Gemsbok
Oryx gazella

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Oryx gazella – (Linnaeus, 1758)
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – BOVIDAE – Oryx – gazella
Common Names: Gemsbok (English, Afrikaans), Gemsbuck, Oryx (English), Inkukhama, iKukhama (Ndebele), None (Sepedi), Kukama (Setswana), Inyamatane (Swati), Mhala (Tsonga), Noni (Venda), Inkukhama (Xhosa), Sudafrikanischer Spiessbock (German)
Synonyms: Capra gazella Linnaeus, 1758; Oryx gazella ssp. gazella (Linnaeus, 1758)
Taxonomic Note:
Previously regarded as a single species, Gemsbok (O. gazella) and Beisa Oryx (O. beisa), from East Africa, are now considered distinct species based on taxonomic results revealing high (40%) genetic divergence between haplotypes (Osmers et al. 2012), morphology and geographic distribution (Grubb 2005). Osmers et al. (2012) do, however, recommend further molecular investigations into the genetics of the species to confirm their results. Currently, no subspecies have been identified within the assessment region.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Patel, T.1 & da Silva, J.2
Reviewer: Mallon, D.3
Institutions: 1Endangered Wildlife Trust, 2South African National Biodiversity Institute, 3IUCN SSC Antelope Specialist Group
Previous Assessors: Relton, C., Selier, J. & Strauss, M.
Previous Reviewer: Child, M.F.
Assessment Rationale
Listed as Least Concern as Gemsbok are numerous and widespread across the arid and semi-arid regions within the assessment region, and population numbers increased over three generations (1992–2015) on formally protected areas, often by over 100%. The future of this species in South Africa is regarded as secure, as they continue to occur in large numbers, are resilient to aridity, and survive successfully without permanent sources of drinking water when free-ranging. The Kgalagadi Transfrontier Park contains the largest subpopulation of Gemsbok, with an estimated size of 5,548 individuals (2019 count). Overall, approximately 9,570 individuals exist in other formally protected areas, and due to their high economic value (particularly within the hunting industry), there were around 37,610 individuals on private land (based on the 2016 assessment). There were at least 55,376 individuals in South Africa during the last assessment in 2016. Recent data were not robust enough to update these estimates; however, any change in population number will not affect the status of the species at present. No direct conservation interventions are currently required, and this species should continue to be a key component in the wildlife economy. The development of a translocation and metapopulation strategy is encouraged to sustain the genetic resilience of the population and to prevent extensive introduction outside its natural range.
Regional population effects: Within the assessment region, migration does take place within the Kgalagadi Transfrontier Park, which is only fenced along its western and southern boundaries, allowing the unrestricted movement of animals between South Africa and southern and central Botswana. Variability in the localised environmental conditions experienced in these areas largely determines the nature and direction of movement across these international borders. Although the Gemsbok subpopulation in Mapungubwe National Park is small, there may be some movement into and out of Botswana. The remaining population is, however, typically restricted to private ranches and reserves, provincial and national protected areas. A degree of genetic dispersal occurs through the translocation of individuals between these protected areas via game sales. No rescue effects are necessary.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Patel T & da Silva JM. 2025. A conservation assessment of Oryx gazella. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
Historically, Gemsbok occurred extensively in the arid and semi-arid savannah territories of the Kalahari and Karoo regions of southern Africa. They ranged expansively in Namibia aside from the Zambezi Region (previously known as the Caprivi Strip) and surroundings, throughout the arid and semi-arid regions of Botswana, extending marginally into western Zimbabwe and southwest Angola (East 1999). During the 19th and 20th centuries, anthropogenic modification and fragmentation of their habitat resulted in a significant reduction in their range. More recently, however, widespread reintroductions have occurred onto both private and formally protected reserves. Currently, their distribution is widespread, although patchy, across the southwest of southern Africa. A healthy population occurs in the Iona National Park of Angola (B.A. Bennett. pers. comm. 2015), a traditional stronghold for the species (East 1999). Gemsbok have also been introduced onto private game ranches in Zimbabwe (East 1999).
Within the assessment region, Gemsbok historically ranged across the arid regions of the Northern Cape, through Namaqualand, and extended partially into the Karoo (Western Cape), Eastern Cape and Free State; the most eastern limit of their range being the confluence of the Orange and Vaal Rivers and small areas of the North West Province. There may be free-roaming herds in North West (outside fenced areas) but they may also be escapees (Power 2014). Gemsbok were historically permitted in Gauteng and are now mainly held on exemption farms, but no further imports are allowed (C.Whittington-Jones pers. comm. 2016). Their populations have recently increased within their historic range as a result of reintroductions onto many formally protected and private properties (Skinner & Chimimba 2005; Power 2014). Additionally, as a consequence of their commercial value, they have now been introduced onto a number of privately owned game ranches situated outside of their natural distribution, such as the eastern parts of Limpopo Province (Knight 1999).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
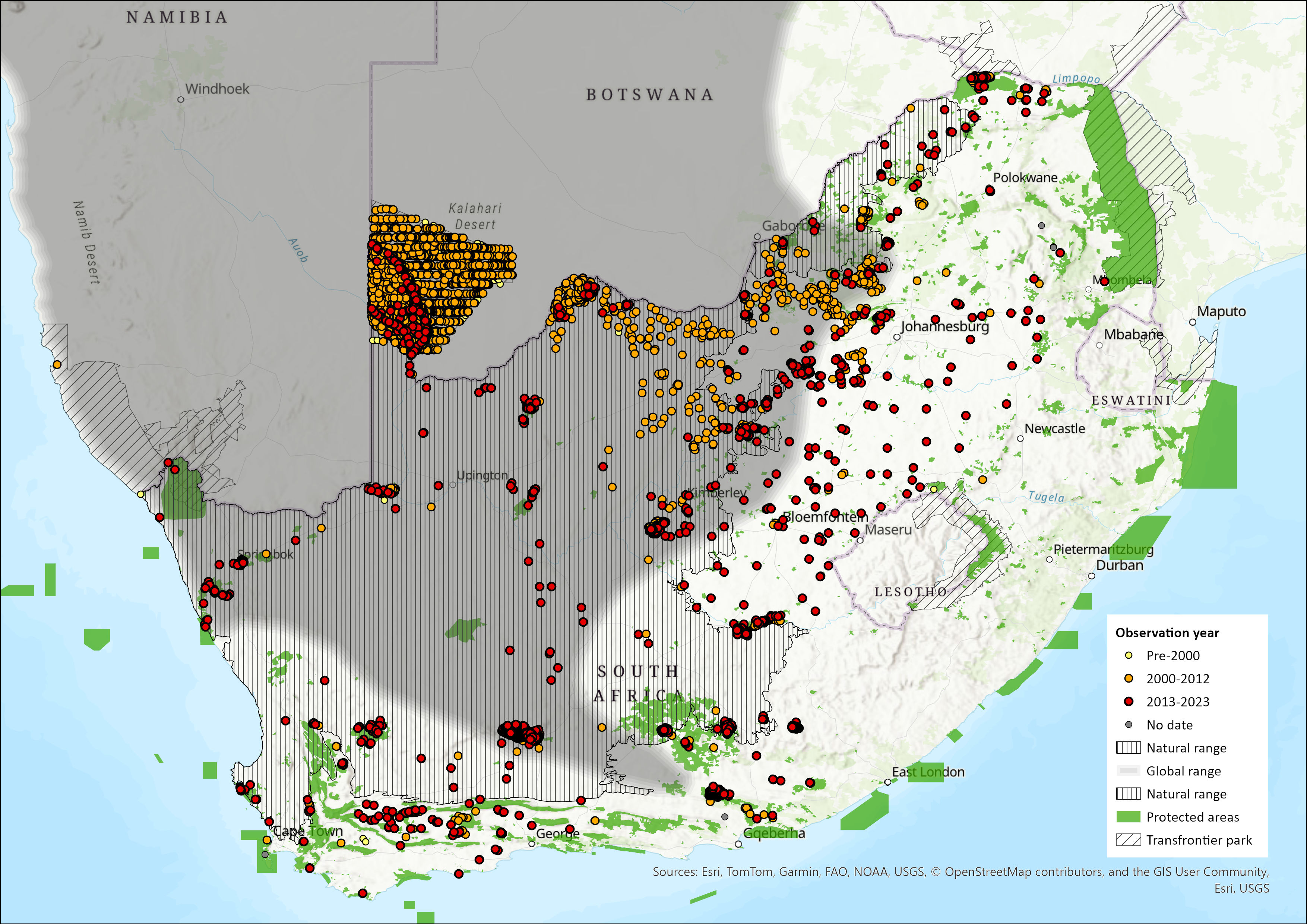
Map
Figure 1. Distribution records for Gemsbok (Oryx gazella) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Possibly Extinct | Native | – | – |
| Botswana | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa -> Eastern Cape Province | Extant | Native | – | – |
| South Africa -> Free State | Extant | Native | – | – |
| South Africa -> Gauteng | Extant | Native | – | – |
| South Africa -> KwaZulu-Natal | Extant | Native | – | – |
| South Africa -> Limpopo Province | Extant | Native | – | – |
| South Africa -> Mpumalanga | Extant | Native | – | – |
| South Africa -> North-West Province | Extant | Native | – | – |
| South Africa -> Northern Cape Province | Extant | Native | – | – |
| South Africa -> Western Cape | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Arid southern African savannahs are particularly vulnerable to climate change and are expected to become hotter and drier with increasingly unpredictable rainfall (Hulme et al. 2001; Hannah et al. 2002; Meadows 2006). Amplified temperatures in association with a decline in the frequency of rainfall could radically constrict key habitats, thus threatening the habitat availability and forage resources of this species.
In the Kalahari, climate change is resulting in higher temperatures and reduced rainfall (New et al. 2006). These hotter and drier conditions can ultimately result in behaviour changes, such as nocturnal feeding, for ungulates like Gemsbok in this region, due to temperature induced stress (Cain et al. 2006). Further research is needed on the effect of climate change on Gemsbok populations in other regions.
Population information
Gemsbok are widespread and common across the dry regions of southern Africa. Naturally, Gemsbok population distribution and birth rates are known to vary with annual rainfall (Mills & Retief 1984). East (1999) estimated 330,000 Gemsbok globally and applied an average correction figure of 1.3 to allow for undercounting bias in aerial surveys to reach a total estimate of 373,000. The same figure was cited in the 2008 IUCN Red List assessment,in Knight (2013) and in the current IUCN assessment (IUCN SSC Antelope Specialist Group 2020). The generation length of Gemsbok has been estimated as 7.8 years (Pacifici et al. 2013), which yields a 23.6 year three- generation window. Within the assessment region, the population is thriving and subpopulations are stable or increasing on protected areas, conservancies and private game ranches. For example, over three generations on Camdeboo National Park, Eastern Cape Province (1997–2020), the subpopulation has increased from 70 to 174 individuals; from 339 to 843 individuals (1998-2021) in Karoo National Park; and from 15 (in 2003) to 183 (2022) individuals in Mountain Zebra National Park (SANParks data). Similar increases on protected areas have occurred in the Northern Cape over three generations for example from 78 individuals to 387 in Goegap Nature Reserve between 1990 and 2013; and from 19 individuals in 2002 to 97 in 2013 at Doornkloof Nature Reserve. The subpopulations on both Augrabies Falls National Park (336 individuals; 2012 count) and Mokala National Park (1,544 individuals; 2016 count) are also stable or increasing (C. Bissett unpubl. data). The largest subpopulation exists in Kgalagadi Transfrontier Park, where the subpopulation is estimated to be 5,548 individuals (2019 count: SANParks data). Thus, on formally protected areas alone, there are well over 10,000 mature individuals in the assessment region. We assume trends are similar on private lands. For example, in the North West Province, while there are an estimated 2,121 individuals on formally protected areas, there are c. 17,440 on private lands (2013 estimates). Overall, it estimated to be a total population size of at least 55,376 individuals across the country in the previous assessment. There are no major threats to this species and its numbers on private lands are increasing. Most of the national population is fragmented except the largest subpopulation in the Kgalagadi Transfrontier Park. Recent data were not robust enough to update these national estimates, however, any change in population number will not affect the status of the species at present.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| No | – | Within protected areas the population is increasing. |
Current population trend: Stable
Number of mature individuals in population: Approximately 55,376 individuals (2016 estimate)
Number of mature individuals in largest subpopulation: 5,548 individuals in the Kgalagadi Transfrontier Park (based on 2019 census data).
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Severely fragmented: Yes, aside from the Kgalagadi Transfrontier Park, the dispersal of Gemsbok is largely limited by fences, and is thus dependent on translocation for dispersal. The Kgalagadi Transfrontier Park is only fenced along its western and southern boundaries, allowing the unrestricted movement of animals between the assessment region and southern and central Botswana. Some movement of animals may also occur between the Mapungubwe National Park and Botswana.
Number of Subpopulations: 33 sites
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years:(Not specified)
Population genetics
To date, only two molecular studies have been conducted on Gemsbok – the one whereby Gemsbok was found to be differentiated from Oryx beisa (Osmer et al. 2012) and a near-chromosome-scale genome assembly of the species (Farré et al. 2019). Consequently, the population genetic structure and diversity within and between the various protected areas, conservancies and private game ranches where it occurs is unknown (however Osmer et al. [2012], did detect genetic differentiation between individuals from Namibia and the Northern Cape). In particular, it is unclear whether any locality, regardless of increases in individual numbers, is genetically healthy (i.e., effective population size greater than 500, minimal to no inbreeding) or compromised (e.g., inbred, hybridised, etc). Given that translocations between farms and reserves are known to take place, and that Gemsbok currently exists in highly fragmented and managed entities, it is important to ensure the genetic integrity of each genetically distinct population is maintained, mimicking the natural movement patterns and gene flow of the species. Consequently, a fine-scale molecular study using genome-wide nuclear markers across the species full distributional range is highly recommended.
Based on the currently available information it is expected that Gemsbok within the assessment region exist as a single metapopulation and exceed 10,000 mature individuals. From this the effective population size (Ne) can be inferred by applying a conversion ratio of Ne/Nc of 0.1-0.3. This yields an Ne estimate between 1,000-3,000 individuals, exceeding the threshold of 500 for healthy stable populations.
Habitats and ecology
This species is exceedingly well-adapted to arid conditions, occurring in semi-arid and arid grass, shrub and woodland savannahs of the Kalahari, Karoo and adjacent regions of southern Africa. In addition, this species makes use of sandy and stone plains, alkaline pans and river valleys, and will ascend mountains to frequent salt licks and springs. In the Kgalagadi Transfrontier Park, Gemsbok show a preference for sandy dune areas of red Kalahari soils sparsely carpeted in short annual grasses (Eloff 1959).
Gemsbok are predominantly grazers, but occasionally supplement their diets with browse material, (Cerling et al. 2003), water-rich fruits and underground tubers strewn throughout dune regions (Dieckmann 1980; Knight 1991; Verlinden & Masogo 1997), particularly during adverse conditions (Williamson 1987; Knight 1995a). They also eat underground tubers, Gemsbok Cucumbers (Acanthosicyos naudinianus) and Tsamma Melons (Citrullus lunatus), which have high water concentrations (Knight 1991; Dieckmann 1980). They may use Tsamma Melons even when surface water is available, suggesting that these additional resources contain other valuable supplementary nutrients (Knight 1995a). When surface water is readily available their diet consists of large quantities of fibre-rich roughage, however, where water is less available they become increasingly more selective. In the Kgalagadi Transfrontier Park, Knight (1991) found the diet of Gemsbok to consist of a high biomass of low quality dune grasses such as Eragrostis lehmanniana and Stipagrostis spp. Their narrow muzzle aids in the selection of taller grasses. Depending on ambient temperature, they typically require between 2.4 and 3.9 litres of water per day (Knight 1995a), and will drink water regularly when available. However, they are able obtain the bulk of their required water intake from grasses and alternative forage resources and, thus, are not dependent on permanent sources of drinking water for survival (Knight 1991, 1995a). Gemsbok conserve body water through selective brain cooling (Maloney et al. 2002; Strauss et al. 2016), seasonal changes in activity pattern (M. Boyers pers. comm. 2016) and microclimate selection (M. Boyers pers. comm. 2016; W. Strauss unpubl. data). With a keen sense of smell, Gemsbok have been observed with their noses to the ground, locating underground roots and tubers that they dig up using their front hooves (Williamson 1987).
It is expected that, along with physiological and behavioural adaptations, the use of these moisture-rich resources enable Gemsbok to survive and reproduce successfully without the need for large-scale migrations seen by other large herbivore species in arid regions (for example, Blue Wildebeest, Connochaetes taurinus). During an extreme drought period in the 1980s, populations of Blue Wildebeest, Red Hartebeest (Alcelaphus buselaphus), Eland (Tragelaphus oryx) and Giraffe (Giraffa camelopardalis) experienced substantial population declines in the Kalahari, while resident Gemsbok populations were much less affected (Spinage & Matlhare 1992; Knight 1995b; Thouless 1998).
Typically, Gemsbok will forage during the early mornings and late afternoons, sometimes continuing deep into the night (Knight 1991; C. Relton pers. obs. 2013), when plants contain more moisture. The hottest parts of the day are usually spent ruminating and resting, often under shaded tree islands of Vachellia (previously Acacia) erioloba (C. Relton pers. obs. 2013). Knight (1991) noted that Gemsbok also consumed pods of V. erioloba to supplement their diets. Considered non-migratory ungulates, Gemsbok do not reveal seasonal trends in their movements (Williamson 1987), but move nomadically in pursuit of spatially and temporally variable resources (C.unpubl. data). Exhibiting nomadic movements, Gemsbok home ranges vary significantly depending on the area and resources available. Males are territorial and have stable territories. In Namaqualand, Dieckmann (1980) reported that the territories of males ranged from 4.2–9.8 km², while in the Kgalagadi Transfrontier Park they increased to an average of 26 km². Knight (1991) found the home ranges of females in the Kgalagadi Transfrontier Park to average 1,430 km².
Gemsbok are gregarious, occurring in unstable mixed or nursery herds of usually between 3–30 individuals, or as solitary males. Herds of up to 300 animals have been documented following rainfall events in the Namib Desert (Skinner & Chimimba 2005). Similar to other arid-adapted species experiencing unpredictable environmental conditions, they are opportunistic breeders, and, as young are often present all year round, there appears to be no definite breeding season (Eloff 1959). However, Skinner et al. (1974) noted a peak calving season in August and September in the North West Province. A gestation period of 264 days was recorded by Brand (1963). Long gestation periods are common in arid and semi-arid herbivore species (compared to seasonal breeders in temperate and tropical areas) (Skinner & Van Jaarsveld 1987).
Ecosystem and cultural services: As an iconic species of southern Africa’s arid regions, the Gemsbok is a valuable flagship species, and may attract public support for the conservation of South Africa’s dry, threatened savannah regions, which are particularly vulnerable to the effects of climate change. They are a valuable prey species to large predators, particularly African Lion (Panthera leo) and Spotted Hyaena (Crocuta crocuta), in South Africa’s arid and semi-arid regions (Périquet et al. 2015). They are economically valuable to the hunting industry of South Africa for venison, skins and trophies.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.5. Shrubland -> Shrubland – Subtropical/Tropical Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
| 8.2. Desert -> Desert – Temperate | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
This is a popular species in the live animal trade industry at game auctions, and has a subsistence value as venison and for national recreational biltong, meat and trophy hunters. The Gemsbok is considered a high value – high return animal in the captive breeding, game ranch and hunting industries of South Africa (Bothma 2005), and has substantial international value as a trophy animal. Hunting and subsistence use does not, however, have a damaging effect on the stability of Gemsbok populations. This is largely a result of its recent large-scale introduction and reintroduction onto private lands.
Patterson and Khosa (2005) reported that Gemsbok generate 8.7% of hunting income for South Africa, and was described as one of the most hunted species in both Namibia (Lindsey et al. 2007) and South Africa (Patterson & Khosa 2005). A minimum ranch size of 1,200 ha is recommended for private Gemsbok owners, and the smallest viable population should include at least three males and seven females (Osmers 2012).
| Subsistence: | Rationale: | Local Commercial: | Further detail including information on economic value if available: |
| Yes | Venison, skins and live animal trade in game auctions. | Yes | Venison, trophies and live sales. |
National Commercial Value: Yes
International Commercial Value: Yes
| End Use | Subsistence | National | International | Other (please specify) |
| 1. Food – human | true | true | true | – |
| 15. Sport hunting/specimen collecting | – | true | true | – |
Is there harvest from captive/cultivated sources of this species? Yes
Harvest Trend Comments: Venison, skins, trophies and live sales. Captive breeding for trophies. In some cases breeding for particular colour variants takes place, such as Golden Gemsbok.
Threats
Currently, there are no major threats to the survival of this species within the assessment region. During the 19th and 20th centuries, the population declined significantly as a direct result of habitat loss due to the expansion of urban and agricultural practices in southern Africa. Since then large scale reintroductions and introductions of Gemsbok have occurred both onto private game ranches, and protected areas. Namibia for example, holds the majority of its Gemsbok on private game ranches, and East (1999) noted that the population increased from approximately 55,000 in 1972 to over 164,000 in 1992. By 2006, the Gemsbok population in Namibia was estimated at more than 388,000 (Mendelsohn 2006). Gemsbok have a number of physiological and behavioural adaptations which allow for its successful survival in fragmented, arid environments, even during harsh drought conditions. Their independence from permanent sources of drinking water, are facilitated through their utilisation of water-rich fruit, and underground roots and tubers, microclimate selection, seasonal changes in activity patterns and selective brain cooling. Livestock farming, particularly in the Kalahari, is a minor threat to this species, as a result of expanding competition with livestock and the subsequent habitat loss and degradation from overgrazing and bush encroachment (Verlinden & Masogo 1997; Verlinden 1998; Bergström & Skarpe 1999; Wallgren et al. 2009). Research suggests that, in the region of Kgalagadi, the majority of the most suitable grazing environments is utilised for livestock grazing rather than wildlife (Verlinden 1998; Darkoh 2003).
Although some illegal poaching of this species exists, mostly in Botswana, it is not considered to be a threat for Gemsbok within the assessment region.
Current habitat trend: Stable (Driver et al. 2012). However, habitat quality may be locally declining as a result of expanding livestock farming and climate change.
Conservation
This species is economically important to southern Africa’s wildlife industry, is a valuable trophy species on game farms, and a sustainable source of protein for local communities. Because of this, it is in great demand among game ranchers, and has been extensively introduced into areas outside of its natural distribution. Gemsbok subpopulations have increased substantially across northern South Africa, due to translocations of Namibian animals onto private game farms.
Gemsbok also occur within a number of formally protected areas within the assessment region, where they are important to South Africa’s ecotourism industry. These include the Kgalagadi Transfrontier Park (which contains South Africa’s largest subpopulation), Karoo National Park, Mokala National Park, Augrabies Falls National Park, Molopo Nature Reserve and Madikwe Game Reserve, among others. Population numbers within these reserves are thought to be stable or increasing, and numbers are additionally increasing within the private sector.
No specific conservation interventions are currently required for this species, and wildlife ranching is considered beneficial for Gemsbok conservation in South Africa, and has effectively increased the extent and abundance of this species. Yet, ranch managers should consider the importance of maintaining genetic diversity in isolated subpopulations of Gemsbok, especially on game farms running breeding programmes selecting for particular traits (Osmers 2012) or colour variants, such as the Golden Gemsbok. Genetic variability plays a vital role in continued reproductive success and fitness, which becomes principally important during adverse environmental conditions. Since arid savannahs are particularly vulnerable to enhanced aridity under current climate change predictions, it is vital that managers prevent genetic degradation of populations by inbreeding (see recommendations for land managers and practitioners). Finally, due to its status in the game industry and attractiveness for trophy and venison hunters, it is important to ensure that habitats and water sources are maintained, particularly on small, private properties, where natural movements are restricted by fences. Population trends and genetic diversity should be monitored to prevent inbreeding and the loss of genetic material.
Recommendations for land managers and practitioners:
- Continued development of Gemsbok as a keystone species within the sustainable, wildlife-based rural economy, by incentivising landowners to supply cheap, low-carbon protein to local communities, thus ensuring that the benefits of this species are shared.
- Osmers (2012) suggested private ranch-owners should maintain subpopulation viability and genetic diversity by:
- Knowing the origin of founder population animals, and establishing when and from where additional genetic material should be inserted into the population.
- Starting with a founder population as large as possible to ensure the effective population is sustainable, and threats of inbreeding are kept to a minimum.
- Periodically supplement the population with genetic material from an alternate large source population.
- Add individuals to the population from other large, well-managed source populations every few years in order to enrich genetic diversity, and compensate for alleles lost through mutation or trait selection.
- Monitor phenotypic data such as horn length and carcass weight.
Research priorities:
- Assessing the vulnerability of Gemsbok to climate change. Considering that the climate of the Northern Cape Province, a stronghold of the Gemsbok, is predicted to become unlike anything currently experienced in South Africa (hotter and drier with increased variability in rainfall), the arid-adapted Gemsbok is an ideal model animal in which to investigate behavioural plasticity – including conservation physiology – in the face of anthropogenic climate change. Studies over the medium-term (c. 5 years) relating individual variability in physiological parameters to reproductive output and success (fecundity) could provide valuable insights into the extent to which ungulates are able to cope with the effects of anthropogenic climate change.
- Quantifying the contribution of Gemsbok to the wildlife economy and in creating sustainable social-ecological systems in South Africa.
Encouraged citizen actions:
- Landowners should create conservancies for this species and engage local stakeholders to create sustainable, wildlife-based rural economies.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas.
Bibliography
Bergström R, Skarpe C. 1999. The abundance of large wild herbivores in a semi-arid savanna in relation to seasons, pans and livestock. African Journal of Ecology 37: 12-26.
Bothma J du P. 2005. Extensive wildlife production in South Africa. Wildlife Seminar, Northern Game Farmers’ Organisation. Pretoria, South Africa.
Brand DJ. 1963. Records of mammals bred in the National Zoological Gardens of South Africa during the period 1908 to 1960. Proceedings of the Zoological Society of London 140: 617-659.
Cain, J., Krausman, P., Rosenstock, S., & Turner, J. 2006. Mechanisms of thermoregulation and water balance in desert ungulates. Wildlife Society Bulletin, 34, 570-581.
Cerling TE, Harris JM, Passey BH. 2003. Diets of East African Bovidae based on stable isotope analysis. Journal of Mammalogy 84: 456-470.
Darkoh MBK. 2003. Regional perspectives on agriculture and biodiversity in the drylands of Africa. Journal of Arid Environments 54: 261-279.
Dieckmann RC. 1980. The ecology and breeding history of the gemsbok, Oryx gazella gazella (Linnaeus, 1758), in the Hester Malan Nature Reserve. Ph.D Thesis. University of Pretoria, Pretoria, South Africa.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
East, R. (compiler). 1999. African Antelope Database 1998. IUCN, Gland, Switzerland and Cambridge, UK.
Eloff FC. 1959. Observations on the migration and habits of the Antelopes of the Kalahari Gemsbok Park-Part I. Koedoe 2: 1-29.
Farré M, Kim J, Proskuryakova AA, Zhang Y, Kulemzina AI, Li Q, Zhou Y, Xiong Y, Johnson JL, Perelman PL, Johnson WE, Warren WC, Kukekova AV, Zhang G, O’Brien SJ, Ryder OA, Graphodatsky AS, Ma J, Lewin HA, Larkin DM. 2019. Evolution of gene regulation in ruminants differs between evolutionary breakpoint regions and homologous synteny blocks. Genome Res. 29(4):576-589. doi: 10.1101/gr.239863.118. Epub 2019 Feb 13. PMID: 30760546; PMCID: PMC6442394.
Ferreira S, Gaylard, A, Greaver, C, Hayes, J, Cowell C, Ellis G. 2013. Animal abundances in Parks 2012/2013. Scientific Services, SANParks, Skukuza, South Africa.
Grubb, P. 2005. Artiodactyla. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World. A Taxonomic and Geographic Reference (3rd ed), pp. 637–722. Johns Hopkins University Press, Baltimore, USA.
Hannah, L.; Midgley, T. L.; Bond, W. J.; Bush, M.; Lovett, J. C.; Scott, D.; Woodward, F. I. 2002. Conservation of biodiversity in a changing climate. Conservation Biology 16(1): 264-268.
Hulme, M., Doherty, R., Ngara, T., New, M. and Lister, D. 2001. African climate change: 1900-2100. Climate Research 17: 145-168.
IUCN SSC Antelope Specialist Group. 2020. Oryx gazella (amended version of 2016 assessment). The IUCN Red List of Threatened Species 2020: e.T15573A166485425.
Knight M. 1990. Oryx gazella (Südafrikanische Oryxantilope). In: Mills MGL, Hes L (ed.), Sӓugetiere des südlichen Afrikas, eine illustrierte Enzyklopӓdie, pp. 278. Struik Publishers, Pty Ltd, Kapstadt.
Knight MH. 1991. Ecology of the gemsbok Oryx gazella gazelle (Linnaeus) and blue wildebeest Connochaetes taurinus (Burchell) in southern Kalahari. Ph.D Thesis. University of Pretoria, Pretoria, South Africa.
Knight MH. 1995a. Tsama melons, Citrullus lanatus, a supplementary water supply for wildlife in the southern Kalahari. African Journal of Ecology 33: 71-80.
Knight, M. H. 1995. Drought-related mortality of wildlife in the southern Kalahari and the role of man. African Journal of Ecology 33: 377-394.
Knight, M. 2013. Oryx gazella Gemsbok (Southern Oryx). In: J. Kingdon and M. Hoffmann (eds), The Mammals of Africa. VI. Pigs, Hippopotamuses, Chevrotain, Giraffes, Deer and Bovids, pp. 572-576. Bloomsbury Publishing, London.
Lindsey PA, Roulet PA, Romañach SS. 2007. Economic and conservation significance of the trophy hunting industry in sub-Saharan Africa. Biological Conservation 134: 455-469.
Maloney SK, Fuller A, Mitchell G, Mitchell D. 2002. Brain and arterial blood temperatures of free-ranging oryx (Oryx gazella). Pflügers Archive 443: 437-445.
Meadows ME. 2006. Global Change and Southern Africa. Geographical Research 44: 135-145.
Mendelsohn J. 2006. Farming systems in Namibia. Research & Information Services of Namibia, ABC Press, Cape Town, South Africa.
Mills MG, Retief PF. 1984. The response of ungulates to rainfall along the riverbeds of the southern Kalahari. Koedoe 27: 129-141.
New, M., et al. 2006, Evidence of trends in daily climate extremes over southern and west Africa, J. Geophys. Res., 111,D14102, doi:10.1029/2005JD006289.
Osmers B, Petersen B-S, Hartl GB, Grobler JP, Kotze A, Van Aswegen E, Zachos FE. 2012. Genetic analysis of southern African gemsbok (Oryx gazella) reveals high variability, distinct lineages and strong divergence from the East African Oryx beisa. Mammalian Biology 77: 60-66.
Osmers KB. 2012. Genetic structuring between Gemsbok (Oryx gazella) populations and the impact of the founder effect on isolated populations. M.Sc. Thesis. University of Limpopo, Polokwane, South Africa.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Patterson, C. and Khosa, P. 2005. Background research paper: A status quo study on the professional and recreational hunting industry in South Africa. Trade Record Analysis for Fauna and Flora in Commerce, Pretoria, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Périquet S, Fritz H, Revilla E. 2015. The Lion King and the Hyaena Queen: large carnivore interactions and coexistence. Biological Reviews 90: 1197-1214.
Skinner JD, Van Jaarsveld AS. 1987. Adaptative significance of restricted breeding in Southern African ruminants. South African Journal of Science 83: 657-663.
Skinner JD, Van Zyl JHM, Oates LG. 1974. The effect of season on the breeding cycle of plains antelope of the western Transvaal Highveld. Journal of South African Wildlife Management and Assessment 4: 15-23.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Spinage CA, Matlhare JM. 1992. Is the Kalahari cornucopia fact or fiction? A predictive model. Journal of Applied Ecology 29: 605-610.
Strauss WM, Hetem RS, Mitchell D, Maloney SK, Meyer LC, Fuller A. 2016. Three African antelope species with varying water dependencies exhibit similar selective brain cooling. Journal of Comparative Physiology B 186: 527-540.
Thouless CR. 1998. Large mammals inside and outside protected areas in the Kalahari. Transactions of the Royal Society of South Africa 53: 245-255.
Verlinden A, Masogo R. 1997. Satellite remote sensing of habitat suitability for ungulates and ostrich in the Kalahari of Botswana. Journal of Arid Environments 35: 563-574.
Verlinden A. 1998. Seasonal movement patterns of some ungulates in the Kalahari ecosystem of Botswana between 1990 and 1995. African Journal of Ecology 36: 117-128.
Wallgren M, Skarpe C, Bergström R, Danell K, Bergström A, Jakobsson T, Karlsson K, Strand T. 2009. Influence of land use on the abundance of wildlife and livestock in the Kalahari, Botswana. Journal of arid environments 73: 314-321.
Williamson DT. 1987. Plant underground storage organs as a source of moisture for Kalahari wildlife. African Journal of Ecology 25: 63-64.