
Flat-headed Free-tail Bat
Sauromys petrophilus

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Sauromys petrophilus – (Roberts, 1917)
ANIMALIA – CHORDATA – MAMMALIA – CHIROPTERA – MOLOSSIDAE – Sauromys – petrophilus
Common Names: Roberts’s Flat-headed Bat, Flatheaded Free-tailed Bat, Rock-dwelling Flat-headed Bat, Rock-loving Flat-headed Bat (English), Platkoplosstertvlermuis (Afrikaans)
Synonyms: Mormopterus petrophilus (Roberts, 1917); Sauromys petrophilis Roberts, 1917 ssp. haagneri; Sauromys petrophilis Roberts, 1946 ssp. erogensis; Sauromys petrophilis Roberts, 1946 ssp. fitzsomonsi; Sauromys petrophilis Shortridge & Carter, 1938 ssp. umbratus
Taxonomic Note:
Initially, this species was described as Platymops petrophilus (Skinner & Chimimba 2005; Cotterill 2013). The family Molossidae was revised by Peterson (1965), and the genus Platymops was geographically restricted to the East African region, while those species from southern Africa were included under the genus Sauromys (Skinner & Chimimba 2005; Monadjem et al. 2010). Although Platymops and Sauromys were previously listed as subgenera under the genus Mormopterus (Freeman 1981; Koopman 1993), following Meester et al. (1986), Jacobs & Fenton (2001), Bronner et al (2003), Skinner & Chimimba (2005) and Monadjem et al. (2010), we consider Sauromys a distinct genus. ACR (2015) lists four subspecies, including S. p. erongensis (Roberts 1946) from Namibia, S. p. umbratus (Shortridge & Carter 1938) from the Western Cape, S. p. haagneri (Roberts 1917) from Namibia and the Northern Cape, and S. p. petrophilus (Roberts 1917) from the Limpopo Province, Botswana, Zimbabwe and Mozambique. However, there is doubt as to their validity (Cotterill 2013). The distribution of this species is fragmented and may have led to genetic isolation of principal populations where the western population is separated from the eastern one by 800 km and future studies may well show these to be genetically distinct (Monadjem et al. 2020).
Red List Status: LC – Least Concern (IUCN version 3.1)
Assessment Information
Assessors: Lötter, C.A.1, Balona, J.2 & da Silva, J.M.3
Reviewer: Richardson, E.J.4
Institutions: 1Inkululeko Wildlife Services (Pty) Ltd, 2Gauteng and Northern Regions Bat Interest Group, 3South African National Biodiversity Institute, 4Independent Consultant at Richardson & Peplow Environmental
Previous Assessors and Reviewers: Jacobs, D., MacEwan, K., Cohen, L., Monadjem, A., Richards, L, Schoeman, C., Sethusa, T. & Taylor, P.
Previous Contributors: Nicholson, S.K., Relton, C. & Raimondo, D.
Assessment Rationale
Roberts’ Flat-headed Bat is listed as Least Concern in view of its wide distribution (extent of occurrence in the assessment region is 809,610 km2), presumed large population, and presence in many protected areas. Long-term passive acoustic bat call monitoring data from various proposed and operational wind farm sites in southern Africa confirms that although this species is widespread in the assessment region, it is closely associated with rocky terrain, where this species’ percentage contribution to all recorded bat calls is low to moderate (0-30%; Inkululeko Wildlife Services unpubl. data). With the rapidly growing number of operational and proposed wind (and solar) farms in the western half of the assessment region, from the Western Cape northwards along the west coast up through Namibia, and eastwards along the Orange River, this could pose a serious threat to this species. Like other aerial-foraging Molossid species, Roberts’s Flat-headed Bat is categorized as having a “High” risk of fatality from wind turbines (MacEwan et al. 2020). If/When there are operational wind farms within the distribution range of this species, fatalities of this species must be carefully monitored and adequately mitigated. If not, this species, like the similar Egyptian Free-tailed Bat (Tadarida aegyptiaca) will likely be heavily impacted by wind farm development (Aronson 2022). Climate change may also present an emerging threat to this species, due to its occurrence predominantly in arid and semi-arid areas where water and food can be scarce, and extreme heat may lead to fatal dehydration. Future work should focus on resolving the taxonomy of this taxon, quantifying and mitigating fatalities of this species at operational wind farms and estimating the population size and trend of this taxon.
Regional population effects: Although the distribution of this species is somewhat patchy through southern Africa, dispersal is possible considering its high wing-loading (Jacobs & Fenton 2001) and its occurrence in transfrontier parks, such as Ai-Ais Richtersveld Transfrontier Park and the Greater Mapungubwe Transfrontier Conservation Area.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Lötter CA, Balona J & da Silva JM. 2025. A conservation assessment of Sauromys petrophilus. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
The species is widely but patchily distributed throughout southern Africa ranging along the west coast from southern Angola through Namibia, south into South Africa. In South Africa, its distribution extends southwards into the Western Cape Province, as well as eastwards along the northern border of South Africa, extending marginally into southern Botswana (Cotterill 2013) and into Zimbabwe and western Mozambique (Monadjem et al. 2020). It is inexplicably absent from northwest Zimbabwe where habitat is thought to be suitable (Monadjem et al. 2020). It is abundant in the drier western regions of Namibia and South Africa (Skinner & Chimimba 2005) and is generally found between 100 m and 2,000 m asl. In South Africa its range is discontinuous, with records from the following provinces and districts: Limpopo, Mpumalanga, the Pretoria area of Gauteng, Western Cape (Clanwilliam and Ceres) and the Northern Cape (e.g. Goodhouse, Augrabies Falls, Groblershoop, and Danielskuil) (Monadjem et al. 2020; Inkululeko Wildlife Services unpubl. data). This species is also present within the Greater Mapungubwe Transfrontier Conservation Area, as well as the Ai-Ais Richtersveld Transfrontier Park (Skinner & Chimimba 2005; Monadjem et al. 2020). The type specimen is from the North West Province, South Africa (Monadjem et al. 2020). The distribution of this species is fragmented owing to the patchy nature of the rocky outcrops in which it roosts (Jacobs and Fenton 2001). The estimated extent of occurrence in the assessment region is 809,610 km2.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 100
Elevation Upper Limit (in metres above sea level): 2000
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
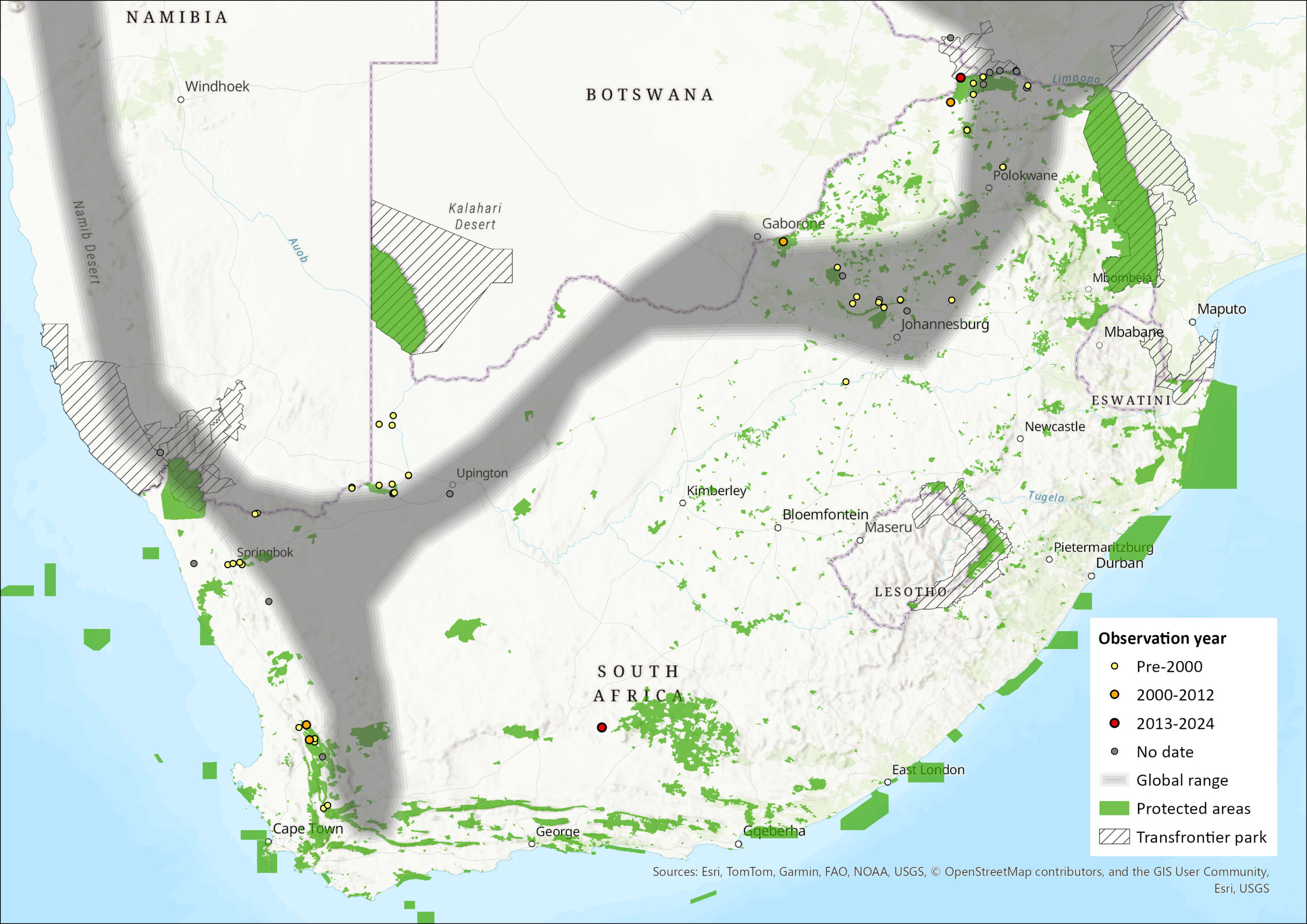 Figure 1. Distribution records for Flatheaded Free-tailed Bat (Sauromys petrophilus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Figure 1. Distribution records for Flatheaded Free-tailed Bat (Sauromys petrophilus) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
In a review study on the impact and implications of climate change for bats, Sherwin et al. (2012) identified seven risk factors for bats encompassing three broad aspects: 1) biogeography – small range size, high latitude or high altitude range and a range occupying a geographic area likely to become water stressed; 2) foraging niche – frugivory and species restricted to aerial hawking; and 3) dispersal ability – species with restricted dispersal behaviour. These identified risk factors and similar research (Adams and Hayes 2021) suggest that S. petrophylis may be impacted if, for example, climate change reduces the availability of water and insect prey in the arid and semi-arid areas where this species predominantly occurs. Furthermore, S. petrophilus was found to exhibit rapid spikes in evaporative water loss during conditions of high ambient temperature (Toussaint & McKechnie 2012). Thus, this species may be especially vulnerable to fatal dehydration during periods of extreme heat (Toussaint & McKechnie 2012; ACR 2015).
Population Information
Roberts’ Flat-headed Bat is locally common in some areas, particularly the arid, western regions of southern Africa (Jacobs & Fenton 2001; Monadjem et al. 2010; ACR 2015), or where roosting sites are abundant, such as the Limpopo Valley and the Cederberg, but rare elsewhere (Cotterill 2013). Generally, this species occurs in colonies consisting of tens of individuals rather than hundreds (Rautenbach 1982; ACR 2015).
Current population trend: Stable
Continuing decline in mature individuals: No
Extreme fluctuations in the number of subpopulations: (Not specified)
Continuing decline in number of subpopulations: (Not specified)
All individuals in one subpopulation: (Not specified)
Number of mature individuals in largest subpopulation: (Not specified)
Number of Subpopulations: (Not specified)
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
To date, a population genetic/genomic study has not been undertaken on the species, yet it would prove invaluable in identifying any genetically distinct units/subpopulations within the assessment region and in verifying possible subspecies. The distribution of this species appears to be fragmented beyond the presumed dispersal capabilities of the species, which could suggest genetic isolation (Monadjem et al. 2020). Based on known records (see Map), there may be at least five distinct subpopulations within the assessment region. It is not possible to estimate the size of each given the limited data available for the various colonies. Both population structure and size should be investigated using more widespread and fine-scale molecular markers.
Habitats and ecology
Occurs in both wet and dry woodlands (including miombo and mopane), shrublands and Acacia-wooded grasslands always in areas with rocky outcrops and hills, especially of Karoo sandstones and granitic intrusions (Cotterill 2013). This species is closely associated with rocky habitats, usually in dry woodland, mountain fynbos or arid scrub (Monadjem et al. 2020). In the assessment region, the species is recorded from the Mopane Bioregion, Central Bushveld, Lowveld, Gariep Desert, Bushmanland, Northwest Fynbos, Namaqualand Hardeveld, and Namaqualand Sandveld. In the Western Cape, it utilises open areas surrounding artificial wetlands (Sirami et al. 2013). Generally restricted to rocky areas, the essential habitat requirements of this species include the presence of narrow rock crevices and fissures for daytime roosting, as well as the availability of an adequate insect food supply (Skinner & Chimimba 2005).
It roosts in crevices, narrow cracks, exfoliated rock, sandstones and granite (Jacobs & Fenton 2001; Monadjem et al. 2020). Its extremely flattened skull is an adaptation to roosting in the narrowest of rock crevices (Jacobs & Fenton 2002). Roosts are made up of small numbers; most records average a roost size of four (Skinner & Chimimba 2005).
Long-term passive acoustic bat call monitoring data from various proposed and operational wind farm sites in southern Africa confirms that although this species is widespread in the assessment region, it is closely associated with rocky terrain, where this species’ percentage contribution to all recorded bat calls is low to moderate (0-30%; Inkululeko Wildlife Services unpubl. data).
The species is an open-air forager with a diet consisting mainly of Diptera, Hemiptera and Coleoptera (Monadjem et al. 2020). The reproductive ecology of this species is largely unknown. However, in Zimbabwe, a pregnant and lactating female was recorded in mid-November (Monadjem et al. 2020).
Ecosystem and cultural services: As this species is insectivorous, it may contribute to controlling insect populations (Boyles et al. 2011; Kunz et al. 2011). Bats often prey on the insect species that destroy crops (Boyles et al. 2011; Kunz et al. 2011): ensuring a healthy population of insectivorous bats can thus result in a decrease in the use of pesticides.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 3.8. Shrubland -> Shrubland – Mediterranean-type Shrubby Vegetation | – | Suitable | – |
| 6. Rocky areas (e.g. inland cliffs, mountain peaks) | – | Suitable | Yes |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: Forearm mean = 3.83 cm (Monadjem et al. 2020)
Size at Maturity (in cms): Male: Forearm mean = 3.95 cm (Monadjem et al. 2020)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Breeding Strategy
| Does the species lay eggs? |
| No |
| Does the species give birth to live young |
| Yes |
| Does the species exhibit parthenogenesis |
| No |
| Does the species have a free-living larval stage? |
| No |
| Does the species require water for breeding? |
| No |
Movement Patterns
Movement Patterns: Unknown
Congregatory: Roosts communally in small groups of up to 10 individuals (Rautenbach 1982 cited in Monadjem et al. 2020).
Systems
System: Terrestrial
General Use and Trade Information
There is no evidence to suggest that this species is traded or harvested within the assessment region.
Local Livelihood: (Not specified)
National Commercial Value: (Not specified)
International Commercial Value: (Not specified)
End Use: (Not specified)
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
Due to the specific roosting requirements, patchy occurrence and small colony sizes of this species, it may be significantly impacted by disturbances that result in degradation or loss of roosting habitat and/or high mortality of individuals. Mining and renewable energy development, in particular, may post a serious threat to colonies or populations in rocky regions. Where wind farms have or will involve construction of turbines on rocky ridges, hills and mountain tops, any associated colonies of Roberts’s Flat-headed Bat could be significantly impacted or even lost by roost disturbance (from e.g. blasting or excavation activities) and/or turbine-related fatality or displacement of individuals. Based on wing morphology and its open-air foraging behaviour, Roberts’s Flat-headed Bat has been categorized as having a “High” risk of fatality from turbines (MacEwan et al. 2020).
The rapidly growing number of operational and proposed wind (and solar) farms in southern Africa could, therefore, pose a serious threat to this species – particularly in the western half of the assessment region, from the Western Cape northwards along the west coast up through Namibia, and eastwards along the Orange River. At least one carcass of this species was reported by Aronson (2022) from data that were collated from 25 operational wind farms in South Africa for the period 2011-2020, and additional turbine-fatalities of this species may have occurred. Additionally, climate change may present an emerging threat to this species due to its occurrence predominantly in arid and semi-arid areas, where water and food can be scarce, and extreme heat may lead to fatal dehydration (Toussaint & McKechnie 2012; ACR 2015).
Conservation
This species is present in several protected areas in Namibia and Angola. In South Africa, it has been reported from the following protected areas: the Greater Mapungubwe Transfrontier Conservation Area, the Ai- Ais Richtersveld Transfrontier Park, the Algeria forestry station, Blouberg Nature Reserve, Baobab Tree Reserve, Kruger National Park, and Augrabies Falls National Park.
To mitigate mortalities of this species from turbine collisions on wind farms, development of wind farms must avoid encroachment into the prescribed buffers around confirmed and potential roosts of this species in suitable rocky habitat (for buffer recommendations see MacEwan et al. 2020 or later). Turbine-fatalities of this species must be reduced below fatality threshold values (as calculated according to MacEwan et al. 2018 or later) with interventions such as: i) ultrasound to deter bats (Weaver et al. 2020); ii) blanket curtailment of turbines at low wind speeds during seasons and hours of the night when this species is most active (Berthinussen et al. 2010; Arnett et al. 2011; Hayes et al. 2019); iii) acoustic smart curtailment of turbines in response to real-time bat activity (https://www.wildlifeacoustics.com/smart-system) ; and/or optimized smart curtailment of turbines during predicted periods of high bat activity based statistical modelling (https://west-inc.com/wp-content/uploads/2024/04/Optimized-Smart-Curtailment-SOQ_FINAL.pdf). As high bat fatalities at wind farms cannot be easily offset (Aronson et al. 2018; Mark Botha pers. comm.), avoidance and minimisation of bat fatalities is critical.
Recommendations for land managers and practitioners:
- Development of wind farms must avoid encroachment into the prescribed buffers around confirmed roosts and suitable rocky habitat (e.g. ridges, gorges, cliff faces, batholiths, etc.) where roosts of this species occur.
- Turbine-fatalities of this species must be reduced with interventions such as ultrasound to deter bats and curtailing turbines at low wind speeds.
- Data sharing by wind farm managers into a national database is needed to be able to calculate cumulative impacts and thereafter implement collaborative mitigation and management efforts.
- Reduce pesticide use in agricultural landscapes.
Research priorities:
- Taxonomic resolution through molecular and morphometric research is needed.
- Further surveys are needed to clarify the distribution of this species.
- Wind farm carcasses of this species could be used to obtain greater insight into the biogeography, genetics, reproductive biology, ecology (e.g. diet), and ecosystem services of this species in eastern southern Africa.
- Systematic monitoring of subpopulation trends is required.
Bibliography
ACR. 2015. African Chiroptera Report 2015. Pretoria, South Africa.
Adams, R.A. and Hayes, M.A. 2021. The importance of water availability to bats: climate warming and increasing global aridity. 50 years of bat research: foundations and new frontiers, pp.105-120.
Arnett, E.B., Huso, M.M., Schirmacher, M.R., Hayes, J.P. 2011. Altering turbine speed reduces bat mortality at wind-energy facilities. Frontiers in Ecology and the Environment 9: 209–214.
Aronson, J. 2022. Current state of knowledge of wind energy impacts on bats in South Africa. Acta Chiropterologica 24: 221-238.
Baerwald, E.F., D’Amours, G.H., Klug, B.J. and Barclay, R.M. 2008. Barotrauma is a significant cause of bat fatalities at wind turbines. Current Biology 18: 695–696.
Berthinussen, A., Richardson, O.C. and Altringham, J.D. 2010. Bat Conservation: Global Evidence for the Effects of Interventions. Synopses of Conservation Evidence. Pelagic Publishing, Exeter.
Boyles, J.G., Cryan, P.M., McCracken, G.F. and Kunz, T.H. 2011. Economic importance of bats in agriculture. Science 332: 41–42.
Bronner, G.N., Hoffmann, M., Taylor, P.J., Chimimba, C.T., Best, P., Mathee, C.A. and Robinson, T.J. 2003. A revised systematic checklist of the extant mammals of the southern African subregion. Durban Museum Novitates 28: 56–106.
Cotterill, F.P.D. 2013. Sauromys petrophilus Robert’s Flat-headed Bat. Pages 486–487 in Happold M, Happold DCD, editors. Mammals of Africa. Volume IV: Hedgehogs, Shrews and Bats. Bloomsbury Publishing, London, UK.
Festa, F., Ancillotto, L., Santini, L., Pacifici, M., Rocha, R., Toshkova, N., Amorim, F., Benítez‐López, A., Domer, A., Hamidović, D. and Kramer‐Schadt, S. 2023. Bat responses to climate change: a systematic review. Biological Reviews, 98(1), pp.19-33.
Freeman, P.W. 1981. A multivariate study of the family Molossidae (Mammalia, Chiroptera): morphology, ecology, evolution. Fieldiana:. Zoology, new series 7: 1–173.
Hayes, M.A., Hooton, L.A., Gilland, K.L., Grandgent, C., Smith, R.L., Lindsay, S.R., Collins, J.D., Schumacher, S.M., Rabie, P.A., Gruver, J.C., and Goodrich-Mahoney, J. 2019. A smart curtailment approach for reducing bat fatalities and curtailment time at wind energy facilities. Ecological Applications, 0(0), 2019, e01881.
Jacobs, D.S. and Fenton, M.B. 2001. The status of Sauromys petrophilus and Chaerephon pumilus (Chiroptera: Molossidae) in the Western Cape Province of South Africa. African Zoology 36: 129–136.
Jacobs, D.S. and Fenton, M.B. 2002. Mormopterus petrophilus. Mammalian Species 703: 1–3.
Koopman, K.F. 1993. Order Chiroptera. Pages 137–241 in Wilson DE, Reeder DM, editors. Mammal Species of the World: Smithsonian Institution Press, Washington, DC, USA.
Kunz, T.H., Braun de Torrez, E., Bauer, D., Lobova, T. and Fleming, T.H. 2011. Ecosystem services provided by bats. Annals of the New York Academy of Sciences 1223: 1–38.
MacEwan, K. 2011. Richtersveld Wind Farm: Bat Assessment Report. Natural Scientific Services, Johannesburg, South Africa.
MacEwan, K.L. 2016. Fruit bats and wind turbine fatalities in South Africa. African Bat Conservation News 42: 3–5.
MacEwan, K., Aronson, J., Richardson, K., Taylor, P., Coverdale, B., Jacobs, D., Leeuwner, L., Marais, W. and Richards, L. 2018. South African Bat Fatality Threshold Guidelines. Edition 2. South African Bat Assessment Association, South Africa.
MacEwan, K., Sowler, S., Aronson, J. and Lötter, C. 2020. South African Best Practice Guidelines for Pre-construction Monitoring of Bats at Wind Energy Facilities. Edition 5. South African Bat Assessment Association, South Africa.
Meester, J.A.J., Rautenbach, I.L., Dippenaar, N.J. and Baker, C.M. 1986. Classification of southern African mammals. Transvaal Museum Monographs 5: 1–359.
Monadjem, A., Taylor, P.J., Cotterill, F.P.D. and Schoeman M.C. 2020. Bats of Southern and Central Africa: a biogeographic and taxonomic synthesis. 2nd Edition. University of Witwatersrand Press, Johannesburg.
Peterson, R.L. 1965. A review of the flat-headed bats of the family Molossidae from South America and Africa. Contributions of the Life Sciences Division, Royal Ontario Museum 64: 1–32.
Rautenbach, I.L. 1982. Mammals of the Transvaal. No. 1, Ecoplan Monograph, Pretoria, South Africa.
Sherwin, H.A., Montgomery, W.I. and Lundy, M.G. 2013. The impact and implications of climate change for bats. Mammal Review 43: 171–182.
Sherwin, H.A., Montgomery, W.I., and Lundy, M.G. 2012. The impact and implications of climate change for bats. Mammal Review. 2012. 10.1111/j.1365-2907.2012.00214.x.
Sirami, C., Jacobs, D.S. and Cumming, G.S. 2013. Artificial wetlands and surrounding habitats provide important foraging habitat for bats in agricultural landscapes in the Western Cape, South Africa. Biological Conservation 164: 30–38.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Toussaint, D.C. and McKechnie, A.E. 2012. Interspecific variation in thermoregulation among three sympatric bats inhabiting a hot, semi-arid environment. Journal of Comparative Physiology B 182: 1129–1140.
Weaver, S.P., Hein, C.D., Simpson, T.R., Evans, J.W., and Castro-Arellano, I. Ultrasonic acoustic deterrents significantly reduce bat fatalities at wind turbines. Global Ecology and Conservation 24: e01099
