
Geoffroy’s Horseshoe Bat
Rhinolophus acrotis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Rhinolophus acrotis – von Heuglin, 1861
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – RHINOLOPHIDAE – Rhinolophus – acrotis
Common Names: Geoffroy’s Horseshoe Bat, Cretzschmar’s Horseshoe Bat, Arabian Horseshoe Bat (English), Geoffroy se Saalneusvlermuis, Geoffroy-saalneusvlermuis (Afrikaans)
Synonyms: Rhinolophus clivosus K. Andersen, 1905; acrotis Heuglin 1861.
Taxonomic Note:
Further taxonomic research is needed into Rhinolophus acrotis. Variability in mitochondrial and nuclear DNA, bacular morphology, pelage colour, habitat preference, echolocation calls, and reproduction strongly suggests R. acrotis as a southern and eastern African species (distinct from R. clivosus which is restricted the Middle East and North Africa) which comprises multiple species (Benda & Vallo 2012; Stoffberg et al. 2012; Dool et al. 2016; Demos et al. 2019, Taylor et al. 2024; Uvizl et al. 2024). Stoffberg et al. (2012) provided genetic evidence for five distinct groups within South Africa corresponding to a Western Cape clade, Knysna region clade, Northern Cape clade, a predominantly KwaZulu-Natal/Mpumalanga mixed group and a Mpumalanga/Limpopo clade (TStoffberg et al. 2012; Taylor et al. 2024). A useful phenotypic character for separating R. acrotis and R. darlingi from all other southern African Rhinolophus species is the absence of the minute first upper premolar in the toothrow; and R. acrotis is larger than R. darlingi (Monadjem et al. 2020).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P1., Lotter, C.2, Balona, J.3 & da Silva, J.M.4
Reviewers: Howard, A1., Bastian, A.5
Institutions: 1University of the Free State, 2Inkululeko Wildlife Services (Pty) Ltd, 3Gauteng and Northern Regions Bat Interest Group, 4South African National Biodiversity Institute, 5University of KwaZulu-Natal.
Previous Assessors and Reviewers: Jacobs, D., Cohen, L., Richards, L.R., Monadjem, A., Schoeman, C., MacEwan, K., Sethusa, T. & Taylor, P.
Previous Contributors: Child, M.F. & Raimondo, D.
Assessment Rationale
This species is listed as Least Concern in view of its wide distribution (estimated extent of occurrence in the assessment region is 1,196,561 km2), and local abundance (colonies can comprise thousands of individuals), and it being recorded from many protected areas in the assessment region. However, taxonomic resolution is required as genetic evidence suggests that five distinct clades exist within South Africa, largely corresponding to different biomes, which should therefore be considered as evolutionarily significant units in conservation planning. If cryptic species are described, a -reassessment will be necessary since distinct units may be facing unique threats. Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to established colonies of this species in old mine tunnels.
Regional population effects: While habitats are largely connected across regions, wing-loading is low (Jacobs et al. 2007), so dispersal effects are presumed to be limited, and thus rescue effects are uncertain. This is compounded by taxonomic uncertainty (Stoffberg et al. 2012). For example, specimens of this species from Namibia have been reclassified as R. damarensis, thus R. acrotis is no longer known to occur in Namibia (Monadjem et al. 2020; Jacobs et al. 2013).
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citaiton: Taylor P, Lötter C, Balona J & da Silva JM. 2025. A conservation assessment of Rhinolophus acrotis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
R.acrotis has a disjunct distribution from Ethiopia and Eritrea through all East African countries to Malawi in the south; in southern Africa, it is present in Mozambique and Zambia in the north, ranging southwards into South Africa. While previously thought to occur in Namibia and Angola (for example, Skinner & Chimimba 2005), specimens from these regions have now been reclassified as a new species called R. damarensis (Stoffberg 2007; Monadjem et al. 2020; Jacobs et al. 2013). It is also absent from Botswana (Monadjem et al. 2020).
In the assessment region, the species is widespread in the eastern regions, recorded from Lesotho, Eswatini and all provinces in South Africa, absent only from parts of the arid interior (Monadjem et al. 2020). However, if R. acrotis comprises a complex of several species, then Stoffberg et al. (2012) or Taylor et al. (2024) should be consulted to provide the composite species distributions in South Africa where five genetically distinct groups are largely linked to different biomes, similar to those reported for Miniopterus natalensis (Miller-Butterworth et al. 2003). Roberts (1951) recognised southern African R. acrotis as an endemic southern African species of R. geoffroyi where four of the lineages identified by Stoffberg et al. (2012) correspond to the geographical distributions of his proposed R. geoffroyi subspecies: clade 1 (R. g. geoffroyi) in the Cape Floral Kingdom (CFK) that covers the extreme southwestern and southern parts of South Africa with winter rainfall season; clade 3 (R. g. augur) in the arid areas on the central plateau of the western half of the country; clade 4 (R. g. zuluensis) in the eastern mesic parts of South Africa; and clade 5 (R. g. zambesiensis) occurring in the northern parts of South Africa (Stoffberg et al. 2012). An additional lineage, clade 2, may represent a unique taxon that occurs in the Knysna Forest comprising patches of indigenous forest in the southeastern parts of the CFK (Stoffberg et al. 2012). The estimated extent of occurrence for R. acrotis within the assessment region is 1,196,561 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0 m asl (Mondajem et al. 2024)
Elevation Upper Limit (in metres above sea level): 3,869 m asl (Mondajem et al. 2024)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
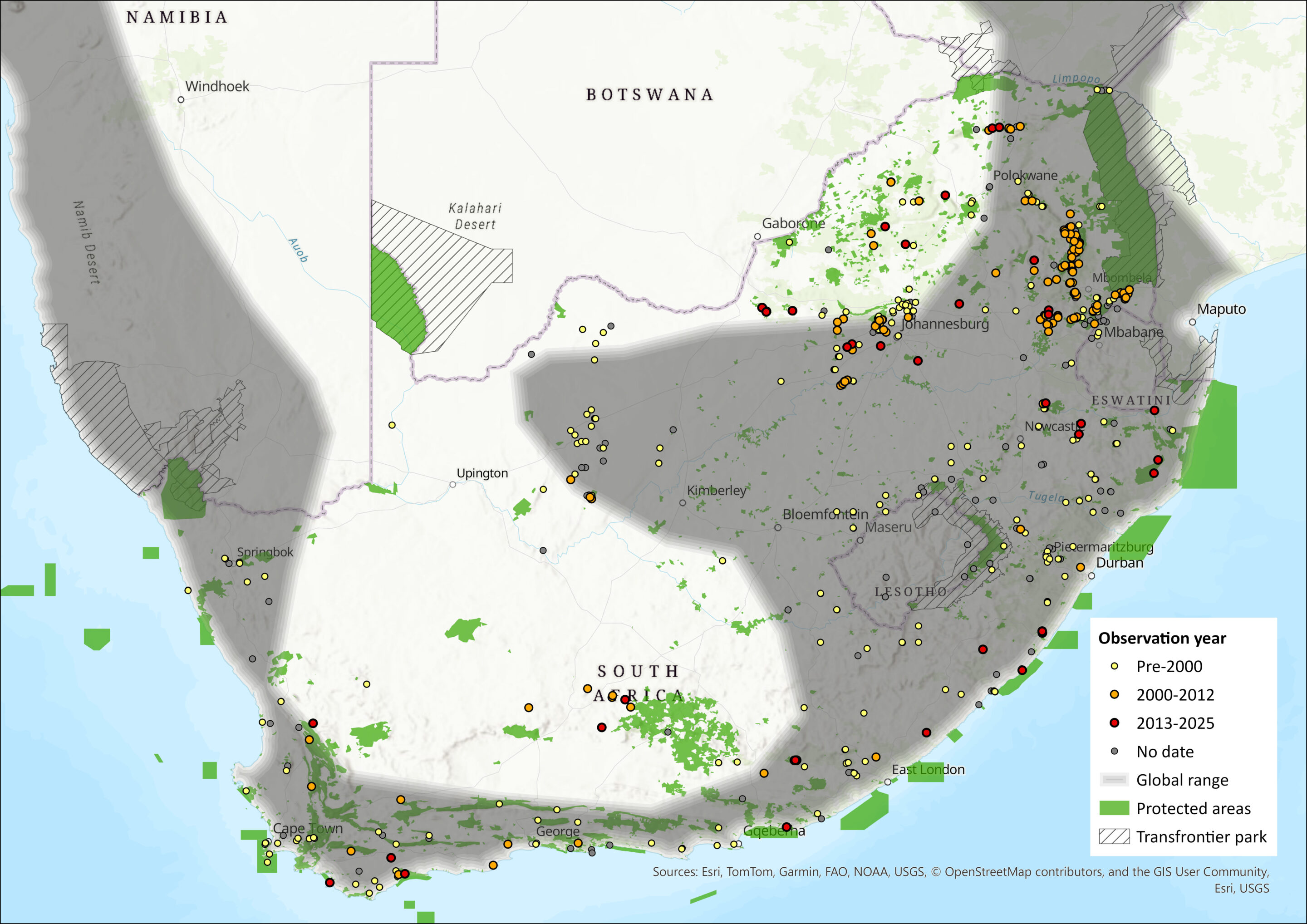 Figure 1. Distribution records for Geoffroy’s Horseshoe Bat (Rhinolophus acrotis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Geoffroy’s Horseshoe Bat (Rhinolophus acrotis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Algeria | Extant | Native | – | – |
| Angola | Extant | Native | – | – |
| Burundi | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Chad | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Djibouti | Extant | Native | – | – |
| Egypt | Extant | Native | – | – |
| Eritrea | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Ethiopia | Extant | Native | – | – |
| Kenya | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| Libya | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Rwanda | Extant | Native | – | – |
| Somalia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Uganda | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Taylor et al. (2024) found that future climate Maxent models did not predict a future reduction by 2070 in the ranges of temperate-montane bat species associated with the higher-elevation Maloti Drakensberg (including R. acrotis); instead, their ranges appear remarkably stable over time. Taylor et al. (20204) proposed that this stability may be at least partly due to the buffering effect of topographic ruggedness and elevational and habitat heterogeneity in high mountains, providing multiple potential refugia in the face of increasingly adverse climate conditions.
Population Information
While abundance is uncertain throughout most of its range, it is generally common in southern Africa (besides Zimbabwe) (Taylor 2000) and can be locally abundant in certain caves (Herselman & Norton 1985), such as De Hoop Guano Cave, where it may form colonies of several thousand individuals (McDonald et al. 1990). Similarly, in Eswatini, three separate populations contained over a thousand individuals (Monadjem 1998; ACR 2015). It is well represented in museums, with over 380 specimens examined in Monadjem et al. (2020).
Current population trend: Stable
Continuing decline in mature individuals: No
Extreme fluctuations in the number of subpopulations: Unknown
Continuing decline in number of subpopulations: Unknown
All individuals in one subpopulation: No
Number of mature individuals in largest subpopulation: Unknown
Number of Subpopulations: Unknown
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Uvizl et al. (2024) showed that R. acrotis (formerly R. clivosus) is broadly distributed throughout southern, central eastern, and northeastern Africa and Taylor et al. (2024) revealed the presence of three distinct, latitudinally structured mtDNA lineages within the species, one of which is isolated to South Africa. Within South Africa, additional substructure was proposed by Stoffberg et al. (2012); however not all genetic groups were geographically differentiated (particularly within KwaZulu-Natal, Limpopo and Eswatini), or genetic support was minimal (between Western Cape and Eastern Cape clades), despite apparent differences in echolocation and wing morphology data. To gain a better perspective of the genetic diversity and structure within this species, a population genomic study utilizing fine-scale nuclear markers and incorporating more comprehensive sampling throughout the assessment region is highly recommended. Until then, it is expected that at least 4 distinct subpopulations exist corresponding to the Western Cape animals (group 1; Stoffberg et al. 2012). Eastern Cape (group 2; Stoffberg et al. 2012); north-western group (group 3; Stoffberg et al. 2012), and a northeastern group (groups 4 and 5; Stoffberg et al. 2012).
Considering the species is considered common and locally abundant, each of its subpopulations are expected to have an Ne >500. However, this will need to be confirmed using molecular techniques.
Habitats and ecology
Rhinolophus acrotis has been recorded from a wide variety of habitats, ranging from savannah woodland, Mediterranean-type shrubland, dry (and possibly moist) savannah, riparian forest, open grasslands and semi-desert to even more arid environments. However, these habitat types may correspond to cryptic species (Stoffberg et al. 2012). It is generally a temperate species, absent from hot, low-lying areas and associated with mountainous areas (such as the Maluti-Drakensberg) in its northerly range (Taylor 2000 p. 200; Monadjem et al. 2020). Its range is most likely dependent on the availability of caves or similar day roosts. Roosting has been recorded in caves, rock crevices, disused mines, hollow baobabs (Adansonia spp.) and various rural and urban buildings. It can also use modified habitats for foraging, such as artificial wetlands (Sirami et al. 2013). It travels up to 10 km between caves (Rautenbach 1982) and undergoes prolonged periods of hibernation during winter in South Africa (R. T. F. Bernard unpubl. data). It is a clutter forager, feeding mainly on Lepidoptera and Coleoptera (Monadjem et al. 2020). It establishes “feeding stations” at night, hanging from branches of trees or from verandas of houses to eat its insect prey, discarding the harder parts in a pile underneath the night roost (Monadjem et al. 2020).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011). Ensuring a healthy population of insectivorous bats can thus result in a decrease in the need and/or use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland | – | Suitable | – |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 2.2. Savanna -> Savanna – Moist | – | Unknown | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 7.1. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Caves | – | Suitable | Yes |
| 7.2. Caves and Subterranean Habitats (non-aquatic) -> Caves and Subterranean Habitats (non-aquatic) – Other Subterranean Habitats | – | Suitable | Yes |
| 8.1. Desert -> Desert – Hot | – | Marginal | – |
| 14.4. Artificial/Terrestrial -> Artificial/Terrestrial – Rural Gardens | – | Marginal | – |
| 14.5. Artificial/Terrestrial -> Artificial/Terrestrial – Urban Areas | – | Marginal | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Forearm mean = 5.41 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 5.31 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Unknown (likely to be limited).
Congregatory: Colonies of several thousand individuals are known (Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
This species is not known to be traded or utilised in any form, although a recent paper indicated that this species and other bats have been imported into the USA for the trade in the bat taxidermy (Tarango et al. 2025).
National Commercial Value: No
International Commercial Value: No
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Implementation of the Mine Closure Strategy of the South African Department of Mineral Resources and Energy (Government Notice 446, 21 May 2021) poses a serious threat to established colonies of this species in old mine tunnels. The rapidly growing number of renewable energy developments across the assessment region could also disturb roosts of this species, but due to the clutter foraging habits of this species, it is regarded as having a “low” risk of fatality from wind turbines (MacEwan et al. 2020). Some colonies may also be threatened by disturbance to their roosting sites in caves, or indirect poisoning resulting from agricultural use of insecticides, pesticides and similar chemicals (ACR 2015). If cryptic species are revealed (Stoffberg et al. 2012), taxon-specific threats will have to be more accurately identified and quantified.
Conservation
R.acrotis occurs in many protected areas within the assessment region including Kruger National Park, iSimangaliso Wetland Park, Garden Route National Park, De Hoop Nature Reserve, Agulhas National Park, Table Mountain National Park and West Coast National Park.
The South Africa Department of Mineral Resources and Energy should engage with the South African Bat Assessment Association with regards to future closure of old mine tunnels in the country, so as to minimise the impact of this on cavity-roosting bat species such as R. acrotis. Renewable energy developments must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (for buffer recommendations see MacEwan et al. 2020 or later). Monitoring of population trends in response to the threat of mine closure or expansion, and renewable energy development, is recommended. Bats of the genus Rhinolophus are generally susceptible to indirect poisoning through the use of insecticides and thus there is a need to evaluate the impact of this threat on populations and to investigate alternative methods of insect control (D. Kock pers. comm. 2004). The strong concordance between genetic and ecological diversity suggests that the five distinct clades are adapted to their respective habitats and should be considered as separate units in conservation planning (Stoffberg et al. 2012).
Recommendations for land managers and practitioners:
- The South Africa Department of Mineral Resources and Energy should engage with the South African bat interest groups, bat conservation groups, cave hobbyists, provincial and national conservation authorities with regards to future closure of old mine tunnels in the country, so as to minimise the impact on cavity-roosting bat species such as R. acrotis.
- Renewable energy development must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species (see MacEwan et al. 2020).
- Identify and protect important roost sites for this species according to conservation units identified by Stoffberg et al. (2012).
- Reduce pesticide use in agricultural landscapes to ensure adequate food availability.
Research priorities:
- Taxonomic resolution is required by incorporating multi-locus DNA sequence data, as well as morphological data, into future research (Stoffberg et al. 2012).
- Monitoring of population trends in response to the threat of mine closure or expansion, and renewable energy development, is recommended.
Encouraged citizen actions:
- Minimise disturbance at caves when visiting, and in particular, avoid hibernation and breeding periods.
- Citizens can report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP).
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Benda, P.and Vallo, P. 2012. New look on the geographical variation in Rhinolophus acrotis with description of a new horseshoe bat species from Cyrenaica, Libya. Vespertilio 16: 69-96.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Demos, TC, Webala PW, Goodman SM, Kerbis Peterhans JC, Bartonjo M, and Patterson BD. 2019. Molecular phylogenetics of the African horseshoe bats (Chiroptera: Rhinolophidae): expanded geographic and taxonomic sampling of the Afrotropics. BMC Evolutionary Biology 19:1-14.
Dool, S.E., Puechmaille, S.J., Foley, N.M., Allegrini, B., Bastian, A., Mutumi, G.L., Maluleke, T.G., Odendaal, L.J., Teeling, E.C. and Jacobs, D.S. 2016. Nuclear introns outperform mitochondrial DNA in inter-specific phylogenetic reconstruction: Lessons from horseshoe bats (Rhinolophidae: Chiroptera). Molecular Phylogenetics and Evolution 97:196-212.
Herselman, J.C. and Norton, P.M. 1985. The distribution and status of bats (Mammalia: Chiroptera) in the Cape Province. Annals of the Cape Provincial Museums (Natural History) 16: 73–126.
Jacobs, D.S., Babiker, H., Bastian, A., Kearney, T., van Eeden, R. and Bishop, J.M. 2013. Phenotypic convergence in genetically distinct lineages of a Rhinolophus species complex (Mammalia, Chiroptera). PloS one 8: e82614.
Jacobs, D.S., Barclay, R.M. and Walker, M.H. 2007. The allometry of echolocation call frequencies of insectivorous bats: why do some species deviate from the pattern? Oecologia 152: 583–594.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association. South Africa.
McDonald, J.T., Rautenbach, I.L. and Nel, J.A.J. 1990. Roosting requirements and behaviour of five bat species at De Hoop Guano Cave, southern Cape Province of South Africa. South African Journal of Wildlife Research 20: 157–161.
Miller-Butterworth, C.M., Jacobs, D.S. and Harley, E.H. 2003. Strong population substructure is correlated with morphology and ecology in a migratory bat. Nature 424: 187–191.
Monadjem, A. 1998. The mammals of Swaziland. Conservation Trust of Swaziland and Big Games Parks, Mbabane, Swaziland.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Monadjem, A., Montauban, C., Webala, P.W., Laverty, T.M., Bakwo-Fils, E.M., Torrent, L., Tanshi, I., Kane, A., Rutrough, A.L., Waldien, D.L. and Taylor, P.J. 2024. African bat database: curated data of occurrences, distributions and conservation metrics for sub-Saharan bats. Scientific Data, 11(1), p.1309.
Rautenbach, I.L. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Roberts, A. 1951. The Mammals of South Africa. The Trustees of the Mammals of South Africa. Central News Agency, Johannesburg, South Africa.
Sirami, C., Jacobs, D.S. and Cumming, G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Skinner, J.D. and Chimimba, C.T. 2005. The Mammals of the Southern African Subregion. Cambridge University Press, Cambridge, UK.
Stoffberg, S. 2007. Molecular phylogenetics and the evolution of high frequency echolocation in horseshoe bats (Genus Rhinolophus). Ph.D. Thesis. University of Cape Town.
Stoffberg, S., Schoeman, M.C .and Matthee, C.A. 2012. Correlated genetic and ecological diversification in a widespread southern African horseshoe bat. Public Library of Science One 7(2): 1-11.
Tarango, M., Kolby, J.E., Goodman, O.L., Anderson, C.J., Tinsman, J., Kirkey, J., Liew, A., Jones, M., Firth, C. and Reaser, J.K. 2025. Going batty: US bat imports raise concerns for species conservation and human health. One Health, 20, p.100999.
Taylor, P.J. 2000. Bats of Southern Africa: Guide to Biology, Identification, and Conservation. University of Natal Press, Pietermaritzburg, South Africa.
Taylor, P.J., Kearney, T.C., Clark, V.R., Howard, A., Mdluli, M.V., Markotter, W., Geldenhuys, M., Richards, L.R., Rakotoarivelo, A.R., Watson, J., Balona, J., Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate‐driven diversification and a buffer against future climate change in bats. Global Change Biology, 30(6), p.e17344.
Uvizl, M., Kotyková Varadínová, Z. and Benda, P. 2024. Phylogenetic relationships among horseshoe bats within the Rhinolophus ferrumequinum group (Mammalia, Chiroptera). Zoologica Scripta, 53(3), pp.249-266.
