Forest Shrew
Myosorex varius

2025 Red list status
Least Concern
Regional Population Trend
Declining
Change compared
to 2016
No Change
Overview
Myosorex varius – (Smuts, 1832)
ANIMALIA – CHORDATA – MAMMALIA – EULIPOTYPHLA – SORICIDAE – Myosorex – varius
Common Names: Forest Shrew (English), Bosskeerbek, Bos-skeerbekmuis (Afrikaans)
Synonyms: No Synonyms
Taxonomic Note:
Significant variation in size is found across its range (Meester et al. 1986). Myosorex varius is comprised of two distinct genetic lineages defined by mitochondrial and nuclear sequences, a northern lineage, comprising individuals from the grassland and savanna biome of Limpopo, Mpumalanga, Gauteng, Free State, KwaZulu-Natal and the Eastern Cape, and a southern fynbos lineage, comprising individuals from the Western and Eastern Cape provinces of South Africa, where there is genetic structuring between the western and eastern areas of the Cape Floristic Region (Willows-Munro and Matthee, 2011). The southern lineage is further separated into a western (winter rainfall areas) and eastern (aseasonal rainfall areas) sublineage. The two lineages diverged about 2 million years ago and most likely represent distinct species. A similar vicariance (splitting) event occurred in rodent genera Rhabdomys and Otomys (Willows-Munro & Matthee, 2011; Taylor et al. 2013).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Taylor, P.1 & da Silva, J.M.2
Reviewer: Russo, I.M.3
Institutions: 1University of the Free State, 2South African National Biodiversity Institute, 3Cardiff University
Previous Assessor: Cassola, F.
Previous Reviewer: Amori, G.
Previous Contributor: Baxter, R.
Assessment Rationale
This endemic species is listed as Least Concern as it has a very wide range within the assessment region, occurring in diverse habitats and many protected areas, and can exist in both intact and agricultural landscapes. It is threatened by ongoing habitat loss and degradation, caused primarily by coastal development, human settlement expansion, forest clear-cutting for agriculture and overgrazing from livestock farming. Although ongoing habitat loss may cause local declines, this is not expected to cause a net population decline for this widespread species in the near future. However, recent molecular work suggests distinct evolutionary lineages corresponding to the Grassland/Savannah and Fynbos biomes, and further molecular and morphological research is needed to resolve the potential species status. Furthermore, recent climate modelling work predicts an ambiguous response to climate change (depending on dispersal capacity), with area of occupancy ranging from a decline of 63–66% by 2050 (from 1975). However, the occupancy of the northern lineage is projected to decline by 23–43% and should be reassessed following taxonomic resolution. The range of the species overall is predicted to shrink in the interior and move towards the coast. However, coastal landscapes are increasingly fragmented by ongoing urban, rural and industrial expansion (for example, urban and rural settlements have expanded by 1.1–8% between 2000 and 2013), which thus represents an outright loss of habitat rather than a range shift. If this species is split into two species pertaining to Grassland/Fynbos evolutionary lineages, it will necessitate reassessment as both (especially the northern lineage) may be threatened by high levels of habitat loss within the contracted ranges.
Key interventions include the protection of forest habitats, and the creation of corridors between patches to facilitate gene flow and allow adaptation to climate change, as well as the enforcement of regulations restricting disturbance to protected forests.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Taylor P & da Silva JM. 2025. A conservation assessment of Myosorex varius. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
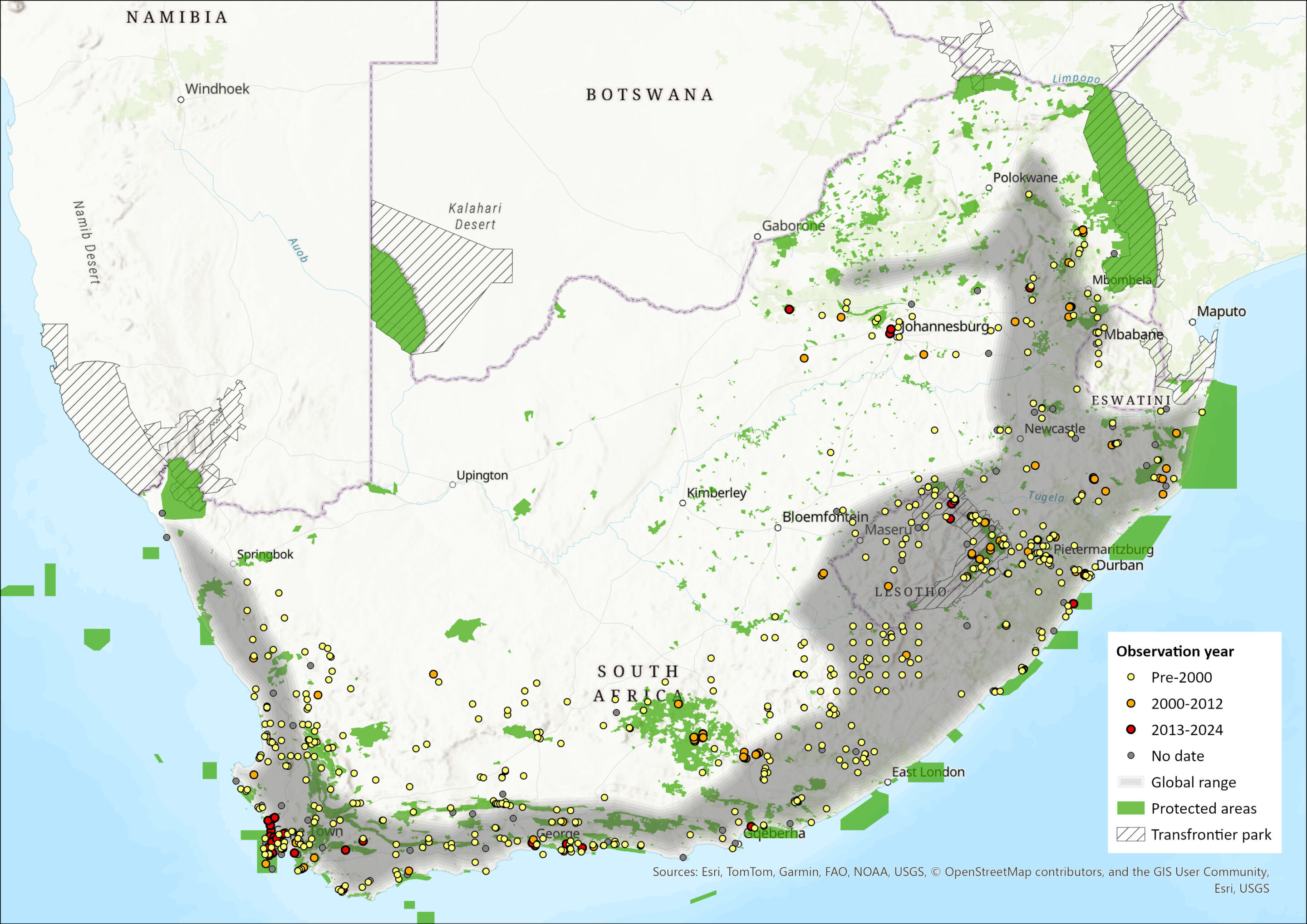
Forest Shrews are endemic to the assessment region, occurring widely across Lesotho, Eswatini and southern South Africa. They occur throughout montane grassland and fynbos habitats in South Africa, including savannah and the Highveld bushveld transition zone (Power 2014), but not in dry areas. It is thought to be a generalist species better able to exist in transformed or agricultural landscapes and better able to tolerate marginal habitats than other Myosorex species (Meester 1958; Skinner & Chimimba 2005), which has enabled them to occupy both the drier fynbos regions of the Western Cape Province and the grasslands of the South African interior. However, although it has been recorded in a number of grassland, forest, savanna, semiarid and fynbos habitats, it is typically found only in cool, moist microhabitats such as river banks or high-mist areas on the west coast of South Africa (Baxter & Dippenaar 2013). It is sympatric with all other Myosorex species. Taylor et al. (2013) established that M. varius does not occur in Limpopo Province, where it is replaced by M. cf. tenuis. Further vetting of museum records pertaining to M. varius in this province will be necessary.
The northern lineage is distributed across the grassland and savannah biomes, from Mpumalanga to Eastern Cape provinces, while the distribution of the southern lineage broadly follows the Cape Fold Mountains of the Western Cape Province (Willows-Munro & Matthee 2011). The contact zone between the two lineages occurs within the Albany thicket region along the Eastern Cape coast, around Port Elizabeth, which corresponds to the meeting point of five biomes (Willows-Munro & Matthee 2011).
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): (Not specified)
Elevation Upper Limit (in metres above sea level): (Not specified)
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Forest Shrew (Myosorex varius) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Eswatini | Extant | Native | – | – |
| Lesotho | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
Climate change is considered to be the principal emerging threat to this species (Ogony 2014), both due to loss of habitat and habitat degradation from drying out of wetlands and because shrews cannot tolerate extremes of temperature for long and thus their foraging time will be reduced. Due to their small size, low dispersal capacity, high metabolism, short life span and sensitivity to temperature extremes, climate change will reduce the amount of suitable habitat available. This is particularly true for the southern lineage that occupies Afromontane grasslands as these areas are likely to become increasingly fragmented.
Climate modelling predicts an ambiguous response to climate change (depending on dispersal capacity), with area of occupancy ranging from a decline of 66% to an increase of 36% by between 1975 and 2050 (Taylor et al. 2016). However, the projected increase is an unlikely scenario due to under-prediction of the model because the species comprises both a northern and southern lineage. Indeed, when the two lineages are modelled separately, model fit is much improved and reveals that the northern (grassland) lineage is more threatened, as it is projected to decline by 23–43%, while the southern (fynbos) lineage shows a mixed response ranging from a decline of 33% to an increase in occupancy.
Predictions show a range shift from high-altitude grasslands towards the coast, but the coast is highly developed and will not result in an occupancy substitution. For example, urban areas have expanded at rates of 5.6–8.6% in KwaZulu-Natal, Eastern and Western Cape provinces between 2000 and 2013 (GeoTerraImage 2015). Thus, unfettered urban expansion, especially coastal development, is likely to synergise with climate change in being the major threat to this species.
Most species within this genus including Myosorex varius are reduced to areas within the interglacial montane refugia (Pahad et al. 2023). This species is therefore likely to be vulnerable to anthropogenic climate change beyond the range of the glacial cycles (Pahad et al. 2023).
Population
In some areas such as the Drakensberg, this species can be the dominant small mammal at higher elevations, with abundance dropping off towards the coast. However, population size in the Kwa-Zulu Natal Drakenberg is reported to fluctuate considerably between drought and non-drought years (Taylor, 1998). Trapping data along an elevational gradient in the Eastern Cape revealed a linear increase in relative abundance (frequency of occurrence in traps) from around 2% at 600 m, 3–8% at 1,500 m to 612% at 1,800 m (Baxter & Dippenaar 2013). Similarly, they were found to be significantly less abundant than M. sclateri in the lowland Dukuduku Forest of KwaZulu-Natal Province (Perrin & Bodbijl 2001). At Seekoeivlei Nature Reserve in Free State Province, it was the third most abundant small mammal sampled, constituting 22% of samples after Crocidura mariquensis (41%) and Rhabdomys pumilio (28%) (Wandrag et al. 2002).
It is an adaptable species, able to adjust reproductive cycles to suit environmental conditions (Baxter 2005). As such, it is thought to be a generalist and thus less affected by habitat fragmentation, which is supported by the lack of population structure in the northern lineage (Willows-Munro & Matthee 2011). However, the varying rainfall regimes and landscape heterogeneity have been shown to correspond to distinct evolutionary lineages (Willows-Munro & Matthee 2011), and, if split into multiple species, the assumption of its generalist nature should be refined.
This may particularly be true for the southern lineage where population structure is apparent between eastern and western populations and which may feel the brunt of fragmentation as suitable habitats shift towards the transformed coast.
Population estimates within each lineage/subpopulation have not been quantified.
Population Information
Current population trend: Declining. Inferred and projected from ongoing habitat loss and degradation.
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No. Thought to be a generalist species that is less affected by habitat fragmentation. However, the southern lineage may be more susceptible to the fragmenting effects of climate change as suitable habitats shift towards the coast.
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
A phylogeographic study based on one mitochondrial and one nuclear marker uncovered substantial genetic structuring within this species across the assessment region (Willows-Munro & Matthee, 2011). Specifically, three genetic clusters were identified: a northern lineage, and two sublineages within the southern Cape Floristic Region (an eastern and western group). Rainfall regimes appear to be the key factor explaining the separation of each lineage. The summer rainfall northern lineage separates from the southern lineage along the dry “Bedford Gap”, that has been identified as an important biogeographic barrier in animal and plant species (Taylor et al. 2024). Similarly, the western CFR sublineage experiences winter rainfall, while the eastern CFR experiences aseasonal rainfall It is not expected that any additional lineages have gone extinct, consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (3/3 subpopulations remaining).
Even though the species is thought to be abundant in parts of its range (e.g., Drakensberg), no estimates are available for each of the subpopulations/lineages or for the species as a whole. Due to this lack of information, it is not possible to quantify the GBF’s headline indicator – proportion of populations with an effective population size ( Ne) > 500. It is highly recommended that a more fine scale population genetic study be undertaken, possibly incorporating samples from Willows-Munro & Matthee (2011) but supplementing them with additional samples. This will enable the quantification of Ne for each subpopulation. It would also be useful in identifying additional genetic structure.
Habitats and ecology
Forest Shrews occur in every biome in South Africa including coastal forests, thickets, grasslands, savannah, Nama Karoo, Succulent Karoo and Fynbos. As such, its name is somewhat a misnomer as it is not restricted to forests (Skinner & Chimimba 2005). It is present in both primary habitats and degraded areas such as suburban gardens where sufficient ground cover occurs (Taylor et al. 1998). For example, it was recently sampled in grasslands (wet and dry), Kikuyu (Pennisetum clandestinum) pastures and disturbed grasslands in Umvoti Vlei Conservancy, Kwazulu-Natal Province (Fuller & Perrin 2001). However, it prefers dense moist grasslands (Rautenbach 1982; Rowe-Rowe & Meester 1982; Taylor 1998; Baxter & Dippenaar 2013), commonly along the banks of rivers or dams. In the KwaZulu-Natal Province, they have also been found in Afromontane and coastal and scarp forests where they may occur in sympatry with M. cafer and M. sclateri. It occurs in coastal mountains of the Eastern, Northern and Western Cape provinces. It is also associated with rocky, high-altitude grassland slopes, occurring at higher altitudes than any other shrew species within the assessment region, from 1,500–2,200 m asl (Rowe-Rowe & Meester 1982, 1985; Lynch 1994). In Dukuduku Forest, KwaZulu-Natal Province, they occurred only in dwarf shrubland (Perrin & Bodbijl 2001).
Captive animals are predominantly nocturnal (Baxter et al. 1979), although they may become predominantly diurnal during winter (Brown et al. 1997). When there is a pair in the nest, they sleep head to tail (Baxter & Meester 1980). They excavate shallow blind tunnels under rocks or other objects, or use existing burrows (such as those from Molerats). They feed predominantly on invertebrates but are also cannibalistic and feed on conspecific and rodent carcasses (Skinner & Chimimba 2005).
Ecosystem and cultural services: This species is an important prey item for the Barn Owl (Tyto alba), the Water Mongoose (Atilax paludimosus), the African Striped Weasel (Poecilogale albimucha) and the Striped Polecat (Ictonyx striatus).
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 1.4. Forest -> Forest – Temperate | Moderately suitable | – | |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 4.4. Grassland -> Grassland – Temperate | – | Suitable | – |
Life History
| Generation Length | Justification | Data Quality |
| 1 | see Pacifici et al. 2013 | medium |
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
There is no known subsistence or commercial use of this species.
General notes regarding trade and use of this species: (Not specified)
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
The main threat this species is the loss or degradation of moist, productive areas such as wetlands and rank grasslands. The two main drivers behind this are abstraction of surface water and draining of wetlands through industrial and residential expansion, and overgrazing of moist grasslands, which leads to the loss of ground cover and decreases small mammal diversity and abundance (Bowland & Perrin 1989). Suppression of natural ecosystem processes, such as fire, can also lead to habitat degradation through bush encroachment or loss of plant diversity through alien invasives, and is suspected to be increasing with human settlement expansion. There are also clear overlaps and synergistic effects between these threats. Shrews have a high metabolic rate and thus rely on highly productive and complex environments, where small mammal diversity is highest (Bowland & Perrin 1993). Wetlands are the most threatened ecosystem within the assessment region: 65% of wetland ecosystem types are threatened (48% Critically Endangered, 12% Endangered and 5% Vulnerable; Driver et al. 2012). Overall, 45% of our remaining wetland area exists in a heavily modified condition, due primarily to onsite modification from crop cultivation, coal mining, urban development, dam construction, and overgrazing (and thus erosion) and off-site modifications from disruptions to flow regime and deterioration of water quality (Driver et al. 2012).
Conservation
The main intervention for this species is the protection and restoration of wetlands and grasslands within and around forest patches. Protected area expansion should attempt to create corridors between high-altitude and coastal habitats. Biodiversity stewardship schemes should be promoted if landowners possess wetlands or grasslands close to core protected areas or remaining forest patches, and the effects on small mammal subpopulations should be monitored. Protecting such habitats may create dispersal corridors between forest patches that will enable adaptation to climate change.
All forests in South Africa are protected by law, although the degree to which they are enforced may vary. Legislation should be enforced to prevent development or human encroachment in key habitats, which includes increased enforcement of forest-related transgressions to minimise disturbance to existing forest patches, as well as stricter zonation on development to decrease fragmentation of remaining forests.
At the local scale, landowners and managers should be educated, encouraged and incentivised to conserve the habitats on which shrews and small mammals depend. Retaining ground cover is the most important management tool to increase small mammal diversity and abundance. This can be achieved through lowering grazing pressure (Bowland & Perrin 1989), or by maintaining a buffer strip of natural vegetation around wetlands (Driver et al. 2012). Research will be needed to set the recommended length of the buffer strip in various habitats, but 500 m may provide a good indication of ecological integrity (Driver et al. 2012). Small mammal diversity and abundance is also higher in more complex or heterogeneous landscapes, where periodic burning is an important tool to achieve this (Bowland & Perrin 1993). Similarly, the specific fire regime thresholds should be calibrated by research. Removing alien vegetation from watersheds, watercourses and wetlands is also an important intervention to improve flow and water quality, and thus habitat quality, for shrews. This can be achieved through the Working for Water programme (for example, Marais et al. 2004). However, the subsequent effects on shrew subpopulations must be monitored to demonstrate success (sensu Richardson & van Wilgen 2004). Education and awareness campaigns should be employed to teach landowners and local communities about the importance of conserving wetlands and moist grasslands.
Recommendations for land managers and practitioners:
- Enforce regulations on developments that potentially impact on the habitat integrity of forests.
- Landowners should be incentivised to stock livestock or wildlife at ecological carrying capacity and to maintain a buffer of natural vegetation around wetlands.
Research priorities:
- Molecular and morphological studies to resolve the taxonomic resolution of the two putative evolutionary lineages.
- Research should be conducted to determine disturbance thresholds in various habitats (for example, ecological stocking rates, amount of natural vegetation needed to sustain a viable subpopulation, and fire intensity and frequency needed to sustain habitat complexity) needed by managers to conserve shrew species.
Encouraged citizen actions:
- Citizens are requested to submit any shrews killed by cats or drowned in pools to a museum or a provincial conservation authority for identification, thereby enhancing our knowledge of shrew distribution (carcasses can be placed in a ziplock bag and frozen with the locality recorded).
Bibliography
Baxter R, Dippenaar N. 2013. Myosorex varius (Smuts). Forest Shrew. Pages 161–163 in Happold M, Happold DCD, editors. Mammals of Africa. Bloomsbury Publishing, New York, USA.
Baxter RM. 2005. Variation in aspects of the population dynamics of the endemic forest shrew Myosorex varius in South Africa. Pages 181–190 in Merritt JJ, Churchfield S, Shefttel B, editors. Advances in the Biology of Shrews II. Carnegie Museum of Natural History, Pittsburgh, USA.
Baxter RM, Goulden EA, Meester J. 1979. Activity patterns of Myosorex varius and M. cafer in captivity. South African Journal of Zoology 14:91–93.
Baxter RM, Meester J. 1980. Notes on the captive behaviour of five species of southern African shrews. Säugetierkundliche Mitteilungen 26:55–62.
Bowland AE, Perrin MR. 1989. The effect of overgrazing on the small mammals in Umfolozi Game Reserve. Zeitschrift für Säugetierkunde 54:251–260.
Bowland JM, Perrin MR. 1993. Wetlands as reservoirs of small mammal populations in the Natal Drakensberg. South African Journal of Wildlife Research 23:39–43.
Brown CR, Hunter EM, Baxter RM. 1997. Metabolism and thermoregulation in the forest shrew Myosorex varius (Soricidae: Crocidurinae). Comparative Biochemistry and Physiology Part A: Physiology 118:1285–1290.
Driver A, Sink KJ, Nel JN, Holness S, Van Niekerk L, Daniels F, Jonas Z, Majiedt PA, Harris L, Maze K. 2012. National Biodiversity Assessment 2011: An Assessment of South Africa’s Biodiversity and Ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Fuller JA, Perrin MR. 2001. Habitat assessment of small mammals in the Umvoti Vlei Conservancy, KwaZulu-Natal, South Africa. South African Journal of Wildlife Research 31:1–12.
GeoTerraImage. 2015. Quantifying settlement and built-up land use change in South Africa.
Heim de Balsac, H. and Meester, J. 1977. Order Insectivora. In: J. Meester and H.W. Setzer (eds), The Mammals of Africa: An Identification Manual, pp. 1-29. Smithsonian Institution Press, Washington, D. C., USA.
Hutterer, R. 2005. Order Soricomorpha. In: D.E. Wilson and D.M. Reeder (eds), Mammal Species of the World, pp. 220-311. Johns Hopkins University Press, Baltimore, Maryland, USA.
IUCN. 2016. The IUCN Red List of Threatened Species. Version 2016-3. Available at: www.iucnredlist.org. (Accessed: 07 December 2016).
IUCN. 2017. The IUCN Red List of Threatened Species. Version 2017-1. Available at: www.iucnredlist.org. (Accessed: 27 April 2017).
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
Marais C, van Wilgen BW, Stevens D. 2004. The clearing of invasive alien plants in South Africa: a preliminary assessment of costs and progress. South African Journal of Science 100:97103.
Meester J. 1958. Variation in the shrew genus Myosorex in southern Africa. Journal of Mammalogy 39:325–339.
Meester JA, Rautenbach IL, Dippenaar NJ, Baker CM. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5:1–359.
Ogony OL. 2014. Potential impacts of climate change on Mysorex species as a model for extinction risk of montane small mammals in South Africa. M.Sc. Thesis, University of Venda, Thoyandou, South Africa.
Pacifici, M., Santini, L., Di Marco, M., Baisero, D., Francucci, L., Grottolo Marasini, G., Visconti, P. and Rondinini, C. 2013. Generation length for mammals. Nature Conservation 5: 87–94.
Pahad, G., Jansen van Vuuren, B., and Montgelard, C. 2023. Palaeoclimatic niche modelling reveals the phylogeogrphic history of the South African shrew Myosorex varius and the colonisation route of the genus Myosorex (Mammalia, Soricidae) from East Afrcia. Mammalian Biology, 103: 579-590.
Perrin MR, Bodbijl T. 2001. Habitat selection and small mammal prey availability of the gaboon adder in Zululand (KwaZulu-Natal), South Africa. South African Journal of Wildlife Research 31:115126.
Power RJ. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng, South Africa.
Rathbun, G.B. (subeditor). 2005. Macroscelidea. In: J.D. Skinner and C.T. Chimimba (eds), The Mammals of the Southern African Subregion, 3rd edition, pp. 22-34. Cambridge University Press, Cambridge, UK.
Rautenbach IL. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph. Pretoria, South Africa.
Richardson DM, van Wilgen BW. 2004. Invasive alien plants in South Africa : how well do we understand the ecological impacts? South African Journal of Science 100:45–52.
Rowe-Rowe DT, Meester J. 1982. Habitat preferences and abundance relations of small mammals in the Natal Drakensberg. South African Journal of Zoology 17:202–209.
Rowe-Rowe DT, Meester J. 1985. Altitudinal variation in external measurements of two small-mammal species in the Natal Drakensberg. Annals of the Transvaal Museum 34:49–53.
Skinner JD, Chimimba CT. 2005. The Mammals of the Southern African Subregion. Third edition. Cambridge University Press, Cambridge, UK.
Taylor, P. J. 1998. The smaller mammals of KwaZulu-Natal. University of Natal Press, Pietermaritzburg.
Taylor, P. J., Kearney, T. C., Kerbis Peterhans, J. C., Baxter, R. M. & Willows-Munro, S. 2013. Cryptic diversity in forest shrews of the genus Myosorex from southern Africa, with the description of a new species and comments on M. tenuis. Zoological Society of the Linnean Society 169: 881-902, https://doi.org/10.1111/zoj.12083https://doi.org/10.1111/zoj.12083.
Taylor, P. J., Ogony, L., Ogola, J., Baxter, R. M. 2017. South African mouse shrews (Myosorex) feel the heat: using species distribution models SDMs and IUCN Red List criteria to flag extinction risks due to climate change and habitat loss. Mammal Research, 62:149-162. DOI 10.1007/s13364-016-0291-z.
Taylor, P. J., Kearney, T. C., Clark, V. R., Howard, A., Mdluli, M. V., Markotter, W. Geldenhuys, M., Richards, L. R., Rakotoarivelo, A. R., Watson, J., Balona, J., and Monadjem, A. 2024. Southern Africa’s Great Escarpment as an amphitheater of climate-driven diversification and a buffer against future climate change in bats, Global Change Biology, 30: e17344, https://doi.org/10.1111/gcb.17344https://doi.org/10.1111/gcb.17344
Wandrag GF, Watson JP, Collins NB. 2002. Rodent and insectivore species diversity of Seekoeivlei Provincial Nature Reserve, Free State province, South Africa. South African Journal of Wildlife Research 32:137–143.
Willows-Munro S, Matthee CA. 2011. Linking lineage diversification to climate and habitat heterogeneity:
phylogeography of the southern African shrew Myosorex varius. Journal of Biogeography 38: 1976–1991.