Four-toed Sengi
Petrodromus tetradactylus

2025 Red list status
Near threatened
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Petrodromus tetradactylus – Peters, 1846
ANIMALIA – CHORDATA – MAMMALIA – MACROSCELIDEA – MACROSCELIDIDAE – Petrodromus – tetradactylus
Common Names: Four-toed Sengi, Four-toed Elephant-shrew (English), Bosklaasneus (Afrikaans)
Synonyms: No Synonyms
For general biological information on all sengi species, consult accounts in: Species accounts: Order Macroscelidea, in: Perrin, M. & Rathbun, G. B. 2013. Mammals of Africa, Volume I (Ed. by Kingdon, J., Happold, D., Hoffmann, M., Butynski, T., Happold, M. & Kalina, J.), pp. 261-278. London: Bloombury. Order Macroscelidea, in: Rathbun, G. B., (subeditor). 2005. The Mammals of the Southern African Subregion, 3rd edition (Ed. by Skinner, J. D. & Chimimba, C. T.), pp. 22-34. Cape Town, South Africa: Cambridge University Press. Species accounts: in: Rathbun, G. B. 2013. Mammals of Africa, Volume I (Ed. by Kingdon, J., Happold, D., Hoffmann, M., Butynski, T., Happold, M. & Kalina, J.), pp. 279-287. London: Bloomsbury. Order MACROSCELIDEA, in: Heritage, S. 2018. Handbook Mammals of the World, 8. Insectivores, Sloths and Colugos (D.E. Wilson, D.E. and R.A. Mittermeier, eds), Lynx Edicions, Barcelona, Spain. For current and more technical information, search the on-line bibliography at www.sengis.org. This genus is in need of a taxonomic revision that would assess the currently recognised 10 subspecies. In addition, molecular genetics and morphology suggests that Elephantulus rozeti from northern Africa and Petrodromus are more closely related to each other than E. rozeti is to any of the other species of Elephantulus, and a new genus, Petrosaltator, has been proposed for E. rozeti. (Dumbacher et al. 2016).
Taxonomic Note:
In the past the single family was included in the order Insectivora, but now the family is in the monophyletic order Macroscelidea and the super-cohort Afrotheria. Currently, there are 20 living species recognised in six genera. The soft-furred sengis or elephant-shrews include five genera: Petrodromus and Petrosaltator are monospecific, Macroscelides has three species, Galegeeska has two species and Elephantulus contains 8 species. The five species of giant sengis belong to the genus Rhynchocyon. The common name “sengi” is being used in place of elephant-shrew by many biologists to try and disassociate the Macroscelidea from the true shrews (family Soricidae) in the order Eulipotyphla. See the Afrotheria Specialist Group web site and www.sengis.org for additional information.
This genus specifically is in need of a taxonomic revision that would assess the currently recognised 10 subspecies. Subspecies are characterised mainly by pelage colour and bristles under the tail. Two subspecies occur within the assessment region, P. t. schwanni in north-eastern Limpopo and P. t. warreni in northern KwaZulu-Natal. The subspecies in KwaZulu-Natal (P. t. warreni) lacks tail bristles, as opposed to other forms (Rathbun 2013).
| Red List Status |
| NT, B2ab(ii,iii,v) (IUCN version 3.1) |
Assessment Information
Assessors: Roxburgh, L.1
Reviewers: Schradin, C.2
Contributors: da Silva, J. M.3
Institutions: 1Endangered Wildlife Trust, 2Institut pluridisciplinaire Hubert Curien, 3South African National Biodiversity Institute
Previous Assessor: Rathbun, G.B.
Previous Reviewer: Smit-Robinson, H.
Previous Contributors: Child, M.F., Davies-Mostert, H. & Roxburgh, L.
Assessment Rationale
This species is at the edge of its range within the assessment region where two disjunct subspecies occur: P. t. schwanni in north-eastern Limpopo province, and P. t. warreni in northern KwaZulu-Natal province, both being restricted to intact riparian and coastal forest. The estimated area of occupancy (AOO) for P. t. schwanni and P. t. warreni, based on remaining forest habitat, within the assessment region is 72 and 192 km2 respectively with a total AOO of 264 km2. Although P. t. warreni might represent a South African endemic, further taxonomic resolution is required before we assess it separately. Overall, the species qualifies for Endangered B2ab(ii,iii,v) based on restricted area of occupancy (264 km2 in South Africa), presumed small population size, and a continuing decline in woodland habitat as a result of human expansion over the past decade. In KwaZulu-Natal alone, there was a 7.6% loss of natural habitat from 2005 to 2011, while in the period from 2014 to 2020, loss of natural habitat within its range in KwaZulu-Natal (KZN) was estimated as 3.7%, while for KZN as a whole it was slightly higher at 4.5%. As such, forest patches are likely to be severely fragmented, hindering dispersal of the species. Fragmentation further opens up forest patches for ongoing anthropogenic disturbances, such as incidental bushmeat hunting and removal of ground cover and thus represents a continuing decline in both mature individuals and habitat quality. However, the species occurs predominantly in large, well-managed protected areas, including the Great Limpopo Transfrontier Park (P. t. schwanni) and the Lubombo Transfrontier Conservation Area (P. t. warreni), so net population decline is unlikely. Additionally, rescue effects are possible (see below). Thus, we downlist to Near Threatened B2ab(ii,iii,v) status, as was done in the previous assessment. The key intervention for this species is further protected area expansion (especially transfrontier expansion) and connection of remaining forest patches.
Regional population effects: This species is the second-most widespread sengi in Africa, occurring from central and eastern Africa south to the north-eastern corner of South Africa. The assessment region thus represents the edge of its range. Although sengis are not long-distance dispersers, the presence of both subspecies in major transfrontier conservation areas, with presumably intact forest corridors, leads us to suspect rescue effects are possible. However, P. t. schwanni may represent a unique subspecies/species, in which case rescue effects are not possible. This should be investigated and may require reassessment.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Roxburgh L. 2025. A conservation assessment of Petrodromus tetradactylus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
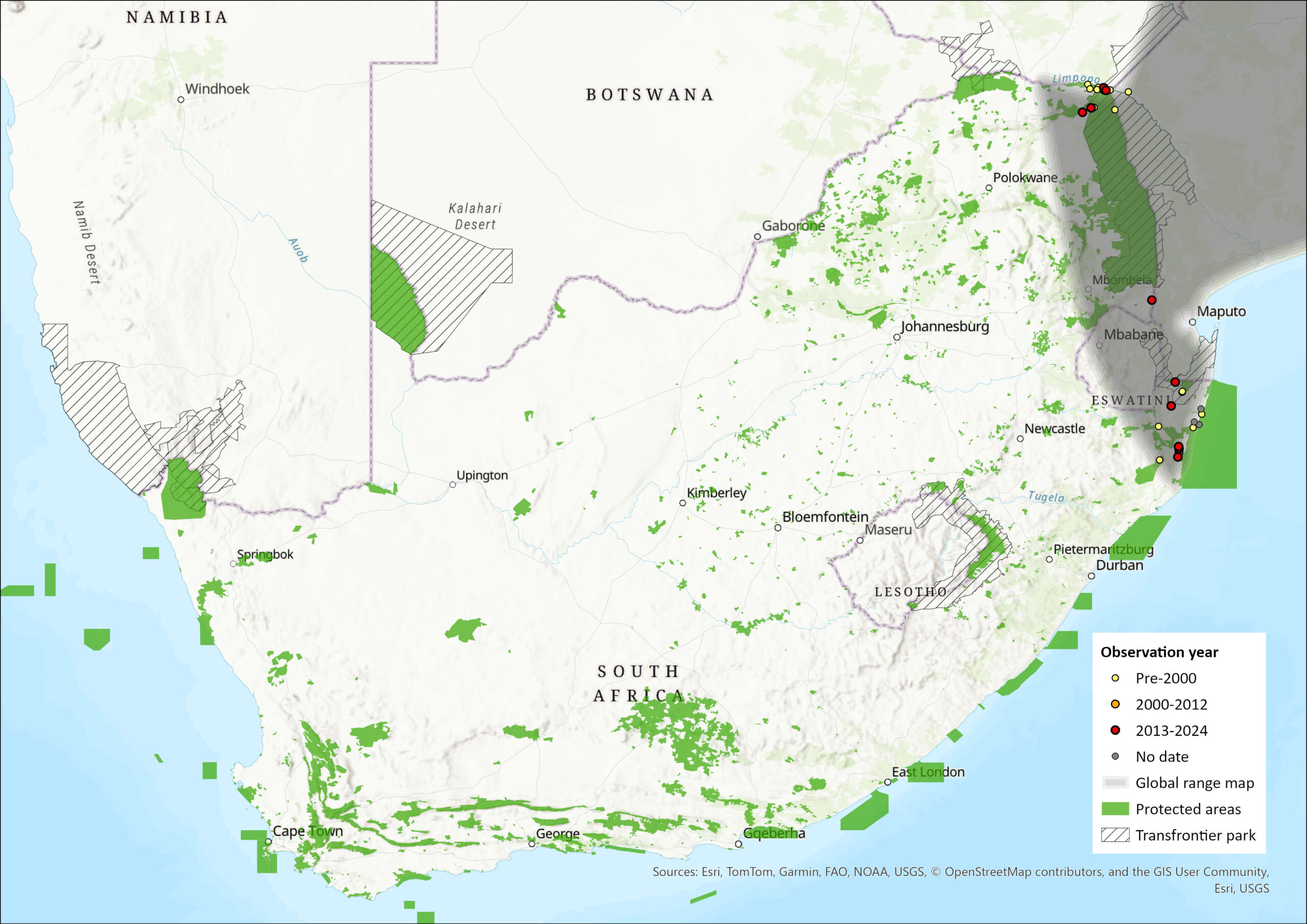
This species is one of the most widespread sengis, occurring in forest, woodland, and thicket habitats in central and eastern Africa from DR Congo to northeastern South Africa. Their occurrence depends on the availability of suitable forest or woodland habitat with dense underbrush, which is discontinuous throughout their range (Skinner & Chimimba 2005). It is an edge-of-range species within the assessment region, occurring in woodland and forest habitats in Limpopo and KwaZulu-Natal provinces and perhaps in Eswatini. The subspecies P. t. warreni occurs in KwaZulu-Natal and may be a South African endemic form (Corbet & Hanks 1968), where their distribution is predominantly coastal (Rathbun 2005), whereas P. t. schwanni occurs in the riparian forests of northern Limpopo and across the border to the north. Although it has not been recorded from Eswatini (Monadjem 1998), it may possibly occur there marginally in suitable forest habitats.
The estimated area of occupancy (AOO) globally is 2,485,700 km2 based on censored minimum convex polygon with no consideration of fragmented habitat. The estimated global extent of occurrence is 5,700,000 km2. Within the assessment region, the AOO is estimated to be 264 km2 based on remaining forest patches within the species range (SANBI 2018). The estimated AOO for P. t. schwanni and P. t. warreni, based on remaining forest habitat, is 72 and 192 km2 respectively. The IUCN recommended methodology of using a 4 km2 grid of occupied cells yields a more conservative total AOO of 172 km2, but levels of occupancy within its range are likely to be underestimated.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Biogeographic Realms
Biogeographic Realm: Afrotropical
Map
Figure 1. Distribution records for Four-toed Sengi (Petrodromus tetradactylus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Presence Uncertain | Native | – | Resident |
| Congo, The Democratic Republic of the | Extant | Native | – | Resident |
| Eswatini | Possibly Extant | Native | – | Resident |
| Kenya | Extant | Native | – | Resident |
| Malawi | Extant | Native | – | Resident |
| Mozambique | Extant | Native | – | Resident |
| Rwanda | Extant | Native | – | Resident |
| South Africa | Extant | Native | – | Resident |
| Tanzania, United Republic of | Extant | Native | – | Resident |
| Zambia | Extant | Native | – | Resident |
| Zimbabwe | Extant | Native | – | Resident |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified
Climate change
This is a wide-ranging species that is clearly able to tolerate higher temperatures and some variation in climate across its broad range. It is unlikely to be severely affected by temperature changes due to climate change in South Africa. However, the impacts of increasing rainfall and rainfall intensity on this species, which are particularly likely in KwaZulu-Natal province (Engelbrecht et al. 2024), might lead to direct mortality, localized destruction of habitat, and food scarcity, as a study (Walker et al. 2023) of the impact of a tropical cyclone on mammals suggests.
Population information
Although widespread, the species is often only locally common because it is restricted to closed-canopy forest, woodlands, and thickets. Little information is available on densities. In Afzelia habitat within Arabuko-Sokoke Forest (Kenya), the estimated density was 1.2 animals / ha (FitzGibbon 1995). Linn et al. (2007) found that home range sizes for two males and two females in dune forest in Sodwana Bay, South Africa were similar to FitzGibbon (1995), at around 1 ha, with overlap between a male and female pair. Using this density for the estimated AOO within the assessment region yields 52,800 individuals.
However, there is no evidence that it is abundant. For example, as there was no overlap between the home ranges of adjoining pairs in Tembe Elephant Park, KwaZulu-Natal, and a low population density was assumed (Oxenham & Perrin 2009). Further studies should be conducted to determine density estimates for both subspecies within the assessment region.
It often foot-drums and thus attracts the attention of people in suitable habitat, but sightings are almost always of individuals. It builds and maintains characteristic and distinct paths through the leaf litter that are often composed of a straight series of clean oval patches, which can be used as an indicator of presence.
Population Information
| Continuing decline in mature individuals? | Qualifier | Justification |
| Yes | – | Suspected from incidental bushmeat hunting |
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population genetics
P. tetradactylus has been studied in a broad biogeographic context and found multiple deeply divergent lineages within the species (Hagemann et al. 2024). Only one specimen was incorporated from South Africa, so nothing can be inferred with respect to the assessment region, but it does highlight the need for a more integrative taxonomic look into the species. To date, no fine scale population genetic studies have been undertaken on the species, yet two sub-species are recognized in South Africa: one in KwaZulu-Natal (P. t. warreni) that is differentiated from t. schwanni in Limpopo by the lack of tail bristles (Rathbun 2013). A population genetic study is highly recommended to confirm the validity of these subspecies (especially given the multiple divergent lineages identified in central and eastern Africa), and to potentially identify other genetically distinct subpopulations. Assuming no other subpopulations exist or have gone extinct, the Convention on Biological Diversity’s Global Biodiversity Framework’s complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (2/2 subpopulations remaining).
Since no estimates of each subpopulation’s effective population size (Ne) exist, we can estimate it using proxy information based on density and area of occupancy. Using the estimated remaining forest habitat and a density of 1 ha per pair, the Limpopo subpopulation is estimated at 14,400 individuals and 38,400 for the KZN subpopulation. Applying a Ne/Nc conversion ratio of 0.1-0.3, Ne is estimated at 1440-4320 for Limpopo and 3,840-11,520 for KZN. While these estimates may be overestimations, both subpopulations are still likely to exceed the Ne 500 threshold, thereby suggesting each is genetically health and stable. From this we can calculate the GBF’s headline indicator – proportion of species with an effective population size greater than 500 – would is valued at 1.0 (2/2 subpopulations with Ne >500).
Habitats and ecology
The species is known from forest, dense woodlands, and thickets (Jennings & Rathbun 2001), where animals probably form pairs (FitzGibbon 1995; Oxenham & Perrin 2009). They prefer habitats usually with surface leaf litter, which makes it easy to identify their characteristic paths that are usually composed of a straight line of oval bare patches in the leaf litter. They are able to live in fallow agricultural areas that have suitable cover and leaf litter, invertebrates for food, and that are adjacent to undisturbed habitats. They are omnivorous, but prey mainly on invertebrates. This is the largest species of sengi in the subregion with a body mass of 130-280g (Rathbun 2005). They are active during day and night and shelter in holes, termite mounds, under fallen logs, hollow logs, patches of dense underbrush or under roots of trees but do not build or use nests (Jennings & Rathbun 2001). When alarmed they rapidly stamp their hind feet on the substrate, which can be heard many metres away, and may utter a loud shrill squeak when in distress (Rathbun 2005). The common name emanates from the fact that, unlike all other members of the family, they have only four toes on their hind feet.
Ecosystem and cultural services: None recorded.
IUCN Habitats Classification Scheme
|
Habitat |
Season |
Suitability |
Major Importance? |
|
1.5. Forest -> Forest – Subtropical/Tropical Dry |
Resident |
Suitable |
Yes |
|
1.6. Forest -> Forest – Subtropical/Tropical Moist Lowland |
Resident |
Suitable |
Yes |
|
1.9. Forest -> Forest – Subtropical/Tropical Moist Montane |
Resident |
Suitable |
Yes |
|
2.2. Savanna -> Savanna – Moist |
Resident |
Suitable |
Yes |
|
3.6. Shrubland -> Shrubland – Subtropical/Tropical Moist |
Resident |
Suitable |
Yes |
Life History
Generation Length: (Not specified)
Age at maturity: female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: 35.5
Size at Maturity (in cms): Male: 35
Longevity: up to 6 years and 7 months in captivity
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
Does the species lay eggs? No
Does the species give birth to live young? Yes
Does the species exhibit parthenogenesis? No
Does the species have a free-living larval stage? No
Does the species require water for breeding? No
Movement Patterns
Movement Patterns: Not a Migrant
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
FitzGibbon et al. (1995) documented subsistence hunting in coastal Kenya, which is assumed to continue to the present, but it is not known whether this species is hunted in South Africa. Given the increase in the human population along protected area edges in general (Wittemyer et al. 2008), one could assume that hunting pressure has increased, and may especially be prevalent in KwaZulu-Natal where many rural communities surround forest patches. However, bushmeat hunting of this species may be lower than expected from relative density based on model outputs (Rowcliffe et al. 2003).
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
May be opportunistically used in bushmeat |
No |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major identified threats range-wide, although habitat loss through woodland clearing and subsistence snaring may have impacts on local subpopulations in the assessment region. If we assume that both woodland clearing and subsistence hunting increase with rural population density and settlement expansion, it is suspected there is an ongoing loss of habitat and mature individuals, especially the forests of KwaZulu-Natal (see below).
Current habitat trend: Declining. Habitat loss is ongoing through woodland clearing, especially around protected area fringes (Wittemyer et al. 2008). There was a 7.6% loss of natural habitat from 2005 to 2011, with an average loss of 1.2% per annum in KwaZulu-Natal (Jewitt et al. 2015). In the period from 2014 to 2020, loss of natural habitat within its range in KwaZulu-Natal (KZN) was estimated as 3.7%, while for KZN as a whole it was slightly higher at 4.5% (DFFE 2022). Additionally, there was an 8% and 1% increase in rural settlements in Limpopo and KwaZulu-Natal, respectively, between 2000 and 2013 (GeoTerraImage 2015).
Conservation
The species occurs in several protected areas (confirmed in northern Kruger National Park and protected areas in KwaZulu-Natal) within the assessment region, including Great Limpopo Transfrontier Park (P. t. schwanni) and the Lubombo Transfrontier Conservation Area (P. t. warreni). For example, they occur in Tembe Elephant Park (Oxenham & Perrin 2009), Isimangaliso Wetland Park and Bonamanzi Game Reserve. This species would benefit from protected area expansion to conserve woodland habitat and connect subpopulations existing in forest fragments. For example, a transfrontier conservation area in Maputaland that connects with the existing Lubombo Transfrontier Conservation Area has been proposed and would add 480 km2 of linking corridors between forest habitats and potentially generate US$18.8 million from game ranching (Smith et al. 2008). Such a protected area network would undoubtedly benefit this species.
Recommendations for land managers and practitioners:
- Land owners and communities should reduce stocking rates to conserve undergrowth.
Research priorities:
- Field surveys to confirm occupancy rate across the assessment region. Continue to accumulate information on occurrence points (see www.sengis.org). Collect evidence to indicate population trends and status of habitats.
- Genetic and phenotypic analyses to determine whether, if any, species-complexes are present across its distribution south of the Sahara.
- Studies to determine the extent and impact of snaring in South Africa.
Encouraged citizen actions:
- Landowners can create conservancies to protect woodland habitat.
- Citizens are encouraged to report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP) with photographic confirmation.
Bibliography
Corbet, G.B. and Hanks, J. 1968. A revision of the elephant-shrews, family Macroscelididae. Bulletin of the British Museum of Natural History (Zoology) 16: 1-111.
Department of Forestry, Fisheries and the Environment (DFFE). 2022. South African National Lanc-Cover 2014 / 2020 Change Assessment Report. DFFE, South Africa.
Dumbacher, J.P, Carlen, E.J. and G.B. Rathbun. 2016. Petrosaltator gen. nov., a new genus replacement for the North African sengi Elephantulus rozeti (Macroscelidea; Macroscelididae). Zootaxa 4136 (3): 567-579.
Engelbrecht, F.A., Steinkopf, J., Padavatan, J., and Midgley, G.F. 2024. Projections of Future Climate Change in Southern Africa and the Potential for Regional Tipping Points. In: von Maltitz, G.P., et al. Sustainability of Southern African Ecosystems under Global Change. Ecological Studies, vol 248. Springer, Cham. https://doi.org/10.1007/978-3-031-10948-5_7.
FitzGibbon, C.D. 1995. Comparative ecology of two elephant-shrew species in a Kenyan coastal forest. Mammal Review 25: 19–30.
FitzGibbon, C.D., Mogaka, H. and Fanshawe, J.H. 1995. Subsistence hunting in Arabuko-Sokoke Forest, Kenya, and its effects on mammal populations. Conservation Biology 9: 1116-1126.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa. Pretoria.
Hagemann J., Nogales, L.V., Hofreiter,M., Arnold, P. 2024. Four-toed sengi (Petrodromus tetradactylus, Afrotheria, Mammalia) museomics reveals a crucial role of East African forests in macroscelidean diversification, Zoological Journal of the Linnean Society 201: zlae081, https://doi.org/10.1093/zoolinnean/zlae081
Heritage, S. 2018. Order MACROSCELIDEA. In Handbook of the Mammals of the World, Volume 8. Insectivores, Sloths and Colugos (D.E. Wilson and R.A. Mittermeier, eds), Lynx Edicions, Barcelona, Spain.
Jennings, M. and Rathbun, G.B. 2001. Petrodromus tetradactylus. Mammalian Species 682: 1-6.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Linn, I., Perrin, M. R., and Hiscocks, K. 2007. Use of space by the four-toed elephant-shrew Petrodromus tetradactylus (Macroscelidae) in Kwazulu-Natal (South Africa). Mammalia, 71(1/2). doi:10.1515/mamm.2007.012.
Monadjem, A. 1998. Distributional patterns and conservation status of mammals of Swaziland, southern Africa. Koedoe 41: 45-59.
Olbricht, G. 2007. Longevity and fecundity in sengis (Macroscelidea).Afrotherian Conservation 5: 3–5
Oxenham, K.H. and Perrin, M. 2009. The spatial organization of the four-toed elephant-shrew (Petrodromus tetradactylus) in Tembe Elephant Park, KwaZulu-Natal, South Africa. African Zoology 44: 171-180.
Perrin, M. and Rathbun, G.B. 2013. Species accounts: Order Macroscelidea, Family Macroscelididae, Genus Elephantulus, E. edwardii, E. intufi, E. myurus, E. rozeti, E. rufescens; Genus Macroscelides; species account M. proboscideus. In: J.S Kingdon, D.C.D. Happold, M. Hoffmann, T.M. Butynski, M. Happold and J. Kalina (eds), Mammals of Africa, Volume I: Introductory Chapters and Afrotheria, pp. 261-278. Bloomsbury Publihing, London.
Rathbun GB. 2013. Species accounts: Genus Petrodromus, P. tetradactylus, Genus Rhynchocyon, R. cirnei, R. chrysopygus, R. petersi. In: Kingdon J, Happold D, Hoffmann M, Butynski T, Happold M, Kalina J (ed.), Mammals of Africa, Volume I, pp. 279-287. Bloomsbury Publishing, London, UK.
Rathbun, G.B. (subeditor). 2005. Order Macroscelidea. In: J.D. Skinner and C.T. Chimimba (eds), The Mammals of the Southern African Subregion, 3rd edition, pp. 22-34. Cambridge University Press, Cape Town, South Africa.
Rowcliffe JM, Cowlishaw G, Long J. 2003. A model of human hunting impacts in multi-prey communities. Journal of Applied Ecology 40: 872-889.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
South African National Biodiversity Institute. 2018. The Vegetation Map of South Africa, Lesotho and Swaziland, Mucina, L., Rutherford, M.C. and Powrie, L.W. (Editors), Online, http://bgis.sanbi.org/SpatialDataset/Detail/18, Version 2018
Walker, R. H., Hutchinson, M. C., Becker, J. A., Daskin, J. H., Gaynor, K. M., Palmer, M. S., … Long, R. A. 2023. Trait-based sensitivity of large mammals to a catastrophic tropical cyclone. Nature, 623(7988), 757-764. doi:10.1038/s41586-023-06722-0
Wittemyer, G., Elsen, P., Bean, W.T., Burton, A.C.O. and Brashares, J.S. 2008. Accelerated human population growth at protected area edges. Science 321: 123-126.