
River Health Programme. 2003. State-of-Rivers Report: Free State Region River Systems. Department of Water Affairs and Forestry, Pretoria.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1983. The Mammals of the Southern African Subregion. University of Pretoria, Pretoria, South Africa.
van Zyl A, van der Merwe M, Blignaut AS. 1999. Meat quality and carcass characteristics of the vondo, Thryonomys swinderianus. South African Journal of Animal Science 29(120–123).
van der Merwe M, Avenant NL. 2004. The greater cane rat, Thryonomys swinderianus, is a pest species that is expanding its range in southern Africa. Navorsinge van die Nasionale Museum Bloemfontein 20: 3–10.
van der Merwe M. 1999. Breeding season and breeding potential of the greater cane rat (Thryonomys swinderianus) in captivity in South Africa. South African Journal of Zoology 34: 69–73.