Fat Mouse
Steatomys pratensis

2025 Red list status
Least Concern
Regional Population Trend
Stable
Change compared
to 2016
No Change
Overview
Steatomys pratensis – Peters, 1846
ANIMALIA – CHORDATA – MAMMALIA – RODENTIA – NESOMYIDAE – Steatomys – pratensis
Common Names: Fat Mouse (English), Vetmuis (Afrikaans), Thilivhe (Tshivenda), Thanyani (Xitsonga), Igundane (Zulu)
Synonyms: S.edulis, leucorhynchus, maunensis, natalensis, nyasae
Taxonomic Note: Similar to Steatomys krebsii, this species requires urgent taxonomic revision. Currently its range is thought to extend from its southern limit in KwaZulu-Natal northwards to Kenya, although Monadjem et al. (2015) suggests that this northernmost specimen may in fact represent the discrete species, S. parvus. Additionally, a record from Ethiopia, identified by Musser & Carleton (1993) as S. pratensis from an owl pellet, warrants further examination (Happold 2013). Although there is some overlap in size, this species is generally larger than S. krebsii, and has 10–14 nipples, compared to eight in S. krebsii (Monadjem et al. 2015). However, such taxonomic issues are unlikely to affect the South African species. Chromosome number: 2n = 68 (Robbins & Baker 1978).
Red List Status: LC – Least Concern, (IUCN version 3.1)
Assessment Information
Assessors: Smith, C.1 & da Silva, J.M.2
Reviewer: Kraai, M.3
Institutions: 1Endangered Wildlife Trust,2South African National Biodiversity Institute,3Sol Plaatje University
Previous Assessors & Reviewers: Shoeman, C., Relton, C., Harvey, J. & Mondajem, A.
Previous Contributors: Child, M.F., Avenant, N., Avery, M., Baxter, R., MacFadyen, D., Palmer, G., Taylor, P. & Wilson, B.
Assessment Rationale
Based on its wide distribution within the assessment region, presence within various protected areas, the fact that it is fairly regularly trapped, and the lack of major threats to this species, we list S. pratensis as Least Concern. There appears to be no population decline of this species. It is able to survive successfully in human-modified habitats, for example, fallow fields and frequently inhabits agricultural lands.
Regional population effects: Rescue effects are possible across Botswana, Zimbabwe and Mozambique. The population is not isolated, and its range is continuous throughout the savanna regions of southern Africa. Dispersal is possible both through agricultural lands and transfrontier reserves.
Reasons for Change
Reason(s) for Change in Red List Category from the Previous Assessment: No change
Red List Index
Red List Index: No change
Recommended citation: Smith C & da Silva JM. 2025. A conservation assessment of Steatomys pratensis. In Patel T, Smith C, Roxburgh L, da Silva JM & Raimondo D, editors. The Red List of Mammals of South Africa, Eswatini and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa.
Regional Distribution and occurrence
Geographic Range
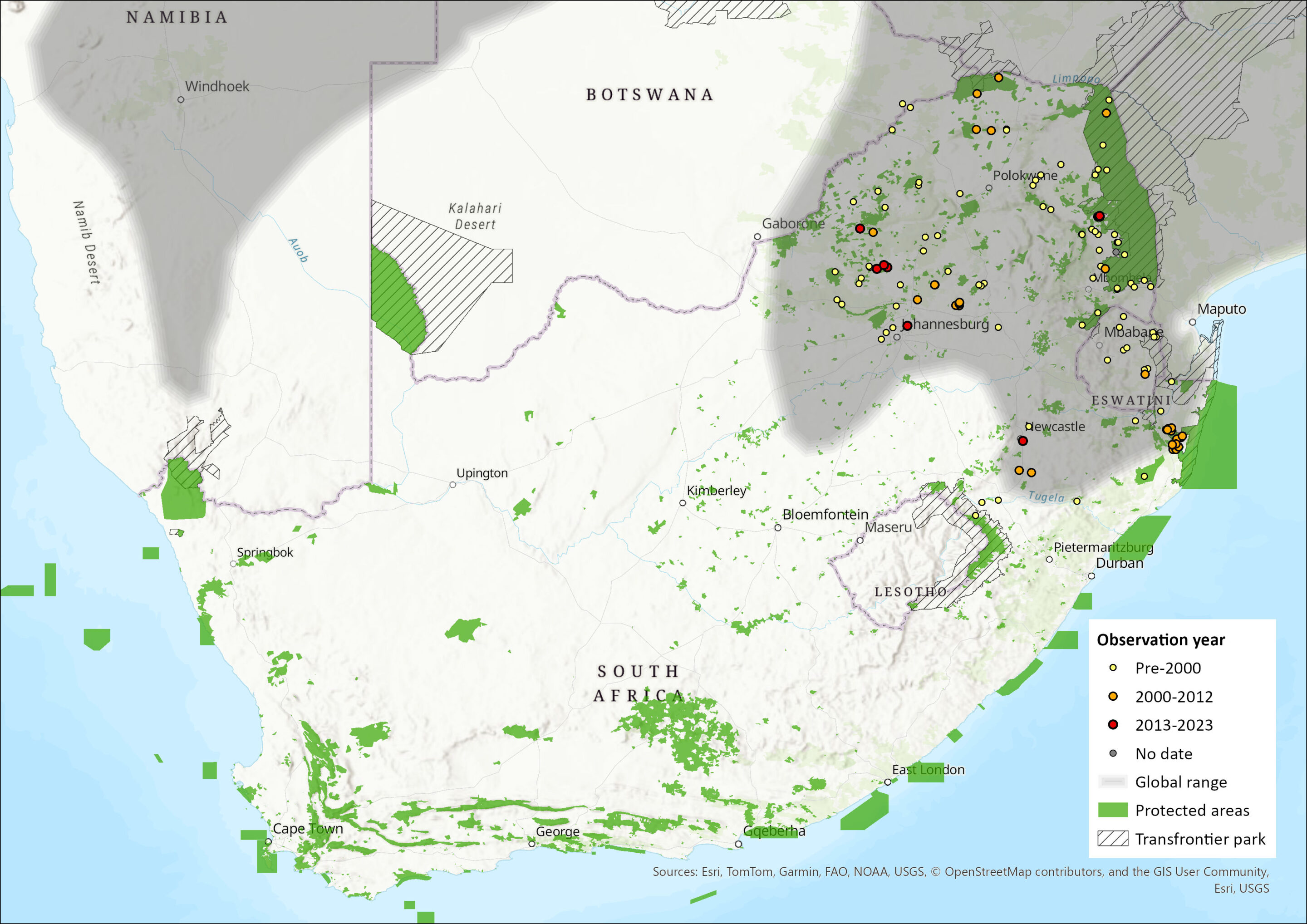
Although the extent of its distribution remains under scrutiny, the range of the Fat Mouse is currently considered to stretch widely across the savannas of southern Africa, and marginally into East Africa. The records from Kenya and Ethiopia may, however, represent misidentified specimens of S. parvus (Happold 2013; Monadjem et al. 2015). In southern Africa, this species has been recorded across northern Namibia and Botswana, throughout Zimbabwe, into parts of Mozambique, extensively into northeastern South Africa, and across eSwatini but not Lesotho (Lynch 1994). In South Africa, it has a relatively wide distribution across the Limpopo, Gauteng, North West, Mpumalanga and KwaZulu-Natal provinces (Skinner & Chimimba 2005). In the North West Province, this species is restricted to the northeastern bushveld regions (Power 2011), and its distribution probably does not extend further west than Pilanesberg National Park (Rautenbach 1982; Power 2014), although Monadjem et al. (2015) show a record more westerly on the border with Botswana.
Elevation / Depth / Depth Zones
Elevation Lower Limit (in metres above sea level): 0
Elevation Upper Limit (in metres above sea level): 1500
Depth Lower Limit (in metres below sea level): (Not specified)
Depth Upper Limit (in metres below sea level): (Not specified)
Depth Zone: (Not specified)
Map
Figure 1. Distribution records for Fat Mouse (Steatomys pratensis) within the assessment region (South Africa, Eswatini and Lesotho). Note that distribution data is obtained from multiple sources and records have not all been individually verified.
Biogeographic Realms
Biogeographic Realm: Afrotropical
Occurrence
Countries of Occurrence
| Country | Presence | Origin | Formerly Bred | Seasonality |
| Angola | Extant | Native | – | – |
| Botswana | Extant | Native | – | – |
| Cameroon | Extant | Native | – | – |
| Central African Republic | Extant | Native | – | – |
| Congo, The Democratic Republic of the | Extant | Native | – | – |
| Eswatini | Extant | Native | – | – |
| Malawi | Extant | Native | – | – |
| Mozambique | Extant | Native | – | – |
| Namibia | Extant | Native | – | – |
| South Africa | Extant | Native | – | – |
| Sudan | Extant | Native | – | – |
| Tanzania, United Republic of | Extant | Native | – | – |
| Zambia | Extant | Native | – | – |
| Zimbabwe | Extant | Native | – | – |
Large Marine Ecosystems (LME) Occurrence
Large Marine Ecosystems: (Not specified)
FAO Area Occurrence
FAO Marine Areas: (Not specified)
Climate change
No recent research or literature has focused on how climate change might affect this species in the future. Climate change driven wildfires in the Okavango Delta are expected to increase microhabitat loss for small mammals including the fat mouse (Kapuka 2021).
Population Information
Although no population estimates are available for this species, the Fat Mouse is considered common within its range, occurring across a range of habitats. For example, Rautenbach et al. (2014) caught this species in a variety of habitats in Phinda Private Game Reserve, KwaZulu-Natal, as well as on surrounding pineapple and cattle farms (Rautenbach 2013). Delcros et al. (2015) caught this species in Acacia woodlands at uMkhuze Game Reserve, KwaZulu-Natal. Power (2011) recorded this species as relatively abundant in Borokalalo National Park, North West province. Additionally, it appears to benefit from agricultural land use and can be the dominant species in areas of subsistence agriculture (P. Taylor unpubl. data). While it does not display population explosions, it is regularly sampled, and therefore considered stable (Monadjem & Perrin 2003; Massawe et al. 2011).
Population Information
Current population trend: Stable
Continuing decline in mature individuals: Unknown
Number of mature individuals in population: Unknown
Number of mature individuals in largest subpopulation: Unknown
Number of subpopulations: Unknown
Severely fragmented: No
Quantitative Analysis
Probability of extinction in the wild within 3 generations or 10 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 5 generations or 20 years, whichever is longer, maximum 100 years: (Not specified)
Probability of extinction in the wild within 100 years: (Not specified)
Population Genetics
While taxonomic issues exist for this species, they are unlikely to impact the species within the assessment region.
To date, no population genetic study has been undertaken on this species making accurate estimates of genetic distinct subpopulations and their diversity not possible. However, because the range of this species is continuous throughout the savanna regions of southern Africa and dispersal is possible both through agricultural lands and transfrontier reserves, it is expected that this species exists as a single metapopulation within the assessment region. Consequently, the Convention on Biological Diversity’s Global Biodiversity Framework’s (GBF’s) complementary genetic indicator – proportion of populations maintained (PM) – would receive a value of 1.0 (all populations remaining).
Because S. pratensis is considered common and occurs across a range of habitats, it is expected to number in excess of 10 000 mature individuals. Based on this assumption, the GBF’s headline genetic indicator – proportion of populations with an effective population size (Ne) above 500 can be quantified using a conversion ratio of 0.1-0.3 Ne/Nc (census size). While the 10 000 value is not an actual census count, the quantity can still be used as a proxy for quantifying the proportion of populations with an Ne > 500 indicator, which is valued at 1.0 (1/1 population with Ne > 500).
It is highly recommended that a population genetic study be undertaken to better understand the population genetic structure and health of the species and to verify the estimated indicator values.
Habitats and ecology
Occurring across a variety of habitat types, Fat Mice are generally associated with open grasslands (subtropical or tropical dry lowland) and dry savannas (Monadjem et al. 2015) and are generally absent from forests and montane grasslands. They have been recorded along the edges of rivers and swamplands in arid regions, within grassland habitats ranging from sparse to densely vegetated cover and are also often associated with cultivated lands (Skinner & Chimimba 2005; Rautenbach 2013). Their affiliation with agricultural areas may be attributed to the soft sandy soils and abundant food supply (Skinner & Chimimba 2005). The species appears to favour recently burnt areas with new grass cover (Monadjem 1999). This species typically occurs at elevations lower than 1,500 m asl (Monadjem et al. 2015).
Fat Mice are nocturnal, live either singly or in pairs (Skinner & Chimimba 2005), and are exclusively terrestrial (Monadjem et al. 2015). Deep burrows with several tunnels are constructed into loose, sandy soils, sloping downwards into a chamber (De Graaf & Nel 1992), where they enter daily torpor during the coolest periods of the year (Richardson & Perrin 1992). The diet of Fat Mice in Kruger National Park in summer comprised 82% seeds, 13% insects and 4% herbage (Watson 1987), which indicates that this species is omnivorous and granivorous. They can gather and store food when it is readily available and do not often leave their burrows during the hot season (Skinner & Chimimba 2005; Kayala 2024). An average of 3.2 young are born seasonally during warm summer months between October and April (Smithers 1971; Kern 1977; Smithers & Wilson 1979; Rautenbach 1982).
Ecosystem and cultural services: This species is a valuable prey resource for nocturnal avian predators (Monadjem et al. 2015), and may contribute to seed dispersal (Linzey & Washok 2000). Fat Mice are hunted locally for bushmeat in certain communities.
IUCN Habitats Classification Scheme
| Habitat | Season | Suitability | Major Importance? |
| 2.1. Savanna -> Savanna – Dry | – | Suitable | – |
| 4.5. Grassland -> Grassland – Subtropical/Tropical Dry | – | Suitable | – |
Life History
Generation Length: (Not specified)
Age at Maturity: Female or unspecified: (Not specified)
Age at Maturity: Male: (Not specified)
Size at Maturity (in cms): Female: (Not specified)
Size at Maturity (in cms): Male: (Not specified)
Longevity: (Not specified)
Average Reproductive Age: (Not specified)
Maximum Size (in cms): (Not specified)
Size at Birth (in cms): (Not specified)
Gestation Time: (Not specified)
Reproductive Periodicity: (Not specified)
Average Annual Fecundity or Litter Size: (Not specified)
Natural Mortality: (Not specified)
Does the species lay eggs? (Not specified)
Does the species give birth to live young: (Not specified)
Does the species exhibit parthenogenesis: (Not specified)
Does the species have a free-living larval stage? (Not specified)
Does the species require water for breeding? (Not specified)
Movement Patterns
Movement Patterns: (Not specified)
Congregatory: (Not specified)
Systems
System: Terrestrial
General Use and Trade Information
Due to their high fat content, Fat Mice are utilised as bushmeat by people in some areas. Usually, they are skewered on a stick, and held over an open flame to burn off their fur. Their meat is considered a delicacy (Skinner & Chimimba 2005). However, this practice has not been reported from the assessment region and is unlikely to affect the population if it does occur locally.
|
Subsistence: |
Rationale: |
Local Commercial: |
Further detail including information on economic value if available: |
|
Yes |
– |
– |
– |
National Commercial Value: No
International Commercial Value: No
|
End Use |
Subsistence |
National |
International |
Other (please specify) |
|
1. Food – human |
true |
– |
– |
– |
Is there harvest from captive/cultivated sources of this species? (Not specified)
Harvest Trend Comments: (Not specified)
Threats
There are no major threats identified for this species. However, grassland transformations, mismanaged fire and over grazing could impact them. It can survive on agricultural and grazed landscapes. Intensive overgrazing may be a minor threat, however, if ground cover is removed or land use is at a high intensity (Starik et al 2020). Anecdotal reports suggest it can also survive in degraded grassland (D. MacFadyen unpubl. data), although more research is necessary. The expansion of mining will also reduce habitat for this species.
Current habitat trend: There is no substantial decline in habitat quality or quantity, as the Savannah Biome is well protected in South Africa (Driver et al. 2012), although mining may cause local declines (MTPA 2014; Jewitt et al. 2015).
Conservation
This species occurs within a number of protected areas within the assessment region, such as Kruger National Park, Borokalalo National Park, Phinda Private Game Reserve, uMkhuze Game Reserve, Nylsvlei Nature Reserve and the Greater Mapungubwe Transfrontier Conservation Area. This species is likely to benefit from protected area expansion to incorporate additional grassland habitats. Additionally, grassland restoration projects and post-mining rehabilitation programmes, which should be the responsibility of mining industries, and enforced by local authorities, would also benefit this species.
Recommendations for land managers and practitioners:
- The species would benefit from suitable land management: land owners should leave corridors of grassland between grazed areas and decrease stocking rates.
- Systematic surveys needed to gather information on population size and trends.
Research priorities:
- Taxonomic resolution of the Steatomys genus.
- Quantification of potential threats.
- Population size, distribution and trend estimates.
- Diet, reproduction and general biology.
Encouraged citizen actions:
- Report sightings on virtual museum platforms (for example, iNaturalist and MammalMAP), especially outside protected areas. However, due to their morphological similarities, misidentification of this species with Steatomys krebsii is common. See taxonomic note for distinguishing characteristics.
Bibliography
Delcros, G., Taylor, P.J., and Schoeman, M.C. 2015. Ecological correlates of small mammal assemblage structure at different spatial scales in the savannah biome of South Africa. Mammalia 79(1): 1-14.
Driver, A., Sink, K.J., Nel, J.N., Holness, S., Van Niekerk, L., Daniels, F., Jonas, Z., Majiedt, P.A., Harris, L. and Maze, K. 2012. National Biodiversity Assessment 2011: An assessment of South Africa’s biodiversity and ecosystems. Synthesis Report. South African National Biodiversity Institute and Department of Environmental Affairs, Pretoria, South Africa.
Jewitt, D., Goodman, P.S., Erasmus, B.F.N., O’Connor, T.G. and Witkowski, E.T.F. 2015. Systematic land-cover change in KwaZulu-Natal, South Africa: Implications for biodiversity. South African Journal of Science 111: 1-9.
Kapuka, A. and Hlásny, T., 2021. Climate change impacts on ecosystems and adaptation options in nine countries in southern Africa: What do we know?. Ecosphere, 12(12), p.e03860.
Kayala, E.N. 2024. Rodent species composition, abundance, richness, and diversity amongst different habitats in two villages in the Mukwe Constituency, Kavango East Region of Namibia (Doctoral dissertation, University of Namibia).
Kern NG. 1977. The influence of fire on populations of small mammals of the Kruger National Park. M.Sc. Thesis. University of Pretoria.
Linzey AV, Washok KA. 2000. Seed removal by ants, birds and rodents in a woodland savanna habitat in Zimbabwe. African Zoology 35: 295–299.
Lynch, C.D. 1994. The mammals of Lesotho. Navorsinge van die Nasionale Museum Bloemfontein 10(4): 177-241.
MTPA. 2014. Mpumalanga Biodiversity Sector Plan Handbook. Mpumalanga Tourism & Parks Agency, Mbombela (Nelspruit), South Africa.
Massawe AW, et al. 2011. Spatial and temporal population dynamics of rodents in three geographically different regions in Africa: Implication for ecologically-based rodent management. African Zoology. 46: 393–405.
Monadjem A, Perrin M. 2003. Population fluctuations and community structure of small mammals in a Swaziland grassland over a three-year period. African Zoology 38: 127-137.
Monadjem A. 1999. Population dynamics of Mus minutoides and Steatomys pratensis (Muridae: Rodentia) in a subtropical grassland in Swaziland. African Journal of Ecology 37: 202–210.
Monadjem, A., Taylor, P.J., Denys, C. and Cotterill, F.P.D. 2015. Rodents of Sub-Saharan Africa: A Biogeographic and Taxonomic Synthesis. . De Gruyter, Berlin, Germany.
Musser, G.G. and Carleton, M.D. 1993. Family Muridae. In: D.E. Wilson and D.A. Reeder (eds), Mammal species of the world: A taxonomic and geographic reference, pp. 501-736. Smithsonian Institution Press, Washington, DC, USA.
Power RJ. 2011. Bojanala Platinum District Mammal Report. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mafikeng, South Africa.
Power, R.J. 2014. The distribution and status of mammals in the North West Province. Department of Economic Development, Environment, Conservation & Tourism, North West Provincial Government, Mahikeng.
Rautenbach A, Dickerson T, Schoeman MC. 2014. Diversity of rodent and shrew assemblages in different vegetation types of the savannah biome in South Africa: no evidence for nested subsets or competition. African Journal of Ecology 52: 30-40.
Rautenbach A. 2013. Patterns and processes of rodent and shrew assemblages in the savanna biome of KwaZulu-Natal, South Africa. M.Sc. Thesis. University of KwaZulu-Natal.
Rautenbach, I. L. 1982. Mammals of the Transvaal. Ecoplan monograph, N.S. Supplemento 1: 111-211.
Richardson EJ, Perrin MR. 1992. Seasonal changes in body mass, torpidity and reproductive activity of captive fat mice, Steatomys pratensis. Israel Journal of Zoology 38: 315–322.
Robbins, L. W. and Baker, R. J. 1978. Karyotypic data for African mammals, with a description of an in vivo bone marrow technique. Bulletin of Carnegie Museum of Natural History 6: 188-210.
Skinner, J.D. and Chimimba, C.T. (eds). 2005. The Mammals of the Southern African Subregion. Cambridge University Press, United Kingdom, Cambridge.
Smithers, R.H.N. 1971. The mammals of Botswana. National Museums of Rhodesia, Museum Memoir 4: 1-340.
Smithers, R.H.N. and Wilson, V.J. 1979. Check List and Atlas of the Mammals of Zimbabwe Rhodesia. Trustees of the National Museums and Monuments of Rhodesia, Salisbury, Rhodesia.
Starik, N., Mbango, O.K., Bengsch, S., Göttert, T. and Zeller, U. 2020. Landscape transformation influences responses of terrestrial small mammals to land use intensity in north-central Namibia. Diversity, 12(12), p.488.
Watson CRB. 1987. The comparative ecology of two small mammal communities in the Kruger National Park. M.Sc. Thesis. University of Pretoria.
de Graaf G, Nel JAJ. 1992. Notes on a single burrow system of the fat mouse Steatomys pratensis in the Kruger National Park. Koedoe 35: 123–124.