
Fraser’s Dolphin
Lagenodelphis hosei

2025 Red list status
Least Concern
Regional Population Trend
Unknown
Change compared
to 2016
No Change
Overview
Lagenodelphis hosei – Fraser, 1956
ANIMALIA – CHORDATA – MAMMALIA – ARTIODACTYLA – DELPHINIDAE – Lagenodelphis – hosei
Common Names: Fraser’s Dolphin, Bornean Dolphin, Sarawak Dolphin, Shortsnout Dolphin, Short-snouted Whitebelly Porpoise, Short-snouted Whitebelly Dolphin, White Porpoise, (English), Fraser se Dolfyn, Fraser-dolfyn (Afrikaans), Dauphin de Fraser (French), Delfín de Borneo (Spanish; Castilian), Lagenodelfino (Italian)
Synonyms: Lagenodelphis hosei Fraser, 1956
Taxonomic Note:
Until 1971 this species was recognised only from a skeleton located in 1895 from Borneo (Findlay et al. 1992). However, it was later sighted at sea off southern Africa, Australia and in the Eastern Pacific (Perrin et al. 1973). The name Lagenodelphis was given to this species by Fraser (1956), because the skull resembles characteristics from both Lagenorhynchus and Delphinus.
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessor: Purdon, J.1,2 & da Silva, J.3
Reviewer: Sathyan, R.4
Institutions: 1TUT Nature Conservation, 2The Whale Unit, University of Pretoria, 3South African National Biodiversity Institute, 4University of the Western Cape
Previous Assessors: Plön, S. & Cockcroft, V.
Previous Reviewer: Child, M.F.
Previous Contributors: Relton, C., Atkins, S., Findlay, K., Meyer, M., Oosthuizen, H. & Elwen, S.
Assessment Rationale
The species is suspected to be widespread and abundant and there have been no reported population declines, or major threats identified that could cause a range-wide decline. Globally, it has been listed as Least Concern and, within the assessment region, it is not a conservation priority and status Least Concern reflects the lack of major threats to the species. The most prominent threat to this species globally may be incidental capture in fishing gear and, although this is not considered a major threat to this species in the assessment region, Fraser’s Dolphins have become entangled in anti-shark nets off South Africa’s east coast. This threat should be monitored.
Regional population effects: Fraser’s Dolphin has a widespread, pantropical distribution, and although its seasonal migration patterns in southern Africa remain inconclusive, no barriers to dispersal have been recognised, thus rescue effects are possible.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citations: Purdon J & da Silva JM. 2025. A conservation assessment of Lagenodelphis hosei. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The distribution of L. hosei is suggested to be pantropical (Robison & Craddock 1983), and is widespread across the Pacific and Atlantic Oceans (Ross 1984), and the species has been documented in the Indian Ocean off South Africa’s east coast (Perrin et al. 1973), in Sri Lanka (Leatherwood & Reeves 1989), Madagascar (Perrin et al. 1994) and the Maldives (Anderson 1996). Although, sightings and strandings records from the east coast of South Africa show a temporal pattern (the majority occurred in summer; Findlay et al. 1992), there is insufficient data to infer the seasonal movements of this species within the assessment region (Ross 1984). Generally, this species appears to be limited to tropical and subtropical regions between 30°N and 30°S (Jefferson & Leatherwood 1994; Dolar 2002) across the three major ocean basins, thus reports from temperate areas may be extralimital records associated with uncommon environmental conditions (Perrin et al. 1994), such as the global El Niño event in 1983–1984.
In South Africa, this species primarily inhabits deep (> 1,000 m) waters off the east coast (north of 34.3°S) beyond the shelf-edge; and possibly has a localized occurrence, which is strongly affiliated with the subtropical Agulhas Current (Findlay et al. 1992). Strandings, bycatch and entanglement (in bather protection nets) records of 27 individuals from South Africa stretch along the coastline from Cape Vidal to Knysna.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
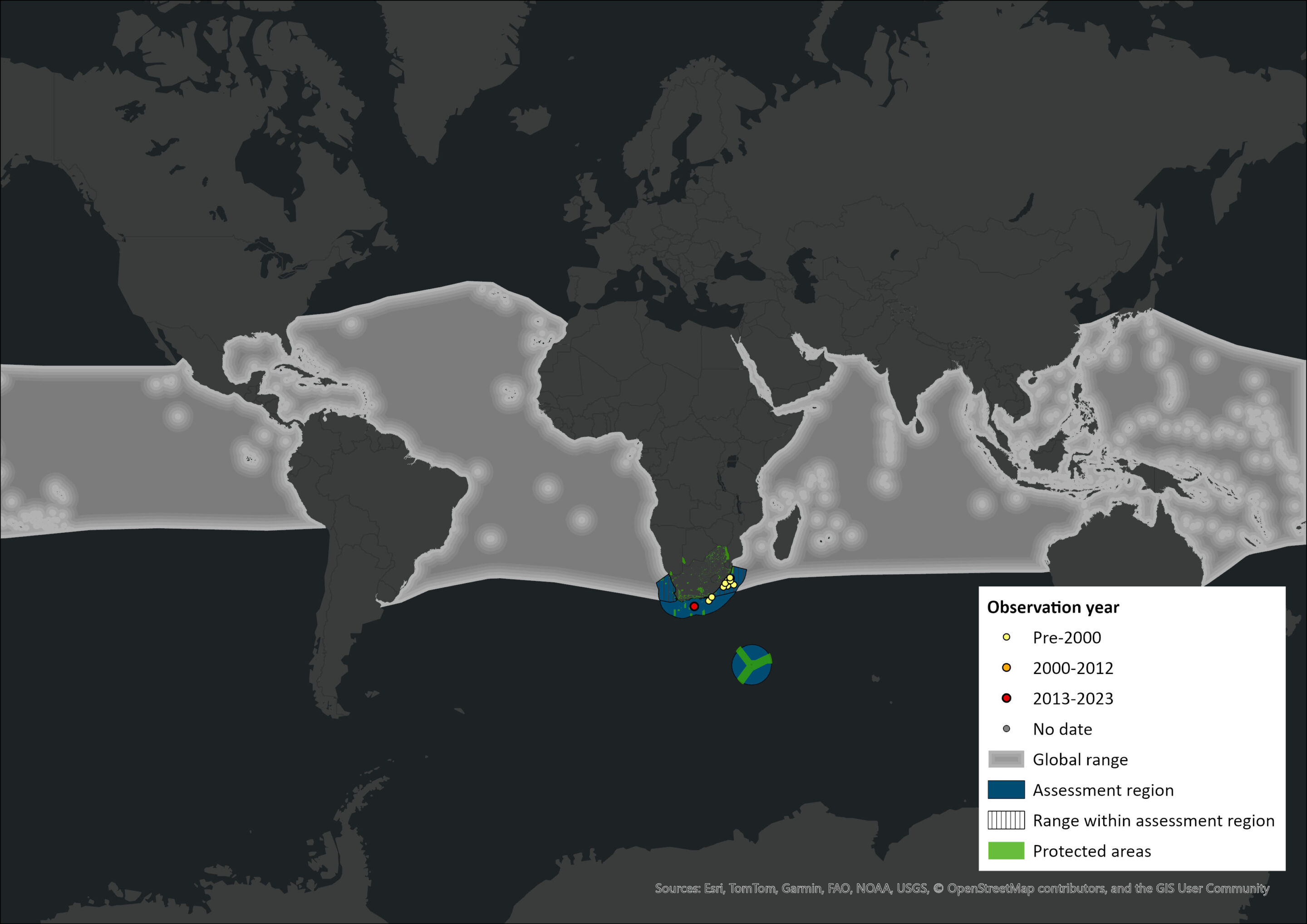 Figure 1. Distribution records for Fraser’s Dolphin (Lagenodelphis hosei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Fraser’s Dolphin (Lagenodelphis hosei) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical, Australasian, Indomalayan, Nearctic, Neotropical, Oceanian
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| American Samoa | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Angola | Extant | Native | – | Resident |
| Anguilla | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Antigua and Barbuda | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Argentina | Extant | Native | – | Resident |
| Aruba | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Australia | Extant | Native | – | Resident |
| Bahamas | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bahrain | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bangladesh | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Barbados | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Belize | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Benin | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bermuda | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Bonaire, Sint Eustatius and Saba | Extant | Native | – | Resident |
| Brazil | Extant | Native | – | Resident |
| Brunei Darussalam | Extant | Native | – | Resident |
| Cabo Verde | Extant | Native | – | Resident |
| Cambodia | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cameroon | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cayman Islands | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| China | Extant | Native | – | Resident |
| Colombia | Extant | Native | – | Resident |
| Comoros | Extant | Native | – | Resident |
| Congo | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Congo, The Democratic Republic of the | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Cook Islands | Extant | Native | – | Resident |
| Costa Rica | Extant | Native | – | Resident |
| Cuba | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Curaçao | Extant | Native | – | Resident |
| Côte d’Ivoire | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Djibouti | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Dominica | Extant | Native | – | Resident |
| Dominican Republic | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Ecuador | Extant | Native | – | Resident |
| Egypt | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| El Salvador | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Equatorial Guinea | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Eritrea | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Ethiopia | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Fiji | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| France | Extant | Vagrant | – | – |
| French Guiana | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| French Polynesia | Extant | Native | – | Resident |
| Gabon | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Gambia | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Ghana | Extant | Native | – | Resident |
| Grenada | Presence Uncertain | Native | – | Seasonal Occurrence Uncertain |
| Guadeloupe | Presence Uncertain | Native | – | Resident |
| Guam | Presence Uncertain | Native | – | Resident |
| Guatemala | Presence Uncertain | Native | – | Resident |
| Guinea | Presence Uncertain | Native | – | Resident |
| Guinea-Bissau | Presence Uncertain | Native | – | Resident |
| Guyana | Presence Uncertain | Native | – | Resident |
| Haiti | Presence Uncertain | Native | – | Resident |
| Honduras | Presence Uncertain | Native | – | Resident |
| Hong Kong | Presence Uncertain | Native | – | Resident |
| India | Presence Uncertain | Native | – | Resident |
| Indonesia | Extant | Native | – | Resident |
| Iran, Islamic Republic of | Presence Uncertain | Native | – | Resident |
| Iraq | Presence Uncertain | Native | – | Resident |
| Israel | Presence Uncertain | Native | – | Resident |
| Jamaica | Presence Uncertain | Native | – | Resident |
| Japan | Extant | Native | – | Resident |
| Jordan | Presence Uncertain | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Kiribati | Extant | Native | – | Resident |
| Korea, Republic of | Presence Uncertain | Native | – | Resident |
| Kuwait | Presence Uncertain | Native | – | Resident |
| Liberia | Presence Uncertain | Native | – | Resident |
| Madagascar | Extant | Native | – | Resident |
| Malaysia | Extant | Native | – | Resident |
| Maldives | Extant | Native | – | Resident |
| Marshall Islands | Presence Uncertain | Native | – | Resident |
| Martinique | Presence Uncertain | Native | – | Resident |
| Mauritania | Presence Uncertain | Native | – | Resident |
| Mauritius | Presence Uncertain | Native | – | Resident |
| Mayotte | Extant | Native | – | Resident |
| Mexico | Presence Uncertain | Native | – | Resident |
| Micronesia, Federated States of | Extant | Native | – | Resident |
| Montserrat | Extant | Native | – | Resident |
| Morocco | Presence Uncertain | Native | – | Resident |
| Mozambique | Presence Uncertain | Native | – | Resident |
| Myanmar | Presence Uncertain | Native | – | Resident |
| Namibia | Presence Uncertain | Native | – | Resident |
| Nauru | Extant | Native | – | Resident |
| New Caledonia | Presence Uncertain | Native | – | Resident |
| Nicaragua | Presence Uncertain | Native | – | Resident |
| Niue | Presence Uncertain | Native | – | Resident |
| Northern Mariana Islands | Presence Uncertain | Native | – | Resident |
| Oman | Extant | Native | – | Resident |
| Pakistan | Presence Uncertain | Native | – | Resident |
| Palau | Extant | Native | – | Resident |
| Panama | Extant | Native | – | Resident |
| Papua New Guinea | Extant | Native | – | Resident |
| Peru | Presence Uncertain | Native | – | Resident |
| Philippines | Extant | Native | – | Resident |
| Pitcairn | Presence Uncertain | Native | – | Resident |
| Portugal | Presence Uncertain | Native | – | Resident |
| Portugal -> Azores | Presence Uncertain | Native | – | Resident |
| Portugal -> Madeira | Presence Uncertain | Native | – | Resident |
| Puerto Rico | Extant | Native | – | Resident |
| Qatar | Presence Uncertain | Native | – | Resident |
| Réunion | Extant | Native | – | Resident |
| Saint Barthélemy | Extant | Native | – | Resident |
| Saint Helena, Ascension and Tristan da Cunha | Presence Uncertain | Native | – | Resident |
| Saint Kitts and Nevis | Presence Uncertain | Native | – | Resident |
| Saint Lucia | Presence Uncertain | Native | – | Resident |
| Saint Martin (French part) | Extant | Native | – | Resident |
| Saint Pierre and Miquelon | Presence Uncertain | Native | – | Resident |
| Saint Vincent and the Grenadines | Extant | Native | – | Resident |
| Samoa | Extant | Native | – | Resident |
| Sao Tome and Principe | Presence Uncertain | Native | – | Resident |
| Saudi Arabia | Presence Uncertain | Native | – | Resident |
| Senegal | Extant | Native | – | Resident |
| Seychelles | Presence Uncertain | Native | – | Resident |
| Sierra Leone | Presence Uncertain | Native | – | Resident |
| Singapore | Presence Uncertain | Native | – | Resident |
| Sint Maarten (Dutch part) | Extant | Native | – | Resident |
| Solomon Islands | Extant | Native | – | Resident |
| Somalia | Presence Uncertain | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Spain | Extant | Native | – | Resident |
| Spain -> Canary Is. | Extant | Native | – | Resident |
| Sri Lanka | Extant | Native | – | Resident |
| Sudan | Presence Uncertain | Native | – | Resident |
| Suriname | Presence Uncertain | Native | – | Resident |
| Taiwan, Province of China | Extant | Native | – | Resident |
| Tanzania, United Republic of | Presence Uncertain | Native | – | Resident |
| Thailand | Extant | Native | – | Resident |
| Timor-Leste | Presence Uncertain | Native | – | Resident |
| Togo | Presence Uncertain | Native | – | Resident |
| Tonga | Presence Uncertain | Native | – | Resident |
| Trinidad and Tobago | Presence Uncertain | Native | – | Resident |
| Turks and Caicos Islands | Presence Uncertain | Native | – | Resident |
| Tuvalu | Presence Uncertain | Native | – | Resident |
| United Arab Emirates | Presence Uncertain | Native | – | Resident |
| United Kingdom of Great Britain and Northern Ireland | Extant | Vagrant | – | – |
| United States Minor Outlying Islands | Presence Uncertain | Native | – | Resident |
| United States of America | Extant | Native | – | Resident |
| Uruguay | Extant | Native | – | Resident |
| Vanuatu | Presence Uncertain | Native | – | Resident |
| Venezuela, Bolivarian Republic of | Extant | Native | – | Resident |
| Viet Nam | Extant | Native | – | Resident |
| Virgin Islands, British | Presence Uncertain | Native | – | Resident |
| Virgin Islands, U.S. | Presence Uncertain | Native | – | Resident |
| Wallis and Futuna | Presence Uncertain | Native | – | Resident |
| Western Sahara | Presence Uncertain | Native | – | Resident |
| Yemen | Presence Uncertain | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
| Presence | Origin | Formerly Bred | Seasonality | |
| 31. Atlantic – western central | Extant | Native | – | Resident |
| 34. Atlantic – eastern central | Extant | Native | – | Resident |
| 41. Atlantic – southwest | Extant | Native | – | Resident |
| 47. Atlantic – southeast | Extant | Native | – | Resident |
| 51. Indian Ocean – western | Extant | Native | – | Resident |
| 57. Indian Ocean – eastern | Extant | Native | – | Resident |
| 61. Pacific – northwest | Extant | Native | – | Resident |
| 71. Pacific – western central | Extant | Native | – | Resident |
| 77. Pacific – eastern central | Extant | Native | – | Resident |
| 87. Pacific – southeast | Extant | Native | – | Resident |
Climate change
Climate change is predicted to expand tropical water species poleward into more preferred water temperatures (van Weelden et al. 2021, Kebke et al. 2022). Fraser’s dolphin is one such species and the study by Chen et al. (2020) also confirms habitat expansion which they suggest is possibly due to climate change and warming waters. Even though the species may be able to tolerate the warmer water temperatures, their prey may not. Fraser’s dolphins typically feed on midwater or deepwater species whose habitat may become restricted due to temperature constraints, or it may shift poleward (Kebke 2022). Ultimately these changes could lead to a reduction in prey availability, habitat loss which could impact Fraser’s dolphin overall health, fecundity and survival rate.
Population
No global estimates or trends are available for Fraser’s Dolphins, and although some regional estimates exist for other areas (for example, there are 289,000 CV = 34% in the eastern tropical Pacific; Perrin et al. 1994), there are none for the assessment region. Strandings records show fairly regular records between 1990 and 2009. However, since 2009 there has been no reported strandings, bycatch or entanglement of this species. Whether this is indicative of a declining population, or just a lack of data, is concerning. By 1986, ten stranding events, including a total of 14 individuals, had been recorded on South Africa’s east coast, associated with the subtropical Agulhas Current (Findlay et al. 1992). Since 1990, an additional 13 Fraser’s Dolphins have been reported, of which two were bycatch in shark nets. Taylor et al. (2007) estimated a generation period of 11.1 years.
Population Information
Current population trend: Unknown
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
Very little is known about the population genetic structure and diversity of Fraser’s dolphins, especially in the assessment area. However, a recent paper by Chen et al. (2020) which looked at 18 microsatellite loci and 1 mitochondrial DNA locus compared 112 Fraser’s dolphins across much of its distribution (the majority of specimens collected mainly from the waters of Japan, Taiwan and the Philippines and to a lesser degree from the Gulf of Mexico and the Caribbean Sea) found The study found differences between Japanese and Philippine L. hosei, which could suggest additional structure between other regions where this species occurs. This will need to be verified using a more comprehensive, fine scale genomic study.
Given that no population size data exists for the assessment region, a measure of effective population size cannot be estimated.
Habitats and ecology
Fraser’s Dolphin is a pantropical, oceanic species, predominantly preferring deep offshore regions (Dolar 1999). In South African waters, Findlay et al. (1992) suggests that this species may have a localised occurrence associated with the subtropical Agulhas Current, which moves southwards in summer. This species has been recorded feeding both at the surface (for example, in the Caribbean, Watkins et al. 1994; and on the east coast of South Africa, Ross 1984), as well as at depths of up to 500 m (Robison & Craddock 1983). In general, their diet is considered to consist mostly of deep-sea fishes and squid, in fact some of the prey recorded by Tobayama et al. (1973) rarely extend higher than 200 m below the surface. There is also a lack of documented association between this species and flocking seabirds or schools of tuna, which substantiates the shortage of surface feeding in this species (Perrin et al. 1994).
Fraser’s Dolphins in the Sulu Sea (southwest of the Philippines) were found to have a more diverse diet, when compared to Spinner Dolphins (Stenella longirostris), feeding on a variety of mesopelagic fishes (primarily myctophid species), cephalopods (including Abraliopsis, Onychoteuthis, Histioteuthis and Chiroteuthis), as well as crustaceans (Dolar et al. 2003). In contrast, the bulk of the diet of South African Fraser’s Dolphins comprised of cephalopods (mostly Chiroteuthidae, Histioteuthidae and Octopoteuthidae), while fish constituted only 4% of their diet and there was no evidence of these individuals feeding on crustaceans (Sekiguchi et al. 1992).
In southern Africa, this species has been recorded in pods consisting of between 7 and 1,000 individuals, with an average group size of 183.1 (Findlay et al. 1992). Although little data is available regarding the reproductive biology of this species, Ross (1984) suggests that conception and calving may take place in summer within the assessment region. However, no clear trends in reproductive seasonality have been documented in other parts of its range.
Ecosystem and cultural services: Marine mammals integrate and reflect ecological variation across large spatial and long temporal scales, and therefore they are prime sentinels of marine ecosystem change (Moore 2008).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 9.1. Marine Neritic -> Marine Neritic – Pelagic | – | Marginal | – |
| 10.1. Marine Oceanic -> Marine Oceanic – Epipelagic (0-200m) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? Yes
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Marine
General Use and Trade Information
Although Fraser’s Dolphins are exploited by small-scale subsistence fisheries in the Indo-Pacific, and in drive fisheries of Taiwan (Perrin et al. 1994), there is no trade or use of this species within the assessment region.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: Yes
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
true |
– |
– |
|
2. Food – animal |
true |
true |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Fraser’s Dolphins are taken incidentally in a variety of fishing gear globally, for example, this species is commonly caught as bycatch in the driftnet fishery off the Philippines (Dolar 1994), and in purse-seine fisheries of the eastern tropical Pacific. In South Africa there appears to be no significant fisheries involvement (Best 2007), although some are killed by anti-shark nets in South Africa (Cockcroft 1990; Perrin et al. 1994).
Considering that this is a deep-diving, offshore species, it is likely that this species may be vulnerable to the effects of marine noise pollution, for example, those produced by navy sonar and seismic exploration. Additionally, as cephalopods constitute a large proportion of the diet of Fraser’s Dolphins, it is likely that the increasing risk of plastic pollution may threaten this species due to accidental ingestion.
Current habitat trend: Stable
Conservation
Fraser’s Dolphins are listed in Appendix II of the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES) and this species is protected by the Marine Living Resources Act (No. 18 of 1998) of the national legislation. No species-specific conservation measures have been identified. However, estimates of population size and trends are needed to determine the status of this species in South African waters. Research is required to assess the impacts and trends of potential threats. This species may benefit from a reduction in the length, modification or compete removal of anti-shark nets off South Africa’s east coast, as recommended for Stenella spp.
Protected Species Observers (PSOs) play a crucial role in monitoring and mitigating anthropogenic impacts on marine mammals (Ganley et al. 2025), including Fraser’s dolphin. PSO data have been instrumental in expanding knowledge on the species’ distribution, particularly in data-poor regions like the eastern tropical Atlantic (Weir et al. 2013). Such data contribute to understanding habitat preferences and inform conservation assessments, supporting Red List evaluations.
Recommendations for managers and practitioners:
- The severity of threats, as well as the potential synergistic effects of those threats on this species, requires investigation.
- Sightings, strandings and bycatch data should be recorded, especially during ship-based surveys aimed at other cetacean species, as well as pelagic commercial fisheries and marine tour operators.
- A portable and accessible rapid DNA amplification assay enables efficient identification of suspicious cetacean products, supporting law enforcement efforts. This tool provides valuable insights into the pathways of illegal trade and consumption, contributing to a more comprehensive understanding of cetacean utilisation. Such information is critical for developing effective management and conservation strategies, particularly for vulnerable species (Endo et al. 2022).
- A study by West et al. (2023) detected cetacean morbillivirus (CeMV) in the faeces of an infected Fraser’s dolphin using RT-qPCR, at a 1:1,000 simulated seawater dilution. This highlights the potential for non-invasive disease screening in wild cetaceans, aiding in pathogen monitoring and health assessments. Such diagnostic advancements are crucial for understanding disease spread and informing conservation strategies for L. hosei populations (West et al. 2023).
Research priorities:
- Genetic sequencing for individuals that have stranded in the past to determine if the population in the assessment region differs from those in the waters of Japan, Taiwan, the Philippines, the Gulf of Mexico and the Caribbean Sea.
- Data on population size and trends, and on subpopulation substructure, could influence the listing of this species, and it may require reassessment as new data emerge.
- Data pertaining to the species’ distribution patterns and the severity of potential threats that could affect the listing of this species.
Encouraged citizen actions:
- Use information dispensed by the South African Sustainable Seafood Initiative (SASSI) to make good choices when buying fish in shops and restaurants, e.g. wwfsa.mobi, FishMS 0794998795.
- Save electricity and fuel to mitigate CO2 emissions and hence rate of climate change.
- Buy local products that have not been shipped.
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) to help with mapping geographical distribution.
- Avoid using plastic bags.
Bibliography
Anderson RC. 1996. First records of Fraser’s dolphin (Lagenodelphis hosei) from the Maldives. Journal of South Asian Natural History 2: 75-80.
Best, P.B. 2007. Whales and Dolphins of the Southern African Subregion. Cambridge University Press, Cape Town, South Africa.
Chen I, Nishida S, Chou LS, Isobe T, Mignucci-Giannoni AA, Hoelzel AR (2020) Population genetic diversity and historical dynamics of Fraser’s dolphins Lagenodelphis hosei. Mar Ecol Prog Ser 643:183 195 https://doi.org/10.3354/meps13268
Cockcroft VG. 1990. Dolphin catches in the Natal shark nets, 1980–1988. South African Journal of Wildlife Research 20: 44-51.
Dolar MLL. 1994. Incidental takes of small cetaceans in fisheries in Palawan, central Visayas and northern Mindano in the Philippines. Report of the International Whaling Commission (Special Issue) 15: 355-363.
Dolar MLL. 1999. Abundance, distribution and feeding ecology of small cetaceans in the eastern Sulu Sea and Tañon Strait, Philippines. Ph.D. dissertation. University of California, San Diego, CA, USA.
Dolar, M. L. L. 2002. Fraser’s dolphin Lagenodelphis hosei. In: W. F. Perrin, B. Wursig and J. G. M. Thewissen (eds), Encyclopedia of Marine Mammals, pp. 485-487. Academic Press.
Dolar, M. L. L., Walker, W. A., Kooyman, G. L. and Perrin, W. F. 2003. Comparative feeding ecology of spinner dolphins (Stenella longirostris) and Fraser’s dolphins (Lagenodelphis hosei) in the Sulu Sea. Marine Mammal Science 19(1): 1-19.
Endo, H., Yamada, T. K., Watanabe, H., & Shinohara, A. 2022. A rapid DNA amplification assay for cetacean product identification: Providing insights for illegal cetacean utilization and implications for conservation. Scientific Reports, 12, 2401. https://doi.org/10.1038/s41598-022-06441-2
Findlay KP, Best PB, Ross GJB, Cockcroft VG. 1992. The distribution of small odontocete cetaceans off the coasts of South Africa and Namibia. South African Journal of Marine Science 12: 237-270.
Ganley, L.C., Sisson, N.B., McKenna, K.R. & Redfern, J.V. 2025. Perspectives on using Protected Species Observer (PSO) data to fill knowledge gaps about marine species distributions and habitat use, ICES Journal of Marine Science, Volume 82, Issue 3, fsae076, https://doi.org/10.1093/icesjms/fsae076
IUCN (International Union for Conservation of Nature). 2012. Lagenodelphis hosei. The IUCN Red List of Threatened Species. Version 3.1. Available at: http://www.iucnredlist.org. Downloaded on 21 February 2016.
Jefferson, T. A. and Leatherwood, S. 1994. Lagenodelphis hosei. Mammalian Species 470: 1-5.
Kebke, A., Samarra, F. & Derous, D. 2022. Climate change and cetacean health: impacts and future directions. Phil. Trans. R. Soc. B 377: 20210249. https://doi.org/10.1098/rstb.2021.0249
Leatherwood, S. and Reeves, R.R. (eds). 1989. Marine mammal research and conservation in Sri Lanka 1985–1986. UNEP Marine Mammal Technical Report 1, Nairobi, Kenya.
Moore SE. 2008. Marine mammals as ecosystem sentinels. Journal of Mammalogy 89: 534-540.
Perrin, W. F., Leatherwood, S. and Collet, A. 1994. Fraser’s dolphin Lagenodelphis hosei Fraser, 1956. In: S. H. Ridgway and R. Harrison (eds), Handbook of marine mammals, Volume 5: The first book of dolphins, pp. 225-240. Academic Press.
Perrin, W.F., Best, P., Dawbin, W.H., Balcomb, R., Gambell, R. and Ross, G.J.B. 1973. Rediscovery of Fraser’s dolphin Lagenodelphis hosei. Nature 241: 345-350.
Robison BH, Craddock JE. 1983. Mesopelagic fishes eaten by Fraser’s dolphin, Lagenodelphis hosei. Fishery Bulletin 81: 283-289.
Ross, G. J. B. 1984. The smaller cetaceans of the south east coast of southern Africa. Annals of the Cape Provincial Museums (Natural History) 15: 173-410.
Sekiguchi, K., Klages, N.T.W. and Best, P.B. 1992. Comparative analysis of the diets of smaller odontocete cetaceans along the coast of southern Africa. South African Journal of Marine Science 12: 843-861.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Taylor, B.L., Chivers, S.J., Larese, J. and Perrin, W.F. 2007. Generation length and percent mature estimates for IUCN assessments of cetaceans. NOAA, Southwest Fisheries Science Center Administrative Report LJ-07-01. La Jolla, California.
Tobayama TM, Nishiwaki M, Yang HC. 1973. Records of Fraser’s Sarawak dolphin (Lagenodelphis hosei) in the western north Pacific. Scientific Reports of the Whales Research Institute, Tokyo 25: 251-263.
van Weelden, Celine, Jared R. Towers, and Thijs Bosker. 2021. Impacts of climate change on cetacean distribution, habitat and migration. Climate Change Ecology 1: 100009.
Watkins, W. A., Daher, M. A., Fritsrup, K. M. and Notarbartolo Di Sciara, G. 1994. Fishing and acoustic behavior of Fraser’s dolphin (Lagenodelphis hosei) near Dominica, southeast Caribbean. Caribbean Journal of Science 30: 76-82.
Weir, C.R., Goncalves, L. & May, D. 2013. New Gulf of Guinea (Africa) range state records for pygmy killer whale (Feresa attenuata) and Fraser’s dolphin (Lagenodelphis hosei). Marine Biodiversity Records 6: e35.
West, K.L., Silva-Krott, I., Clifton, C.W., Humann, C. & Davis, N. 2023. Detection of cetacean morbillivirus in dolphin feces and the potential application for live cetacean health monitoring. Marine Mammal Science, 40(1), 262-269. DOI: https://doi.org/10.1111/mms.13064
