
Giant Golden Mole
Chrysospalax trevelyani

2025 Red list status
Endangered
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Chrysospalax trevelyani – (Günther, 1875)
ANIMALIA – CHORDATA – MAMMALIA – AFROSORICIDA – CHRYSOCHLORIDAE – Chrysospalax – trevelyani
Common Names: Giant Golden Mole (English), Reuse-Gouemol, Reuse-Kruipmol (Afrikaans), Intuku-yehlathi (Xhosa)
Synonyms: Bematiscus (Cope 1892); Chrysochloris trevelyani Günther, 1875
Taxonomic Note:
Generic synonym = Bematiscus Cope, 1892 (see Ellerman et al. 1953). No subspecies. One sister species in the genus, C. villosus (A. Smith 1833; see Bronner, 2024).
Red List Status: EN – Endangered, B2ab(i,ii,iii,iv,v) (IUCN version 3.1)
Assessment Information
Assessor: Mynhardt, S.1 & da Silva, J.2
Reviewer: Asher, R.J.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3University of Cambridge
Previous Assessor: Bronner, G.
Previous Reviewers: Asher, R.J. & Taylor, A.
Assessment Rationale
Although recorded from 17 localities in the Eastern Cape, this species is now probably locally extinct at many sites, and possibly survives only in larger patches of indigenous Afromontane forest on the eastern slopes of the Amathole mountains. Although the historical extent of occurrence (EOO) is >20,000 km2, including museum records as far east as “Port St Johns, W. Pondoland” (AMNH 54365) and “Notinsila” (probably Ntsila, UMZC E.5470.H), it has very specific habitat requirements and its total area of occupancy (AOO) is estimated to be 140 km2. Its distribution is severely fragmented (even if historical records are included as many of these were from small and isolated indigenous forest patches). The species does not occur in commercial forestry plantations which abut, or have replaced, many of the remaining patches of natural habitat. Some of the larger indigenous forests are officially “protected”, but management and conservation actions on the ground are often poor. Many of these “protected forests” fall under the jurisdiction of local tribal chiefs, and even in some state-managed forests cattle are allowed to range freely, and trample/degrade the habitat of this species. Recreational hunting by young herdsmen and pack-hunting by domestic/feral dogs pose a threat at some locations, whereas bark-stripping of trees for traditional medicines, collection of firewood and burning of unprotected forest patches degrade their preferred habitats. Recent surveys (2021 and 2022) conducted in these indigenous forests, involving field trapping and eDNA sampling, failed to detect this species, indicating that it may already be locally extinct at these localities. Ongoing urbanisation in the vicinity of East London/Buffalo City, and coastal tourism developments have disturbed many of the coastal forests in which this species may have occurred historically. Climate change may also represent an emerging threat. Given the restricted area of occupancy, probable reduction in number of locations to <10, and the varied and probably increasing threats to this species, it is listed as Endangered.
Regional population effects: All known populations are endemic to South Africa. Range is highly fragmented and restricted to patches of Afromontane and Transkei Coastal Scarp forests, many of which are small and isolated by unsuitable or transformed habitats that probably prevent migration and gene flow among demes of this lowly vagile species.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Mynhardt S & da Silva JM. 2025. A conservation assessment of Chrysospalax trevelyani. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
This species is endemic to South Africa. Recorded from indigenous Afromontane forests in Eastern Cape from East London northwards along coast to Port St Johns, and inland to Amathole and Kologha Mountains near King Williams Town and Stutterheim.
Estimated Area of Occupancy: 140 km2
Continuing decline in AOO: yes
Estimated extent of occurrence: 21,311 km2
Continuing decline in EOO: yes
Number of locations: 10
Continuing decline in number of locations: yes
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
MAP
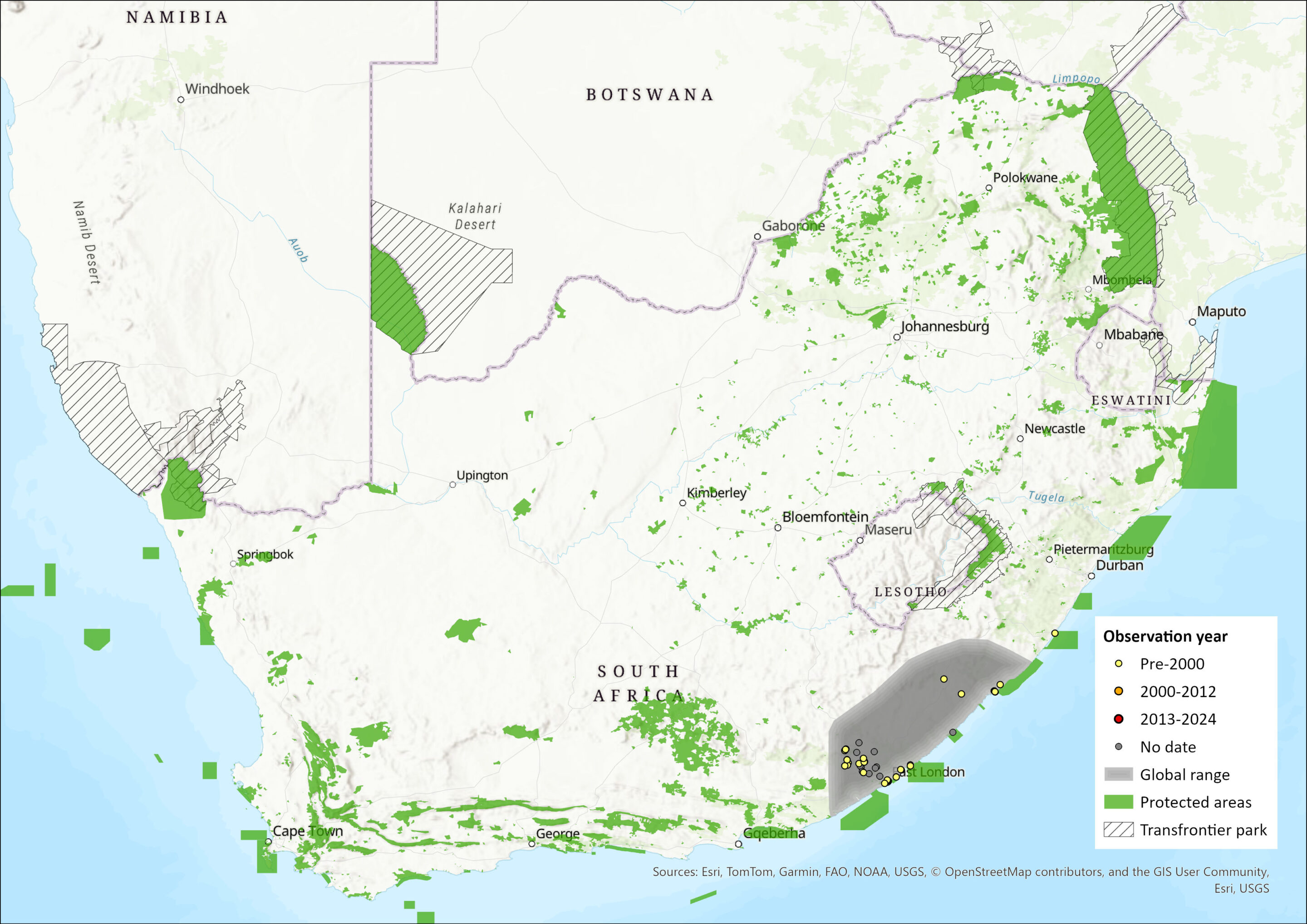 Figure 1. Distribution records for Giant Golden Mole (Chrysospalax trevelyani) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Giant Golden Mole (Chrysospalax trevelyani) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| South Africa | Extant | Native | – | Resident |
| South Africa -> Eastern Cape Province | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Although the potential impacts of climate change on this species have not been directly assessed, climate change is expected to impact most golden mole species. Since these subterranean animals are restricted to soft soils for burrowing, and have very low vagility, their ability to migrate or shift their distribution ranges in response to climate change is very limited. Climate change is likely to impact golden moles through changes in vegetation type, soil moisture and prey availability. Erasmus et al. (2002) projected the extinction of this species due to a 2° C increase in temperature from climate change.
Population information
Locally common, but with a clumped dispersion.
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | Inferred | Suspected |
| Extreme fluctuations in the number of subpopulations | Justification |
| Unknown | – |
| Continuing decline in number of subpopulations | Qualifier | Justification |
| Yes | Inferred | Suspected |
Current population trend: Unknown, but expected to be declining due to ongoing loss of habitat.
All individuals in one subpopulation: No
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Severely fragmented: Yes, this species occurs in specific microhabitats within Afromontane forests and some Coastal Scarp forests of the Eastern Cape, which are highly fragmented owing to both historical and anthropogenic factors.
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
While phylogenetic assessments have included C. trevelyani (e.g., Bronner et al. 2024), the species has not been subject to a population genetic investigation. Given that the distribution is thought to be severely fragmented, it is possible that multiple genetically distinct subpopulations exist and while they might be extirpated from some indigenous forests, it is unknown is these sites may have contained genetically distinct subpopulations.
Given the uncertainty in the number of existing and extinct populations, and their potential size, the two genetic diversity indicators in the Convention of Biological Diversity’s Global Biodiversity Framework cannot be quantified. It is highly recommended that continued survey attempts persist and if possible, a population genomic study conducted across the species range.
Habitats and ecology
Occurs in Transkei Coastal Scarp forests and Amathole Mistbelt forests, sometimes marginally into adjacent grassland habitats (Maddock 1986). Not present in commercial forestry plantations, which abut or have replaced many indigenous forest patches. They have specific habitat requirements, selecting areas in forest patches with soft soils, well-developed undergrowth, and deep leaf-litter layers; but avoid steep slopes and rocky terrain (Bronner 2013). Apparently restricted to larger forest patches (Castley et al. 2000). They make short tunnels (1-13 m long) amidst the roots of trees, and forage mainly above ground in leaf litter, rooting for small invertebrates (especially giant earthworms and omniscomorph millipedes) and may even consume any small vertebrates they encounter (Bronner 2013). They are predominantly nocturnal, but Maddock and Hickman (1985) reported some minor diurnal activity in captive animals. Although having been observed to enter water and use its forefeet as paddles, Hickman (1986) found that due to their size and small body surface to mass ratio, buoyancy, endurance, manoeuvrability and speed is low, thus, in the wild, swimming is likely to be limited to short distances.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.5. Forest -> Forest – Subtropical/Tropical Dry | Resident | Suitable | Yes |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | Resident | Suitable | Yes |
| 4.4. Grassland -> Grassland – Temperate | Resident | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: 21-23
Size at Maturity (in cms): Male: 22-24
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): 8
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: Litter size 1-2
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
General notes regarding trade and use of this species: This species does not appear to be utilised or traded in any form. There is no evidence of this species being used in traditional medicine or as bushmeat. They are hunted recreationally by some local youths, however they do not seem to have any cultural significance and many local communities seem to be unaware of their presence.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Likely major threats include: habitat loss owing to fragmentation of forests, which is ongoing in coastal forests as a result of urbanisation (East London / Buffalo City district) and ubiquitous coastal tourist resorts; and degradation of remaining forests as a result of forest clearance, collection of firewood, bark stripping, cutting for construction, allegedly sustainable timber harvesting and livestock overgrazing/trampling. The species may now be locally extinct at many locations where it occurred formerly (even within the Amathole forests they have disappeared at sites where they were collected in the early 1990s). Erasmus et al. (2002) projected the extinction of this species due to a 2° C increase in temperature from climate change.
Continuing decline in area, extent and/or quality of habitat: Yes. Based on the results of Berliner and Desmet (2007), it can be deduced that 2% of the natural area of the Eastern Cape was lost during the period 2007 and 2015 at the rate of 0.24% per year. Based on the analysis of the proposed mining applications and municipal spatial planning products, it is estimated that over 20,000 km2 (12% of the total area) may be lost between 2015 and 2045.
Conservation
Giant Golden Moles are possibly present in a few small nature reserves within their range, and state-managed forest reserves, but conservation efficacy in such areas appears to be dubious. The indigenous forests in the Kologha Kabusi, Pirie and Isidenge region of the Amathole Mountains contain most of the remaining populations. Enhanced management and improved protection of these indigenous forests is necessary to prevent habitat degradation, especially by cattle. Additionally, field surveys are needed to establish the conservation status and threats faced by subpopulations at the 17 localities this species is known to have occurred at in the past. Recent surveys (2021 and 2022) conducted in these indigenous forests, involving field trapping and eDNA sampling, failed to detect this species, indicating that it may already be locally extinct at these localities.
Reserve expansion through land acquisition and stewardship are mechanisms that are employed to protect threatened ecosystems in the Eastern Cape and these mechanisms will be maximised in an effort to minimise habitat loss. To achieve this, plans are in place to double the protected area to over 1 million hectares over the next decade (K. Mangwale pers. comm. 2015). However, it is important to note that reserve expansion is only likely to benefit this species if the specific forests containing this species are expanded.
Recommendations for land managers and practitioners:
- Field surveys are needed to establish the conservation status and threats faced by populations at the 17 localities this species is known to have occurred at in the past.
- A systematic monitoring scheme should be established to determine changes in conservation status and habitat status of the preferred forest patches with soft soils, well-developed undergrowth and deep leaf-litter layers.
Research priorities:
- Field surveys to determine population size, niche requirements, distribution and trends.
- Studies to quantify the threat levels to the species.
- Studies to generate evidence for the effectiveness of improving protected area security and connectivity.
Encouraged citizen actions:
- Look for distinctive signs of activity (surface runways through leaf litter leading to open burrow holes) and report these to local conservation authorities if found – particularly in the coastal scarp forests where rapid urbanisation is degrading available habitat.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
- Deposit any dead specimens found in a state or provincial museum, together with information on the date and site where found.
Bibliography
Afrotheria Specialist Group. 2014. Specialist Group website. Available at: http://www.afrotheria.net/index.php.
Berliner, D. and Desmet, P. 2007. Eastern Cape Biodiversity Conservation Plan: Technical Report. Department of Water Affairs and Forestry, Pretoria, South Africa.
Bronner, G.N. 1992. Notes on the early post-natal development of a giant golden mole Chrysospalax trevelyani (Güinther, 1875) born in captivity (Mammalia: Insectivora; Chrysochloridae). Koedoe 35: 57-58.
Bronner, G.N. 2013. Chrysospalax trevelyani. In: J. Kingdon, D. Happold, T. Butynski, M. Hoffmann, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria, pp. 246-2480. Bloomsbury Publishing, London.
Bronner, G.N. and Jenkins, P.D. 2005. Order Afrosoricida. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 70-81. The Johns Hopkins University Press, Baltimore, MD, USA.
Bronner, G.N., Mynhardt, S., Bennett, N.C., Cohen, L., Crumpton, N., Hofreiter, M., Arnold, P. and Asher, R.J., 2024. Phylogenetic history of golden moles and tenrecs (Mammalia: Afrotheria). Zoological Journal of the Linnean Society, 201(1), pp.184-213.
Castley, J.G., Kerley, G.I.H. and Simelane, T.S. 2000. Forest vertebrate diversity: status, threats and priorities in the Eastern Cape. Proceedings of the Natural Forests and Savana Woodlands Symposium II.
Ellerman, J.R., Morrison-Scott, T.C.S. and Hayman, R.W. 1953. Southern African Mammals 1758 to 1951: A Reclassification. British Museum (Natural History), London, UK.
Erasmus, B.F.N., van Jaarsveld, A.S., Chown, S.L., Kshatriya, M. and Wessels, K.J. 2002. Vulnerability of South African animal taxa to climate change. Global Change Biology 8: 679-693.
Hickman, G.C. 1986. Swimming of Amblysomus hottentotus (Insectivora: Chrysochloridae) with notes on Chrysospalax and Eremitalpa. Cimbebasia A 8: 55-61.
IUCN. 2015. The IUCN Red List of Threatened Species. Version 2015.2. Available at: www.iucnredlist.org. (Accessed: 23 June 2015).
Maddock, A.H. 1986. An unknown and rare mammal endemic to southern Africa. Cimbebasia Series 8: 88–90.
Maddock, A.H. and Hickman, G.C. 1985. A preliminary report on locomotory activity in wild and captive Chrysospalax trevelyani (Mammalia: Chrysochloridae). South African Journal of Zoology 20: 271-273.
Poduschka, W. 1980. Notes on the giant golden mole Chrysospalax trevelyani Günther, 1875 (Mammalia: Insectivora) and its survival chances. Zeitschrift für Säugtierkunde 45: 193-206.
